Abstract
Background
Ionizing radiation (IR) has the potential to harm cellular macromolecules, raising concerns across a range of exposure contexts. This study investigates the radioprotective potential of Gentiana lutea root extract (GRE), a natural product known for its antioxidant properties.
Research Design
The GRE composition was analysed using ultra-performance liquid chromatography (UPLC). Peripheral blood mononuclear cells were treated with 0.25-2 mg/mL GRE 24 hours prior to 0.5 and 2 Gy gamma radiation exposure (Co-60 source). Oxidative stress and antioxidant responses were evaluated via lipid peroxidation products, reduced glutathione, and pro-oxidant/antioxidant balance assays. Genomic damage was assessed using the micronuclei test, comet and colorimetric DNA fragmentation assay, and DNA repair was assessed through evaluation of gene expression levels that are involved in base excision repair.
Results
The presence of gentiopicroside, loganic acid, sweroside, swertiamarin, and rosmarinic acid was confirmed by UPLC analysis. GRE pretreatment significantly improved cell viability, reduced oxidative and genomic damage, and enhanced antioxidative responses in irradiated cells.
Conclusions
Natural compounds in GRE induce cellular responses that attenuate sublethal radiation damage in human PBMCs in vitro, supporting its further evaluation as a potential radioprotective strategy.

Introduction
The living world has been exposed to natural radiation since its inception. Natural radiation comes from three main sources: the cosmos, primary natural radionuclides, and secondary radionuclides derived from the decay of primordial radionuclides. 1 Furthermore, the development of human civilization and nuclear technology created artificial sources of ionizing radiation (IR), which include, among others, X-rays in medicine and radioactive medical waste, products created by nuclear tests in the atmosphere, radioactive waste from industry, industrial gamma rays, and various objects produced for human use.2,3 Ionizing radiation has a direct and indirect impact on cell molecules. The direct impact is caused by the transfer of energy to cell molecules and leads to the breaking of bonds within the biomolecules, affecting genetic material by creating DNA damage. The indirect effects are caused by hydroxyl radicals (•OH), generated in the process of water radiolysis, and the interaction of reactive oxygenated species (ROS) with cell molecules leading to the breakdown of nucleic acids, lipids, and proteins and forming additional reactive molecules such as lipid hydroperoxides (LOOH), hydroxynonenal (HNE), malondialdehyde (MDA), acrolein, and protein radicals, which further interact with cellular structures and create additional damage. 4 Hematopoietic tissue is particularly radiosensitive, where after exposure to IR, a significant decrease in the number of lymphocytes can be observed. 5 Hence, peripheral blood mononuclear cells (PBMCs) are commonly used as a good and reliable model system for testing the radioprotective effect in vitro. 6 Mechanisms of protection against IR include the activation of antioxidant mechanisms and the repair of damaged cellular molecules. The radioprotective agent needs to have a stable formulation, offer substantial protection, be simple to use, and not be extremely harmful to especially vulnerable tissues. Special attention is devoted to the study of radioprotectors of plant origin. Of the 1144 new drugs developed in the last 25 years, about 60% come from natural resources. To date, about 74 herbal products have been tested for their radioprotective potential in various in vitro and in vivo studies. 7 Gentiana lutea root extract (GRE) was shown to activate antioxidant and repair mechanisms in damaged cells.8,9 Antioxidant defense (AO) is achieved either directly by scavenging free radicals, or indirectly by activating enzymes or their de novo synthesis, which is regulated by the activation of transcription factors such as nuclear factor-erythroid factor 2-related factor 2 (Nrf 2). 10 GRE’s chemical composition demonstrated antioxidant and genoprotective properties, which represent strong indications that its application can achieve radioprotective effects.11,12 The finding of natural products that are accessible and not harmful to exposed tissues, to be used as radioprotectors, could offer a simple solution to prevent cellular damage induced by IR. The development of nuclear technology in diagnostic, therapeutic and industrial applications justifies research on the radioprotective potential of plants and may represent a safe preventive measure against radiation-induced damage. The main goal of this research is to analyze the effects of GRE on genomic damage and oxidative stress parameters induced by ɤ radiation in PBMCs, starting from the assumption that GRE pretreatment can contribute to the reduction of genomic damage induced by IR.
Materials and Methods
Materials
The plant extract was prepared from a commercially available Gentiana lutea root, a product of the Institute for the Study of Medicinal Plants “Dr. Josif Pančić” Belgrade, Republic of Serbia (Management of product safety and quality policy: SPRS ISO 9001:2015, codex alimentarius [CAC/RCP 1-1969, rev. 4-2003)]. Chemical substances used for cell culture were mononuclear isolation medium (LSM-A), RPMI 1640 medium, and phytohemagglutinin (PHA-M) (Capricorn Scientific, Germany). Trypan blue dye (TB) was used for the cell viability test (Stemcell Technologies, Vancouver, Canada). Chemical substances used in the examination of oxidative stress parameters: 5,5′-dithio-bis-(2-nitrobenzoic acid) (DTNB), reduced glutathione (GSH), horseradish peroxidase (HRP), 3,3′,5,5′-tetramethylbenzidine (TMB), chloramine T, hydrogen peroxide, uric acid, 1-methyl-2- phenylindole, tris base, acetonitrile, Triton X-100, 1,1,3,3-tetramethoxypropane, methanesulfonic acid, acetaldehyde, as well as for cytokinesis blocking micronucleus assay (CBMN): Cytochalasin B, Giemsa stain, are products of Sigma Aldrich (St. Louis, MO, USA). Chemicals used for quantitative real-time PCR: Тhe High Capacity cDNA Reverse Transcription Kit, TaqMan assays and TaqMan Universal Master Mix were produced by Applied Biosystems (Foster City, CA, USA).
GRE Synthesis
The aqueous extract was prepared by heating ground Gentiana lutea roots for 10 min in a 1:5 (m/V) ratio. The obtained infusion was filtered, and afterward, GRE was lyophilized and stored in sample tubes at 4°C until use. Prior to analysis lyophilized powder was dissolved in water to obtain a concentration of 50 mg/mL, passed through 0.2 μm Minisart filter (Göttingen, Germany), and used as a stock solution in further experiments.
Ultra-Performance Liquid Chromatography Analysis
Gradient Condition Details Used for UPLC Separation

Isolation, Cultivation, and Treatment of Human Peripheral Blood Mononuclear Cells
Peripheral blood mononuclear cells (PBMCs) were isolated from blood samples obtained from three healthy volunteers, and collected in Li-heparin vacutainers using a lymphocyte separation medium. The study was conducted in accordance with the Declaration of Helsinki, and was approved by the Ethics Committee of the “Vinča” Institute of Nuclear Sciences – National Institute of the Republic of Serbia (Ethical approval number 116-5-2/2023-000). The study duration was from May 2024 to January 2025. PBMCs were cultivated in RPMI 1640 medium supplemented with 10% fetal bovine serum (FBS), at a concentration of 1 × 106 cells/mL. Part of the isolated cells was additionally stimulated to divide with 3% PHA-M 24 hours before pretreatment. All cultures were pretreated with equal volumes of a series of GRE water stock solutions to obtain final concentrations of 0, 0.25, 0.5, 1, and 2 mg/mL and avoid the effects of dillution. Pretreatment lasted for 24 hours. Following 24-hour treatment, cultures were irradiated with doses of 0.5 and 2 Gy of ɤ radiation, originating from Co-60 at the dose rate of 3.5 Gy/h. According to the bio-dosimetry curves, applied doses are considered as sublethal while provoking measurable DNA damage when applied in in vitro PBMC models. 13 The experimental irradiation of the samples was carried out in the reference field of gamma radiation Co-60 in the Secondary Standard Dosimetry Laboratory (SSDL) of the Institute of Nuclear Sciences “VINČA”. The absorbed dose of gamma radiation (D(Gy)) was determined using an ionization chamber with an electrometer for the reference position on which the experimental samples were placed. Ionization chamber PTW-30013 has been calibrated at the IAEA (International Atomic Energy Agency). After irradiation, the cell cultures were cultivated at a temperature of 37 °C, in a humid atmosphere and with 5% CO2 (CO2 incubator, ESCO, Singapore) for an additional 24 hours.
Examination of Cell Viability by the Strober Procedure
Trypan blue (TB) dye was used to assess cell viability 24 hours after treatment with ionizing radiation according to Strober’s procedure. 14 PBMCs suspensions and 0.4% TB were mixed in a 1:1 ratio, applied to a hemocytometer (Cambridge Instruments Inc., United States of America), and unstained, living cells were counted using a light microscope (Optech, Germany) at a magnification of 400 x. The number of live cells in mL was calculated as the mean value of counting 4 x in 16 squares, multiplied by a volume correction factor of 104 and a correction factor for dilution of the sample with a color of 2.
Analysis of the Prooxidant/Antioxidant Balance
The prooxidant/antioxidant balance (PAB) in lymphocyte lysates was analysed according to the procedure of Alamdari et al.. 15 The PAB test is based on the dual reactivity of the chromogen 3,3′5,5′-tetramethylbenzidine (TMB) in the reaction catalysed by the enzyme peroxidase with the pro-oxidant hydrogen peroxide (H2O2), to form a blue-colored product, and uric acid as an antioxidant to form a colorless product. The staining intensities of the standard solutions are proportional to the ratio of the added amounts of H2O2 and uric acid. The intensity of the colored product, measured at 450 nm wavelength using a microplate reader (Sunrise, Tecan Group Ltd, Männedorf, Switzerland), reflects the relative oxidative/antioxidant state of the sample. The PAB values were calculated from the standard curve of known H2O2/uric acid ratios and expressed relative to the untreated control (set at 100%).
Lipid Peroxidation Product (LPP) Assay
Lipid peroxidation product (LPP) assay was used for the detection of malondialdehyde (MDA) and 4-hydroxyalkenals (HNE) levels in PBMCs culture lysates, obtained by freeze-thaw cycles after applied treatments. 16 Optical density (OD) of the colored product resulting from the reaction of N-methyl-2-phenylindole and LPP in samples was measured at 586 nm on the Sunrise absorbance microplate reader (Tecan). The LPP values in the samples were calculated based on the standard curve of the 1,1,3,3-tetramethoxypropane (TMOP) standard and expressed in relation to the protein concentration in the sample.
Determination of Reduced Glutathione Levels
The levels of reduced glutathione (GSH) in cell lysates were determined by the Ellman method. 17 The assay is based on the reduction of 5,5-dithio-bis-nitrobenzoic acid (DTNB) by GSH, forming the strong yellow product 2-nitro-5-mercaptobenzoic acid. Samples OD were read at 412 nm, on a colorimetric reader, and the concentration of GSH in the samples was calculated based on the equation of the standard curve, in the range 0–1.25 mM. The final concentration of GSH was calculated as nmol/mg of total proteins in the cell lysate and presented as a relative value to the control (100%).
Comet Assay
Alkaline and neutral comet assays were conducted as described by Shahidi et al. 18 Following treatment, cells were submerged in the 1% low-melting point agarose and distributed on the agarose-coated glass. When agarose was set, the slides were immersed in an alkaline lysis solution (2.5 M sodium chloride, 100 mM EDTA, 10 mM Tris base, 10% dimethylsulfoxide, 1% sodium N-lauroyl sarcosinate, and 1% Triton X-100, Merck, Germany, pH 10) for 1 h at 4°C. The unwinding step was done by immersing the slides in a freshly prepared alkaline solution (0.3 M NaOH and 1 mM EDTA, Merck, Germany, pH > 13) in a horizontal gel electrophoresis tank (Bio-Rad Laboratories, CA, USA) for 40 min at 4°C. Electrophoresis was done at 0.75 V/cm for 30 min at 4°C. The slides were then washed three times in neutralization buffer (400 mM Tris buffer, pH 7.5) and rinsed in absolute ethanol for 5 min, then air-dried and stained with DAPI (Sigma-Aldrich Co, MO, USA) in Vectashield solution (Vector Laboratories, GRB). For the neutral comet assay, the lysis step was followed by neutralization in TBE buffer (90 mM Tris, 2 mM EDTA, 90 mM boric acid, Merck, Germany, pH 8.4) at 4°C for 5 min. The electrophoresis was done at 0.75 V/cm for 20 min at 4°C. The slides were rinsed in ethanol for 5 min, air-dried and stained. Analysis was done using an epifluorescent automated Zeiss-Axioimager Z2 microscope (Carl Zeiss, Jena, Germany) and Metafer CometScan software (MetaSystems, Jena, Germany). DNA damage was analysed as a percentage of DNA in the comet tail on at least 150 cells per sample.
Cytokinesis-Block Micronucleus Assay
The Cytokinesis-block micronucleus assay (CBMN) was performed as described by Fenech.
6
Slides were scored using an Axioimager 1 microscope (Carl Zeiss, Jena, Germany), with magnification 400x and 1000x, following the scoring criteria outlined by the International Human Micronucleus (HUMN) Project.
19
Each sample was evaluated for the frequency of micronuclei in at least 1000 binuclear cells (MNi-BN) and cytokinesis-block proliferation index (CBPI) was calculated according to Eq. 1:
MI-MIV represent a number of cells with one to four nuclei, and N is total number of cells.
DNA Fragmentation Colorimetric Assay
The activation of programmed cell death was assessed using a colorimetric technique for the detection of DNA molecular fragments, a hallmark of apoptosis.
20
The cells were lysed by 1 mL of TTE solution (0.2% Triton X-100, 10 mM Tris-HCl, 1 mM EDTA, pH 7.4), and fragmented chromatin was released from nuclei in the supernatant. After centrifugation for 20 min at 20000 g at 4 °C (Eppendorf 5417R, Hamburg, Germany) the supernatant with fragmented DNA was transported to separate tubes, and 1 mL of TTE solution was added to the pellet containing intact chromatin. Then DNA molecules were precipitated and hydrolyzed by 5% TCA for 15 minutes at 90°C. The diphenylamine solution was added to the tubes, mixed vigorously, and left overnight at RT until the color development. Optical density (OD) was read at 600 nm on a colorimetric reader. The proportion of fragmented DNA was calculated according to Eq. 2 as the ratio of fragmented DNA to total DNA:
Gene Expression Analysis by Quantitative Real-Time PCR
Thermal Cycler Setup
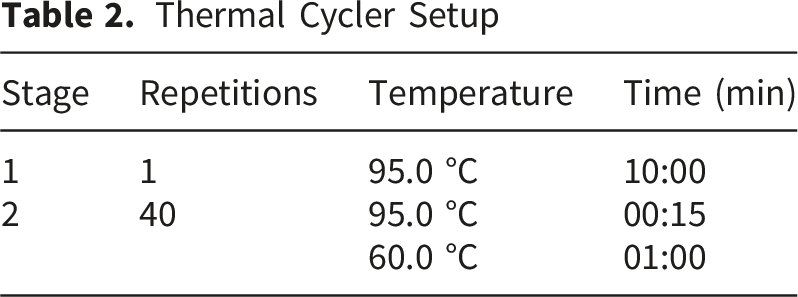
Statistical Analysis
All experiments were repeated three times in duplicate. The results are presented as means ± standard deviation (SD). All statistical analyses were run on SPSS 10 for Windows (IBM, Armonk, NY, USA), using ANOVA test. Statistical analysis of comet assay data was performed using the Mann–Whitney U test for pairwise comparisons between treated and control groups, as the data did not follow a normal distribution. The level of significance was noted as xxx p < 0.001; xx p < 0.01; x p< 0.05, statistical significance for pretreated samples compared to untreated control; ***p< 0.001; **p<0.01; *p< 0.05 statistical significance for pretreated samples compared to irradiated samples (0.5 and 2 Gy treatment).
Results
Ultra-Performance Liquid Chromatography Analysis
The water extract of Gentiana lutea root was analysed by UPLC (Figure 1). UPLC chromatogram of Gentiana lutea root extract: 1-loganic acid, 2-swertiamarin, 3-gentiopicroside, 4-sweroside, and 5-rosmarinic acid
Quantitative Analysis of Gentiana lutea Root Extract (GRE) Water Solution

Cytotoxicity and Genotoxicity Testing
To select the GRE concentrations that could be used for the radiation pretreatment, a cell survival test was performed in PBMCs exposed to GRE for 48 hours at concentrations of 0.25; 0.5; 1 and 2 mg/mL. Since none of the applied treatments led to a significant reduction in the number of viable cells (Figure 2A, control bars), all tested concentrations were used as a 24-hour pretreatment to radiation exposure in further experiments. Compared to the cultures irradiated with 0.5 Gy, pretreatment with 0.25; 0.5 and 1 mg/mL GRE significantly improved cell survival after exposure to radiation with a dose of 0.5 Gy (Figure 2A). Cell cultures pretreated with GRE at concentrations of 0.25 and 0.5 mg/mL and then irradiated with 2 Gy had a better survival rate when compared to the cultures that were not exposed to GRE treatment (Figure 2A). PBMCs viability and genomic damage pretreated with 0.25, 0.5; 1 and 2 mg/mL of Gentiana lutea root extract and exposed to 0.5 and 2 Gy irradiation dose: A-viability, B-DNA fragmentation, C-CBMNs assay representative photomicrographs of micronuclei and nuclear buds (top panel); cells in different stages of apoptosis (lower panel), recorded after 2 Gy irradiation of PHA-activated lymphocytes
Frequency of Micronuclei and Proliferation Index in PHA-PBMCs Pretreated With 0.25, 0.5, 1 and 2 mg/mL of Gentiana lutea Root Extract and Exposed to 0.5 and 2 Gy Irradiation Dose
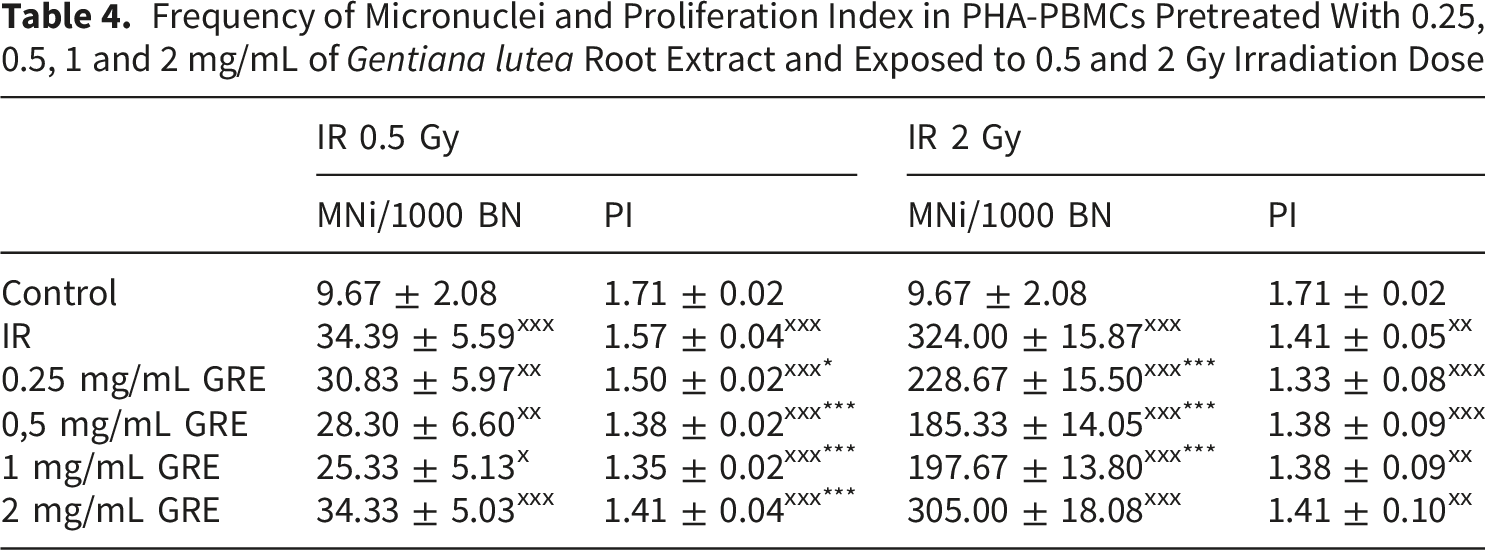
Irradiation of PHA-PBMCs led to a significant increase in MN frequency, nearly 3-fold after 0.5 Gy, and more than 30-fold upon 2 Gy irradiation (Table 4). A significant decrease in the frequency of MN was observed in all PHA-PBMCs that were exposed to pretreatment with GRE and irradiation with 2 Gy (Table 4). Similar trend was noticed for cell cultures pretreated with GRE and irradiated with 0.5 Gy, however, the observed MN frequency decrease was not significant (Table 4.).
PI was significantly lower in all PHA-PBMCs that were exposed to both doses of radiation (Table 4). GRE pretreatment further reduced the PI in 0.5 Gy irradiated cultures. As treatment with selected concentrations of GRE did not affect cell survival, we can assume that after radiation-induced DNA damage, the pretreatment affected the activation of repair mechanisms and either prevented damaged cell division and/or activated cell death of excessively damaged cells. Since lower PI (Table 4) detected in PBMCs that were pretreated with GRE could be linked not just to cell death, seen as apoptotic features of PHA-PBMC (Figure 2C), but also to cell cycle arrest due to activation of DNA repair, expression patterns of genes involved in DNA repair were further surveyed.
Unlike the CBMN assay, the alkaline comet assay showed a significant difference between 0.5 Gy irradiated cells and cells pretreated with GRE. GRE lowered the percentage of DNA damage by approximately 4% in all pretreated PBMCs. Although the mean value of DNA damage does not differ between groups treated with the different concentrations of GRE, the median value of % DNA in the comet tail for cells pretreated with 0.5 mg/mL GRE was the lowest - 5.4 vs. 9.9 in irradiated cells without pretreatment, indicating that cellular response could be dependent on the applied concentration (Figure 3A). The neutral comet assay detected the lowest % of DNA damage upon the same GRE concentration, also reducing both the mean DNA damage from 10% in 2 Gy irradiated cells to 6% in 0.5 mg/mL GRE pretreatment, and the median value from 8.8% to 5.2%. Additionally, cells with significant DNA damage were found after all applied treatments in both alkaline and neutral comet experiments. The excessive DNA damage in these cells could present the apoptotic events which were also noticed in cells by CBMN assay. DNA damage presented as % DNA in comet tail in PBMCs pretreated with 0.25, 0.5, 1 and 2 mg/mL of Gentiana lutea root extract: A-alkaline comet assay in PBMCs exposed to 0.5 Gy; B-neutral comet assay in PBMCs exposed to 2 Gy irradiation dose
Expression of Parp1 was elevated in PBMCs compared to untreated control following 1 mg/mL GRE and 0.5 Gy exposure and all treatments exposed to 2 Gy. The highest elevation was detected in 0.5 mg/mL GRE treatment and 2 Gy dose exposure - 1.7-fold. Significant changes in Ogg1 expression were not detected, while Xrcc1 expression was higher in PBMCs treated with 0.25, 0.5 mg/mL and 1 mg/mL GRE and then irradiated with 0.5 Gy, and in all GRE-pretreated followed by 2 Gy irradiation dose. Pretreatment with 1 mg/mL GRE and 2 Gy exposure led to the highest change, to 1.3-fold (Figure 4). Relative fold change of genes expression involved in DNA repair processes: Ogg1, Parp1, and Xrcc1 in PBMCs pretreated with 0.25, 0.5, 1 and 2 mg/mL of Gentiana lutea root extract and exposed to 0.5 and 2 Gy irradiation dose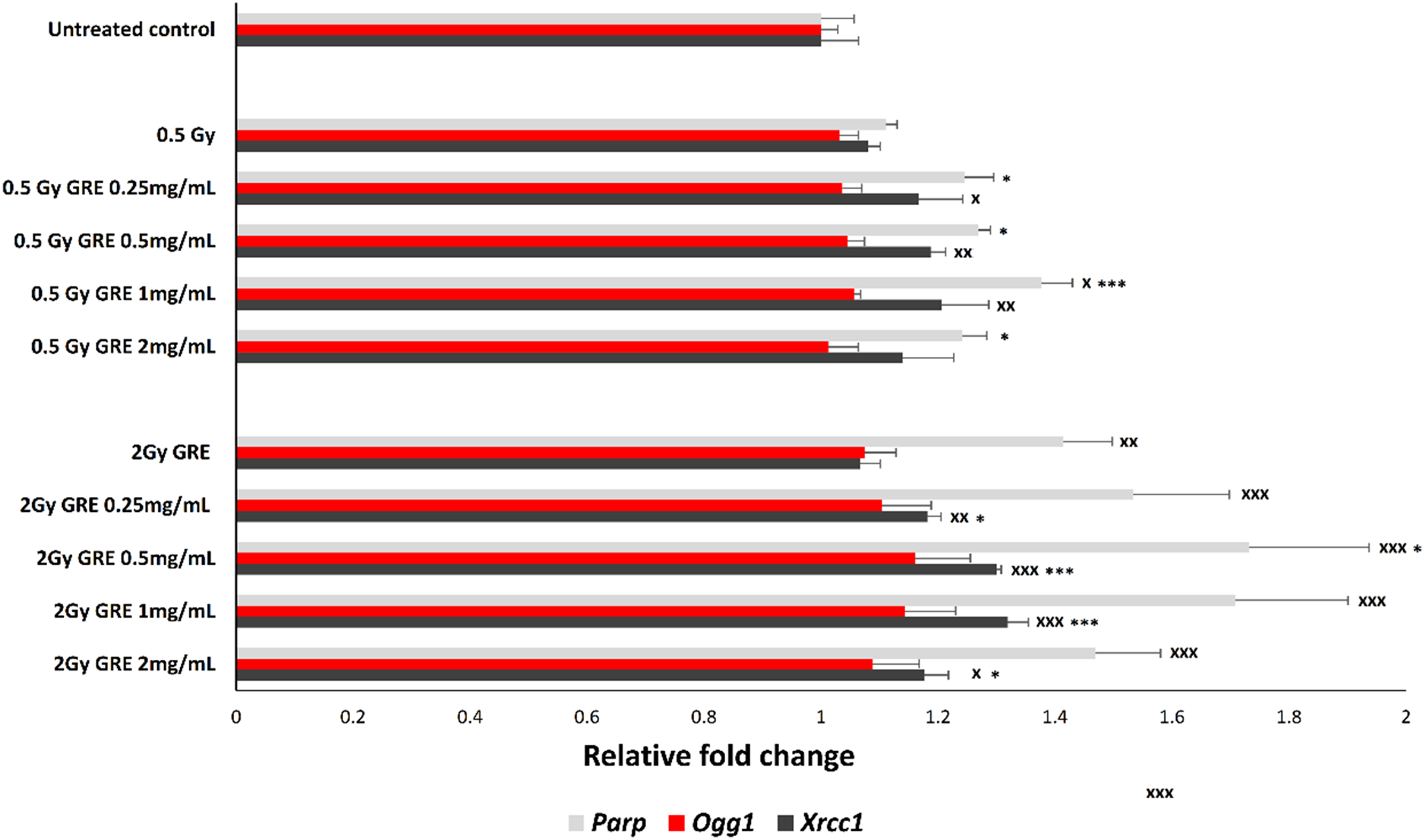
The cellular response to oxidative stress can be monitored by measuring levels of LPPs, the reduced form of GSH, and PAB values to assess redox homeostasis (Figure 5). Irradiation by 0.5 Gy significantly reduced GSH and elevated PAB values, while exposure to 2 Gy led to significant elevation of LPP and PAB levels. In 0.5 Gy irradiated cultures, GRE pretreatment with 0.5 and 1 mg/mL lowered LPP levels, and 0.5 mg/mL elevated GSH, while the PAB values were lower following all pretreatment concentrations. In 2 Gy dose irradiated PBMCs, GRE pretreatment with 0.5 mg/mL also reduced LPP and elevated GSH values (Figure 5A). Generally, GRE pretreatment had a positive effect on oxidative stress parameters that was more pronounced in 0.5 Gy exposed cultures. Oxidative stress parameters in PBMCs pretreated with 0.25, 0.5, 1 and 2 mg/mL of Gentiana lutea root extract and exposed to 0.5 and 2 Gy irradiation dose: A - lipid peroxidation products (LPP); B - reduced glutation (GSH) level; and C - prooxidative/antioxidative balance (PAB)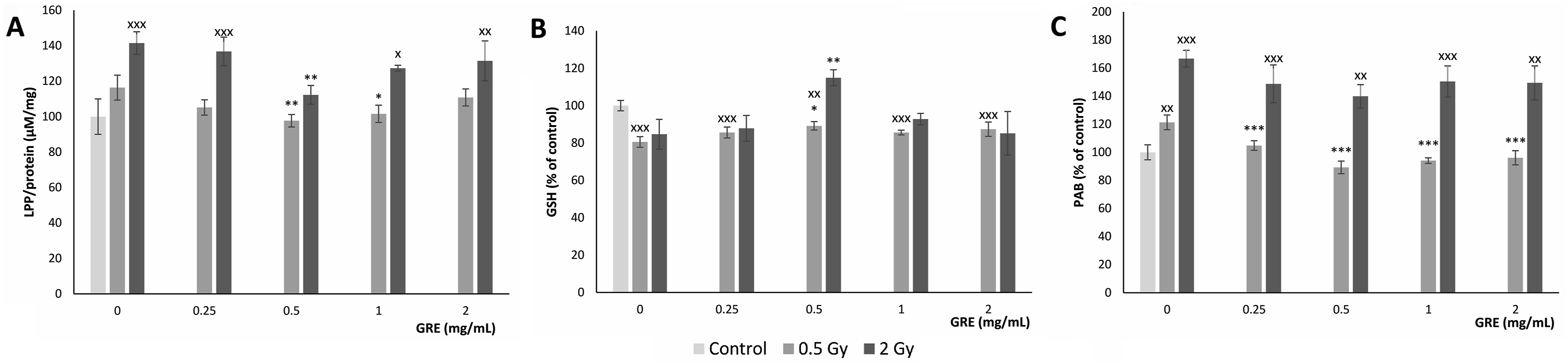
Discussion
GRE chemical composition was characterized by high yields of secoiridoids, mainly gentiopicroside and sweroside, and iridoid loganic acid. The roots of Gentiana lutea are known for their high content of iridoids and secoiridoids and their glucosides. 12 All detected chemicals are acknowledged to manifest antioxidative bioactivity. Application of gentiopicroside was shown to reduce oxidative stress through activation of Nrf2. 21 Nrf2 was also reported as a key target for other GRE constituents: sweroside, 22 swertiamarin, 23 and loganic acid. 24 Initiation of antioxidant defence by Nrf2 activation was commonly reported as the role of radioprotectors.25-27 The radioprotective potential of rosmarinic acid was detected by Xu et al (2016) after in vivo exposure of mice to both radiation and rosmarinic acid, presented as a better 30-day survival rate and higher white blood cell counts, haemoglobin, and platelets than the radiation-only group. 28 Our previous research showed that plant extract, due to its complex chemical composition, could have a more beneficial effect on cells than a single compound present in the same concentration as in the extract. 29 Presumably, since all detected components could act in a radioprotective manner, GRE may be highly beneficial in radiation-induced injury. The radioprotective properties of orally consumed water-ethanol extract of Gentiana lutea root, analysed by Menković et al (2010), showed that the extract has the potential to reduce the cytotoxic effect of X-radiation on PBMCs of healthy people. 30 The ether and methanol fractions of G. austriaca extract showed a radioprotective effect reflected as the reduction of the MN frequency in human lymphocytes exposed to ɤ-rays. 31 Since the variety of extracted compounds from plant material and their yield depend on the solvent used for extraction, 32 we aimed to analyse the radioprotective properties of water extract that is commonly used as a tea infusion. Radioprotective properties were evaluated through cellular survival, e.g., cytotoxicity analysis, genotoxicity, cell proliferation, and detection of fragmented DNA, which represent parameters influenced by the direct effect of ionizing radiation. Oxidative status was analysed by multiple methods in order to evaluate IR-induced damage by its indirect effect. Our results showed that GRE pretreatment with lower concentrations increased cell viability and lowered DNA fragmentation induced by irradiation. Such a cytoprotective effect can be achieved by activation of DNA repair mechanisms and/or antioxidant defence. 33 Activation of homologous recombination (HR) involved in DSB DNA repair processes in molecules in PHA-PBMCs after GRE treatment has been documented in our earlier research. 9 The GRE’s genoprotective influence may be due to the presence of gentiopicroside and swertiamarin, since in our previous research activation of BER damage repair mechanisms was detected. 34 The gene expression analysis of selected roll-players in the BER mechanism, PARP, OGG1, and XRCC1, revealed the elevated expression of Parp and moderate activation of Xrcc1 expression, induced by pretreatment and more pronounced after the application of a higher radiation dose. Analysis of the transcription profile of BER genes involved in radioadaptive response also showed elevation of Parp and Xrcc1 transcripts as in our GRE-pretreated cultures. 35 Expression profile of genes analysed in the present study showed a reasonably high variability in individual responses, which could be expected since the level of activation of genetic damage repair, induced by IR, could be influenced by the genetic polymorphisms in DNA repair genes. 36 Also, a response to acute intense IR injury could affect the level of cell death due to the interindividual levels of repair genes. 37 Along with its role in BER, PARP-1 can take part in the repair of DNA double-strand breaks (DSBs) via the HR and NHEJ pathways. 38 Although a parallel rise of gene expression involved in a specific repair mechanism could be expected, much higher activation of Parp may be a result of its multiple roles in damaged cells. PARP-1 activation after oxidative insult also reduces cell viability. 39 DNA damage results in parthanatos, a type of cell death mediated by hyperactivation of PARP1, which also activates apoptosis-inducing factor (AIF) nucleus translocation, which results in DNA fragmentation that was also detected in our experimental setting. 40 Activation of DNA repair results in the stalling of the cell cycle, 41 so we can reason that the detected reduction of PI is partly due to the arrest of the cell cycle. Cells with apoptosis features were also observed in the analysis of MN preparations (Figure 2C), indicating that part of the cells were removed by apoptosis, which can also affect the PI value. Nevertheless, based on the obtained results of the CBMN test, we can conclude that GRE has genoprotective potential, which is more clearly manifested after exposure to higher doses of radiation.
Comet assay detects DNA damage on the cellular level and it is known for its sensitivity since very low level of damage can be detected in both alkaline and neutral conditions. 42 The principal difference between these two assay variants lies in the type of DNA lesions detected: the alkaline comet assay primarily detects single-strand breaks (SSBs) and alkali-labile sites, including unwound double-strand breaks (DSBs), whereas the neutral comet assay predominantly detects DSBs, which are typically induced by higher doses of ionizing radiation. 18 In our study, DNA damage detected by CBMN assay after 2 Gy dose was ten times higher than in 0.5 Gy irradiated cultures, which guided the use of alkaline conditions for assessing DNA damage at 0.5 Gy and neutral conditions for 2 Gy-irradiated cells. After irradiation with 0.5 Gy, the alkaline comet assay revealed a consistent reduction of primary DNA damage in GRE pretreated cultures at all concentrations, without pronounced dose dependence, indicating a GREs protective and/or effect on the activation of DNA repair mechanisms. Protection against oxidative DNA lesions by extract of Gentiana asclepiadea, plant with comparable phytochemical profile to GRE, 43 was previously reported by Hudecová et al. 44 In our study, the simultaneous upregulation of Parp1 and Xrcc1 supports the activation of base excision repair (BER) pathways, indicating that direct radioprotection and enhanced DNA repair likely act in parallel. Efficient repair of SSBs may prevent the accumulation of complex DNA lesions, thereby reducing the formation of DSBs and micronuclei. After irradiation with 2 Gy, the neutral comet assay demonstrated a more pronounced reduction in DNA double-strand breaks, consistent with the decreased micronucleus frequency observed in the CBMN assay, particularly in cultures pretreated with 0.5 mg/mL GRE. Higher radiation dose induces severe DNA damage which consequently leads to higher activation of repair, as it was detected in our study by more pronounced expression of Parp1 and Xrcc1 upon 2 Gy irradiation. Activation of the primary DNA damage repair moderates cellular damage, but does not reverse it to the non-irradiated control level. In proliferating cells, the persistence of unrepaired SSBs may result in replication fork collapse and subsequent DSB formation, 45 triggering homologous recombination repair, 9 and contributing to micronucleus formation, as observed in this study.
It has been shown that polyphenols detected in GRE can activate different signaling pathways in the cell that influence the activation of DNA damage repair. 46 Other natural radioprotectors studied in vitro, such as curcumin and resveratrol, following 2 Gy radiation exposure, displayed a similar level of genoprotection by reducing chromosomal aberrations by roundly 50%. 47 The increase of free radicals in cells, as the indirect effect of IR, can cause significant genomic damage. 48 Increased genomic damage could initiate cell apoptosis, which is characterized by excessive DNA fragmentation, as was reported in haematopoietic cells exposed to IR doses in the range of 1 to 4 Gy. 49 Lipid peroxidation products (LPPs), such as malondialdehyde (MDA) and 4-hydroxynonenal (4-HNE), are widely used as indicators of oxidative damage to cells, since intense lipid peroxidation can lead to cell membrane and/or organelle damage and activate various types of cell death. 37 Phytochemicals could potentially modulate radiation-induced damage by their antioxidant activity. 50 The main mechanism of action of most radioprotectors is to maintain the intracellular level of GSH, a molecule involved in the direct inactivation of ROS. Glutathione redox status (GSH/GSSG) decreases after irradiation, mainly due to an increase in glutathione disulphide (GSSG) levels. 51 GSH is also necessary to activate the proliferation and repair of damaged tissues, thus preventing the activation of cell death mechanisms. 52 Pretreatment with 0.5 mg/mL GRE showed a promising antioxidant effect after a 0.5 Gy irradiation dose since the applied GRE concentration raises GSH levels and lowers LPP and PAB values. PAB and LPP levels were also lower after 0.5 mg/mL and 2 Gy dose treatment. However, the protective capacity of GRE was less potent at higher radiation doses (2 Gy), as evidenced by incomplete reversal of oxidative imbalance and persistent elevation of prooxidative/antioxidative balance (PAB) values, probably due to the limited capacity of GRE to prevent excessive oxidative damage. The obtained results indicate that exposure to the plant extract enabled cells to cope better with oxidative stress. The rosmarinic acid, detected as a component of GRE, displays antioxidant potential as a scavenger of reactive species of molecules, which represents the direct mechanism by which GRE acts in the antioxidant protection of cells. 53 In addition to the direct removal of ROS, literature data indicate that treatments with GRE affect the activation of the gene expression of the transcription factor Nrf2, which leads to increased synthesis of enzymes involved in the response to oxidative stress. 54 The bioactive compounds, such as gentiopicroside, sweroside, swertiamarin, and loganic acid, detected in GRE, likely contribute to these protective effects via known antioxidant and Nrf2-activating mechanisms.21-25 Radiation exposure affects an organism’s ability to survive by causing a number of pathophysiological conditions, such as oxidative damage, inflammation, and fibrosis. It was observed that several plant extracts may scavenge radiation-induced free radicals, inhibit radiation-induced lipid peroxidation, reduce the level of inflammatory cytokines, and prevent fibrosis. This is especially relevant since, in addition to its potential application in preconditioning regimens for radiodiagnosis and radiotherapy, occupational workers may also benefit from its use. 55 The change in blood oxidative stress biomarkers like MDA, total antioxidant capacity (TAC), GSH, and others was detected in medical radiation workers following exposure to low-dose ionizing radiation. 56 Since Gentiana lutea root extract lowered LPP (MDA, 4-HNE) and PAB levels following 0.5 Gy dose exposure, data obtained in our study indicate its radioprotective validity.
While promising positive effect of GRE pretreatment on radiation induced injury was detected in vitro, as it was previously discussed,36,37 the level of DNA damage repair activation could be influenced by the genetic polymorphisms in DNA repair genes leading to high variability in individual responses. Additionally, because of differences in concentration-time profiles, absorption efficiency, metabolic processes, and other factors that may impact the bioavailability of active compounds present in GRE, the effects of GRE infusion in vivo may differ significantly from those seen in vitro. 57 Since the levels of secondary metabolites in GRE water infusions depend on the solid-to-solvent ratio, extraction period, and extraction temperature, determining the quantity with the best biological effect in non-laboratory conditions may be difficult. 58 Consequently, further studies are required to elucidate the mechanisms of action, determine optimal compound ratios for maximal efficacy, and achieve formulation standardization.
Conclusion
The protective effects of Gentiana lutea root extract (GRE) pretreatment were evidenced by reduced DNA fragmentation, decreased DNA damage measured by the comet assay, and a lower frequency of micronuclei in γ-irradiated GRE-pretreated PBMCs. At the molecular level, the observed upregulation of Parp1 and Xrcc1 suggests the involvement of base excision repair (BER) pathways in the moderation of radiation-induced DNA damage. In parallel, GRE modulated oxidative stress parameters, most notably following 0.5 Gy irradiation, indicating an antioxidant effect manifested through the maintenance of redox balance and activation of antioxidant responses. Taken together, these findings reflect a cytoprotective effect of GRE under ionizing radiation exposure, which supports its radioprotective potential. Nevertheless, in vitro experimental approach and the use of GRE infusion, whose composition may vary, may pose limitations, so further studies should elucidate its molecular mechanisms of action, optimise dosage, and assess its long-term safety and efficacy in vivo.
Footnotes
Ethical Considerations
The study conducted in accordance with the Declaration of Helsinki, and was approved by the Ethics Committee of the “Vinča” Institute of Nuclear Sciences – National Institute of the Republic of Serbia (Ethical approval number 116-5-2/2023-000). All participants were detaly informed about sampling procedure and goals of our research before giving written informed consent to participate.
Author Contributions
All authors contributed to the study creation and realisation. All authors approved the final manuscript. Mila Kostić: Formal analysis; Methodology; Writing - original draft; Validation. Jelena Filipović Tričković: Investigation; Methodology; Writing - review & editing. Ksenija Radošević: Formal analysis; Methodology; Validation. Tamara Lazarević Pašti: Formal analysis; Methodology; Validation. Srboljub Stanković: Formal analysis; Methodology; Validation. Ivana Kolić: Methodology; Resources; Formal analysis. Aleksandra Stanković: Methodology; Resources; Formal analysis. Ana Valenta Šobot: Investigation; Project administration; Supervision; Writing - original draft.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia, Grant no. (451-03-33/2026-03/200017; 451-03-3627/2025-03/3456) and Science fund of the Republic of Serbia (Proof of concept, Grant no. 14874), #fondzanauku #GentiGoldCell #dokazkoncepta.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be available from the corresponding author on reasonable request.
