Abstract
Traumatic brain injury (TBI) is an important condition with high rates of disability and mortality worldwide. Post-traumatic seizure (PTS) frequently occur following TBI, manifesting in both early and late stages. Recurrent PTS without timely intervention may progress to post-traumatic epilepsy (PTE), which defined as the occurrence of two or more unprovoked seizures. Early pharmacological intervention is essential to mitigate the risk of PTE and enhance the prognosis for patients with TBI. Antiepileptic drugs (AEDs) offer a viable strategy for managing PTS. Recent studies indicated that AEDs are more effective in early post-traumatic seizure compared to late post-traumatic seizure, and their efficacy and safety require further evaluation. As research advances in the pathophysiological changes after TBI and the pathogenesis of PTS, current investigations are increasingly focused on neurological damage. Novel compounds targeting various pathways, including antioxidants, anti-neuroinflammatory agents, glutamate modulators and anti-oxidative stress compounds, have demonstrated promising potential in preclinical studies for PTS intervention. This review focuses on the research progress of different AEDs in PTS intervention and discusses the recent developments of emerging PTS intervention strategies based on multiple pathways, providing insights into the clinical application of AEDs and new directions for the development of new drugs for PTS intervention.

Keywords
Introduction
Traumatic brain injury (TBI) is typically defined as damage and disruption to brain structure and function caused by external forces. TBI is now widely recognized as the predominant cause of trauma-related death and disability.1-3 It is reported that approximately 69 million individuals worldwide experience TBI each year. In China, an estimated 13.9 million individuals sustain such injuries annually, representing approximately 20% of the global incidence, which contributes to adverse clinical outcomes and imposes a significant economic burden. 4 After suffering a brain injury, brain tissue may undergo pathophysiological alterations, such as increased neural excitability, neuroinflammation, and oxidative stress. These changes are influenced by factors such as the injury type, trauma severity and age, which may induce the occurrence of post-traumatic seizures (PTSs).5,6
PTS, a prevalent and severe clinical manifestation of TBI, is characterized by hypersynchronous cortical excitability. 7 Based on the latency period, PTS can be categorized into immediate post-traumatic seizures (IPTSs), early post-traumatic seizures (EPTSs) and late post-traumatic seizures (LPTSs). 8 IPTSs occur within 24 hours after TBI, indicating severe cranial brain injury and necessitating immediate evaluation and intervention to mitigate neurological damage and preserve the patient’s life. EPTSs are typically defined as seizures occurring within 7 days after TBI. In certain patients, active pharmacological treatment can facilitate neural tissue repair, leading to a favorable prognosis. LPTSs are seizures that manifest more than 7 days after TBI, which are characterized by recurrence and persistence. LPTSs often necessitating prolonged pharmacological management or surgical intervention when necessary.
The Definition of Terms in the Manuscript

Current studies have identified a correlation between EPTS and LPTS. Some researchers have posited that EPTS may serve as a risk factor that elevates the likelihood of developing LPTS.17,18 Studies indicate that approximately 25% of individuals experiencing EPTS may subsequently develop LPTS. 19 Therefore, timely interventions in EPTS following TBI are of critically important. Given that PTS may represent a continuous and prolonged process, pharmacologic intervention and management are vital to reducing long-term mortality risk and enhancing the prognosis of TBI. Currently, clinical intervention for PTS primarily involves prophylactic treatment with antiepileptic drugs (AEDs) administered within 7 days after TBI. However, the therapeutic efficacy is controversial and limited for LPTSs.14,20 Furthermore, the absence of a standardized medication regimen for PTS intervention underscores the inadequacy of current treatment strategies in meeting clinical demands. In the past decade, numerous researchers have endeavored to develop innovative strategies for PTS prevention. Recently developed compounds, such as free radical scavengers and antioxidants, have demonstrated potential efficacy in PTS. Although many of these studies remain in the preclinical stage, they hold promise for future clinical application.
In this review, we have selected systematic reviews, randomized controlled trials (RCTs), cohort studies, high-impact clinical studies, as well as preclinical and basic studies for a retrospective review, with a focus on their study designs, sample sizes, findings, and conclusions. We summarized the research progress on the pharmacological intervention of PTS, and further introduced and compared the effects of different AEDs applied in PTS intervention. Subsequently, we reviewed the clinical intervention for PTS and explored the latest advancements in novel strategies. Our primary objective is to present novel perspectives on pharmacological interventions for PTS and improve the disease prognosis.
Current Pharmacological Intervention Strategies for PTS
The Effects of Different AEDs for PTS Interventions

Abbreviations: AEDs, antiepileptic drugs; PHT: phenytoin; LEV, levetiracetam; VPA: valproate; TBI: traumatic brain injury; PTS: Post-traumatic seizures; EPTS: early post-traumatic seizures; LPTS: late post-traumatic seizures. GOS-E: glasgow outcome scale-extended; EEG: electroencephalogram.
Pharmacologic Characteristics of Common AEDs for PTS Interventions
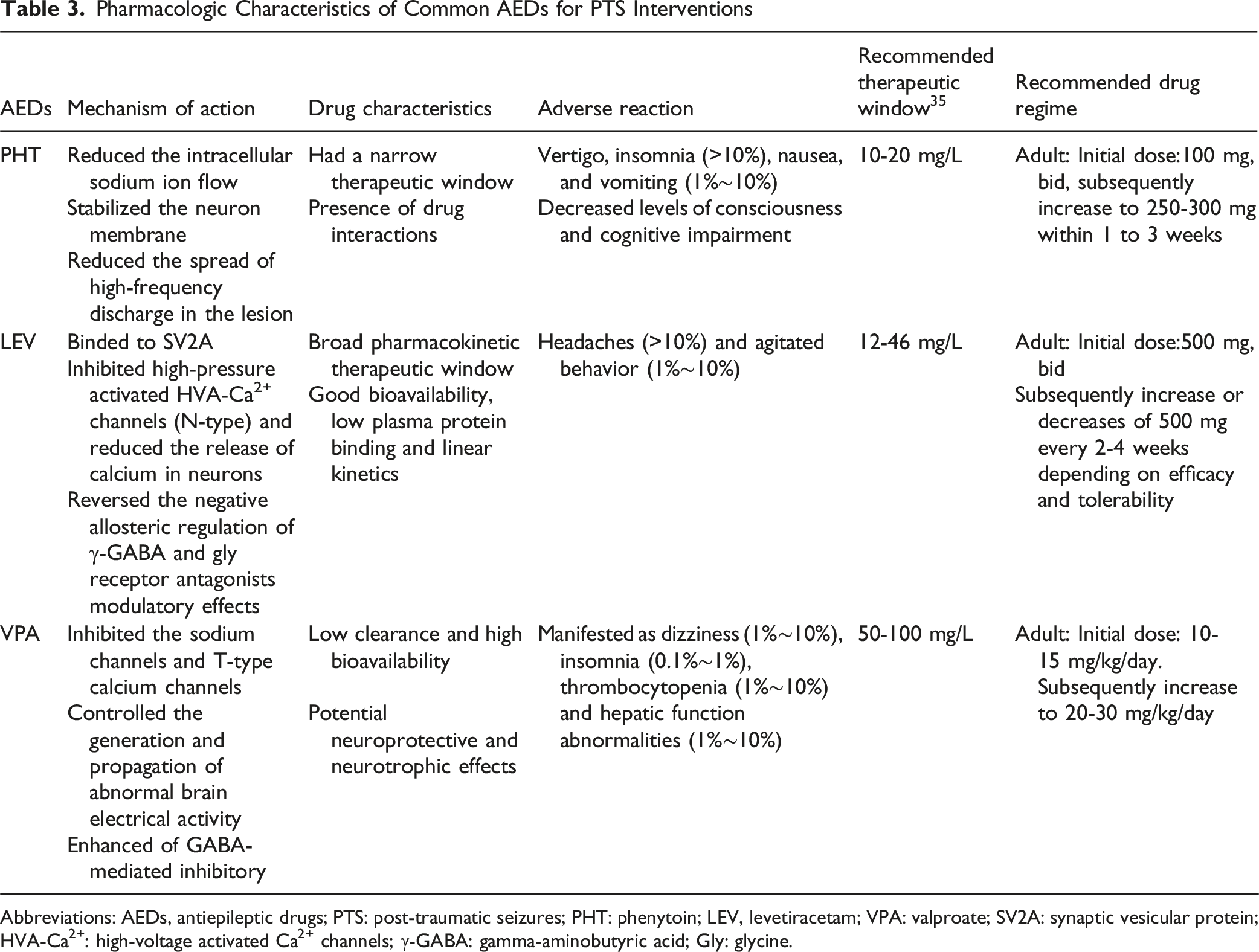
Abbreviations: AEDs, antiepileptic drugs; PTS: post-traumatic seizures; PHT: phenytoin; LEV, levetiracetam; VPA: valproate; SV2A: synaptic vesicular protein; HVA-Ca2+: high-voltage activated Ca2+ channels; γ-GABA: gamma-aminobutyric acid; Gly: glycine.
Phenytoin
PHT is one of the most extensively studied and widely used AEDs. PHT exerts antiepileptic effects by reducing intracellular sodium ion flow, stabilizing the neuronal membrane, and reducing the spread of high-frequency discharges in lesions. Previous clinical studies and meta-analysis have shown that PHT was effective in suppressing seizures during the first week after TBI compared with placebo.26-29 However, the effect of PHT is mainly in reducing the risk of EPTS after TBI, but not in reducing the risk or mortality of LPTS.23,30 Therefore, the fourth edition of the National Brain Injury Foundation (BTF) Guidelines recommended the interventional treatment of PHT within 7 days after TBI, but did not provide definitive recommendations for the development of LPTS beyond the 7-day treatment period. 31 Several specialty guidelines also recommended PHT as the preferred choice for EPTS intervention.32,33
With the emergence of a new generation of AEDs such as LEV, the limitations of PHT have become increasingly apparent, thereby restricting its clinical application. Notably, PHT is characterized by hepatic enzyme induction and a narrow therapeutic window. The former results in frequent drug interactions, as PHT accelerates the metabolism of numerous medications, thereby complicating its concurrent use with other drugs. The latter characteristic, involving nonlinear pharmacokinetics and a narrow therapeutic window of 10 to 20 μg/mL, necessitates therapeutic drug monitoring (TDM).34,35 This monitoring is crucial to ensure therapeutic efficacy and to mitigate adverse effects, requiring dosage adjustments tailored to the patient’s condition. However, the limited range for concentration adjustment poses challenges in meeting individualized clinical needs. Furthermore, the adverse reactions associated with PHT should not be overlooked. Compared to placebo, PHT is linked to a higher incidence of adverse effects, including vertigo, insomnia, nausea, and vomiting, with the most pronounced being impairments in consciousness and cognitive function.
Levetiracetam
LEV represents a newer generation AED. Studies have demonstrated that LEV exerts its antiepileptic effect through three primary mechanisms: (i) LEV binds to synaptic vesicular protein (SV2A), thereby playing a neuroregulatory role. 36 (ii) LEV inhibits high-voltage activated Ca2+ channels (HVA-Ca2+), leading to a reduction in calcium release within neurons; and (iii) LEV reverses the negative allosteric modulation of gamma-aminobutyric acid (γ-GABA) and glycine (Gly) receptor antagonists. 37
Numerous studies, including a network meta-analysis, have demonstrated that LEV and PHT exhibit comparable efficacy in EPTS intervention, yet neither shows an effect on the risk of LPTS.38-40 In particular, LEV has been demonstrated to not only not only reduce the duration of intensive care unit stays for TBI patients 23 and alleviate balance disorders, 41 but also enhance the safety of long-term medication, thereby contributing to the improvement of long-term prognosis in TBI patients. 42 Therefore, it has been proposed that LEV serves as an alternative intervention strategy to PHT for PTS. 43 However, the long-term safety of this medication requires further validation. 44 Several preclinical studies have demonstrated that.
LEV can exert beneficial effects on structural, molecular, and behavioral aspects of the nervous system after TBI by modulating neuroinflammatory pathways in TBI rat models. Nonetheless, the precise mechanisms underlying its neuroprotective effects and their clinical validation necessitate further elucidation.47,48
LEV was considered an attractive AED for PTS intervention due to its pharmacokinetic properties, including high bioavailability, a relatively short half-life, and a predictable and broad pharmacokinetic therapeutic window.37,49,50 Unlike other AEDs, the most important characteristic of LEV are its wide therapeutic window and minimal reliance on hepatic metabolism. This means that LEV has a wider range of dosage adjustment, and the blood concentration of LEV is more stable when combined with other antiepileptic drugs. These characteristics render LEV particularly suitable for individualized treatment, especially in complex cases involving polypharmacy.51,52 Additionally, LEV has almost no drug interactions, which is critical for medication management in patients with TBI. Common adverse effects of LEV include headache and behavioral agitation, with incidence rates exceeding 10% and ranging from 1%–10%, respectively. 51 Compared to other second-generation AEDs such as lamotrigine and oxcarbazepine, LEV has a lower rate of treatment discontinuation due to adverse effects.53,54 Furthermore, LEV has demonstrated superior safety profiles in pregnant women and children, providing distinct advantages in clinical applications for these specific populations.55,56
Valproic Acid
VPA is a broad-spectrum AED that was first introduced into clinical practice in 1967. VPA involves the inhibition of sodium channels and T-type calcium channels, which controls the generation and propagation of abnormal brain electrical activity, as well as the enhancement of γ-GABA-mediated inhibitory effects to control seizures. 57 Clinical trials have demonstrated that VPA exhibits a broad spectrum of efficacy against various types of seizures when compared to other AEDs.58,59 For PTS intervention, Eastman et al. experimental results showed that VPA (480 mg/kg/day for 7 days) reduced the frequency and duration of seizures in patients with PTSs. 60 In addition, only a few clinical trials have shown that VPA may be effective in reducing the risk of EPTS in patients with severe TBI, although high-quality clinical studies are required to substantiate these findings.45,46 Relevant preclinical studies have shown that VPA can inhibit apoptosis, 61 lipid peroxidation 62 and inflammation 63 after central nervous system injury through variety molecular mechanisms, thereby exhibiting neuroprotective properties. 64 At the epigenetic level, VPA has been found to exert potentially neurotrophic effects by modulating brain neurogenic trophic factor (BDNF), a neurotrophic factor found to be associated with the development of focal epilepsy.65-67 These studies suggested that VPA favors the prognosis and recovery of TBI patients. 57
The adverse effects associated with VPA typically manifest early in treatment and include symptoms such as dizziness, insomnia, thrombocytopenia, and hepatic function abnormalities, with a relatively lower incidence of cognitive-related adverse effects. 66 These adverse effects can be mitigated by adjusting the dosage or discontinuing the medication, without compromising the overall treatment efficacy. 68 VPA is characterized by low clearance and high bioavailability, ranging from 96% to 100%. Given the substantial interindividual variability in blood concentrations of VPA, it is essential to monitor blood levels and adjust dosages accordingly in clinical practice to ensure safe and effective individualized treatment. 69 Importantly, the long-term efficacy and safety of VPA require further investigation, particularly in patients with pre-existing liver disease or cognitive decline, as these conditions may elevate the risk of adverse effects.
Other Antiepileptic Drugs
In addition to the extensively studied AEDs previously mentioned, other AEDs such as CBZ, oxcarbazepine and PB have also been examined in a limited number of studies. In a zebrafish model, CBZ has been shown to be effective in intervening in PTS, specifically by reducing tonic-clonic seizures; however, it was ineffective against irregular movement or myoclonic seizures and did not prevent the progression to PTE. 70 PB has similarly shown potential for PTS intervention, and its combination with PHT has been associated with a reduced incidence of epilepsy following severe craniocerebral injuries, with reported rates between 2% and 5.6%. 71 Despite these findings, the limited evidence-based research and significant side effects associated with both drugs preclude definitive conclusions regarding their efficacy in PTS intervention. 22 Furthermore, a case study involving pharmacological intervention with fosphenytoin and phenobarbital in PTS patients yielded unsatisfactory outcomes, with seizure relief ultimately achieved only through surgical means. This underscores the need for further investigation into the effectiveness of phenobarbital in PTS management. 72 Gabapentin, a GABA analog, is primarily advantageous due to its ability to traverse the blood-brain barrier without acting as an inhibitor or inducer of heparanase, and it exhibits relatively few drug interactions. 73 Gabapentin was found to reduce epileptiform discharges and have neuroprotective effects in an undercut model of PTS, suggesting that period has a greater potential for PTS prevention. 74 There are also clinical studies showing that gabapentin significantly improves Glasgow Coma Scale (GCS) scores and mortality in comatose patients with TBI. 75 With the development of new AEDs, including topiramate, lamotrigine, and gabapentin, promising antiepileptic effects have been realized. However, the role of these AEDs in mitigating secondary brain injury and PTS intervention remains underreported and is still in the early stages of investigation.
Emerging Strategies for Intervention of PTS
Analogous to other forms of acquired epilepsy, PTS are marked by a latency period between TBI and the onset of seizures. Recently, numerous clinical and basic studies have been devoted to exploring the biological processes that might be involved in the progression from TBI to epilepsy. As the pathophysiological mechanisms underlying TBI and PTS are progressively elucidated, the physiological changes induced by tissue damage following TBI present a critical temporal window for pharmacological intervention, thereby offering a potential strategy for the intervention and management of PTS.
PTS Intervention Strategies Based on Emerging Compounds
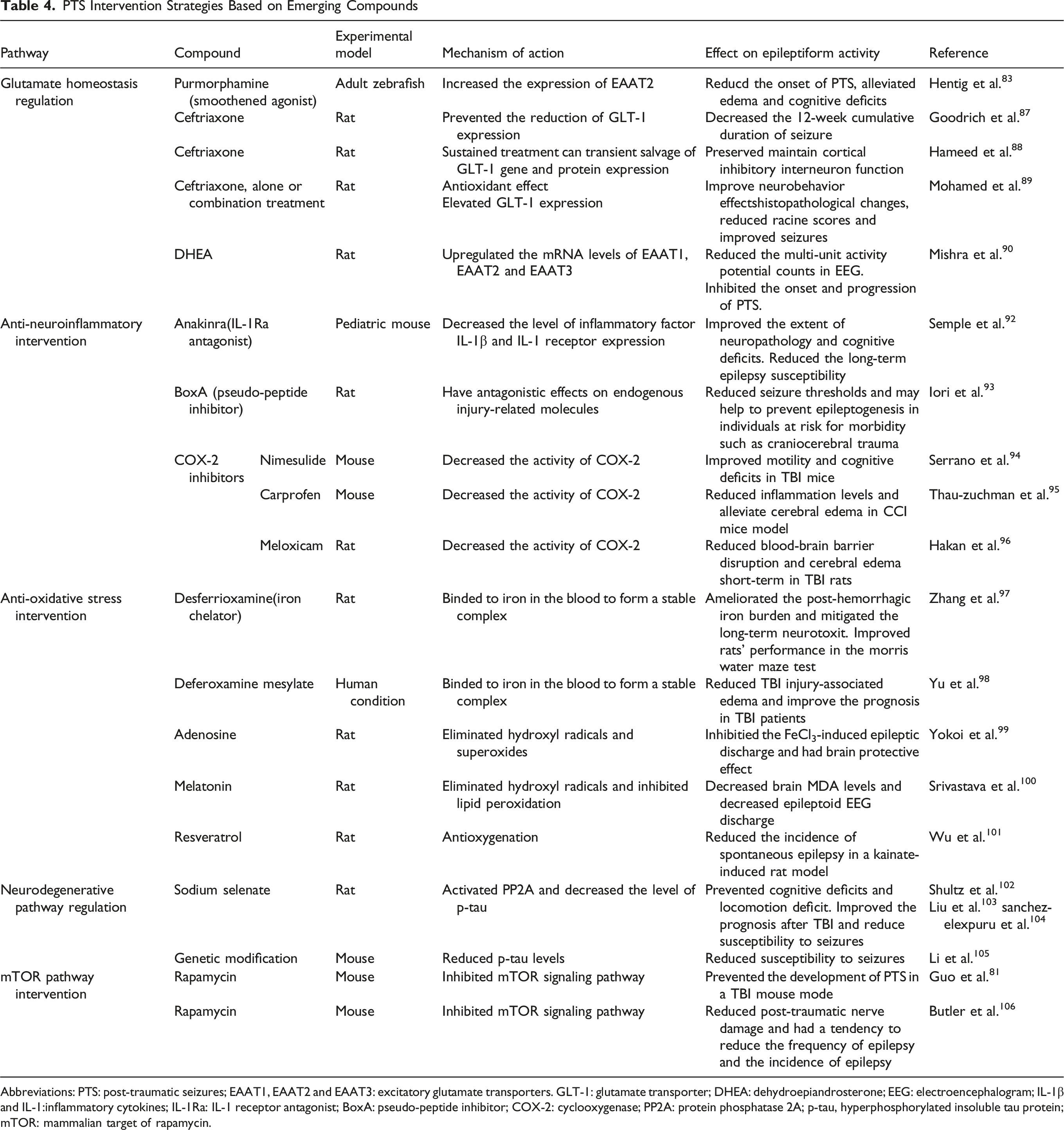
Abbreviations: PTS: post-traumatic seizures; EAAT1, EAAT2 and EAAT3: excitatory glutamate transporters. GLT-1: glutamate transporter; DHEA: dehydroepiandrosterone; EEG: electroencephalogram; IL-1β and IL-1:inflammatory cytokines; IL-1Ra: IL-1 receptor antagonist; BoxA: pseudo-peptide inhibitor; COX-2: cyclooxygenase; PP2A: protein phosphatase 2A; p-tau, hyperphosphorylated insoluble tau protein; mTOR: mammalian target of rapamycin.

Emerging Strategies for PTS Intervention Based on Different Pathways. Compounds Such as Antioxidants/free Radical Scavengers, Glutamate Modulators, Neuroprotective Agents, and Anti-inflammatory Compounds Have Been Explored as Potential Preclinical Therapeutic Candidates. Abbreviations: TBI, Traumatic Brain Injury; PTS, Post-traumatic Seizures; EAAT1, EAAT2 and EAAT3: Excitatory Amino Acid Transporters; GLT-1: Glutamate Transporter; HMGB1, High Mobility Group Box 1; BoxA: Pseudo-peptide Inhibitor; COX-2: Cyclooxygenase; ROS, Reactive Oxygen Species; PP2A: Protein Phosphatase 2A; P-tau, Hyperphosphorylated Insoluble Tau Protein; mTOR:mammalian Target of Rapamycin
PTS Intervention Based on Glutamate Homeostasis Regulation
Glutamate, the most abundant excitatory neurotransmitter in the central nervous system, is associated with a variety of neurological disorders, including Parkinson’s disease, cognitive impairments, and epileptic seizures. 80 The post-traumatic brain results in the abnormal accumulation of extracellular glutamate in surrounding tissues, which induces secondary excitotoxic effects mediated through glutamate receptors. This mechanism is considered one of the main factors for the onset and progression of PTS after TBI. 81 Molecular mechanism studies have indicated that reducing glutamate levels after TBI can prevent the development of PTS. 82 For example, Hentig et al found that the smooth receptor agonist purmorphamine in a zebrafish animal model can increase the expression of excitatory amino acid transporters (EAAT2). And the EAAT2 receptors can scavenge glutamate by reuptake, thereby reducing glutamatergic neuro-excitotoxicity in the brain after suffering TBI and decreasing the onset of PTS and cognitive impairment. 83
Ceftriaxone is a β-lactam antibiotic with cerebrospinal fluid-brain barrier permeability. Multiple studies have demonstrated that ceftriaxone exhibits neuroprotective effects in various rodent models.84-86 In a mouse model of amyotrophic lateral sclerosis (ALS), ceftriaxone can mitigate glutamate-induced excitotoxicity, delay neuronal loss, and improve mouse survival rates. 86 In rodent models of hypoxic-ischemic brain injury, ceftriaxone also effectively reduces neuronal death. 85 Recent studies have found that ceftriaxone also shows promising effects in TBI models. Goodrich et al found that administration of ceftriaxone postoperatively in TBI rats could prevent the reduction of glutamate transporter protein-1 (GLT-1, an EAAT2 homologs in mice and rats) expression and the 12-week cumulative duration of seizure after TBI in electroencephalogram (EEG) recordings. They demonstrated that ceftriaxone exhibits neuroprotective and antiepileptic properties, implying that ceftriaxone may be a safe and promising prophylactic treatment after TBI. 87 In addition, other studies have indicated that sustained treatment with ceftriaxone can preserve and maintain the function of inhibitory interneurons in the cortex of rats with TBI by transiently rescuing GLT-1 gene and protein expression in rat TBI model, thereby alleviating the gradual loss of inhibitory interneurons in the subacute and chronic post-traumatic periods and providing lasting neuroprotection.88,89 While additional evidence is required to substantiate these findings, the underlying mechanism of ceftriaxone indicates significant potential for intervention in PTS. Dehydroepiandrosterone (DHEA) is a natural steroid hormone. In the FeCl3-induced PTS rat model, DHEA was found to have the ability to upregulate the mRNA levels of various excitatory amino acid transporters, such as EAAT1, EAAT2, and EAAT3, thereby decreasing glutamate levels. This result suggested that DHEA can inhibit the onset and progression of PTS. 90
The aforementioned studies found that among several subtypes of EAAT, EAAT2 has the highest correlation with PTS, which is due to the fact that EAAT2 is mainly distributed in the brain, astrocytes and other regions, and is able to uptake glutamate efficiently, and 90%–95% of forebrain glutamate uptake is accomplished via EAAT2. 91 Although the study of EAAT2 on PTS is still only at the stage of animal model, the current study verifies the feasibility of intervening in PTS through EAAT2. The development of pharmacological interventions for PTS based on glutamate levels presents a promising strategy for not only preventing the onset and progression of PTS, but also for mitigating associated neurological impairments. However, the majority of the findings remain confined to preclinical studies based on animal models and necessitate further clinical validation.
PTS Intervention Based on Anti-Neuroinflammation
Neuroinflammation serves as both an indicator of secondary injury in TBI and a critical causative factor in the pathogenesis of various seizure disorders.107,108 Studies have demonstrated that neuroinflammation is induced by the release of cytokines from astrocytes and microglia. Cytokines, including interleukin-1 (IL-1), interleukin-1β (IL-1β) and high mobility group box 1 (HMGB1), acting as inflammatory mediators and are increasingly recognized for their role in modulating neuronal excitability and facilitating the onset and progression of epilepsy. 109 Therefore, researchers have sought to mitigate seizures by modulating cytokine processes in TBI animal models.
Semple et al found that levels of the inflammatory factor IL-1β and IL-1 receptor expression were significantly increased after TBI in a mouse model. Compared with the control group, treatment with the interleukin-1 receptor antagonist (IL-1Ra) Kineret® (Anakinra) (100 mg/kg, s.c) not only improved the extent of neuropathology and cognitive deficits in TBI mice, but also reduced the long-term epilepsy susceptibility. 92 This study was based on immature mouse brains which presents limitations. The pseudo-peptide inhibitor BoxA has been shown to have antagonistic effects on the signaling molecules HMGB1, which could reduce seizure thresholds and may help to prevent epileptogenesis in individuals at risk for morbidity (eg, craniocerebral trauma). 110 In addition, targeted modulation of multiple inflammatory cytokine receptors by the combination of anti-inflammatory agents (Anakinra, Box A, ifenprodil) has been proven to delay the onset of spontaneous seizures and the disease process in rats models. 93 This strategy of combining multiple anti-inflammatory agents acts as a more effective anti-inflammatory synergy through a multi-targeted mechanism of action, which may hold promising research translational prospects for PTS intervention.
Research on inflammation has indicated that the activity of the inflammatory mediator cyclooxygenase-2 (COX-2) may increase after TBI, which implied that COX-2 may play an important role in neuroinflammation and the development and progression of PTS.94,111 Studies have investigated the efficacy of common COX-2 inhibitors as anti-inflammatory agents in animal models of TBI, for example, (i) nimesulide could improve motility and cognitive deficits in mice suffered from TBI. 94 (ii) Carprofen could reduce inflammation levels and alleviate cerebral edema in controlled cortical impact (CCI, a craniocerebral trauma model) mice model, of which can improve neurologic function in CCI mice. 95 (ii) Meloxicam could reduce short-term blood-brain barrier disruption and cerebral edema in TBI rats. 96 These compounds exhibit anti-inflammatory properties; however, they may also induce adverse effects on the central nervous system and other bodily systems. Therefore, a comprehensive evaluation of their benefits and drawbacks is warranted. Currently, there is a lack of systematic studies to elucidate the role and efficacy of these anti-inflammatory compounds in the progression of PTS.
Neuroinflammation frequently plays a role in the long-term recovery process after TBI, which may lead to secondary physical, mental and cognitive impairments. 112 Anti-neuroinflammation intervention strategies aim to mitigate the neuroinflammatory response after TBI through several different mechanisms including the suppression of pro-inflammatory factor release and the modulation of pertinent inflammatory pathways. Intervention with anti-neuroinflammatory compounds at the appropriate stage may reduce the risk of PTS, minimize secondary damage after TBI, and improve the long-term prognosis of patients. 113 Therefore, although the current studies are limited to preclinical studies based on animal models, the intervention of anti-neuroinflammatory compounds on PTS is still one of the highly promising strategies. And further clinical studies are necessary to substantiate their therapeutic efficacy.
PTS Intervention Based on Antioxidant Stress
Currently, the recognized mechanisms underlying the induction of oxidative stress after TBI are as follows: severe TBI can lead to intracranial and subdural hemorrhages, resulting in the release of iron compounds that cause cellular damage and neurotoxicity. 114 Furthermore, iron accumulation can induce mitochondrial damage and generate ROS, thereby exacerbating neuroinflammation. 115 This process not only contributes to secondary damage in TBI but also increases the risk of PTS. 116 Therefore, one strategy to prevent the development of PTS involves mitigating iron accumulation. Another strategy is to counteract oxidative stress by reducing ROS levels, thereby reducing secondary damage in TBI and achieving PTS intervention. 24
Zhang et al demonstrated that desferrioxamine, an iron chelator, was effective in ameliorating the post-hemorrhagic iron burden and reducing the levels of ferritin and transferrin in rats. Additionally, it mitigated the long-term neurotoxity induced by iron in TBI rats and improved their performance in the Morris water maze test. 97 A clinical randomized controlled study indicated that the administration of deferoxamine mesylate (20 mg/kg/day) via intravenous injection for 5 days can reduce TBI injury-associated edema, which was beneficial for the improvement of prognosis in TBI patients. 98 Studies have suggested that minocycline, as an iron chelator, can be beneficial to improve iron loading in iron toxicity models and cerebral hematoma models. 114 In recent decaeds, numerous studies have validated the intervention of antioxidants in the process of epilepsy in animal models. Early studies have shown that adenosine scavenges hydroxyl radicals and superoxide, as well as possess cerebroprotective effects. In animal models, adenosine can inhibit FeCl3-induced epileptic discharges induced by FeCl3, and prevented the onset of PTS. 99
Melatonin as a potent antioxidant not only scavenges hydroxyl radicals but also possesses the property of inhibiting lipid peroxidation. Studies have shown that melatonin demonstrates efficacy in various experimental epilepsy models. Yokoi and Kabuto et al found that in a posttraumatic epilepsy model, melatonin can inhibit iron-induced seizures through its antioxidant action of scavenging free radicals. 117 A study compared the effects of melatonin and phenytoin on an FeCl3-induced posttraumatic seizures model. They evaluated the oxidative stress outcomes by measuring malondialdehyde (MDA) concentrations in whole brain tissue. The results showed that, compared with the phenytoin group, melatonin treatment significantly reduced brain MDA levels and did not show epileptiform EEG discharges, indicating a superior antioxidant protective effect of melatonin compared with phenytoin. 100 This finding shows that melatonin is a promising antioxidant for PTS. Resveratrol, a natural compound with antioxidant properties, was observed to reduce the incidence of spontaneous seizures in a kainate-induced rat model. 101
The treatment of antioxidants in PTS intervention is still only in animal models, and no validation experiments have been conducted at the clinical stage. Iron chelators are in a similar situation, and clinical studies have only shown that they can facilitate the absorption of intracerebral hematoma masses and promote healing in patients with TBI. 98 Although there are no direct studies currently evaluating and verifying the effects of iron chelators and antioxidants in the development and progression of PTS, the mechanisms associated with oxidative stress after TBI suggest that they may have potential value on the PTS intervention and the prognosis of TBI.
PTS Intervention Based on Regulation of Neurodegenerative Pathways
The stability of microtubule proteins within cells, particularly in the axons of neuronal cells, is regulated by the tau protein and its phosphorylation state. The structural integrity of neurons and axonal transport is critically dependent on the regulation of tau proteins. Hyperphosphorylation of tau proteins diminishes neuronal cell stability and increases the risk of extracellular neural progenitor fibers becoming entangled.118,119 Neurodegeneration is frequently observed after sustaining TBI, and pathologically associated with the aggregation of hyperphosphorylated insoluble tau protein (p-tau) and the formation of neurofibrillary tangles. Several studies have indicated that neurofibrillary tangles composed of p-tau may be linked to the development of seizures after TBI. 120 The regulation of neurodegenerative pathways by modulating p-tau levels is expected to be one of the strategies for PTS intervention. 121
Sodium selenate, an oxidized selenium form compound, has been demonstrated to reduce p-tau levels through the activation of protein phosphatase 2A (PP2A). Several studies have indicated that treatment with sodium selenate can decrease hyperphosphorylated p-tau levels, thereby improving prognosis following after TBI and delaying epileptogenesis in an acquired epilepsy model.102,103 Additionally, it has been shown to reduce susceptibility in pentylenetetrazol-induced models. 104 Furthermore, genetic modification aimed at decreasing p-tau levels has been shown to lower susceptibility to seizures. 105 Although sodium selenite therapy holds considerable promise, the precise mechanism by which sodium selenate intervenes in PTS through p-tau inhibition remains unclear. And the high toxicity of sodium selenate presents a significant barrier to its clinical application. A potential strategy to mitigate this issue could involve optimizing the dosage and frequency of sodium selenate administration to reduce its toxicity. Future studies are required to substantiate and validate the therapeutic potential of sodium selenate.
PTS Intervention Based on Mammalian Target of Rapamycin (mTOR) Pathway
The mTOR signaling pathway is an important cell signaling pathways involved in various physiological processes, including cell growth and survival, proliferation, metabolism, and neuronal morphology. 122 Dysfunction of mTOR signaling pathway has been implicated in numerous diseases, such as diabetes, cancer, neurodegeneration, and epileptic seizures. 123 An increasing number of studies supports the association between PTS and the activation of the mTOR signaling pathway. Inhibition of the mTOR signaling pathway may play a role in mitigating epilepsy and enhancing the prognosis of TBI.124,125
Rapamycin, an inhibitor of mTOR, has been demonstrated to prevent the development of PTS in a TBI mouse model. Compared to control groups, the incidence of epilepsy in mice treated with rapamycin decreased significantly from 50% to 13%. 126 Butler et al. demonstrated that rapamycin treatment mitigated post-traumatic neurological damage in a TBI mouse model, and prolonged continuous rapamycin (3 mg/kg) treatment indicating a trend towards reducing both the frequency and incidence of epilepsy. 106 Subsequently, another study from the same group revealed that rapamycin treatment prevented the reduction of tension currents induced by the exogenous agonist after craniocerebral injury, which presented its potential application in PTS intervention. 127 Campolo M et al found that administration of rapamycin (1 mg/kg) and KU0063794 (a dual inhibitor of mTORC1 and mTORC2, 1 mg/kg) significantly alleviated the inflammatory response and neuronal death after TBI, with KU0063794 exhibiting a more pronounced therapeutic effect. Park J et al confirmed these findings in TBI mice through the concomitant administration of mTOR and AKT inhibitors. 128 Overall, considering the link between dysfunction in the mTOR signaling pathway and PTS, mTOR inhibitors emerge as a promising intervention strategy for PTS. Nonetheless, existing research is predominantly confined to animal models, necessitating further investigation into the underlying mechanisms of mTOR inhibitors in future studies.
Therapeutic Treatment of PTE
PTS interventions are particularly crucial for patients with TBI, as a significant proportion of these individuals are at risk of developing PTE. Achieving seizure control and a complete cure in patients with PTE presents considerable challenges. 14 Therefore, the primary objectives in the management of PTS are to control seizures and to delay the progression of PTE, which encompasses reducing the frequency of seizures, shortening their duration, and mitigating the severity of the seizure types. 129 Once PTE is diagnosed, timely standardized treatment with AEDs remains the preferred strategy for patients diagnosed with PTE. While some patients with PTE may exhibit resistance to pharmacological interventions, the majority can still derive benefit from AED therapy.
In clinical practice, the selection of monotherapy or combination therapy is commonly guided by the type of seizures and the severity of PTE. Studies have indicates that LPTSs are predominantly partial seizures. Consequently, a variety of AEDs are commonly used in clinical practice, including traditional AEDs such as CBZ, VPA, PHT, as well as newer AEDs like lamotrigine and LEV. It is crucial to regularly monitor patients’ serum AED concentrations during treatment and to adjust the types and dosages of AEDs in response to the patient’s clinical condition. Gradual dose adjustments are recommended during AED treatment for PTE, as abrupt discontinuation can exacerbate seizure frequency and severity. Furthermore, approximately 33% of PTE patients may develop resistance to AEDs. For those with refractory PTE, alternative interventions such as surgery, neural stimulation, and the ketogenic diet should be considered. 130 Surgical treatment can ameliorate or eliminate seizure symptoms by resecting the epileptogenic focus or blocking the propagation of localized epileptic activity. However, for patients with PTE, surgical treatment poses significant challenges and may be associated with increased risks and reduced efficacy.
Discussion
TBI can lead to the development of epileptogenicity, characterized by complex chronic neural hyperexcitability. PTS are frequently observed following TBI and can be categorized into EPTS and LPTS based on the timing of the initial seizure event. Pharmacological intervention of PTS is crucial for enhancing the prognosis and quality of life in patients with TBI. Among the AEDs considered, PHT and VPA demonstrate efficacy in EPTS after TBI, without significant impact on LPTS. In contrast, LEV has been identified as effective and well-tolerated in both EPTS and LPTS. However, these conclusions are primarily derived from retrospective, uncontrolled observational studies with limited sample size studies. Additionally, the variability in study populations, the heterogeneity of clinical data, the timing of AED intervention, and the duration of treatment further influence the outcomes.
Current research indicates that excitotoxic environment resulting from tissue injury, neurotransmitter release, and Ca2+ influx after TBI is considered a primary cause of EPTS. 131 AEDs may provide short-term intervention for EPTS by inhibiting neuronal hyperexcitability and modulating ion channels within this timeline. However, with the long-term pathophysiological changes of progressive injury, including neuroinflammation, oxidative stress, and neuronal regeneration and synaptic remodeling, AEDs demonstrate limited efficacy in intervening these complex pathological processes. 78 The results of these studies may be the reason why AEDs are effective for EPTS intervention but not for LPTS.
Overall, the available clinical studies do not conclusively support the long-term benefits of pharmacological interventions using AEDs. Interventional medication regimens for AEDs still need to be further explored and validated, and high-quality randomized controlled studies with large sample sizes and prospective studies are necessary. A viable approach may involve conducting comprehensive risk/benefit assessments of patients during the development of long-term interventional protocols for AEDs. For patients at high risk of TBI who are predicted to develop PTE, long-term intervention should be implemented as much as possible. Moreover, the selection of new-generation antiepileptic drugs (AEDs) that enhance long-term prognosis while minimizing adverse effects is recommended. PTS interventions for specific patient populations, such as pediatric and geriatric groups, necessitate increased vigilance concerning the potential effects of medications on cognitive function in the developing brains of children, as well as the complex drug interactions and reduced drug tolerance risks associated with physiological decline, comorbidity, and polypharmacy in the elderly. Therefore, in the selection of antiepileptic drugs for pediatric and geriatric populations, preference should be accorded to medications that exhibit minimal cognitive side effects, a low propensity for drug interactions, and favorable safety profiles, exemplified by lEV and lamotrigine. Dosages ought to be meticulously adjusted in accordance with individual patient circumstances, with careful monitoring of both therapeutic efficacy and potential adverse reactions.
As the physiologic mechanisms of TBI and the pathogenesis of PTS continue to be elucidated, the physiological alterations induced by tissue damage after TBI present both a latency period and an opportunity for pharmacological intervention. Novel compounds including antioxidants, anti-neuroinflammatory compounds, antioxidants, and glutamate modulators have demonstrated efficacy in various animal models by reducing the susceptibility of PTS, thereby mitigating the onset and progression of PTS. These selective, emerging, and targeted intervention strategies may provide multiple protective mechanisms against the pathophysiological changes associated with TBI, potentially benefiting PTS intervention and the reduction of secondary brain damage after TBI. In particular, anti-neuroinflammatory compounds and anti-oxidative stress compounds appear to be promising strategies for TBI recovery and PTS intervention. Although some of these compounds have the potential to be used in clinical practice in the future, these results are still based on animal studies, which are still in the pre-exploratory stage, and the specific mechanisms of intervention are still unclear. More preclinical and clinical studies are still needed to validate the intervention effects of these compounds in the near future. Overall, these intervention strategies provide new directions for PTS interventions and hold the potential to become significant enhancements to future clinical practice, particularly in contexts where existing therapeutic options are constrained.
Conclusion
In summary, although recent studies have provided promising evidence and strategies for the intervention and treatment of PTS over the past decades, numerous questions remain to be addressed. The intervention mechanism of AEDs primarily involves the regulation of neuronal excitability, whereas the emerging multi-targeted PTS intervention strategy is predicated on understanding neurological injuriousness, with both approaches exhibiting mutual synergistic properties. The synergistic combination therapy of AEDs and emerging compounds holds promise as an effective strategy for PTS intervention and represents a significant direction for future research. Multidisciplinary collaborative research combining mechanisms, therapeutic targets, and drug discovery should be directed to discover the key of PTS intervention.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article:This work was funded by Shenzhen Longgang District Economic and Technological Development Special Funds grant number (No. LGKCYLWS2021000038) and the 2024 Healthcare Quality (Evidence-Based) Management Research Project of the National Institute of Hospital Administration, National Medical Commission of the People’s Republic of China (No.YLZLXZ24G030).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Conceptualization W.Z., J.O. and R.L.; software J.O. X.L and R.L.; validation W.Z. and M.T.; investigation W.Z., X.L and Y.Y; resources W.Z. and Y.Y; data curation, W.Z., J.O. and R.L.; writing—original draft preparation W.Z., J.O. and R.L.; writing—review and editing R.L., Y.L. and M.T.; supervision W.Z. and M.T.; project administration W.Z. and Y.Y; funding acquisition W.Z. and Y.Y. All authors have read and agreed to the published version of the manuscript.
