Abstract
Background
Sacubitril/valsartan is recommended for patients with New York Heart Association class II to III heart failure (class 1 recommendation). The objectives of the study were to evaluate the effectiveness and safety of metoprolol against sacubitril/valsartan in patients with heart failure and reduced ejection fraction (HFrEF).
Methods
In retrospective study, patients aged ≥18 years with heart failure and less than 40% of left ventricular ejection fraction received 25 mg metoprolol once daily (ML cohort, n = 117) or 50 mg sacubitril/valsartan twice daily (SV cohort, n = 128) for 6-months.
Results
Systolic and diastolic blood pressures, heart rates, N-terminal pro-brain natriuretic peptide values, and left ventricular ejection fraction values improved across both cohorts, especially in the ML cohort after treatments for 6-months as compared to before treatments (P < .05 for all). Death was higher in the SV cohort than in the ML cohort over 15 months (10 (8%) vs. 2 (2%), P = .0362). Fatigue, depression, shortness of breath, and wheezing have been reported in patients in the ML cohort. Dizziness, hyperkalemia, fatigue, abdominal or stomach pain, and blurred vision have been reported in patients in the SV cohort.
Conclusions
Metoprolol may offer superior safety with comparable efficacy.

Keywords
Introduction
The heart failure is one type of the clinical condition in which ventricular filling and/or ejection functions are impaired 1 due to various cardiac structural and/or functional pathologies or conditions. 2 The global prevalence of heart failure ranges between 1-3%, 3 and 20% of heart failure patients are available in China mainland itself. 4 Heart failure generally increases the financial burden on the healthcare systems. 5
In the last few years, various options have become available for the preventions and treatments of patients with heart failure. 6 Evidence-based medical treatments generally reduce hospitalizations due to clinical conditions of heart failure, significantly improve the quality of life of patients with heart failure, and generally increase the probabilities of survival of patients with chronic heart failure with reduced ejection fraction (HFrEF). 7 Sacubitril and valsartan are angiotensin receptor–neprilysin inhibitor type drug. 2 They could decrease possibilities of death due to all-reasons and requirements of hospitalization due to all-reasons in patients suffering from heart failure 8 and is superior to valsartan alone because of the strong lowering action of N-terminal pro-brain natriuretic peptide (NT-proBNP) in patients with advanced stages of heart failure (by sacubitril). 8 Sacubitril/valsartan prolongs survival of patients suffering from HFrEF. 9 In addition, sacubitril/valsartan is recommended for patients with New York Heart Association class II to III heart failure (this is ‘Class 1 recommendation’). 10 Sacubitril and valsartan reduces the risk of hospitalization and death due to long lasting (chronic) heart failure conditions.8,11 Sacubitril and valsartan are effective in patients suffering from heart failure through relaxation of the blood vessels and pumping of blood throughout body by heart becomes easy. 11 In contrast, very high numbers of unwanted adverse effects are generally observed during sacubitril/valsartan treatments (e.g., hypotension (low blood pressure), cough, dizziness, high blood serum potassium level, decreased hematocrit level, acute renal failure, and high blood serum creatinine level). 6 On the other side of the treatments available metoprolol (a specific β-blocker) can reduce mortality due to all-cause and requirements of hospitalization due to all-cause in patients with HFrEF or heart failure with preserved ejection fraction (HFpEF). 12 The occurrence of ischemic type heart disease and atrial fibrillation of heart are positively associated with β-blocker use during conditions HFrEF or HFpEF, while increased frequencies of hospitalization and diuretic overdose are negatively associated with β-blocker use in patients with HFrEF or HFpEF. 13 Sacubitril/valsartan and metoprolol both are currently used in clinical practice especially in our institute. The PARADIGM-HF trial 14 established sacubitril/valsartan as superior to enalapril, but no direct comparison with metoprolol exists. This gap should be addressed.
The objectives of this retrospective study were to evaluate the effectiveness (NT-proBNP values, mortality, hospitalization) and safety (adverse effects and death rate) of metoprolol against low-dose (50 mg) sacubitril/valsartan sodium (sacubitril 24 mg and valsartan 26 mg) in Chinese HFrEF (<40% of the left ventricular ejection fraction) patients in the north-central region of China. In addition, the study evaluated independent parameter for all-cause of death.
Materials and Methods
Ethics Approval and Consent to Participate
The designed protocol of the current study (Approval number 15BMChH dated January 15, 2020) was prepared by the authors themselves and approved by the Baoji Maternal and Child Health Hospital review board. The study follows the laws of China and the v2008 Declarations of Helinski. The need for informed consent for participation in the study and publication of the study (in one or more forms) were waived by the Baoji Maternal and Child Health Hospital review board because of the retrospective nature of the study.
Design, Setting, and Period
The retrospective collection of the electronic medical records of patients of the Baoji Maternal and Child Health Hospital, Baoji, Shaanxi, China from January 14, 2020, to December 16, 2021.
Inclusion Criteria
Patients aged 18 years or older, with heart failure, and less than 40% of left ventricular ejection fraction (during the last 6 months) 13 who required treatments for heart failure were included in the study analyses (physician’s decision to put on treatments for heart failure).
Exclusion Criteria
Patients those who have incomplete medical records in institutes (three or more vital parameters which affect study results) were excluded from the study. If patients discard any treatments, have therapies interruption, or erroneous administration, then they were excluded from analyses. Patients with severe renal dysfunction and arrhythmias were excluded from the study.
Sample Size Calculations
Based on the assumption that with treatments for HFrEF, there would be a 20% decrease in NT-proBNP values of patients after 6-months of treatments for HFrEF compared to before treatments conditions (BT conditions; primary outcome; effect size). 7 In addition to these assumptions, using OpenEpi software, 15 two-sided significance level (1-α): 95%, and power (1-β, % chance of detection): 80%, and 95% confidence interval the sample size was 117 (minimum patients required in each cohort).
Cohorts
Hundred and seventeen patients received immediate release of 25 mg metoprolol succinate once daily for six months (ML cohort). 16 Hundred and twenty eight patients received low-dose (50 mg) sacubitril/valsartan sodium tablets (sacubitril 24 mg and valsartan 26 mg) twice daily for six months (SV cohort). 16 All patients included in the study were treated with a fixed dose of metoprolol or sacubitril/valsartan for six months. During the treatments period of six months, patients have not received additional sodium-glucose transport protein inhibitors (SGLT-2 inhibitor) or leucovorin to enhance heart failure prognosis.
Outcome Measures
Outcome measures are data those were collected from medical records of patients of hospital.
NT-proBNP Values
An electrochemiluminescent assay (monoclonal assay) was performed to evaluate serum NT-proBNP values. 17 This finding was associated with the primary outcomes of the study.
Secondary Outcomes
Data of hospitalization due to heart failure within treatments and all-cause death within 15 months, including 6 months of treatments, were extracted and evaluated.
Adverse Effects
Any unwanted adverse effects due to prescribed treatments within 6 months of treatments and follow-up (9 months) were extracted from the hospital records of the patients and evaluated.
Clinical Benefits for Treatments
The clinical benefits of the treatments of patients with HFrEF were evaluated as a function of the beneficial scores. Beneficial scores for treatments were calculated from the risk of under-treatments, as expressed in equation (1). The risk of undertreatment was defined using a calculation that involves the percentage of decrease in NT-proBNP values of patients after 6-months of treatments (AT conditions), above which treatments was terminated for patients with reported HFrEF (and underwent treatments for the same; equation (2)). The percentage decrease in NT-proBNP values was considered a numerical value from 0% to 100%. The beneficial score for the treatments of patients with HFrEF was the area above the curve of the method of treatments adopted for patients, and the working area was the area under the curve of the method of treatments adopted for patients. For all methods adopted for treatments, a 20 percentage positive decrease in NT-proBNP values (compared to BT conditions) was used as the reference standard (effect size primary endpoint).
18
The percentage decrease in NT-proBNP values of the patients was calculated using equation (3).
Detrimental Score for Death
Similar to the clinical benefits for the treatments of patients with HFrEF, the detrimental effects lead to the death of the method of treatments adopted for HFrEF were evaluated as a function of the detrimental score for death. The detrimental scores for the treatments related death of patients with HFrEF were calculated from the risk of death, as expressed in equation (4). The risk of death was defined with a calculation that involved the percentage of death during 15-months of follow-up including 6-months of treatments period for treatments of patients with HFrEF (equation (5)). The percentage of deaths was considered to be a numerical value ranging from 0% to 100%. The detrimental score of treatments for HFrEF is the area above the curve of the method of treatments adopted for HFrEF, and the working area is the area below the curve of the method of treatments adopted for HFrEF. For all treatments adopted for HFrEF, no risk of death (0% death) was used as the reference standard (secondary endpoint).
18
Statistical Analyses
InStat 3.01; GraphPad Software, (San Diego, CA, USA) was used for statistical analyses. Fisher’s exact test or chi-square test (χ2-test with or without Yate’s corrections) was used for the statistical analysis of categorical variables. The Kolmogorov and Smirnov method was used to check the normality of the continuous variables. The Mann–Whitney test or Kruskal–Wallis’ test (nonparametric analysis of variance (ANOVA); between cohorts) or Friedman test (within cohort) was performed for continuous non-normal variables for statistical analyses. An unpaired t-test with or without Welch corrections or repeated measures of ANOVA (between cohorts) was performed for continuous normal variables for statistical analyses. Tukey’s test (for continuous normal variables) or Dunn’s test (for continuous non-normal variables) multiple comparison methods were used for post hoc analyses (when P < .05; to check type-I error). Multivariate logistic analyses were performed after univariate statistical analyses to evaluate independent parameters for all-cause of death during 15 months, including 6 months of treatments. 4 All results were considered significant if the threshold P-value was less than .05.
Results
Study Population
From January 14, 2020, to December 16, 2021, 252 patients (aged 18 years or older) with heart failure and less than 40% of left ventricular ejection fraction (during last 6 months results; data collected from hospital records of the Baoji Maternal and Child Health Hospital, Baoji, Shaanxi, China and the referring hospitals) receiving treatments for HFrEF at the Baoji Maternal and Child Health Hospital, Baoji, Shaanxi, China, and the referring hospitals were included. Among them (252 patients), the complete medical records of seven patients were not available to the hospitals. Therefore, data of these patients (seven patients) were excluded from the analysis. Data on NT-proBNP values, hospitalization due to heart failure within treatments, all-cause death during 15 months (time of treatments + follow-up period), and adverse effects of treatments during 15 months (time of treatments + follow-up period) of 245 patients with HFrEF who taking treatments for the same at the Baoji Maternal and Child Health Hospital, Baoji, Shaanxi, China and the referring hospitals were collected from the hospitals’ records of patients and analyzed (anonymous procedures were used to collect data from electronic medical records). The retrospective collection of the medical records of patients and their analyses in the current study is presented as a flow chart in Figure 1. Presentation of Electronic Data for the Study. BT: Before Treatments, AT: 6-Months After Treatments. The Red Color Indicates Worse Parameter Outcomes. Black Color Indicates Not Worse and Not Better Parameter Outcomes.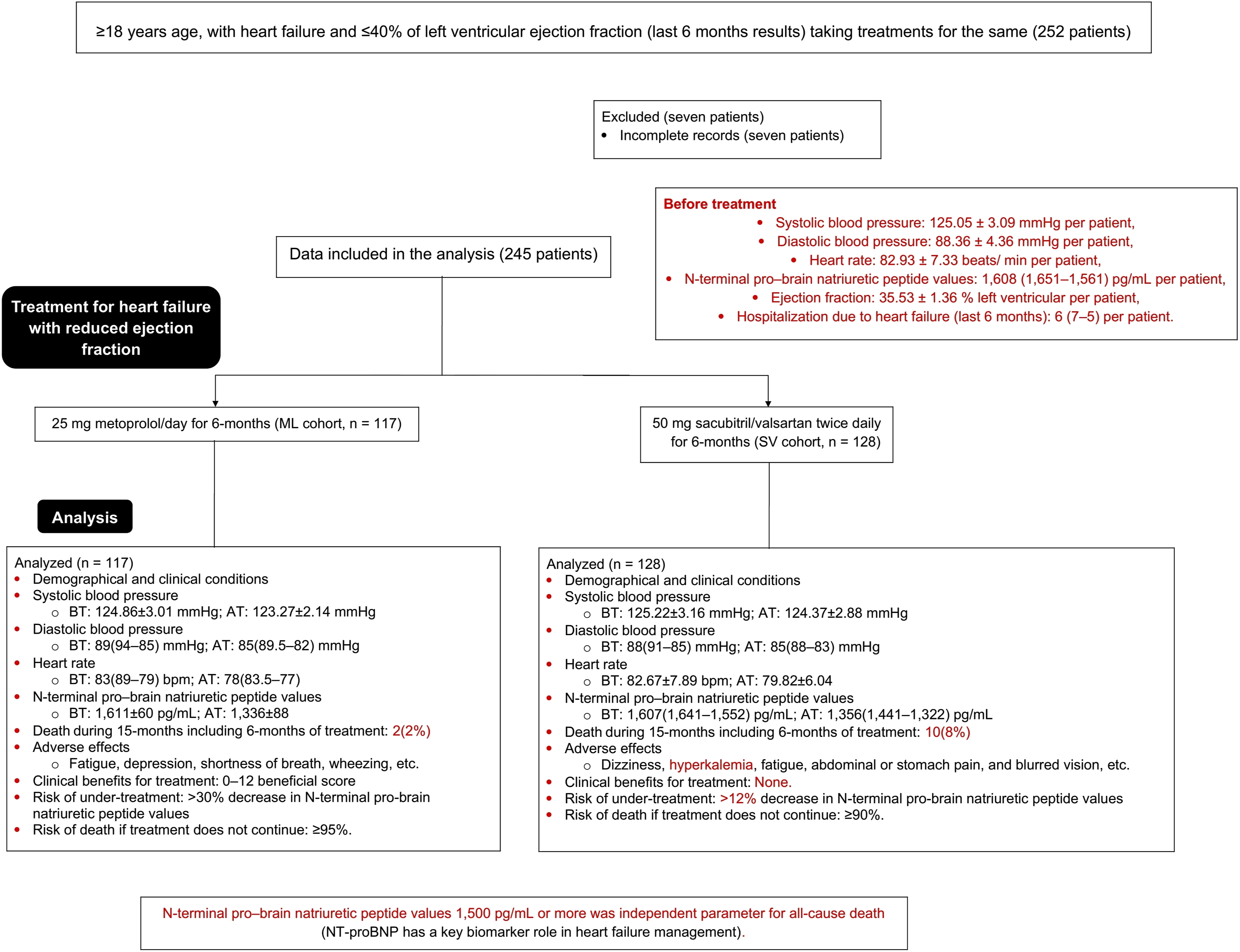
Demographic and Clinical Characters
Demographical and Clinical Conditions of the Enrolled Patients Before Treatments for Heart Failure and Reduced Ejection Fraction.
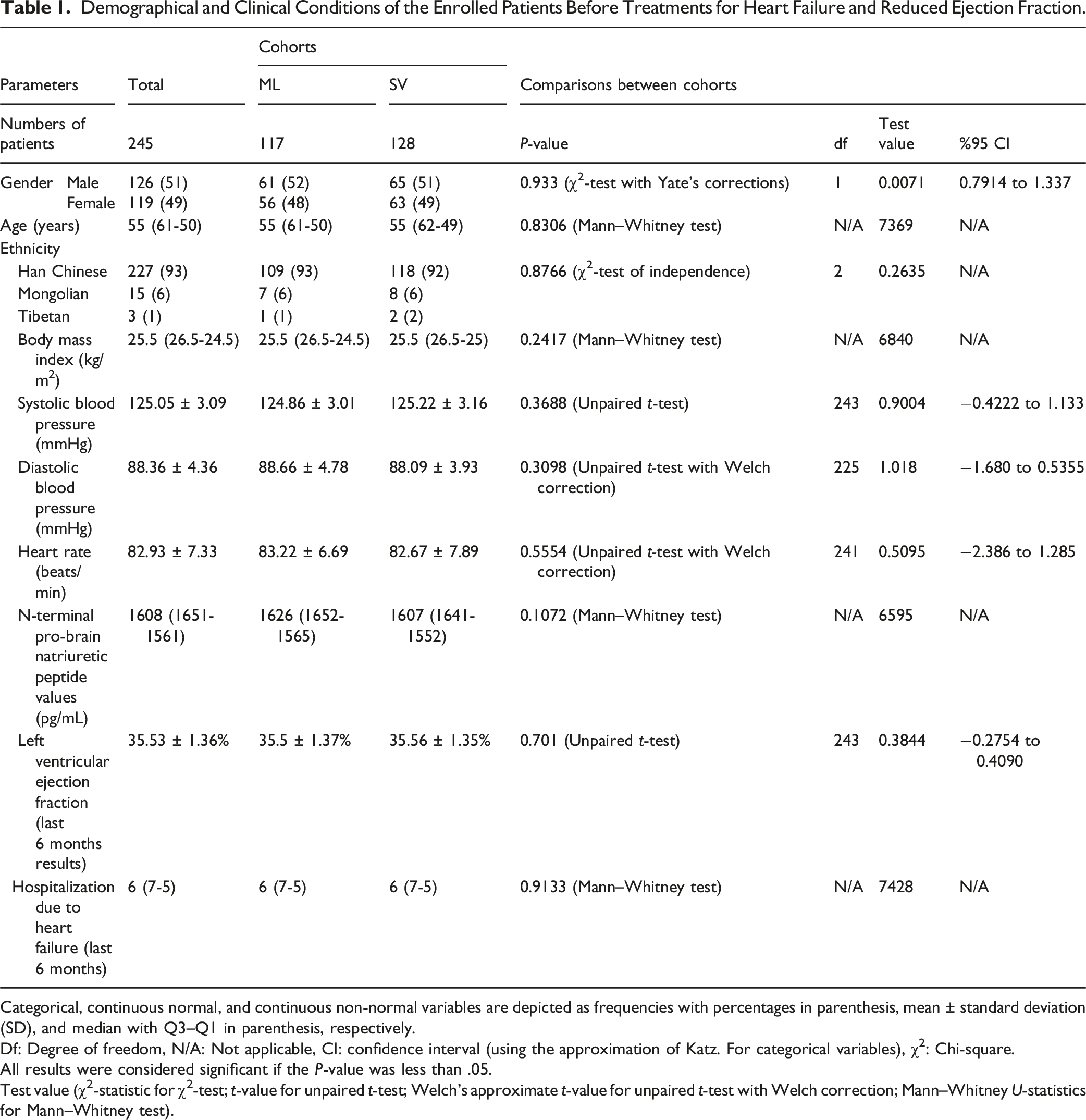
Categorical, continuous normal, and continuous non-normal variables are depicted as frequencies with percentages in parenthesis, mean ± standard deviation (SD), and median with Q3–Q1 in parenthesis, respectively.
Df: Degree of freedom, N/A: Not applicable, CI: confidence interval (using the approximation of Katz. For categorical variables), χ2: Chi-square.
All results were considered significant if the P-value was less than .05.
Test value (χ2-statistic for χ2-test; t-value for unpaired t-test; Welch’s approximate t-value for unpaired t-test with Welch correction; Mann–Whitney U-statistics for Mann–Whitney test).
Outcome Measures
Results Measures of the Enrolled Patients for Primary and Secondary Outcomes
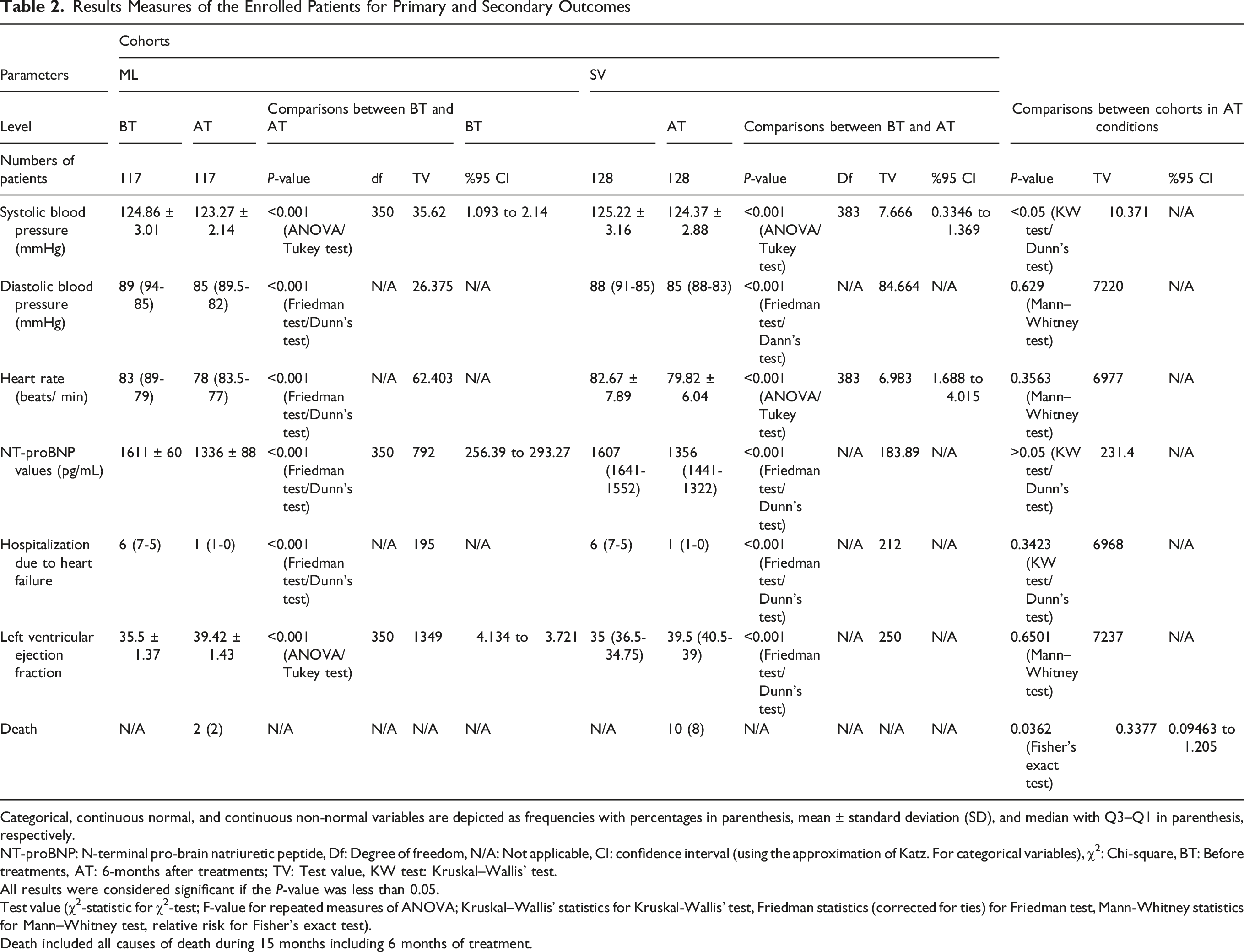
Categorical, continuous normal, and continuous non-normal variables are depicted as frequencies with percentages in parenthesis, mean ± standard deviation (SD), and median with Q3–Q1 in parenthesis, respectively.
NT-proBNP: N-terminal pro-brain natriuretic peptide, Df: Degree of freedom, N/A: Not applicable, CI: confidence interval (using the approximation of Katz. For categorical variables), χ2: Chi-square, BT: Before treatments, AT: 6-months after treatments; TV: Test value, KW test: Kruskal–Wallis’ test.
All results were considered significant if the P-value was less than 0.05.
Test value (χ2-statistic for χ2-test; F-value for repeated measures of ANOVA; Kruskal–Wallis’ statistics for Kruskal-Wallis’ test, Friedman statistics (corrected for ties) for Friedman test, Mann-Whitney statistics for Mann–Whitney test, relative risk for Fisher’s exact test).
Death included all causes of death during 15 months including 6 months of treatment.
Results of the Assumption Test Adopted in the Study.

NT-proBNP: N-terminal pro-brain natriuretic peptide.
Adverse Effects
Adverse Effects of the Enrolled Patients due to Treatments Within 6 Months of Treatment and Follow-Up (9 Months).

Variables are depicted as frequencies with percentages in parentheses.
Fisher’s exact test was used for statistical analysis.
All results were considered significant if the P-value was less than .05.
CI: confidence interval (using the approximation of Katz).
Clinical Benefits for Treatments
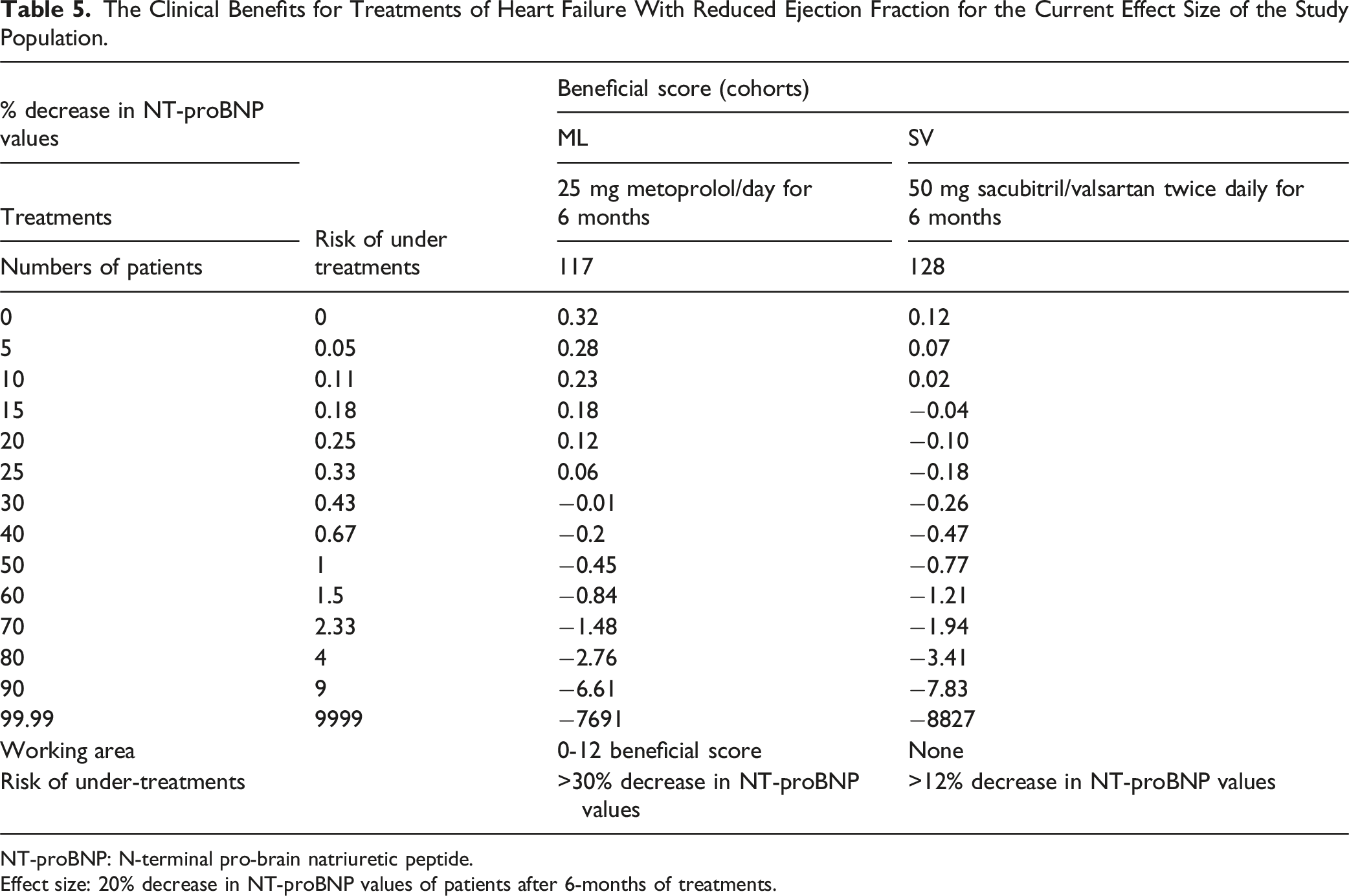
The patients in the SV cohort had working areas of −0.1 to 0.11 beneficial scores, and more than a 12% decrease in NT-proBNP values had the risk of undertreatments. The patients in the SV cohort had no beneficial scores for the current effect size of the study population. Working areas for patients in the ML cohort had 0 to 0.12 beneficial scores, and for the current effect size of the study population, a more than 30% decrease in NT-proBNP values had a risk of undertreatments. A graphical presentation of the clinical benefits of the treatment of heart failure with reduced ejection fraction is presented in Figure 2. The details of the clinical benefits of the treatments of heart failure with reduced ejection fraction for the current effect size of the study population are presented in Table 5. The Graphical Presentation of Clinical Benefits for Treatments of Heart Failure With Reduced Ejection Fraction. The Clinical Benefits for Treatments of Heart Failure With Reduced Ejection Fraction for the Current Effect Size of the Study Population. NT-proBNP: N-terminal pro-brain natriuretic peptide. Effect size: 20% decrease in NT-proBNP values of patients after 6-months of treatments.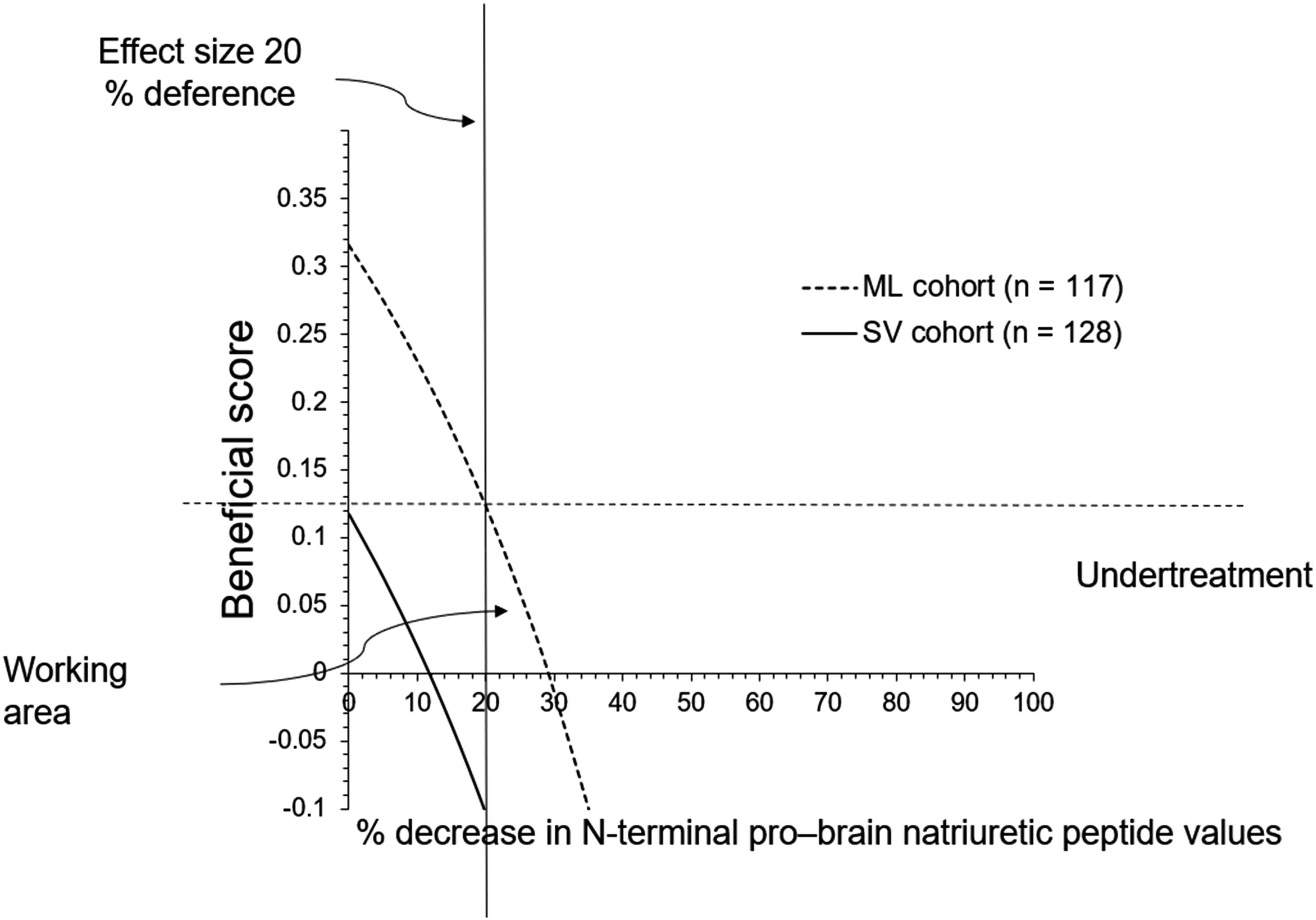
Detrimental Score for Death
The working area had a 0-0.92 detrimental score for death for patients in the SV cohort and a 0-0.98 detrimental score for death for patients in the ML cohort. Patients in the SV cohort had a 90% or higher risk of death, and those in the ML cohort had a 95% or higher risk of death if treatments was not continued. A graphical presentation of the detrimental score for death in the treatments of heart failure with reduced ejection fraction is presented in Figure 3. The details of the detrimental score for the treatments of heart failure with reduced ejection fraction for the current effect size of the study population are presented in Table 6. The Graphical Presentation of the Detrimental Score for Death for Treatments of Heart Failure With Reduced Ejection Fraction. The Detrimental Score of Death for Treatments of Heart Failure With Reduced Ejection Fraction for the Current Effect Size of the Study Population. Reference: No death.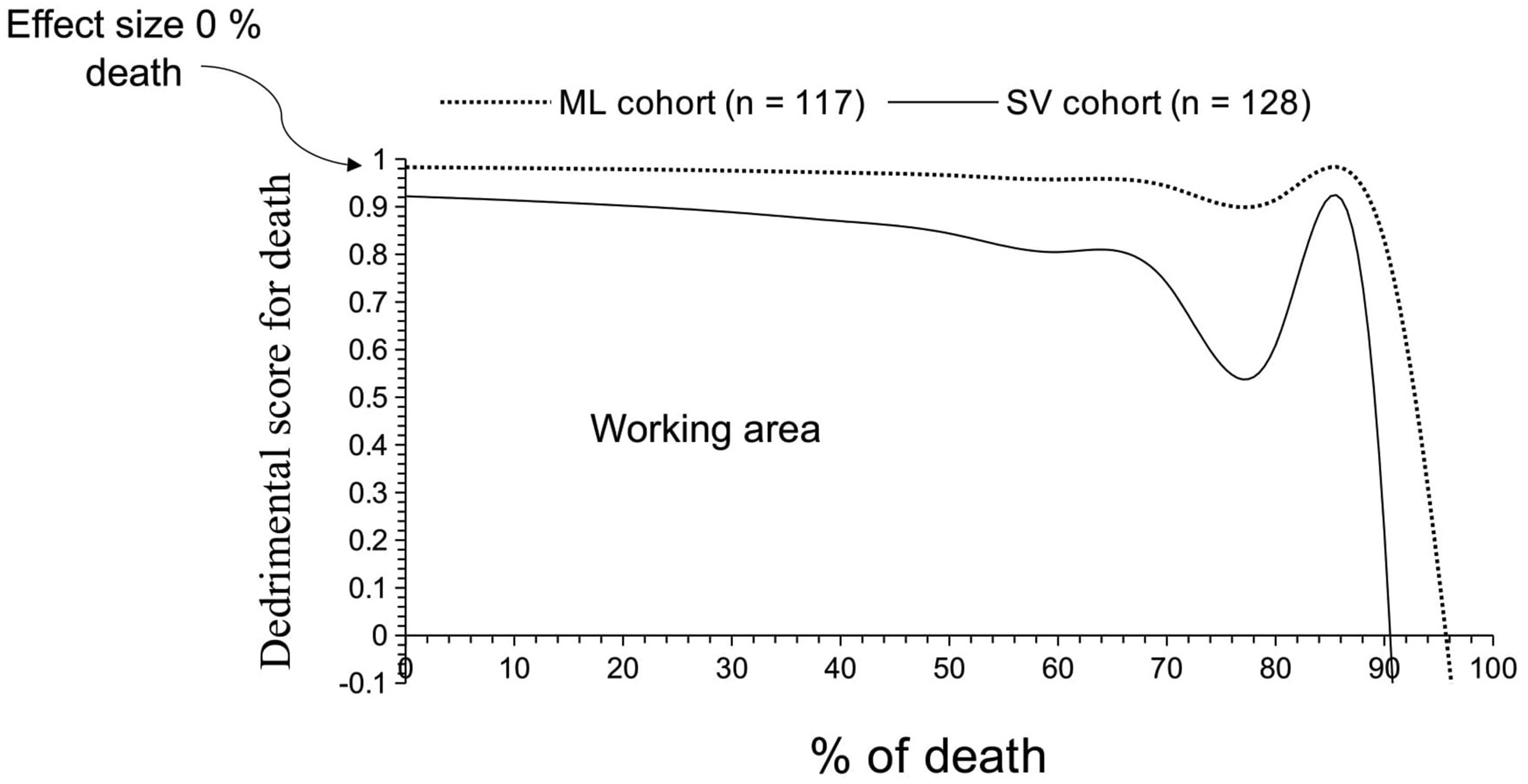

Independent Parameter for All-Cause of Death
Univariate Following Multivariate Logistic Analysis.

Patients who survived (n = 233) were considered as reference standard.
P-value less than .05 and an odd ratio more than was considered significant.
NT-proBNP: N-terminal pro-brain natriuretic peptide, CI: confidence interval.
*Significant parameter for all-cause mortality.
Discussions
Both treatments (low-dose sacubitril valsartan and metoprolol), especially metoprolol, improved all parameters (primary and secondary objectives). Metoprolol is optimally used in patients. 13 Low-dose sacubitril and valsartan are reported to be effective in patients with HFrEF.19,20 However, metoprolol is more likely to be preferred by heart failure patients with ischemic heart disease and atrial fibrillation, while sacubitril and valsartan are not preferred by patients with heart failure and renal compromise patients. 13 Our study recommended low doses of sacubitril and valsartan (50 mg) and metoprolol in patients with heart failure and ischemic heart disease (or any one treatment among recommended four pillars of patients with HFrEF).
Only metoprolol had clinical benefits in achieving the primary objectives (20% or more decrease in NT-proBNP values for the BT conditions). Sacubitril and valsartan degrade biologically active natriuretic peptides (atrial natriuretic peptide, β-type natriuretic peptide, and C-type natriuretic peptide), but they could not degrade biologically inert NT-proBNP. 21 Thus, the current study recommends metoprolol over low doses (50 mg) of sacubitril and valsartan for effective decrease of the NT-proBNP levels in management of HFrEF.
The high levels of NT-proBNP level were associated with death. The results of the independent parameter of death in patients with HFrEF were consistent with those of a multicenter study. 4 A high NT-proBNP level is a prognostic factor in patients with HFrEF.
The patients had elevated systolic and diastolic blood pressures, heart rates, NT-proBNP values, and left ventricular ejection fraction before the start of treatments. Patients with HFrEF have hypertension, chances of worsening renal function, or both. 8 The current study provides in the state-of-the-art about the topic. There is a need for the proper treatments of patients with HFrEF.
In the current study, patients were received low doses of sacubitril and valsartan (24 mg /26 mg) twice daily for 6-months in the SV cohort. New recommendations of the 2022 guidelines from the American College of Cardiology suggested the low doses of sacubitril and valsartan (24 mg/26 mg) twice daily in HFrEF (this recommendation is not incorporated solely sacubitril and valsartan but rather be applied as part of an approach for maximizing dosage of the quadrable therapy, which is essential to be implemented in every patient. In addition, the 2022 guideline emphasizes the benefit of “quadruple therapy” in patients with symptomatic HFrEF, i.e., sacubitril-valsartan, an evidenced-based beta-blocker, a mineralocorticoid antagonist, and a sodium-glucose transport protein inhibitors-2 inhibitor. In clinical practice, it is essential to implement these guidelines in every patient at every visit with a stepwise approach: Step 1. Is the patient on optimal guideline-directed medical therapy? Step 2. If not, justify why (prior intolerance, cost, allergy) and document it. Step 3. Is the patient on maximum-tolerated dosages of guideline-directed medical therapy? Step 4. If not, either increase dosages in a stepwise fashion or document that why further titration is not possible (limiting heart rate, blood pressure, potassium, or creatinine). This would include stepwise initiation, every 1 to 2 weeks, of the following: Low-dose sacubitril-valsartan (sacubitril 24 mg and valsartan 26 mg, twice daily), followed by, a beta-blocker (carvedilol 3.125 twice daily or metoprolol succinate 25 mg daily), then, a mineralocorticoid antagonist (spironolactone 25 mg daily or eplerenone 50 mg daily), and an SGLT-2 inhibitor (dapagliflozin 10 mg daily or empagliflozin 10 mg daily). After all four pillars of treatment are initiated, then sacubitril-valsartan and beta-blocker could be doubled every 1 to 2 weeks as tolerated by heart rate, blood pressure, and serum potassium and creatinine levels). 16 In contrast, there are similar improvements in all types of outcome parameters across high and low doses of sacubitril and valsartan combinations in patients with HFrEF. 19 The expected required benefits in improvement in health status could be gained at low doses of sacubitril and valsartan in patients with HFrEF. 20
Comparative Studies on Heart Failure Treated With Different Forms of Drugs in Different Settings.

The study in question investigates the effectiveness and safety of metoprolol vs sacubitril/valsartan in Chinese patients with HFrEF. Both medications have been established as beneficial in managing heart failure, but the study aims to provide insights into their comparative outcomes, specifically in a cohort of Chinese patients, which is valuable given potential ethnic and genetic variability in drug responses. However, there are certain limitations to this study, such as retrospective cohorts’ analyses and lack of prospective randomized trials. The justifications for the same is that prospective randomized trials results are far away from the real-world clinical care. 8 All patients were Chinese and a single center study, therefore, issues of generalizability of results. Health level of knowledge professionals on the utilization of treatments of heart failure with reduced ejection fraction (<40% of left ventricular ejection fraction) have effects on study results. 13 These were not evaluated in the study. Additionally, the study does not account for potential confounding factors, such as coexisting comorbidities or the use of other heart failure medications. The longer-term effects of both treatments beyond the 15-month follow-up period are not explored, leaving uncertainties regarding the durability of the observed benefits. Moreover, the dosages of both medications, the ML cohort received a lower starting dose of metoprolol (25 mg/day) compared to standard clinical practice (50-200 mg/day; particularly sacubitril/valsartan; the SV cohort’s dosage (50 mg twice daily) corresponds to sacubitril 24 mg/valsartan 26 mg, which is lower than the USFDA-approved 97/103 mg twice daily) doses were lower than the recommended target doses in guidelines, potentially influencing the outcomes of the current study (possible justification is that this is our institute protocol for treatment of HFrEF). This may limit generalizability. The discrepancy in mortality rates may be partly attributable to suboptimal dosing in the SV cohort, although this is speculative. Future studies should examine whether higher doses of sacubitril/valsartan would yield different results or if the safety concerns are intrinsic to the drug.
Conclusions
There is a need for proper treatments of patients with heart failure with reduced ejection fraction (<40% of left ventricular ejection fraction). A total of 25 mg metoprolol once daily and 50 mg sacubitril/valsartan twice daily for 6 months both have effects in the management of heart failure with reduced ejection fraction (<40% of left ventricular ejection fraction) and manageable adverse effects. Metoprolol effectively decreased N-terminal pro-brain natriuretic peptide values more than that does through sacubitril/valsartan in the management of heart failure with reduced ejection fraction (<40% of left ventricular ejection fraction). A high N-terminal pro-brain natriuretic peptide level is a prognostic factor for patients with heart failure with reduced ejection fraction (<40% of left ventricular ejection fraction). The study highlights the prognostic value of N-terminal pro-brain natriuretic peptide levels, as values ≥1500 pg/mL were associated with all-cause mortality. This finding reinforces N-terminal pro-brain natriuretic peptide’s role as a key biomarker in heart failure management, particularly in risk stratification for adverse outcomes. The elevation of N-terminal pro-brain natriuretic peptide in both cohorts underscores its importance in monitoring treatment response and adjusting therapeutic strategies accordingly.
Footnotes
Authors’ Note
All the data and related metadata underlying the reported findings already provided as part of the submitted article.
Acknowledgments
The authors are thankful to the medical and non-medical staff of the Baoji Maternal and Child Health Hospital, Baoji, Shaanxi, China.
Author Contributions
All authors have read and approved the manuscript for publication. YH was the project administrator and contributed to the supervision, resources, methodology, validation, and literature review of this study. QL contributed to the conceptualization, methodology, investigation, software, visualization, and literature review of this study. YL contributed to the resources, methodology, formal analysis, software, visualization, and literature review of the study. All authors contributed to drafting and editing of the manuscript for intellectual content. All authors agree to be accountable for all aspects of this work, ensuring its integrity and accuracy. YH and QL confirmed the authenticity of raw data.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.





