Abstract
The aim of this study was to evaluate the toxic effects of drilling fluid (DF) on the skin in a subacute experimental model. Sexually mature outbred rats of both sexes were used (8 males and 8 females per group), weighing 180–220 g. Dermal exposure was performed for 14 days with monitoring of general condition, behavior, and feed and water intake. Body weight, vertical rearing, and grooming behavior were recorded on days 1, 7, and 14. At the end of the experiment, animals were euthanized for biological sample collection. Peripheral blood parameters, including hemoglobin, erythrocytes, leukocytes, platelets, erythrocyte sedimentation rate (ESR), alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), gamma-glutamyl transferase (GGT), and C-reactive protein (CRP), were analyzed. Morphohistological examination of the epidermis, dermis, hair follicles, liver, and kidneys was performed. The elemental composition of the drilling fluid and drill cuttings was determined using atomic absorption and emission spectroscopy. Experimental animals exhibited reduced mobility and decreased feed and water intake by day 7. Body weight initially declined and subsequently increased. Dermal exposure to DF resulted in a significant increase in leukocyte, neutrophil, and eosinophil counts and was associated with alterations in detoxification enzyme activity (ALT, AST, and LDH). Histopathological changes were observed in the skin, liver, and kidneys, indicating dermal-resorptive toxicity. The results demonstrate that drilling fluid possesses dermal-resorptive toxicity following cutaneous exposure, causing not only local skin alterations but also systemic disturbances involving the hematopoietic, immune, and detoxification systems. The combination of clinical-behavioral, hematological, biochemical, and morphological changes confirms the potential occupational hazard of dermal contact with drilling fluids and substantiates the need to consider this route of exposure in occupational risk assessment. The findings provide an experimental basis for improving sanitary and hygienic standards, developing preventive measures, and conducting further toxicological studies, including investigations of chronic exposure, dose–response relationships, and the contribution of individual drilling fluid components.
Introduction
In Kazakhstan, hydrocarbon production has been increasing steadily in recent years, accompanied by environmental pollution.1,2 This pollution affects not only atmospheric air, soil, water, and vegetation but also the health of adults and children. Modern scientific literature provides substantial evidence of pronounced metabolic disturbances in workers engaged in oil production. Drillers and operators involved in underground well repair have been reported to exhibit impaired cytolytic enzyme activity, intensified lipid peroxidation processes, and significant disorders of the overall functional state of the body.3,4 Workers at oil production facilities and populations residing in hydrocarbon-producing regions demonstrate elevated rates of allergic, cardiovascular, pulmonary, and oncological diseases, and new mono- and multifactorial diseases have been reported.5-8
Functional disturbances in drillers and underground operators are directly associated with exposure to hydrocarbons, including vapor–gas mixtures, volatile organic compounds (benzene, toluene, ethylbenzene, and xylenes), polycyclic aromatic hydrocarbons, and aerosols of drilling fluids (DF). 9 Experimental and epidemiological studies have shown that acute inhalation of high concentrations of crude oil and related chemicals is associated with impaired neurological function, manifested by clinical symptoms of neurotoxicity, alterations in electrophysiological parameters of peripheral nerve function, and reduced performance in neurobehavioral tests.10,11 During oil spills in marine environments, significant deterioration of general somatic health has been observed among oil workers, accompanied by neurological symptoms. 12 In addition to neurological manifestations, impairments in respiratory function and immune reactivity have also been reported.9,13 Oil and gas exploration activities are likewise associated with adverse health effects and decreased immune resistance in populations living in proximity to production facilities. 2
Drilling fluids used in technological processes contain a wide range of chemical substances, including weighting agents and foaming agents, corrosion and hydrate inhibitors, flocculants and emulsifiers, and crude oil components.14,15 In recent years, polymers, nanomaterials and nanocomposites, ionic liquids, and silicates have been incorporated into DF formulations to improve drilling efficiency.16-23 Furthermore, technological advances have enabled the use of so-called smart polymers (smart polymer gels and membranes, and biomimetic polymeric materials) in drilling fluids.24-28 Nanomaterials and nanocomposites comprise a broad group of materials, including colloidal/nano-SiO2 (silica), TiO2, Al2O3, ZnO and other metal oxides, carbon-based materials (graphene and graphene oxide, carbon nanotubes (CNTs), g-C3N4), polymer-coated nanoparticles, nanoporous materials, and silicate-based nanocomposites. 29 Ionic liquids include imidazolium-based, phosphonium/phosphonate-based, and quaternary ammonium/phosphonium compounds. 30
Given the multicomponent composition of drilling fluids and drilling cuttings (DC), many of which contain chemicals with insufficiently studied toxicological properties, it is important to evaluate their hazard potential through different routes of entry into the body. Considering the presence of crude oil and heavy metals in DF and DC,31,32 which are integral components of these mixtures, their toxicological characteristics require comprehensive assessment. In addition, DF contains significant amounts of nanomaterials, nanocomposites, and ionic liquids, whose biological effects and toxicity remain poorly understood.
The specific nature of professional activities of drillers, driller assistants, and other technical personnel involved in oil and gas production is associated with frequent contamination of the skin by drilling fluids and drilling waste. Non-compliance with hygienic and sanitary requirements increases the risk of direct cutaneous exposure to numerous chemicals present in DF and DC. Available literature indicates that one of the most common adverse effects of DF is skin irritation and the development of contact dermatitis. Other reported toxic effects include headache, nausea, eye irritation, and cough. 33 Individual hydrocarbons and petroleum products are also known to cause skin irritation. Repeated contact with petroleum hydrocarbons not only exerts an irritant effect but also impairs the barrier function of the skin, thereby increasing its susceptibility to other irritants, sensitizing agents, and microbial contamination.
It has been reported that aromatic hydrocarbons induce more pronounced skin irritation than aliphatic hydrocarbons. In addition to their irritant properties, repeated exposure to petroleum hydrocarbons disrupts the skin barrier, rendering the skin more vulnerable to other irritants, sensitizers, and bacteria. 34
Researchers emphasize the importance of studying both the irritating and resorptive effects of chemicals on the skin.35,36 However, methodological approaches to identifying dermal contamination, assessing the risk of percutaneous absorption of harmful substances, and evaluating their final toxic effects remain heterogeneous. The skin is a complex multifunctional organ that constitutes an integral part of the organism and participates in maintaining systemic homeostasis through shared metabolic processes. At the same time, its barrier and protective functions depend on intrinsic biological properties and on the physicochemical characteristics of harmful substances that may penetrate through it. 37 In general, three principal types of chemical–skin interaction are recognized: direct local effects, immune-mediated skin reactions, and systemic effects resulting from percutaneous absorption.34,38 The available evidence indicates that the dermal route of exposure plays an important role in the development of both general toxic and specific effects of a wide range of xenobiotics, including drilling fluids.
The aim of this study was to characterize the toxic effects of drilling fluid on the skin under conditions of subacute experimental exposure.
Material and Methods
Animals
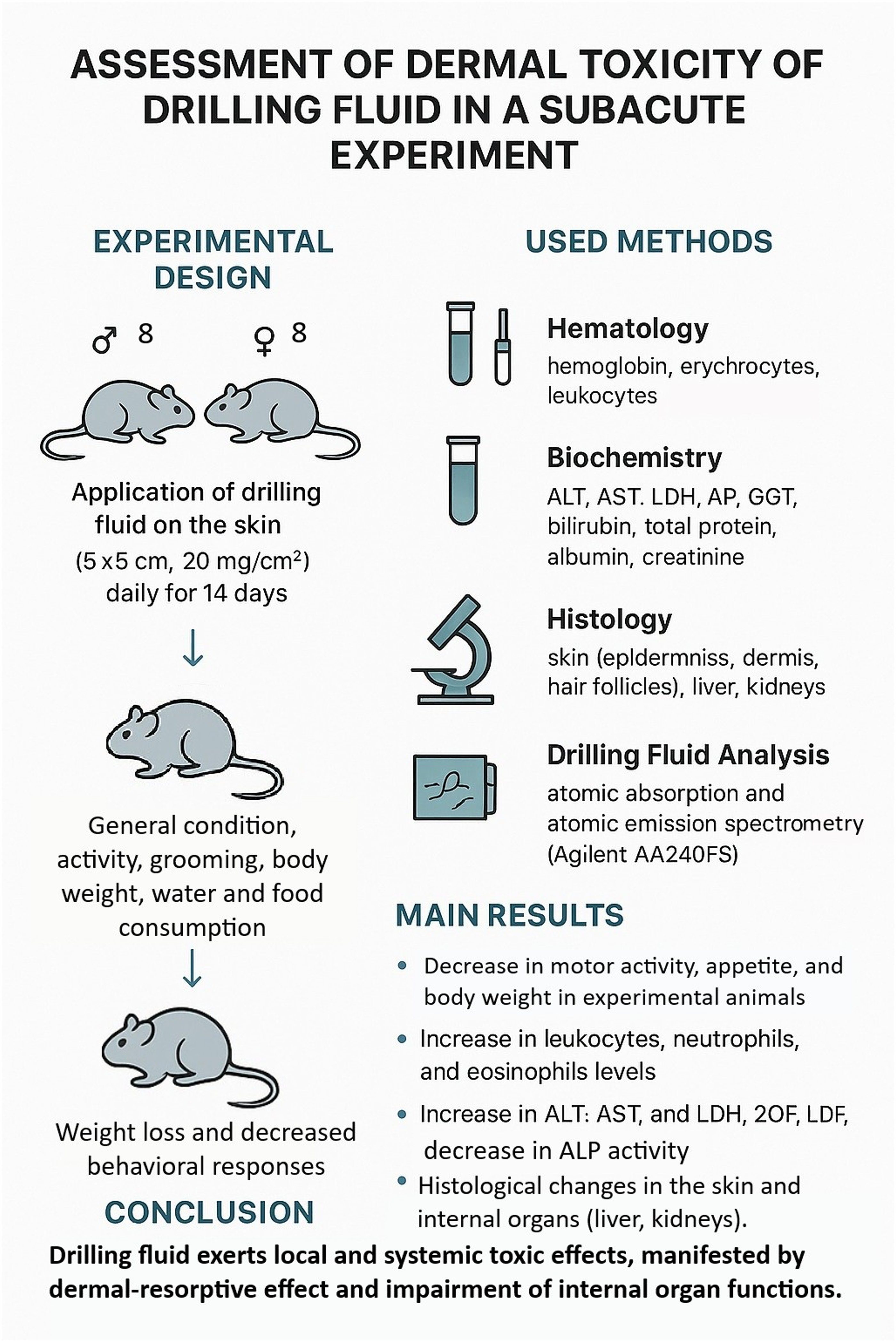
To model subacute dermal exposure, sexually mature outbred rats of both sexes were used. The experimental group consisted of 8 males and 8 females (n = 16), and the control group included 16 animals (8 males and 8 females). The body weight of the animals ranged from 180 to 220 g. The use of outbred rats ensured genetic heterogeneity and increased the extrapolative validity of the findings. Animals were randomly assigned to groups with consideration of body weight; intergroup differences in body weight did not exceed 10%. Species, strain, sex, and age were taken into account.
Sample size was determined using three complementary approaches: (i) a regulatory approach based on standard OECD guidelines, (ii) a statistical approach employing calculations derived from pilot studies and relevant literature, and (iii) an ethical approach in accordance with the principles of the “3Rs” concept (Replacement, Reduction, Refinement).
Inclusion criteria were as follows: age 8–12 weeks; outbred rats of both sexes; body weight 180–220 g; absence of clinical signs of disease; no previous exposure to experimental procedures, chemical agents, or stress; and normal behavioral characteristics. Exclusion criteria included deviations in age or body weight, presence of disease symptoms, abnormal behavior, physical defects, pregnancy, or lactation.
All experimental procedures were performed in strict accordance with the principles of bioethics and laboratory animal welfare, Good Laboratory Practice (GLP) standards, the ARRIVE 2.0 guidelines, and the “3Rs” concept. The study protocol was approved by the Local Bioethics Committee of the Non-Profit Joint-Stock Company “West Kazakhstan Marat Ospanov Medical University” (approval dated March 15, 2023; Protocol No. 3 (3/14)).
Drilling fluid (DF) samples were collected from the Zhanazhol oil field located in Western Kazakhstan and were used in the experiment.
Animal Housing Conditions and Dermal Exposure Protocol
The experimental animals were housed in a specialized university vivarium in compliance with national sanitary regulations (Sanitary Rules for the Design, Equipment, and Maintenance of Experimental Biological Clinics (Vivaria), dated 19.06.1997 No. 8.01.004.97). Animals in both the experimental and control groups had free access to standard chow and water. Hygienic parameters, including natural lighting and ambient temperature, were maintained in accordance with established standards.
All procedures were performed in accordance with the Guidelines P1.2.31.56-13 “Assessment of toxicity and hazard of chemicals and their mixtures for human health” (Moscow, Rospotrebnadzor, 2014, 639 p.). These guidelines are fully consistent with the international OECD Test Guideline No. 427 (Skin Absorption: In Vivo Method). In addition, GOST 33216-2014 (Guidelines for the maintenance and care of laboratory animals – Rules for the maintenance and care of laboratory rodents and rabbits, adopted by the Interstate Council for Standardization, Metrology and Certification, Protocol No. 73-P of December 22, 2014) was followed.
Dermal exposure to the drilling fluid was performed for 14 consecutive days by daily non-occlusive topical application of native drilling fluid to a depilated skin area (5 × 5 cm, approximately 5% of the total body surface area) on the right flank. One to two days prior to exposure, the hair was removed using clippers or scissors. The drilling fluid was applied in its native form at a dose of 20 mg/cm2 using a pipette and spatula.36,39 Non-occlusive conditions were used to more closely simulate occupational exposure scenarios. To minimize grooming behavior and accidental oral ingestion, the substance was applied to an area inaccessible to licking and previously cleared of fur; animals were kept under visual observation after application.
The selected dose and dermal route of exposure are consistent with internationally accepted OECD toxicological approaches (in particular, TG 427), thereby enhancing the regulatory relevance of the study and the comparability of the results with those of other investigations. The use of a single fixed dose corresponds to a screening subacute hazard assessment applied in preventive toxicology. Exclusion of other exposure routes (oral and inhalation) minimized confounding factors and allowed isolated evaluation of dermal absorption and systemic effects.
Throughout the 14-day observation period, the general condition of the animals was monitored, including signs of discomfort or toxicity, as well as food and water intake. The condition of the fur and skin and the intensity and nature of spontaneous motor activity were assessed by visual inspection. Body weight was measured before exposure and on days 1, 7, and 14 of the experiment, and body weight gain was calculated. Motor activity (duration and number of vertical rearings) and the emotional component of behavior (number of grooming episodes) were recorded. The duration of rearings was assessed for 5 minutes on days 1, 7, and 14, while the number of rearings was recorded for 1 hour on the same days.
At the end of the experiment, animals were euthanized using a humane method in accordance with the recommendations of FELASA and ICLAS and the AVMA Guidelines for the Euthanasia of Animals (2020 Edition). To reduce distress, animals were pretreated with an intramuscular injection of xylazine at a dose of 4 mg per 100 g of body weight, followed by decapitation, which ensured rapid loss of consciousness and absence of pain.
Biochemical Analysis and Complete Blood Count
Clinical and laboratory analyses of peripheral blood were performed, including determination of hemoglobin concentration, erythrocyte count, color index, hematocrit, platelet count, and erythrocyte sedimentation rate (ESR). Leukocyte parameters, including total leukocyte count, monocytes, and lymphocytes, were also assessed. All hematological parameters were measured using an automated hematology analyzer (Sysmex XN-550, Japan).
The activity of cytolytic and detoxification-related enzymes—alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), lactate dehydrogenase (LDH), and gamma-glutamyl transpeptidase (GGT)—as well as the concentration of C-reactive protein (CRP) were determined in peripheral blood serum using an automatic modular analyzer (Cobas 6000, immunochemical module C501-1, Switzerland).
Histological Examination
Morphohistological examination of the skin and internal organs was performed in the morphological laboratory of the Department of Histology of the Non-Profit Joint-Stock Company “West Kazakhstan Marat Ospanov Medical University.” The liver and kidneys were additionally sampled for morphological analysis to evaluate potential systemic effects of drilling fluid exposure.
Tissue specimens (∼1 cm3) were fixed in 10% neutral buffered formalin and processed according to standard operating procedures of the morphological laboratory using the paraffin embedding technique.40,41
From the paraffin blocks, histological sections 4–5 μm thick were prepared using a rotary microtome (Accu-Cut SRV 200, Sakura Finetek, USA). For histomorphological evaluation, the sections were stained with hematoxylin and eosin (H&E), both manually and automatically, using a BIO-OPTICA AUS 240 staining system (Bio-Optica, Italy), following previously described protocols.42,43
Microscopic examination was performed using an AxioLab A1 digital light microscope (Zeiss, Germany) at magnifications of ×40, ×100, and ×400. Histological images were captured using an AxioCam ERc5s digital camera (Zeiss, Germany). Morphological assessment included evaluation of the epidermis and dermis, the condition of hair follicles, and measurement of epidermal thickness (stratum corneum, granular, and basal layers) and dermal thickness. Morphometric analysis was performed using ImageJ software.
The severity of histopathological alterations in the liver and kidneys was assessed using a four-point semi-quantitative scoring system as follows: 0, absence of pathological changes with preserved normal tissue architecture; 1 (mild), minimal focal alterations involving limited tissue areas or isolated cells; 2 (moderate), clearly detectable changes affecting a substantial portion of the organ parenchyma; and 3 (severe), diffuse and pronounced pathological alterations accompanied by marked disruption of tissue architecture.44,45
Elemental Analysis of Drilling Fluid and Drill Cuttings
Atomic absorption spectroscopy (Agilent AA240FS, USA) was used to determine the concentration of heavy metals in drilling fluid (DF) and drill cuttings (DC) collected from well SIV No. 64013. Heavy metal content in DF and DC was analyzed at the Regional Center for Sanitary and Epidemiological Expertise (Aktobe, Kazakhstan). Sample preparation and mineralization were carried out according to GOST 26929-94 (Raw materials and food products. Sample preparation. Mineralization for determination of toxic element content; Moscow: Standartinform, 2010, 122 p.), which specifies procedures for sample digestion and analytical conditions.
Atomic emission spectroscopy was used to analyze the elemental composition of DC samples (both unwashed and in liquid form). These analyses were conducted at the Geoplasma LLC laboratory in accordance with methodological guidelines MVI No. 01-IL-2016 and MVI No. 499-AES/MS MKHA. Chemical analysis for oxides and metals was performed in 10 samples.
Statistical Analysis
Statistical analysis of the study data was performed using methods of variational statistics. For all quantitative variables, the arithmetic mean (M) and standard deviation (SD) were calculated. Intergroup comparisons between the control and experimental groups were conducted separately for males and females using the parametric Student’s t-test for normally distributed data and the nonparametric Mann–Whitney U test for non-normally distributed data or small sample sizes. For multiple comparisons, the Bonferroni correction was applied to reduce the risk of type I error. A p value ≤ 0.05 was considered statistically significant. Levels of statistical significance were defined as p < 0.05, p < 0.01, and p < 0.001. Normality of data distribution was assessed using the Shapiro–Wilk test.
This research was conducted within the framework of the scientific project IRN AP19676915, “Development of toxicometric criteria for the hazard and toxicity of drilling fluid and drill cuttings,” funded by the Science Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan.
Results
The results of the subacute dermal toxicity study indicate that drilling fluid exhibits measurable toxic effects. Compared with the control group, experimental animals demonstrated reduced mobility, signs of functional depression, and decreased food and water intake on day 7 of exposure. By day 14, these changes showed a tendency toward normalization.
In the control group, food consumption on days 1, 7, and 14 of the experiment was 24.40 ± 1.07, 24.00 ± 1.25, and 24.40 ± 0.52 g/day in male rats, and 24.40 ± 0.70, 24.20 ± 1.03, and 24.60 ± 0.70 g/day in female rats, respectively.
In the experimental group, food consumption on days 1, 7, and 14 was 24.20 ± 0.79 g/day (p = 0.357), 15.20 ± 0.92 g/day (p < 0.001), and 24.30 ± 0.67 g/day (p = 0.796) in male rats, and 23.50 ± 1.18 g/day (p = 0.074), 14.20 ± 1.13 g/day (p < 0.001), and 24.80 ± 0.42 g/day (p = 0.549) in female rats, respectively.
As shown in Figure 1, feed consumption in the experimental groups decreased markedly in the middle of the experimental period, followed by recovery toward control values by day 14. Feed consumption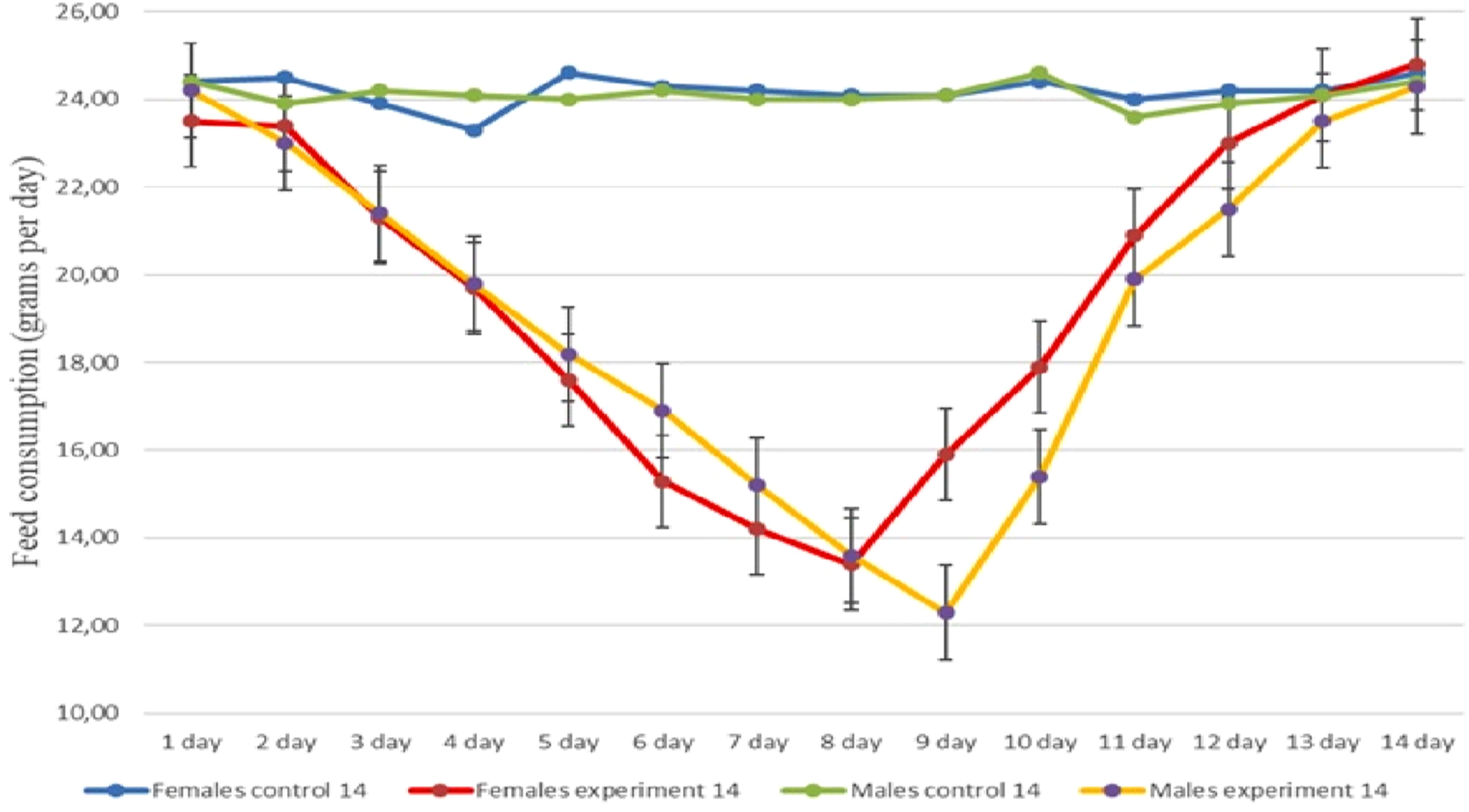
Dynamics of Body Weight Changes

Note: *Statistically significant differences between groups were identified using the Mann–Whitney U test (p < 0.05).
Behavioral Reactions
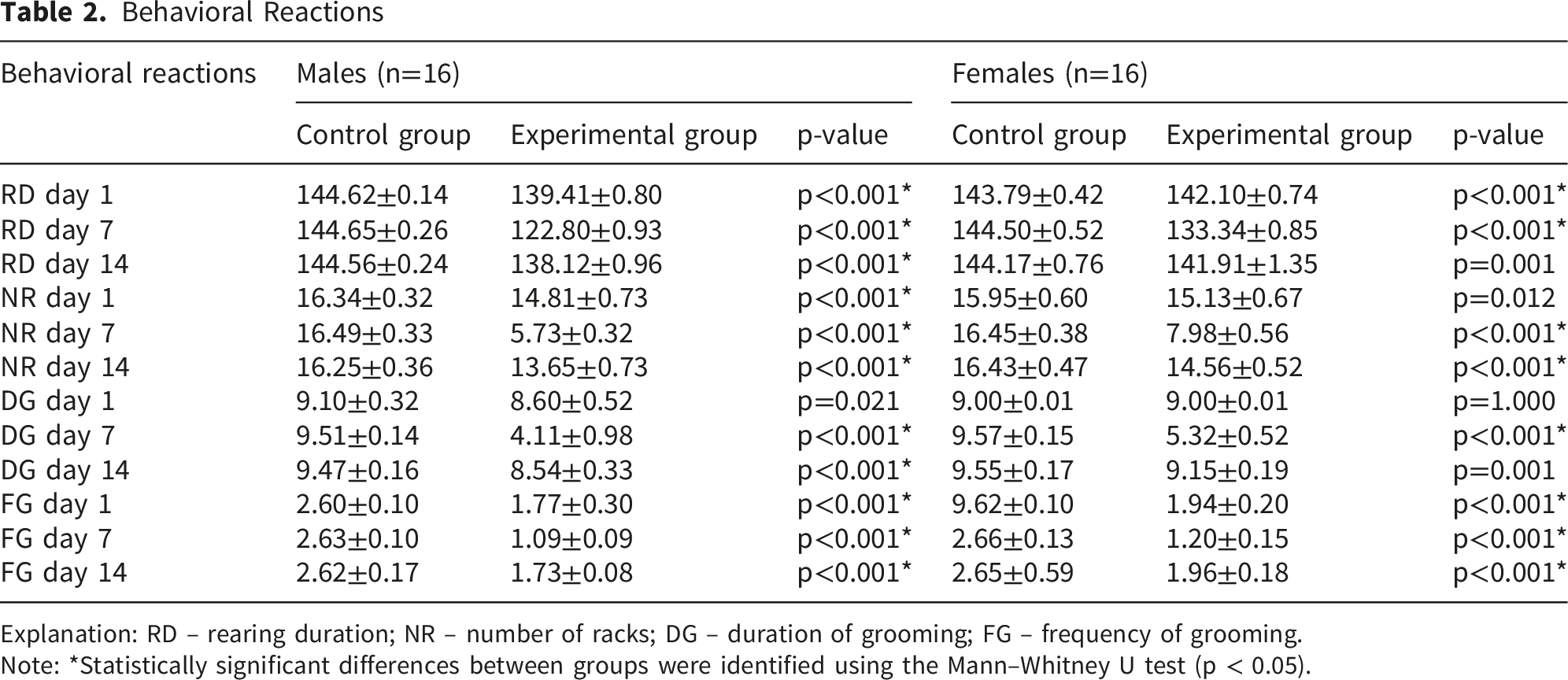
Explanation: RD – rearing duration; NR – number of racks; DG – duration of grooming; FG – frequency of grooming.
Note: *Statistically significant differences between groups were identified using the Mann–Whitney U test (p < 0.05).
Assessment of grooming behavior also indicated toxic effects of dermal exposure to drilling fluid. Both the duration and frequency of grooming decreased by the middle of the experiment, with subsequent restoration toward control values by day 14.
Organ Weight Coefficients (M ± SD)
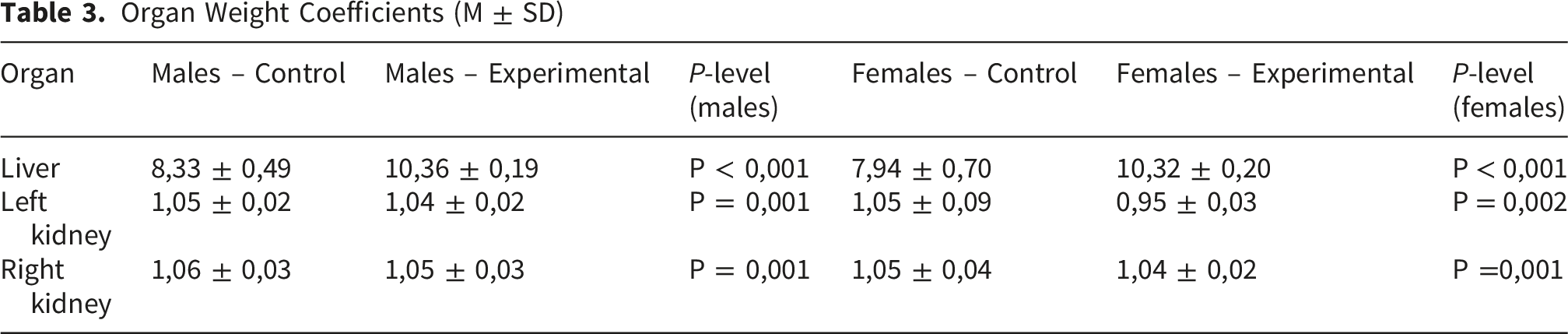
In the control group, the liver weight coefficient was M = 7.94, SD = 0.70 in males and M = 8.33, SD = 0.49 in females. In the experimental group, this parameter was significantly higher compared with controls, reaching M = 10.36, SD = 0.19 in males (p < 0.001) and M = 10.32, SD = 0.20 in females (p < 0.001).
No statistically significant changes in the weight coefficient of the kidneys were observed.
Blood plays essential transport, respiratory, nutritive, excretory, thermoregulatory, protective, and homeostatic roles, which depend on the functional state of the organism and exposure to chemical factors. In the present study, morphological blood indices and selected biochemical parameters were therefore analyzed.
Dermal exposure to drilling fluid resulted in a statistically significant increase in total leukocyte counts in experimental animals compared with controls: M = 7.80, SD = 0.09 in males (p < 0.001) and M = 7.68, SD = 0.09 in females (p < 0.001), versus M = 7.34, SD = 0.14 in control males and M = 7.28, SD = 0.13 in control females. The absolute numbers of neutrophils and eosinophils were also elevated in the experimental groups.
Peripheral Blood Parameters in Experimental and Control Animals (M ± SD)

Significant alterations were also detected in erythrocyte hemoglobin indices. These parameters appeared sensitive to the toxic effects of drilling fluid under dermal-resorptive exposure. In the control group, mean hemoglobin concentration in erythrocytes was M = 337.20, SD = 1.32 in males and M = 335.80, SD = 1.14 in females. In the experimental group, this parameter increased to M = 344.30, SD = 4.19 in males (p < 0.001) and to M = 340.90, SD = 3.60 in females (p = 0.003).
Statistically significant changes were also observed in total bilirubin levels. In experimental males, bilirubin concentration increased to M = 1.70, SD = 0.03 (p < 0.001), compared with control values of M = 1.43, SD = 0.08. Other peripheral blood parameters in experimental animals did not differ significantly from control values.
Biochemical Blood Parameters of Control and Experimental Animals (M ± SD)

An increase in lactate dehydrogenase (LDH) activity was also detected (control: M = 675.80, SD = 17.12; experimental: M = 782.90, SD = 69.60; p = 0.001). In contrast, alkaline phosphatase (ALP) activity decreased significantly in the experimental group compared with controls (control: M = 113.70, SD = 8.17; experimental: M = 91.10, SD = 6.39; p < 0.001). The activity of γ-glutamyl transpeptidase (GGT) remained essentially unchanged.
A tendency toward reduced total protein and creatinine concentrations was observed in experimental animals. Conversely, bilirubin levels increased significantly (control: M = 1.43, SD = 0.08; experimental: M = 1.70, SD = 0.03; p < 0.001). In addition, a decrease in serum albumin concentration was detected (control: M = 61.02, SD = 6.56; experimental: M = 55.50, SD = 0.74; p = 0.064). Similar trends in these biochemical parameters were also observed in female rats exposed to drilling fluid compared with their respective controls.
Histological examination of the skin of experimental rats revealed several characteristic alterations affecting all layers of the epidermis and dermis. Compared with the control group, moderate epidermal thickening was observed after 14 days of drilling fluid application, mainly due to an increased thickness of the stratum corneum. These changes exhibited a mosaic pattern and were occasionally accompanied by focal epidermal thinning.
Pronounced cellular polymorphism of basal epidermal cells was noted, associated with a reduction in their linear dimensions. In addition, rare focal mitotic figures were detected during the experimental period compared with controls, indicating suppression of basal cell proliferative activity. According to these observations, such alterations resulted in focal thinning of the epidermis (Figure 2). Mosaic pattern of epidermal alterations in rat skin following drilling fluid (DF) exposure (day 14 of the experiment). (A) Normal skin structure in control rats; (B) focal thickening and stratification of the stratum corneum (FTh-SC); (C) focal inflammatory infiltrate (FI) and moderate dermal edema (ME); (D) thinned epidermis (TEpi). Staining: hematoxylin and eosin. Magnification: ×40
Morphometric analysis of epidermal layers on day 14 of the experiment demonstrated a significant thickening of the stratum corneum in male rats of the experimental group (M = 31.64 μm, SD = 8.75, p = 0.001) compared with controls (M = 16.59 μm, SD = 2.33). A statistically significant reduction in the thickness of the germinal layer of the epidermis was observed in females of the experimental group (M = 14.22 μm, SD = 2.73, p < 0.001) relative to the control group (M = 14.90 μm, SD = 1.10)
Histological Structure of the Skin in Animals of the Control and Experimental Groups (M ± SD)

Note. M — mean value; SD — standard deviation; P — level of statistical significance of differences between the control and experimental groups; ♂ — males; ♀ — females.
The general toxic effects of drilling fluid (DF) were further evaluated in a subacute experiment by assessing morphological changes in internal organs (liver and kidneys) following 14 days of dermal exposure. Histological examination revealed distinct microscopic alterations in both organs.
After 14 days of dermal application of the drilling fluid (DF), histological examination demonstrated that the liver architecture of male and female rats in the control group remained within physiological limits (score 0). The central vein was centrally located within the hepatic lobule and surrounded by radially arranged hepatic cords. Hepatocytes in all lobular zones exhibited well-defined round nuclei with visible nucleoli and homogeneous cytoplasm, while portal tracts preserved normal histological organization without evidence of vascular congestion, inflammatory infiltration, or degenerative changes (score 0).
In contrast, histopathological evaluation of the liver in male rats on day 14 of exposure revealed treatment-related alterations of mild to moderate severity. Semi-quantitative assessment showed uneven dilation and congestion of venous vessels, including central veins, corresponding predominantly to mild-to-moderate vascular changes (scores 1–2). Mild lymphocytic infiltration was observed in portal tracts and was graded as score 1. Additionally, isolated hepatocytes exhibiting ballooning degeneration were identified, consistent with mild hepatocellular degenerative changes (score 1). No diffuse architectural disruption or extensive hepatocellular necrosis was detected (Figure 3). Morphological alterations in rat liver following drilling fluid (DF) exposure: mild lymphocytic infiltration in the portal tract (black arrows) and congestion of the central vein (blue arrows). Staining: hematoxylin and eosin. Magnification: ×100 Morphological alterations in rat kidney following drilling fluid (DF) exposure: expansion of the space between the glomerular tuft and Bowman’s capsule (black arrows) and vascular congestion (blue arrows). Staining: hematoxylin and eosin. Magnification: ×100
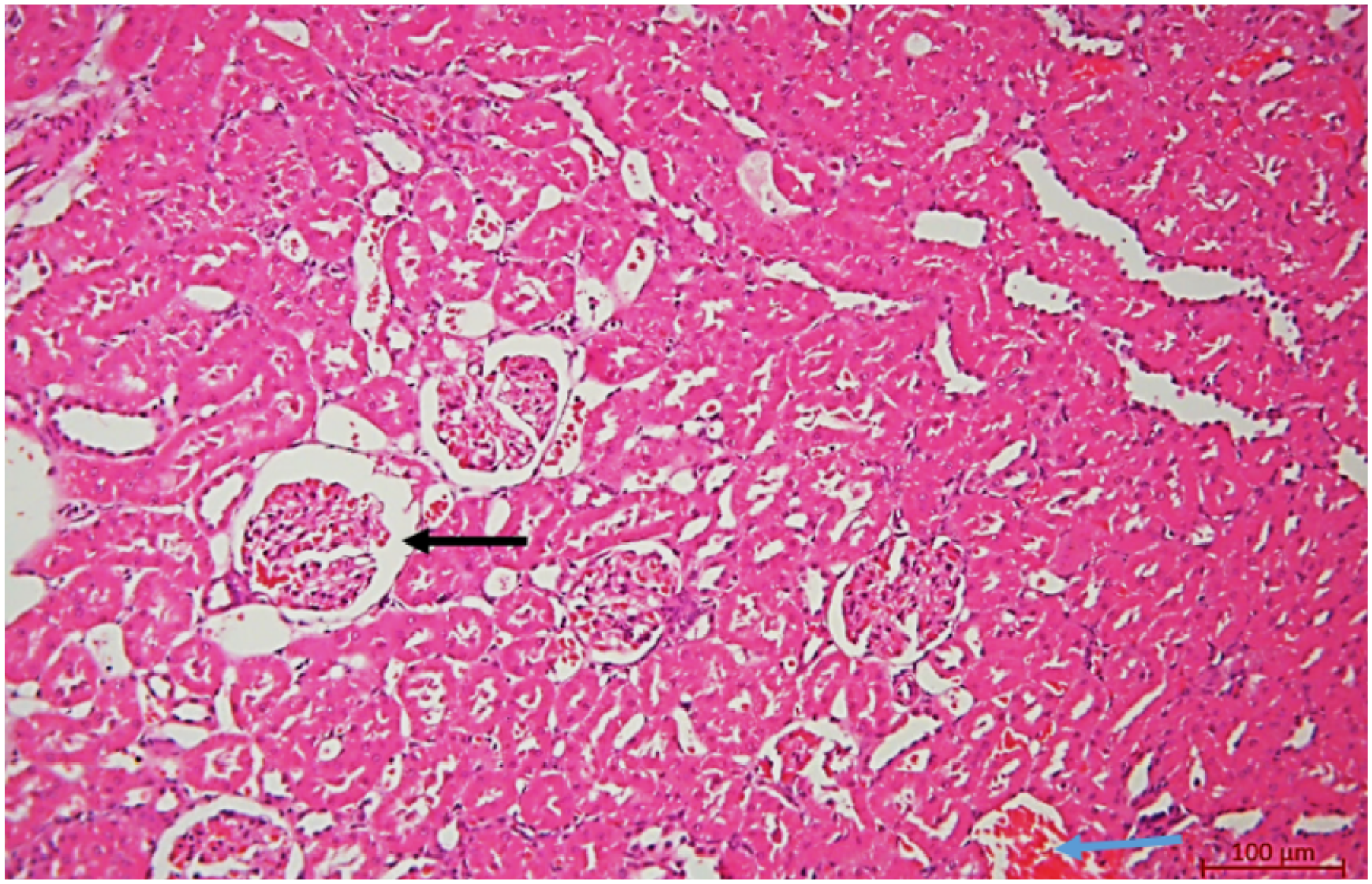
Histological examination of the kidneys in control animals revealed a normal microscopic structure (score 0). In experimental male rats, after 14 days of exposure to the drilling solution, mild to moderate histopathological changes were observed. Specifically, there was a mild to moderate expansion of the space between the vascular glomerulus and Bowman’s capsule due to edema (score 1–2), moderate capillary congestion in the glomeruli (score 2), and mild vacuolization of epithelial cells in some renal tubules (score 1). A weak inflammatory infiltrate (score 1) and mild erythrostasis in the lumens of interstitial vessels (score 1) were also noted (Figure 4). Overall, the morphological alterations in the liver and kidneys were primarily associated with mild to moderate disturbances in organ blood circulation (score 1–2).
In the present study, the concentrations of heavy metals in drilling fluid (DF) and drill cuttings (DC) were determined. In DF, the lead (Pb) concentration was 0.073 ± 0.006 mg/dm3 (maximum allowable concentration (MAC) for drinking water: 0.03 mg/L; p < 0.05), while the arsenic (As) concentration was 0.00014 ± 0.0004 mg/dm3 (MAC: 0.05 mg/L; p < 0.05).
The content of metal oxides in drill cuttings (%) was as follows: phosphorus pentoxide (P2O5, M = 0.114, m = 0.006992), silicon dioxide (SiO2, M = 33.79, m = 2.023995), calcium oxide (CaO, M = 20.51, m = 1.412209), sodium oxide (Na2O, M = 1.104, m = 0.083293), and potassium oxide (K2O, M = 0.756, m = 0.04402).
Metal Concentrations in Drilling Sludge Expressed in Different Units of Measurement

Note. M — mean metal concentration; SD — standard deviation; ppm = mg/kg; ‰ = g/kg.
Discussion
The results of the present study demonstrate that drilling fluid (DF), under subacute experimental conditions with dermal exposure, induces adverse changes in experimental animals. Cutaneous application of DF was associated with deterioration of the general condition, manifested by reduced mobility and food intake, impaired behavioral responses, decreased body weight in experimental groups compared with controls, and alterations in organ weight coefficients. Notable changes were also observed in peripheral blood parameters and in the activity of cytolytic enzymes. Histomorphological examination of the skin, liver, and kidneys revealed pathological alterations indicative of the toxic effects of DF in rats of both sexes.
A comprehensive assessment of systemic and local effects of DF, integrating behavioral, hematological, biochemical, and histological parameters within a single experimental model, convincingly demonstrates not only local skin damage but also the development of systemic toxicity. These effects include hepatotoxicity, nephrotoxicity, and induction of an inflammatory response, as reflected by leukocytosis, increased enzyme activity, and structural changes in the skin. The observed histological alterations in the skin, together with morphofunctional disturbances in internal organs, indicate penetration of DF components through the skin barrier and their subsequent systemic action. The obtained data provide a basis for classifying DF as a potentially hazardous agent upon dermal exposure.
The skin performs not only a barrier function but also numerous physiological roles linking it with internal organs and the external environment.46,47 All structural elements of the skin form an integrated system characterized by metabolic unity and continuous adaptive and compensatory responses. Of particular importance are studies demonstrating the regulatory role of the skin in immune reactivity. The skin represents the largest immunologically active organ, consisting of a complex network of immune and non-immune cells and mediators. 48 At the same time, its barrier-protective properties may be disrupted by exposure to a wide range of chemicals, including DF. Percutaneous absorption of DF components may occur directly through the epidermis, with individual constituents penetrating via the stratum corneum and papillary dermis, as well as through hair follicles, sebaceous glands, sweat gland ducts, and the walls of blood vessels.
It is well recognized that hydrocarbon extraction processes expose workers to a complex combination of harmful occupational factors, among which the toxicological characteristics of DF, drill cuttings (DC), and drilling waste remain insufficiently studied. Because drilling operations are conducted predominantly in open environments, workers’ skin is subjected not only to chemical exposure but also to adverse meteorological factors, including ultraviolet radiation, which may modulate physiological and pathological processes in the skin.49,50 Inadequate use of protective clothing, poor hygienic conditions, and mechanical damage to the skin may further enhance the adverse effects of DF. In some cases, the dermal route of exposure may be as important as, or even more important than, the oral route for certain chemicals. 51
Epidemiological and experimental assessments of occupational exposure to crude oil vapors indicate that oil production workers are at risk of both dermal contact with crude oil and inhalation of its vapors. Prolonged exposure to crude oil and oil production waste is accompanied by significant changes in clinical and biochemical parameters and in the functional state of various organs and systems. 52 Combined and synergistic effects of crude oil vapors, noise, and unfavorable microclimatic conditions substantially aggravate pathological processes in exposed workers. 53 Individuals employed in this sector are subjected to multiple stressors, including chemical agents, noise, thermal load, and psychological strain, and their working conditions are therefore classified as hazardous. 54 The cumulative impact of gasoline, crude oil, asbestos, and welding aerosols has also been documented. 55 These findings emphasize the multifactorial nature of occupational exposure in hydrocarbon production and highlight the importance of toxicological assessment of DF and drilling sludge as key industrial hazards.
Numerous studies have demonstrated that, despite anatomical and physiological differences between the skin of experimental animals and that of humans, rodents remain reliable in vivo models for toxicological investigations.55,56 Accordingly, the present findings provide experimental evidence of both local dermal and systemic effects of DF.
Previous studies on dermal absorption of crude oil in rats showed that 13 weeks after topical application, systemic effects developed, including reduced body weight gain, increased liver mass, decreased thymus mass, and hematological alterations. 57 Reviews of dermal penetration of oil products indicate that aromatic hydrocarbons penetrate the skin to a significantly greater extent than aliphatic hydrocarbons, and that absorption depends strongly on chemical composition and experimental conditions.
In the present study, histological examination of the skin revealed characteristic alterations affecting both the epidermis and dermis, including focal thickening and stratification of the stratum corneum and thinning of the germinal layer. Together with the presence of focal inflammatory infiltrates and moderate dermal edema, these findings indicate the manifestation of dermal toxicity induced by DF. Factors such as skin integrity and hydration, density of hair follicles and sebaceous glands, dermal thickness at the exposure site, physicochemical properties of individual components, their concentration, and exposure duration play a crucial role in the development of dermal toxicity.58-60 Only a limited number of studies have directly addressed dermatitis and dermal toxicity specifically associated with DF. 61 At the same time, extensive evidence demonstrates that crude oil, its constituents, and aromatic hydrocarbons exert pronounced dermal toxicity. 34 This is of particular relevance because the most effective DF formulations from an engineering perspective often contain oil components. Mechanisms of apoptosis in skin cells exposed to bituminous products have been convincingly demonstrated. 62 The liver and kidney alterations, changes in peripheral blood indices, and increased cytolytic enzyme activity observed in the present study further indicate the development of systemic toxicity following dermal exposure to DF.
Substantial evidence has also accumulated regarding the adverse environmental effects of DF on soil, surface water, and groundwater.63,64 These effects are accompanied by phytotoxicity, 65 disruption of soil biota, and changes in physicochemical soil properties. 66 Aquatic organisms, including invertebrates, crustaceans, mollusks, and fish, have been shown to be highly sensitive to DF exposure.67-69 Considering the presence of heavy metals and nanoparticles in DF and drilling waste, this mechanism of toxicity is also relevant for environmental and population health. Given the persistence of heavy metals and their capacity for bioaccumulation in ecosystems,70-72 there is a substantial risk of adverse effects via industrial waste, water, air, and food chains. In this context, particular attention should be paid to metal-containing nanoparticles used in DF formulations.
Nanoparticles possess unique physicochemical properties and are often more toxic than their bulk counterparts.73,74 Several studies have demonstrated that pathological skin conditions associated with barrier disruption, such as dermatitis or ultraviolet irradiation, increase the penetration of topically applied metal oxide nanoparticles.75,76 Dermal exposure to zinc oxide nanoparticles has been shown to induce melanoma-like skin lesions accompanied by oxidative stress. 77 Topical application of silver nanoparticles for 14 days caused concentration-dependent skin reactions characterized by intra- and intercellular epidermal edema and focal epidermal and dermal inflammation, sometimes accompanied by epidermal hyperplasia and parakeratosis. 78 After penetrating the skin, nanoparticles may induce oxidative stress, apoptosis, mitochondrial dysfunction, cytomembrane damage, and DNA injury. 79 Therefore, assessment of the toxicity of DF and drilling sludge should consider not only the native materials but also the complex multicomponent nature of their chemical composition.
This study makes an important interdisciplinary contribution to toxicology, occupational hygiene, industrial hygiene, and ecotoxicology. It provides experimental evidence of both local and systemic toxic effects of drilling fluid under subacute dermal exposure, which is critical for occupational risk assessment, development of sanitary standards, improvement of workplace safety in the oil production industry, and establishment of a national toxicological database. The use of a single dose level of drilling fluid administered via the dermal route in a subacute experimental study is methodologically justified and regulatorily relevant for the primary assessment of substance toxicity and hazard. Employing a single dose reduces intragroup variability associated with different exposure levels and facilitates the interpretation of biochemical, hematological, and morphological changes. At the same time, the absence of multi-dose exposure limits the analysis of dose–response relationships, quantitative risk assessment, and extrapolation of the obtained data, thereby necessitating further studies using an expanded dose range and combined exposure routes.80,81
The limitations of the present experimental model are also related to the use of outbred rats, which restricts extrapolation of the findings to other animal species and humans. The subacute nature of the experiment, with a drilling fluid exposure duration of 14 days, allows assessment of only subacute toxic effects. In contrast, chronic, cumulative, and delayed toxic consequences of prolonged dermal contact under long-term exposure conditions remain insufficiently investigated. Interspecies differences in skin permeability, metabolism, and detoxification systems may further influence the severity of toxic effects. 82 Moreover, the complex and variable chemical composition of drilling fluids complicates the identification of specific components responsible for the observed toxic effects and limits the reproducibility of results when drilling fluids obtained from different oil fields are used. 83
Conclusion
An in vivo model of subacute dermal exposure to drilling fluid (DF) was developed in this study in compliance with OECD, GLP, and ARRIVE guidelines, with strict adherence to the 3R principles. The obtained toxicometric data indicate the presence of both dermal-resorptive and systemic toxic effects of the drilling fluid, manifested by alterations in the general condition of the animals, body weight parameters, relative organ weights, peripheral blood indices, and the activity of cytolysis-related enzymes. Morphological and histological changes observed in the skin, liver, and kidneys further confirm the systemic nature of the toxic effects.
The results of this study may be used to substantiate hygienic standards, assess occupational health risks, develop safety data sheets, and support the inclusion of drilling fluids and their components in the National Register of Chemical Substances.84-87
A representative in vivo model of subacute dermal exposure to drilling fluid was established in accordance with international OECD, GLP, and ARRIVE requirements and based on the 3R principles. It was demonstrated that subacute dermal exposure to drilling fluid exerts an adverse effect on experimental animals, manifested by impairments in general condition, spontaneous locomotor activity, feed consumption, and body weight dynamics.
Statistically significant changes in relative organ weight indices, peripheral blood parameters, and the activity of enzymes involved in cytolytic processes were identified, indicating the toxic effects of drilling fluid. Morphological and histological alterations in the skin, liver, and kidneys confirm the systemic toxic impact of drilling fluid following dermal exposure. Analysis of the chemical composition of the drilling fluid and drilling cuttings complements the toxicological characterization of the investigated substance and may be applied in the development of safety data sheets, establishment of hygienic regulations, and assessment of occupational risks in the oil and gas industry.
Footnotes
ORCID iDs
Ethical Considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Local Commission on Bioethics of the West Kazakhstan Medical University (meeting No. 3 (3/14) dated 15 March 2023).
Authors Contributions
Conceptualization, Zh.K., A.M., S.B.; Methodology, Zh.K., A.M., and S.B.; Formal Analysis, Zh.K., A.Zh.; Investigation, A.Zh., A.K.; Data Curation, A.Zh., and F.R.; Writing – Original Draft Preparation, Zh.K., A.Zh., A.M., A.K., E.G.; Writing – Review and Editing, Zh.K., A.Zh., S.B., E.G., F.R., and Zh.Sh., B.N.; Supervision, A.M.; Project Administration, A.M.; Funding Acquisition, A.M. All authors have read and agreed to the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was funded by the Science Committee of the Ministry of Science and Higher Education of the Republic of Kazakhstan (Grant No. AP19676915).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data are available from the corresponding author upon request.
