Abstract

Introduction
The findings reported in this paper are a consequence of many years of scientific research and I have relied on a number of publications1-45,45-70,71-90,91-120,121-166 relevant to topics addressed. Although now retired, I continue to follow key findings related to the ongoing debate about health risks associated with exposure to low radiation doses.
The principal objective of this paper is to address what is wrong with the System of Radiological Protection (SRP) and to point out what changes could be made to make it better. The four quotations of outstanding scientists that follow help to understand my concern about the current link of the SRP to linear no-threshold (LNT) risk models for radiation-induced cancer that clearly promotes radiophobia. “Largely unnoticed, all life on earth is constantly exposed to low levels of ionizing radiation” (Maier et al.
82
). “The LNT model has not been validated at low doses. Its indiscriminate use to predict an increase in cancer risk following a low-dose exposure tends to cause more harm than it is intended to prevent … Other models of the multiresponse type have been proposed to reflect biological reality much more accurately …” (Feinendengen and Cuttler
41
). “Preconceived concepts impede progress; in the case of the LNT model, they have resulted in substantial medical, economic, and other societal harm” (Tubiana et al.
152
). “It is high time to replace the LNT paradigm by a scientifically based dose-effect relationship where realistic quantitative hormetic or threshold models are exploited” (Janiak and Waligórski
66
).
The core of the SRP consists of three fundamental principles: justification, optimization, and applying ionizing radiation dose limits.84,163 The principle of justification specifies that any activity (or intervention) that changes the radiation-exposure scenario should be overall beneficial to individuals and/or society; the principle of optimization specifies that radiation doses should be as low as reasonably achievable (ALARA), considering both economic and societal factors; the principle of dose limitation applies to planned radiation exposures (other than medical and environmental) and instructs that doses should not exceed established radiation dose limits. Unfortunately, the SRP promotes radiophobia in cases of population exposure to low radiation doses above natural background radiation exposure.
LNT as the Null Hypothesis
In this paper I point out the main cause of radiophobia, the related harm to society, and what can be done to address the problem. The main cause of radiophobia is the invalid28,136 LNT hypothesis for harm to health from exposure to ionizing radiation or genotoxic chemicals. My use of the terminology “LNT hypothesis” rather than “LNT theory” is because there is no established scientific basis for the LNT concept in the risk assessment field. Using LNT as the null hypothesis (Ulsh155,156) allows for utilization of one LNT model formulation as well as for simultaneous use of multiple LNT model formulations in dose-response modeling.48,77,100,139 Use of LNT as the null hypothesis also allows linear interpolation between cancer risk associated with a single high dose of radiation and the corresponding risk for an assigned unexposed group (used for baseline risk assessment).
Neglected Genomic Stress Adaptation
Largely unrecognized is that no LNT model used for health risk assessment related to exposure to low radiation doses accounts for evolution-associated genomic stress adaptation (gensadaptation). This is a new terminology. Note that gensadaptation is progressive and may in some species be facilitated by genes located near each other on chromosomes evolving in a coordinated manner over many generations. 81
For mammals, genomic-stress-related damage to non-coding DNA is repaired less efficiently in slowly proliferating tissue than rapidly proliferating tissue, 108 which points to gensadaptation evolution occurring at different rates for different organs. As different organs age at different rates (related to rate of mutations accumulation), this suggests that organ aging rate may be linked to gensadaptation status.
An example of gensadaptation is the creation of free floating, ephemeral (i.e., short-lived) genes by some bacteria to counter genomic stresses via virus infection not managed via genetic instructions on the bacterial chromosome as discussed by Tang et al 145 More specifically, defense-related reverse transcriptase systems carryout DNA synthesis in order to prevent viral infection. Thus, there are protective genetic instructions not coming from the one-dimensional axis of the genomic DNA (on the chromosome) that are essential for bacterial cell survival when facing viral threats.
A second example of gensadaptation is the brooding brittle star (Amphipholis squamata), which has a genome that is much larger than that in other brittle star species. To increase its species survival probability, the brittle star undergoes a process called polyploidization. 60 With polyploidization a single chromosome is duplicated multiple times. Rather than evolving into different species over time, lineages readily hybridize with each other, leading to large genetic diversity, facilitating the particular species surviving genomic stresses over many generations. 60
A third example of gensadaptation is the bacterium Deinococcus radiodurans (D. radiodurans), which has the ability to survive more than 5000 times the radiation dose that would destroy a regular human cell.30,144 This remarkable survival ability relates to what I call super DNA damage repair. The tolerance to severe genomic stress relates to the robust antioxidant systems that protect the highly efficient DNA repair mechanisms (gifts of evolution) found in the Deinococcus species. DNA damage repair protein C (DdrC) has been found to help in the repair process via sensing and stabilizing DNA breaks through a novel lesion-recognition mechanism. 144 Szabla et al 144 found that the DdrC homodimer (two polypeptide chains identical in the number, order, and kind of amino acid residues) detects DNA strand breaks and binds to two single-strand or double-strand breaks. The resultant immobilization of DNA break pairs leads to circularization of linear DNA and the compaction of nicked DNA, aiding survival of the bacterium. 144
Note that gensadaptation helps to explain humans and other mammals existing today. Indeed, evolution has led to adaptation 143 to genomic stresses posed by radiation and other environmental carcinogens so that the body’s multiple natural defenses (cancer barriers) today efficiently protect from low-dose radiation harm 89 and this protection is enhanced via intercellular signaling stimulated by low radiation doses in a nonlinear dose-dependent manner. 136 Thanks to prior gensadaptation over many prior generations, no pathological or heritable effects were observed in the offspring of mice drinking water containing 100 Bq/ml 137Cs over 25 subsequent generations. 89
Radiation Hormesis
As might be expected, radiation levels well above natural background radiation (e.g., from very high dose radiotherapy) suppress the body’s natural defenses. Low-dose-radiation enhancement of natural defenses and high-dose-radiation suppression of these defenses is a form of radiation hormesis.17-20,35,79,90,123,125
Note that radiation hormesis is not a biological mechanism, but rather an outcome of different protective biological mechanisms that evolution has provided. Long ago when mammals first appeared on our planet, the biological mechanisms associated with radiation hormesis were likely more primitive than today. Far into the future, the mechanisms are likely to be even more sophisticated than today, if planet Earth continues to support abundant life.
Indeed, for humans and other mammals today, there are multiple natural defense mechanisms against cancer (missing from the LNT concept as applied to cancer induction) that include the following39,40,89,123,125,136: (1) epigenetically-regulated DNA repair and antioxidant production (protects from oxidative damage); (2) selective removal via apoptosis (p53-independent) of aberrant cells (e.g. transformed cells); (3) inflammation suppression (reduces cancer risk); and (4) anticancer immunity (destroys cancer cells). These natural barriers to cancer are enhanced by radiation doses that are modestly above natural background radiation levels today at the surface of Earth. Natural background radiation may play an important role in maintaining the baseline level of one or more cancer barriers,
16
and below natural background radiation level studies in progress88,101 may shed light on this possibility. The following quote is relevant here: “We hypothesize that natural background radiation is essential for life and maintains genomic stability and that prolonged exposure to sub-background environments will be detrimental to biological systems” (Pirkkanen et al
101
).
For cancer absolute risk (AR) characterization in epidemiological studies, the baseline risk (>0) relates to different causes of cancer. Only added risk above the baseline risk is attributed to above background radiation exposure. With LNT used as the null hypothesis,155,156 epidemiologists are permitted to assign increased risk of harm for any radiation dose, no matter how small. However, because of natural defenses to environmental carcinogens that have evolved, the AR can actually decrease at low doses (due to enhancement of natural defenses), return to the baseline risk at moderate doses (due to reduced enhancement in natural defenses), and then increase progressively as radiation dose increases more (due to natural defenses suppression). As already implied, such nonlinear radiation dose-response relationships are characterized as being hormetic.
The nonlinear hormetic dose-response relationships for cancer AR and relative risk (RR) are usually characterized by hormesis experts as being U-shaped or J-shaped. Note that when RR has a U shape, a function representing “1 – RR” has an inverse U shape (clarified in this paper).
A simulated hormetic AR dose-response relationship for cancer risk is presented in Figure 1 (upper J-shaped curve) and the assigned absorbed dose D of 0 mGy is for natural background radiation exposure along with exposure to other carcinogens. Thus, except for the assigned zero absorbed dose group, the radiation dose D (in mGy) is in excess of natural background radiation exposure. Simulated (not based on any data) hormetic dose-response relationship (upper curve) for cancer absolute risk, for a given cancer type (e.g., lung cancer) as a function of the absorbed radiation dose D. Here D = 0 mGy is assigned to natural background radiation exposure. Also shown (straight line with positive slope) is the corresponding simulated dose-response for radiation-caused cancers; a threshold-linear relationship with threshold absorbed dose Dt = 100 mGy is used here, but the dose-response for radiation-caused cancer may actually be nonlinear (e.g., threshold-sigmoidal) for doses below the population threshold for lethal deterministic effects.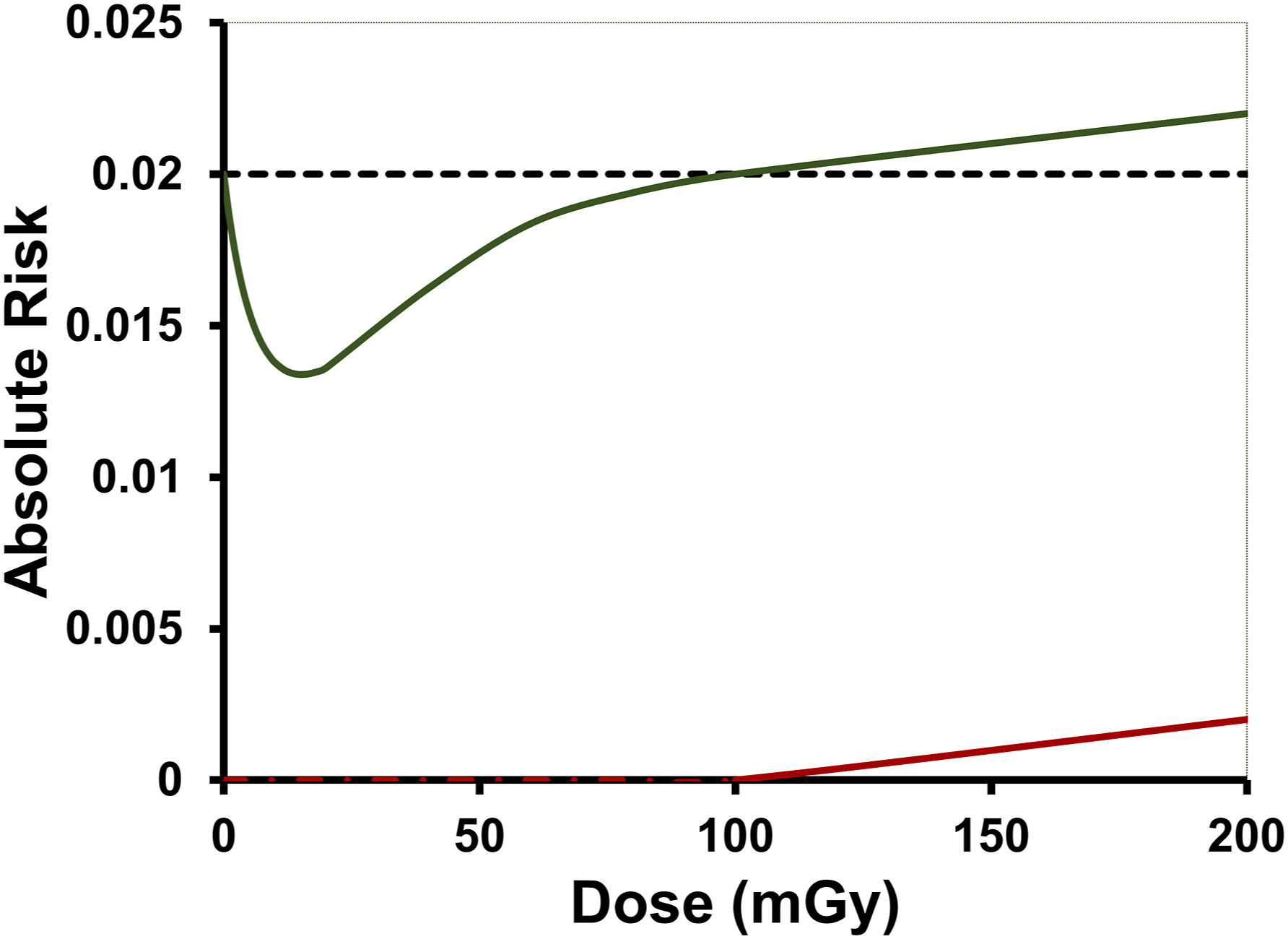
The AR in Figure 1 associated with the simulated hormetic dose-response relationship relates to all causes of the type of cancer of interest, not just radiation exposure. Also shown (lower threshold-linear response) is a simulated possible corresponding AR dose-response relationship for cancer induction by radiation only. Note that the baseline AR (for D = 0 mGy) for the hormetic dose-response curve is not considered to apply to radiation-caused cancer. For radiation-caused cancer, the AR starts at zero. Thus, in this hypothetical example, there are no radiation-caused cancers for low radiation doses, but some cancers that would normally occur (e.g., smoking related lung cancer), do not occur due to natural defenses being enhanced by low radiation doses. Unfortunately, it is difficult to see such hormetic dose-response relationships in epidemiological studies due to the way the studies are designed to adjust for competing risk factors, e.g., adjusting for smoking when evaluating lung cancer risk. 132
Cancer Relative Risk Evaluation in Low-Dose Epidemiological Studies
Epidemiological studies generally focus on cancer occurrence RR and excess relative risk (ERR), as a function of the level of radiation exposure. Note that there are no biomarkers for radiation-caused cancers, even though there are biomarkers for cancer occurrence. 38 Inference about the risk of radiation-caused cancer must therefore be based on RR > 1, i.e., using ERR > 0 not ERR < 0. In looking for evidence for radiation-caused cancer I therefore have focused on RR > 1 and ERR > 0.
The indicated approach therefore allows for focusing only on radiation-caused harm (illustrated with lower line in Figure 1), rather than both radiation benefits and harm (upper J-shaped curve in Figure 1) at the same time. In my opinion focusing on the upper curve would be a big mistake when interested in limiting radiation exposure. For the hormetic doses zone, one can focus on health benefits of low radiation doses only (i.e., RR < 1), which is relevant for disease prevention and disease therapy. For this zone, there is no evidence for radiation caused cancer, since RR < 1. The shape of the dose-response relationship for radiation-caused cancer needs to be established by new radiobiological research.
In this paper I initially address radiation-caused harm (cancer related) to humans, rather than radiation health benefits. Thus, ERR dose-response relationships are used, but adjusted in such a way (e.g., addressing key uncertainties) so as to focus on evidence-based, radiation-caused harm. Some animal (mice) studies data for low-dose gamma rays greatly elevating the body’s natural defenses against cancer leading to decreased cancer risk are also discussed. The data (for thousands of mice) support a population threshold dose for low-LET or high-LET radiation-caused cancer. The possibility of using low-dose radiation for cancer and other disease therapy is also discussed.
Methods
Natural Background Radiation Sources Investigation
Peer-reviewed publications on natural background ionizing radiation sources were relied on. This includes literature related to both external (local and from space) and internal radiation sources.
Radiation Dose Characterization
Key publications related to radiation dose characterization were relied on. This includes publications related to the absorbed dose D, equivalent dose HT, and the effective dose E. All doses D used are in excess of that associated with natural background radiation exposure. Thus D = 0 (mGy or related unit) corresponds to exposure to natural background radiation. For below natural background radiation studies considerations, D < 0 (mGy or related unit).
Unfortunately, values assigned for wR are influenced by RBE of the given radiation type, 63 which differs for different endpoints (e.g., DNA damage, cell survival, neoplastic transformation, cancer induction, etc.), as well as can differ for different radiation doses. Because of uncertainty about what value to assign for wR for a given radiation type, in International Commission on Radiological Protection (ICRP) Publication 92 63 values appear to be assigned based on best scientific judgement. The following warning was provided 63 related to their use: “w R is designed for the practice of radiological protection, not for specific risk assessment. Even the RBE values from experimental systems have limited applicability to risk assessment.” Interestingly, among the scientific community that uses wR in their publications where HT is employed, there appears to be little knowledge of the indicated ICRP warning as equivalent dose is used sometimes in health risk assessment.
There is another widely used dose type called effective dose E. This dose is derived from equivalent doses via applying a second weighting factor called the tissue weighting factor w T . This factor reflects organ and tissue susceptibility. E is currently expressed in the same units as HT (e.g., sievert), which is quite confusing to the public as well as some scientists. Also, since E depends on HT which depends on wR, effective dose E also should not be used in radiation health risk assessment. However, this constraint is often not known (or is ignored) by many who use E in cancer risk assessment.
New Doses Used
A new dose with no units called relative dose (RD) is introduced. RD = 1 represents the population threshold for radiation-caused cancer and applies to all ionizing radiation types and all combinations of different ionizing radiation types. The corresponding absorbed dose threshold is represented by Dt and the corresponding equivalent dose threshold is HT,t. Note that like Dt and HT,t, RD is tissue or organ specific. RBE evaluated based on Dt for different radiation types relative to gamma rays is indicated here by RBEt and may differ from RBE based on a different criterion.
A value RD = 0.5 represents ½ of the population absorbed dose threshold Dt for a specific radiation type, and also ½ of the population equivalent dose threshold HT,t for mixed radiations, and ½ of the RBEt -weighted absorbed dose threshold Dt,rbe for mixed radiations with RBEt evaluated based on population thresholds relative to a reference radiation type (gamma rays assumed here). Note that RD = RDt = 1 corresponds to Dt, and to HT,t, and to Dt,rbe.
One can easily go from RD to the corresponding D, or corresponding HT, facilitating limiting radiation exposures based on threshold dose-response relationships, while not abandoning use of HT. However, and quite important, radiation weighting factors for high-LET radiation types may need to be altered somewhat for application to population thresholds, to be scientifically credible for use in limiting radiation exposure. Revised values for wR could be assigned based on values for RBEt, which may differ significantly from current values for wR.
Small mammals (e.g., mice) studies data could be used for evaluating RBEt, as values would be expected to be independent of species. For accurate assessment, large sample sizes will likely need to be used. In vitro neoplastic transformation studies could possibly also be used for evaluating RBEt.
Another new dose, the exceedance absorbed dose ∆D = D − Dt, is also introduced for cancer risk assessment for threshold dose-response relationships. For D < Dt, ∆D = 0 mGy (or a corresponding dose unit, e.g., Gy), and there is no risk of radiation-caused cancer of the type of interest.
RBE weighting of ∆D for different radiation types contributing to ∆D leads to the new exceedance RBE-weighted dose ∆Drbe, use of which could help in countering radiophobia, as ∆Drbe = 0 mGy is likely for many low-dose radiation exposure scenarios.
Dose-Response Function Characterization
Relevant publications that focused on dose-response function characterization for cancer absolute risk AR(D) and relative risk RR(D) as a function of D were relied on, as well as publications related to radiation exposure limitation. For risk assessment related to jointly inducing cancers at different body sites, the single dose D is replaced by the multiple doses D1, D2, …, Dm, for the m different sites of interest within the body.
Microsoft Excell was used for dose-response function graphing of cancer risk vs radiation doses D, ∆D, ∆Drbe, and RD. Cancer risk distribution percentiles 2.5 % (percentile) and 97.5 % (percentile) were evaluated and used along with data means and linear interpolation, related to dose-response function characterization.
For LNT dose responses for RR or ERR derived from results of an epidemiological study of all-solid-cancer mortality risk by Sposto and Cullings, 139 adjusting for missing risk uncertainty at low doses was based on a Poisson distribution of cases in the zero-dose group, since Poisson regression was employed by the researchers in their analyses. Standard Monte Carlo (MC) evaluations were used to implement the Poisson distribution analyses. The MC analyses were performed using WinBUGS 137 software.
Where hormetic RR dose-response data were analyzed based on a study of Ullrich et al 154 using RFMf/Un mice, my hormetic model122,129 for RR was used. Standard MC via WinBUGS 137 was used to characterize the RR dose-response uncertainty, based on a binomial distribution of cases.
Results
The subsections here relate to the following: (a) the natural sources of ionizing radiation we are all exposed to throughout life, but the public is not inform about; (b) uses of equivalent and effective dose; (c) how LNT-related radiophobia is linked to the SRP; (d) how LNT-hypothesis-related health detriment risk is evaluated within the SRP; (e) major problems with misinforming epidemiological studies of cancer risk that inappropriately use LNT as the null hypothesis; (f) hormetic dose-response relationships at low doses; (g) use of the new relative dose (RD); (h) severing the link between SRP and LNT; (i) new health protection principles; (j) dose rate influences on population thresholds; (k) threshold-linked relative biological effectiveness for mixed radiations.
Natural Background Radiation Sources
Radiation from far away places
“The Earth is constantly bombarded by primary cosmic rays (CRs), which originate either from objects, in our own galaxy (and sometimes in distant outer galaxies) or from the solar wind” (Hubert et al.
59
)
The above quote of Hubert et al 59 helps introduce this subsection as our planet is constantly exposed to cosmic rays, which originate either from objects in our own galaxy (and sometimes in other distant galaxies 92 ) or from what is called the solar wind. Hubert and colleagues 59 point out that both primary and secondary cosmic rays interact with the atoms present in earth’s atmosphere. These interactions then lead to the production of additional cosmic-ray types. Particles included in these types are neutrons, protons, muons, and cosmogenic nuclides. 50 Note that a free neutron breaks down in a rather short time into a proton, an electron, and an antineutrino. 73 Thus, here on Earth, each of us is being exposed to external natural radiation sources from space, but presently at doses likely too small to be harmful, thankful in part to the body’s natural defenses derived from gensadaptation over many generations. In fact, life evolved in a background of significant levels of natural ionizing radiation, 151 and it is questionable whether this life as we know it today, could actually exist in the absence of natural ionizing radiation exposures during evolution. 101
Supernova remnants are a source of some cosmic rays.6,9,55 However, some cosmic rays have much higher energies than supernova remnants are considered to generate, and where the ultra-high energy rays come from is presently unresolved.
Radiation from our sun
Our Sun is known to also produce very energetic particles which contribute to solar cosmic rays. According to Hubert et al, 59 solar cosmic rays are accelerated by magnetic reconnection during solar flares or by shock waves associated with coronal mass ejections (CMEs). When these solar energetic particles (SEPs) have sufficient energy to cause an increase in secondary cosmic rays at ground level here, they are referred to as ground-level enhancements (GLEs). 59 In fact, 73 GLE events were detected from February 1942 to October 2021. 51
A somewhat recent GLE (indicated as GLE73) occurred on 28 October 2021 and was also detected on our moon and on Mars with ground detectors previously placed there. GLE73 is the first GLE event for what is called Solar cycle 25. 51 The solar cycle represents the cycle that the magnetic field of the Sun goes through about every 11 years.
Teraelectronvolt (TeV; one thousand trillion eV) gamma rays are now known to occasionally come from our sun.1,29 The TeV gamma rays were detected at the High-Altitude Water Cherenkov Gamma-Ray Observatory, located on the flanks of the Sierra Negra volcano near Puebla, Mexico. The observatory was designed to detect gamma and other cosmic rays with energies between 100 GeV and 100 TeV. 1 For such high energy gamma rays, special considerations are needed related to assigning radiation doses. 85
Other external radiation sources
The Van Allen radiation belts located above our planet have trapped energetic charged particles that are mainly electrons and protons.96,104 Most of the trapped charged particles originate from the solar wind. The indicated source of natural radiation exposure is relevant for space exploration by astronauts.
What many around the world are not aware of is that high-energy ionizing radiation is now known to be associated with thunderstorms.23,95,102,140,160 Terrestrial gamma-ray flashes (TGFs), which are short (less than 1 millisecond) bright bursts of gamma rays produced inside thunderstorms, are quite powerful and sometimes temporally blind gamma-ray instruments in low-Earth orbit above the storms. 95 As the TGF-related gamma rays travel through air, they produce beams of high energy electrons (and positrons) that are called terrestrial electron beams (TEBs).
Thunderstorm-associated gamma-ray glows also occur and are longer lasting (seconds to minutes) than TGFs but are not as bright. Thunderstorm-associated gamma ray flickers 93 also occur and are between TGFs and gamma-ray glows in terms of brightness and duration.
Going back in time to the Ediacaran Period more than 500 million years ago, Earth’s protective magnetic field was reduced compared to today 58 ; thus, the level of external ionizing radiation exposure of all early life forms at the surface of our planet was likely much higher than today, and this may have accelerated the evolution of radiation tolerance by many lifeforms.
Everyone is naturally radioactive
So far only external natural radiation sources have been discussed. However, there are naturally occurring radioisotopes on our planet and in our bodies that originated during the formation of the solar system and through the interaction of cosmic rays with molecules in the atmosphere. In fact, everyone is naturally radioactive because we eat naturally radioactive foods, drink naturally radioactive liquids, and inhale naturally radioactive air (e.g., a source of radioactive radon gas). Radioisotopes such as polonium-210, carbon-14 and potassium-40 are naturally present in harmless amounts in our bodies. Polonium-210 is found in tobacco. Carbon-14 is found in atmospheric carbon dioxide. Potassium-40 is found in many foods.
Radionuclide effective half-life
The amount of time a given radionuclide remains in the body depends on both its physical half-life and also the biological half-life. 103 The biological half-life is the time needed in order to remove half of the body burden of the radionuclide via excretion and metabolic turnover. Thus, the radiation dose to a tissue/organ from a radionuclide inside the body depends on both the biological and physical half-lives of the radionuclide.
The effective half-life (Te) accounts for both the physical (Tp) and biological (Tb) half-life. It can be evaluated based on the following inverse relationship:
Use of Equivalent and Effective Doses
Assigned Tissue Weighting Factors Related to Radiation Effective Dose Evaluation, Based on ICRP Publication 103. 64

aRemaining tissue: heart, kidneys, adrenals, extra thoracic region, gall bladder, lymphatic nodes, oral mucosa, muscle, pancreas, prostate (males), small intestines, thymus, spleen, and uterus/cervix (females).
Unfortunately for society, calculated values for HT and E reported in publications including news media publications are viewed by the public as both real and reliable doses. However, neither are real doses but rather assigned doses, based on radiological protection principles that need revising in order not to unnecessarily be radiation phobia promoting.
As examples of use of E promoting radiation phobia, I point out its use related to annual exposure to natural background radiation sources. 158 The calculated annual effective dose for people depends on location on our planet. For Canada, the calculated value is stated as being about 2 mSv, and for the United States, it is stated as being about 3 mSv. 21 In our planet’s high natural background radiation areas, such as Ramsar (in Iran), Guarapari (in Brazil), Orissa and Kerala (in India), and Yangjiang (in China), calculated annual effective doses can be > 20 mSv 13 ; supposedly implying more harm (induced cancers) to the indicated populations than for populations with annual effective doses < 10 mSv. The implied more harm relates to effective dose E unfortunately being linked to the invalid66,113,136 LNT hypothesis as used for cancer as well as health detriment risk assessment.
The main use of E is in radiological protection applications, related to limiting planned exposure of humans to ionizing radiation (other than medical and environmental exposures). This appears to be acceptable because use of E (not a real dose) in radiation exposure limitation has helped in preventing harm from exposure to moderate and high real absorbed radiation doses D. Whether harm (e.g., lethal cancer induction) is associated with low and very low radiation doses is highly debated,41,66,91,136,152 although harm is implied by the SRP being linked to the LNT hypothesis as applied to health detriment risk assessment.
A Main Cause of Radiophobia
The following two quotes are relevant to this subsection: “Radiophobia does far more harm to human health than the radiation released by nuclear accidents. In some cases, the harm results from disaster response” (Ropeik
106
). “The harm that society expects from ionizing radiation does not match experience. Evidently there is some basic error in this assumption” (Allison
2
).
The main objective of the ICRP is to advance for public benefit the science of radiological protection. 110 This is to be achieved via providing recommendations and guidance on all aspects of protection against ionizing radiation harm, without unduly curtailing beneficial practices that relate to radiation exposure. 110 Unfortunately, the indicated “without unduly curtailing beneficial practices” has been unachieved due to radiophobia linked to use of LNT risk models for radiation-caused harm to health.
The ICRP-based, LNT-linked SRP evolved over many years4,12 and is likely to continue its evolution in the future. 52 The SRP is multiplex, cautious, and for too long has unintentionally been an impediment to scientific, medical (e.g., low-dose-radiation therapy), and industrial (e.g., climate-change-protecting nuclear power) progress. Radiophobia linked to the SRP has also led to the unnecessary loss of lives as well as harm to health and wasting of large amounts of financial and other resources related to the April 1986 Chernobyl nuclear power plant accident68,78,124,161 and to the March 2011 Fukushima nuclear power plant disaster.141,146,161
Chernobyl-accident-related studies supposedly revealed excess numbers of abortions of healthy human fetuses for Denmark, Finland, Italy, and Greece.78,97,99,138,149
LNT-Hypothesis-Related Health Detriment Risk
The current SRP is based on LNT-hypothesis-related health detriment risk rather than cancer risk alone.26,61,62,64,161 The SRP allows for assigning combined risks for different detrimental effects (cancer, heritable effects, life shortening), even though there is no evidence 150 of such health effects occurring after very low radiation doses (e.g., radiation exposures with assigned E < 10 mSv).
LNT Hypothesis and Effective-Dose Related, Health Detriment Risk Coefficients (10−2·Sv−1), Based on ICRP Publication 103. 64

aBased on information provided in this paper.
The statements in Table 3 that relate to radiation phobia harm caused by assigning health detriment risk based on risk coefficients in the table are my statements, not statements of the ICRP, which has the enormous task of developing an acceptable SRP for all. Although I along with many colleagues are quite concerned about the current SRP’s link to the LNT hypothesis, we respect those members of the ICRP who have worked very hard to develop the current version of the radiological protection system. Developing an SRP that is acceptable to the public and scientific community, and that is scientifically credible, is a major challenge. The ICRP consists of policy makers, practitioners, eminent scientists, and other experts in the field of radiological protection. Members of the Main Commission, committees, and task groups are volunteers, for which they should be thanked.
Figure 2 demonstrates why the LNT-linked risk coefficients in Table 3 related to health detriment risk promote radiation phobia (potentially harmful), even when members of a population are assigned an effective dose no larger than 1 mSv. What is plotted is the LNT-based fold increase in the number of cases of health detriment, as defined in ICRP 103,
64
compared to that assigned for the irradiated population when the effective dose is 0.001 mSv. Note that for E = 1 mSv, the assigned health detriment cases in an irradiated population attributed to radiation exposure is 1000 times larger than for an effective dose of 0.001 mSv. A dash line was used in Figure 2 to emphasize that the assigned fold increases for such very small radiation doses are not supported by modern science or real data.
136
ICRP-103
64
-based, fold increase in the number of people in an irradiated population that are assigned radiation-caused health detriment as a result of having been exposed to a low radiation dose compared to the number of cases attributed to an effective dose of 0.001 mSv. The ICRP-assigned 1000-fold increase in the harm count at 1 mSv (likely a harmless exposure)
150
clearly promotes radiophobia among the public.
The public being afraid (radiophobia) of an effective dose of 1 mSv is understandable, although there are no data 150 demonstrating health detriment from such a low-level radiation exposure.
Misinforming Epidemiological Studies That Use LNT Models
Recent and Somewhat Recent Studies Designed to Yield LNT Outcomes for Relative Risk for Cancer Induction (or Cancer Mortality) That Would Likely Change to Threshold-Linear (e.g., Reference. 132) for Significant Excess Relative Risk or Excess Relative Mortality When Adjusted for Missing Baseline Risk Uncertainty. a

aAll listed studies employed the low-dose vanishing uncertainty stratagem (VUS), via use of LNT as the null hypothesis.
All of the studies in Table 4 were essentially designed to yield LNT outcomes, as LNT was the implied null hypothesis. 156 Quite important, none of the studies addressed uncertainty in the baseline risk so that when RR was evaluated, no uncertainty was assigned to RR = 1 for the baseline group, which is problematic for risk assessment at low doses. 132 This means that with adjustment for the indicated missing uncertainty, the LNT dose-response relationships derived would be expected to transition to threshold linear dose-response relationships, thereby being more in line with modern radiobiology as it relates to radiation-caused harm to health. 132 Based on modern radiobiology, radiation protective processes (thanks to evolution-associated gensadaptation) are enhanced 136 by low radiation doses. Some data strongly supporting this view from a large cancer risk assessment study using mice is presented in this paper.
Given this, why does the ICRP still support the link of the SRP to misinforming epidemiological-studies-based cancer risk modeling that is based on the LNT hypothesis? The answer is revealed in the following quote: “From a pragmatic point of view, no other dose-risk relationship seems more suitable or justifiable for radiological protection purposes” (Laurier et al 76 ). Note that the reason for the link is not that the LNT hypothesis is supported by modern science.
Epidemiological study on solid cancer among a-bomb survivors
I now focus on findings from a study of Sposto and Cullings 139 that simultaneously applied several LNT models in their analysis of aggregate endpoints (multiple causes of death) in a Radiation Effects Research Foundation (RERF) cohort cancer mortality study. The study relates to A-bomb survivors (exposed to gamma rays and neutrons) and when missing risk uncertainty at low doses is addressed, an LNT relationship for ERR transitions into a threshold-linear relationship.
The LNT-based study design of Sposto and Cullings 139 essentially guaranteed that the dose-response relationships generated overestimated cancer mortality risk at low doses, though apparently not realized by the researchers. I make use of this overestimation in deriving upper bound ERR vs dose relationships (threshold-linear, after adjusting for missing uncertainty) that could help in deriving new radiation exposure limits that are less likely to promote radiophobia.
Sposto and Cullings 139 in evaluating all-solid-cancer mortality risk used Poisson regression, linked to multiple LNT RR models for cancer induction. Their analyses were based on assigned surrogate radiation doses (neutron RBE = 10 used for weighting) and the following two forms of mortality risk assessment: “usual aggregate endpoint analysis” with assigned dose not organ-specific and “joint analysis” with the same assigned dose or different assigned doses for each organ. The RBE weighted doses were reported in grays, so I have also used grays or milligrays, and this is not a major issue due to the small neutron contribution to dose for A-bomb survivors. As already indicated, the stated forms of analysis were applied to RERF cohort studies data, 94 with the results obtained compared. My comments relate specifically to the risk assessments performed related to RERF data using five different LNT model formulations (i.e., five different LNT models), which is allowed 100 under the LNT hypothesis for cancer induction, and also for jointly assessing cancer and heritable effects risks. 63
With the usual aggregate endpoint analysis 139 the deaths from different causes were simply added, which mathematically allows a person to die more than once from different causes (e.g., lung cancer, liver cancer, etc.) and allows assigning the same radiation dose (surrogate dose) to all organs. Different causes of death are competing risks. When modeling different causes of death, one should adjust for the different competing risks. This was not recognized by the researchers as being important, so that the mathematics used is the same as if allowing for a person to die more than once from different causes. Note that without adjusting for competing risks, the risk estimates generated could be considered upper bounds and useful for that purpose. Here I make use of this benefit.
Note that using LNT dose response as the null hypothesis (as implicated by using only LNT model formulations) is a form of LNT model idolization. 128 There is now abundant evidence that the LNT hypothesis for cancer induction by ionizing radiation is invalid; the hypothesis has no links to modern radiobiology, radiation physics, and radiation chemistry.28,41,54,66,136,150
Use of multivariate hazard function for risk characterization
With the joint analysis used by Sposto and Cullings,
139
based on the LNT hypothesis for J >1 modes of cancer death, the AR for death from cancer can unscientifically take on values much larger than 1 for high radiation doses (actually a problem with all LNT models that are based on AR). However, this AR problem does not occur when relying on a multivariate hazard function HFLNT(D1, D2, …, Dm), where “D1, D2, …, Dm” are the radiation doses to the m different sites in the body of interest and HFLNT(D1, D2, …, Dm) increases linearly as a given single dose Di (for i = 1, 2, …, m) increases. The multivariate AR for only radiation-caused cancers can be evaluated as follows:
Sposto and Cullings 139 in their analyses focused on here, used a single assigned dose, called the surrogate dose (represented here as DS) and expressed in gray, to be representative of the different radiation doses of interest. Thus, AR(D1, D2, …, Dm) is essentially replaced with AR(DS), RR(D1, D2, …, Dm) is essentially replaced with RR(DS), and ERR(D1, D2, …, Dm) is essentially replaced with ERR(DS). The LNT hypothesis as it applies to cancer induction and cancer mortality allows for such replacements in epidemiological studies, since each dose Di can be replaced with “ai × DS” where “ai = Di/Ds”, making DS the single independent variable.
Dose-response relationships linked to vanishing uncertainty stratagem
Figure 3 shows the assigned RR(DS) vs assigned surrogate dose DS for all-solid-cancer mortality (radiation-caused) for the A-bomb survivors based on findings of Sposto and Culling,
139
for mainly gamma-ray exposure. The dose is an RBE weighted dose expressed in milligray and all deaths from solid cancer irrespective of the type were included. The uncertainty presented represents the assigned 95% CI for the LNT dose-response relationship. Note that as DS decreases, so does the assigned risk uncertainty, so that at a 1 mGy dose essentially no uncertainty is calculated to remain and with certainty a risk increase is assigned, but not proven. I refer to the vanishing of risk uncertainty at low doses as the vanishing uncertainty stratagem (VUS) used in many epidemiological studies linked to LNT. The assigned relative risk RR(DS) for all-solid-cancer mortality, for A-bomb survivors, as a function of the assigned surrogate dose DS, based on RR (per unit surrogate dose) of 0.000232 mGy−1 (S.E., 0.0000454 mGy−1).
139
The assigned surrogate dose is a weighted dose in Gy (based on RBE = 10 for neutrons).
Figure 4 shows a plot of the assigned uncertainty size (95% CI width) US95(DS) vs assigned DS, for RR(DS), for all-solid-cancer mortality for A-bomb survivors, based on the LNT response slope parameter of 0.000232 mGy−1 (S.E., 0.0000454 mGy−1), which is based on Table 2 of Sposto and Cullings.
139
Figure 4 applies to all of their results (usual-aggregate-endpoint as well as three different joint analyses) obtained using the assigned weighted surrogate colon doses. Note again that the assigned uncertainty size US95(DS) is progressively and improperly reduced, as the assigned dose decreases, so that there is essentially no uncertainty about a supposed RR(DS) increase, even for very small surrogate radiation doses DS. However, the uncertainty should not approach zero at very low doses, as the AR(D) estimates for both the numerator and denominator for RR(DS) evaluation have >0 uncertainties.
132
Addressing uncertainty propagation for RR(DS) estimation for low and very low radiation doses is quite important, because when doing so, a possible zero or negative slope for the dose-response relationship for RR(DS) is likely to not be ruled out.54,56,113,136 The assigned uncertainty size (95% CI width) US95(DS) in Figure 3, for the excess relative risk ERR(DS) estimate, for all-solid-cancer mortality, for A-bomb survivors, as a function of the assigned surrogate dose DS, based on an ERR per unit surrogate dose
139
of 0.000232 mGy−1 (S.E., 0.0000454 mGy−1). A dashed line is used to emphasize that key uncertainty is missing for assigned doses <100 mGy.
The importance of the missing uncertainty in Figure 3, for low doses, is illustrated in Figure 5, which presents estimates of the dose-dependent, uncertainty size underestimation factor (USUF(DS)) that I obtained for assigned surrogate radiation doses DS up to 100 mGy. The results are based on the 95% CI (0.97, 1.03) for the estimate RR(DS) = 1, for the zero surrogate dose group, obtained from 20,000 MC realizations (MC error < 0.001). The MC results were obtained, assuming a Poisson distribution of deaths, evaluated based on the reported 7507 solid cancer deaths among 51,707 individuals used by Brenner et al
14
for the unexposed group in their study of A-bomb survivors. A Poisson distribution was employed to be consistent with Poisson regression use by Sposto and Cullings.
139
Similar results would be expected had a binomial rather than a Poisson distribution been used in the MC analysis. The calculated dose-dependent uncertainty size underestimation factor, USUF(DS), for results in Figure 4, as a function of the assigned surrogate dose DS.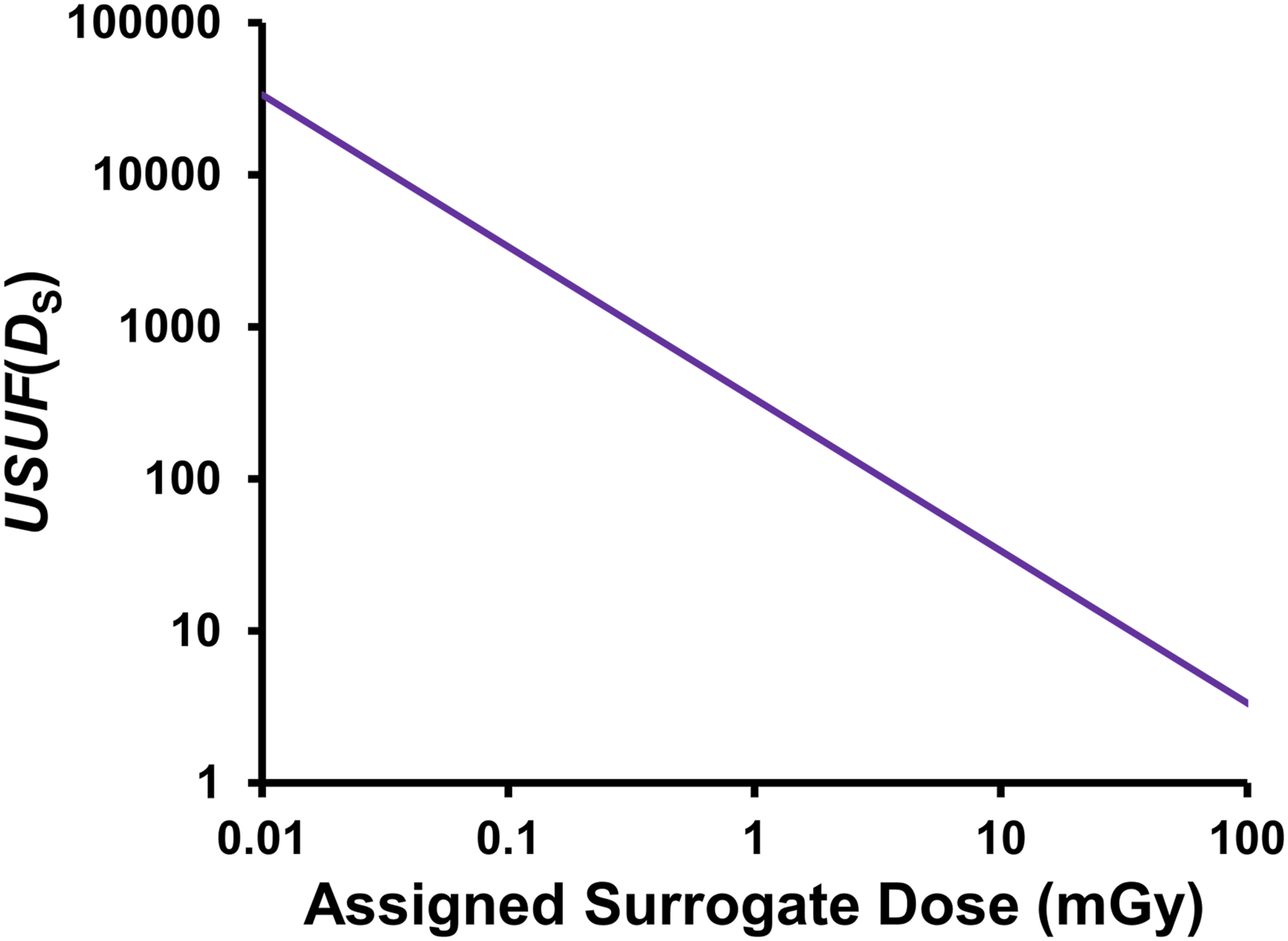
Accounting for missing uncertainty for RR(DS) = 1
The missing uncertainty (%) in Figure 3 can be calculated as [1 – USUF(DS)−1] × 100%. For an assigned surrogate dose of 0.01 mGy, USUF(DS) = 33,708, thus the percentage of the uncertainty that is missing is [1 − (1/33708)] × 100% = 99.997%, rounded! When the missing 99.997% of the uncertainty is addressed (as elsewhere
132
) there is no evidence for an RR(DS) > 0 for assigned surrogate doses DS < 93 mGy (see Figure 6). Excess relative risk ERR(DS), adjusted for uncertainty in RR(DS) = 1, for all-solid-cancer mortality, for A-bomb survivors, as a function of the assigned surrogate dose DS; based on an LNT-associated ERR per unit dose
139
of 0.000232 mGy−1 (S.E., 0.0000454 mGy−1).
The results in Figure 6 based on the study by Sposto and Cullings
139
were used to evaluate and express excess relative risk as a function of the exceedance surrogate dose and results obtained are presented in Figure 7. Note that the value for the threshold dose estimate used for the exceedance dose assignment differs for the three lines plotted. Note also that by adding 1 to the excess relative risk in Figure 7 you get the same numerical results as in Figure 3 (no threshold result) for the relative risk dose-response relationship. This means that for all previous epidemiological studies (e.g., studies in Table 4) where LNT dose-responses have been used for relative risk and excess relative risk without adjusting for uncertainty in RR = 1 for the assigned zero-dose group, one should replace the word “dose” with the words “exceedance dose” on the dose axis; even though the value for Dt (likely > 0 mGy) is not yet determined. In this case, the LNT dose-response relationship transitions to a threshold dose-response relationship. Excess relative risk for all-solid-cancer mortality in Figure 6 based on Sposto and Cullings,
139
replotted as a function of the exceedance surrogate dose (RBE weighed): (A) assigned upper bound risk, based on exceedance surrogate dose ∆D
rbe
= D
rbe
− 93 mGy; (B) central risk estimate, based on ∆D
rbe
= D
rbe
− 129 mGy; (C) assigned lower bound risk, based on ∆D
rbe
= D
rbe
− 210 mGy. Negative values for ∆D
rbe
were set to 0 mGy in all cases.
Note that the use of exceedance doses ∆D and ∆Drbe in Bayesian analysis44,98,137 of dose-response relationships for radiation-caused cancer or cancer mortality should facilitate assigning distributions (i.e., posterior distributions) to thresholds Dt and Drbe,t.
Model uncertainty, dose uncertainty, and missing dose from fallout
Sposto and Cullings 139 did not address model uncertainty (i.e., uncertainty about the correct model to use). The fact that five different LNT model formulations were used, while nonlinear and threshold-linear models were not considered, points to the need to formally address model uncertainty, which can be addressed using Bayesian44,98 and Edisonian 162 analyses. This includes Bayesian semiparametric modeling. 44 With these analyses, different dose-response models can be compared for their performance in explaining the dose-response data used for risk assessment.98,162 With the indicated approaches, the threshold dose Dt can be treated as a free parameter and estimated, along with uncertainty being assigned based on the posterior distribution generated.
Regarding the use of the usual-aggregate-endpoint and joint analysis approaches, Sposto and Cullings 139 state: “… for neither approach is inference about dose response well defined.” The quote is quite important, especially since uncertainty related to the estimate RR(DS) = 1, model uncertainty, and radiation dose uncertainty were not formally addressed. For A-bomb survivors, there is also an issue related to the missing dose from fallout radionuclides as revealed by Sutou, 142 which points to assigned doses as being too small.
Regarding radiation dose uncertainty not being addressed by Sposto and Cullings,
139
the following quote is quite relevant: “Epidemiological studies of stochastic radiation health effects such as cancer, meant to estimate risks of the adverse effects as a function of radiation dose, depend largely on estimates of the radiation doses received by the exposed group under study. Those estimates are based on dosimetry that always has uncertainty, which often can be quite substantial” (Bellamy et al.
5
).
Hormetic Dose-Response Relationships for Doses < Dt
I now return to the simulated hormetic curve in Figure 1, which reflects my view on possible health benefits (cancer prevention and/or elimination) from low radiation doses. For the dose range 0 ≤ D ≤ Dt, AR(D) for cancer occurrence after radiation exposure can be evaluated as follows.
122


DPF(D) is a result of gensadaptation-related multiple protective processes and is a hormetic function (upside-down U shape in this case) of radiation dose (i.e., reflecting low-dose enhancement of protective processes to varying degrees depending on the dose). The upside-down U (i.e., inverted U) shape is due to RR(D) having a U shape. Note that high dose suppression of protective processes is not relevant for the dose range 0 (natural background radiation only) to Dt; only the differential enhancement of the protective processes is relevant. Quite important, DPF(D) applies to a population rather than to an individual and takes on the value 0 for D > Dt. Note also that for D = 0 (e.g., mGy), protective processes are at baseline, not absent, thanks to progressive gensadaptation over many earlier generations. This has implications for below natural background radiobiological studies where D < 0 (e.g., mGy) is now allowed, which will be important in LNT model predictions testing.
As the immune system is part of the protective processes against cancer and its functioning changes with age, DPF(D) likely varies for different age distributions for different irradiated populations. Importantly, the dose range for which DPF(D) > 0 applies likely depends on the age makeup of the irradiated population, the radiation type (e.g., low-LET, high-LET), endpoint considered and exposure scenario (e.g., brief exposure at high rates, chronic exposure at low rates).
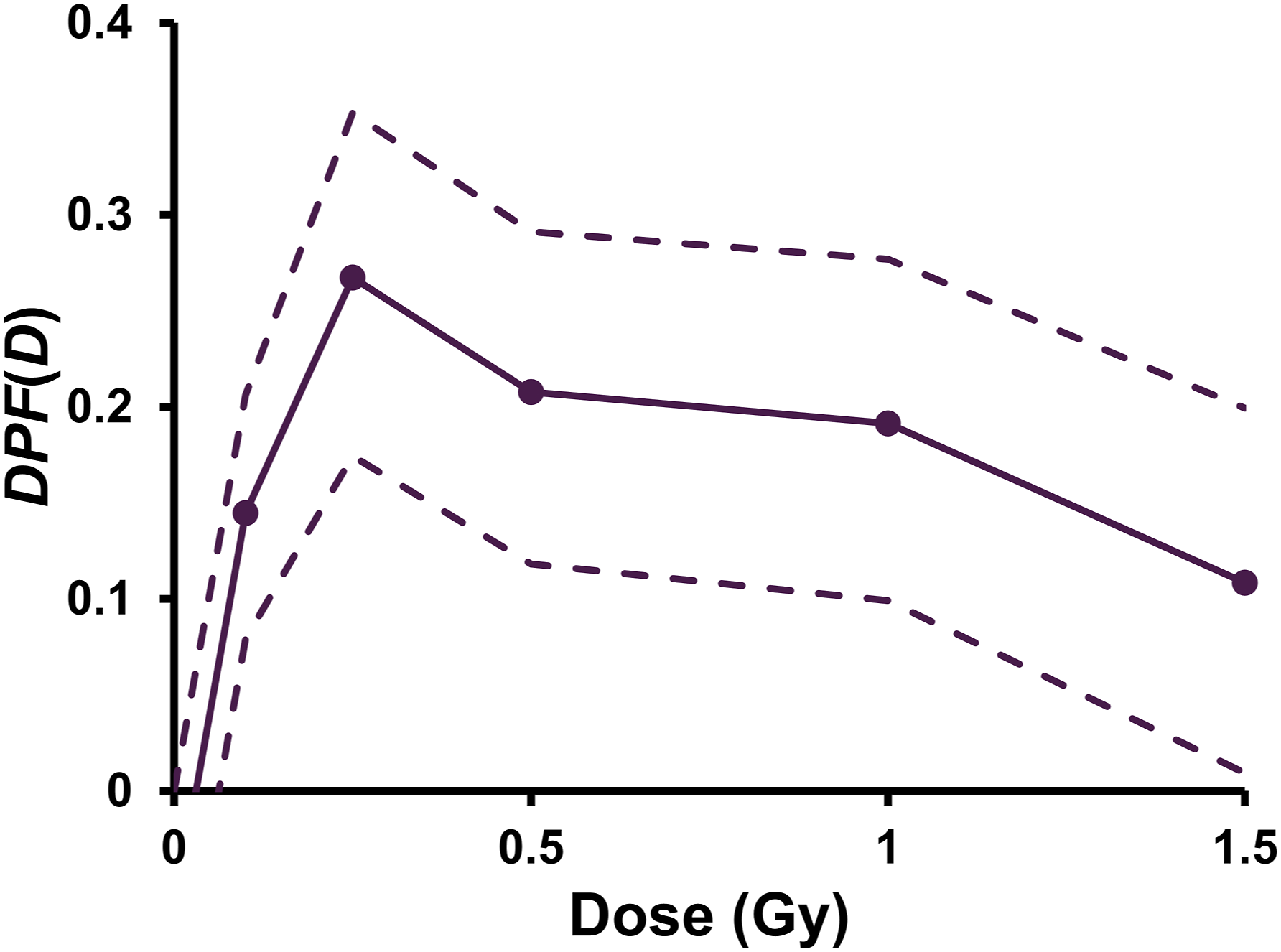
Simulated dose-response relationship for DPF(D)
Figure 8 shows a simulated dose-response for DPF(D), based on the simulated hormetic dose-response in Figure 1 for AR(D) for cancer. Note that the dose-response shape is the inverse of that in Figure 1 for doses in the hormetic zone where AR(D) is below the baseline level. With DPF(D) taking on values > 0, it becomes possible for disease prevention (e.g., cancer prevention) and disease therapy (e.g., cancer therapy) using essentially harmless low radiation doses or somewhat higher doses, as is being explored46,71 by others. Note that for doses in the hormetic zone where DPF(D) > 0, when based on real data, there would be no evidence of harm from radiation exposure since RR(D) < 1. Also, for RR(D) > 1, there is no evidence of health benefits of radiation exposure. This is why DPF(D) = 0 is used for D > Dt. Simulated (not based on any data) disease prevention function DPF(D) for preventing or eliminating cancer of a given type. Note that were the simulation based on actual data, there would be no evidence of radiation caused harm for the indicated dose range.
DPF(D) is characterized as the product of a population-specific benefit function B(D) (where 0 ≤ B(D) ≤ 1), which represents the dose-dependent probability of the body’s natural defenses being enhanced, and a population-specific, dose-independent protection factor PROFAC (where 0 ≤ PROFAC ≤ 1) representing the probability that the enhanced natural defenses are successful in preventing cancer occurrence or eliminating an already present cancer. Thus, the following equation122,129 applies:
DPF(D) therefore accounts for disease prevention occurring in some, but not necessarily all, when the body’s natural defenses are enhanced by a low radiation dose < Dt. In the case of an already existing cancer, the enhancement of natural defenses could eliminate54,56,122 the existing cancer as already pointed out, and this would be reflected by the estimate of DPF(D) obtained from cancer RR analysis. However, both cancer prevention and elimination are stochastic outcomes. Natural defenses that are enhanced by low radiation doses are reviewed in detail elsewhere.54,119,123,128,136
Population relative risk RRp and disease prevention function DPFp
Central Estimates of the Population DPFp for Cancer Prevention via Radiation Doses in the Hormetic Zone for Four Populations.

New research by others (e.g., a graduate student) is needed related to generating modern science based mathematical relationships for RR(D) and DPF(D) for the populations in Table 6 as well as other populations. A useful starting point may be mathematical relationships presented in Bugała and Fornalski. 16 However, the use of E or HT as the independent variable should be avoided.
Regarding the three worker populations in Table 6, where exposure to low radiation doses is associated with their profession, some have claimed what is called the healthy worker effect25,114,126 being responsible for RRp < 1 (note DPFp > 0). Related to the healthy worker effect, supposedly the workers are less susceptible to cancer induction than the average person because they are assumed healthier than the average person. However, according to Fornalski and Dobrzyński, 42 this explanation for results such as in Table 6 has no credibility. Note that there is no evidence for their DNA damage repair being more efficient than for the average person nor is there evidence for their elimination of aberrant cells via apoptosis being more efficient than for the average person.
Use of Relative Dose, RD = D/Dt
Use of unitless dose call normalized dose was demonstrated to be a reliable approach to lethality risk assessment for threshold radiation deterministic effects (e.g., hematopoietic syndrome lethality), where combined exposure to high- and low-LET radiations related to nuclear accidents are involved and risk of harm dose-response relationships were sigmoidal (i.e., S-shaped), with population thresholds.116,117,134 With lethal deterministic effects risk modeling, a normalized dose of 1 corresponds to a median lethal (i.e., lethal for 50% of the population) radiation absorbed dose. For high dose rates and acute lethality via a given mode (e.g., hematopoietic syndrome), normalized dose is evaluated as the ratio “D / LD50”, where LD50 (also indicated with the more general notation D50) is the population median lethal absorbed dose to an organ.
For the stochastic effect considered here (cancer induction), RD is used rather than normalized dose. Thus, for RD <1, risk of radiation-caused harm (cancer induction) is zero. However, Dt is not precisely known for a given population and radiation exposure scenario and likely differs for different populations and different radiation types as well as for mixed radiation types. Unlike for usual use of E, variability and uncertainty can be assigned to RD. Where uncertainty has not formally been characterized, judgmental upper and lower bounds can be assigned.
Like is the case for threshold radiation deterministic effects,116,117 for combined exposure of an organ to radiation types 1, 2, …, n, at high dose rates, the solution for RD for the threshold stochastic effect cancer induction (or cancer mortality) is uncomplicated:
Thus, for combined exposure to external gamma rays (γ) and neutrons (N), the following relationship applies for a given exposure scenario and irradiated organ or tissue:
The corresponding equation for internal alpha (α) + beta (β) + γ irradiation is as follows:
It’s Time to Sever the Link Between the SRP and LNT
The following three quotes are relevant to the information in this section: “A conventional approach to radiation dose-response estimation based on simple parametric forms, such as the linear nonthreshold model, can be misleading in evaluating the risk and, in particular, its uncertainty at low doses” (Furukawa et al.
44
). “… the linear-no threshold theory (LNT) fails very badly in the low-dose region, grossly overestimating the risk from low-level radiation. This means that the cancer risk from the vast majority of normally encountered radiation exposures is much lower than given by usual estimates, and may well be zero or even negative” (Cohen
28
). “As the scientific community discovers new information and gains a deeper understanding of radiation exposures and associated health effects, especially in low-dose environments, it should adapt and evolve accordingly” (Cardarelli II
22
).
Currently, the state of knowledge, with respect to the health risks for humans related to exposure throughout life to very low doses of ionizing radiation, is unreliable. 88 This relates in part to influential organizations such as the ICRP mainly relying on LNT-based epidemiological studies that employ the misinforming procedures presented in Table 5 and possibly others, for low-dose radiation risk assessment. Some of the misinforming procedures in Table 5 are also discussed elsewhere.129,130,132,133
It is informative to provide a radiophobia-related example (not related to radiological protection) of the use of LNT-linked effective dose in assigning cases of health detriment due to low-dose radiation exposure. The hypothetical example relates to emitted solar SEPs (i.e., solar energetic particles) causing a GLE (i.e., ground-level enhancement) event involving radiation exposure of a very large population (millions), among which 1 million people of all ages are considered here. The assigned effective dose for this subpopulation of 1 million is E = 0.01 Sv for each person. The assigned cases of health detriment for the radiation exposure scenario and subpopulation, based on the ICRP SRP risk coefficients (see Table 3), is as follows: cases = (1,000,000 persons) × [(0.01 Sv) × (5.7 × 10−2 Sv−1)] = 570 cases. Note that absorbed radiation doses to different tissues of the body associated with a 0.01 Sv effective dose are unlikely harmful to anyone, given the body’s natural defenses against cancer, which would likely be enhanced by the small, absorbed doses to the different tissues. 136 Clearly the SRP associated health detriment risk coefficient unintentionally promotes radiophobia, even though not intended for such an application.
Below and slightly above natural background radiobiology and biophysics studies may reveal important roles of low dose radiation and microdose distribution 88 in the maintenance and evolution of mammalian life (e.g., via beneficial epigenetic changes7,121,136,159). Based on the new knowledge gained, perhaps our current LNT-based system of radiological protection, which unintentionally promotes harmful radiophobia 91 will have its link to the invalid LNT hypothesis for cancer induction severed. A possible replacement would be the threshold-linear hypothesis 132 for harmful stochastic health effects of radiation doses, below the minimum dose for a severe tissue reaction (deterministic effect). This would allow for continuing the use of radiation weighting (for HT) and dose rate effectiveness factors, although the values assigned may need to be adjusted for use with threshold dose-response relationships.
Recommended New Health Protection Principles
Here I introduce the following health protection principle related to preventing harm to an individual from the stochastic radiation effect cancer induction: RD should be < 1 with high credibility. For evaluating the credibility related to RD < 1, uncertainties related to estimates of both D and Dt will need to be considered because these uncertainties will impact RD uncertainty.

Key Benefits and Constraints for Use of Relative Dose (RD) and the Relative Dose Limit (RDlim) in Limiting Radiation Exposure.

Corresponding equivalent doses to equation (9) for an irradiated person could also be assigned. In this case, the following equivalent dose limitation applies:
The population threshold equivalent dose HT,t is tissue/organ, and exposure-scenario specific.
Organizations, Groups, and Others That Could Help the ICRP to Bring About an Improved System of Radiological Protection That Does Not Promote Radiophobia-Related Harm.

Including additional health endpoints in the SRP
If warranted based on science, including additional health endpoints in the SRP could possibly be addressed via the values assigned to RDlim and HT,lim. This includes cardiovascular disease, neurological disorders, immune dysfunction, and cataracts. Quite important, wR values in Table 1 may need to be revised to be more scientifically credible for threshold health risk vs dose relationships. This view is based on assuming that values in Table 1 are based on LNT dose-response relationships, which may not be the case.
Critically reviewing questionable methods used in epidemiological studies
One effort that would be quite beneficial to society is for a group or organization in Table 8 to critically review data analysis methods used in epidemiological studies of health risks associated with low radiation doses. A second effort that would also be beneficial is for an agency that funds epidemiological studies to oversee (via funded research) testing the reliability of data analysis methods used by epidemiologists in studies of low-dose radiation health risks.
An approach to risk-analysis-methods reliability assessment for epidemiological studies of cancer risk after exposure to low radiation doses was previously proposed 131 and would involve use of simulated (via Monte Carlo calculations) noisy dose-response data. A group of dose-response modelers could produce data (e.g., persons with cancer and those without cancer in a population) for different radiation exposure scenarios and different populations using different plausible dose-response models for AR(D). To be realistic, data noise (e.g., dose errors) will need to be included in the data provided to others (without revealing the noise level), but not used in Monte Carlo evaluations for cancer data generation.
Monte Carlo methods can be used by the modeling group to generate a Bernoulli random variable (1 for cancer, 0 for no cancer) distribution for a given population size and dose distribution over the population, with the probability used based on AR(D) for each individual-specific dose D considered. Both radiation-caused cancer (D > 0 mGy) and cancer not caused by radiation (D = 0 mGy) would be included in the data generated. The number of Bernoulli random variables generated would match the size of the population studied.
A group of epidemiologists would then use their preferred data analysis methods to analyze different simulated data sets produced by the modeling group to generate dose-response relationships for cancer AR(D), RR(D), and ERR(D). Results could then be compared to the correct dose-response relationships, thereby allowing evaluation of epidemiological study methods reliability (or unreliability) for assessing health risks of low radiation doses. The indicated simulated-data-based research could be employed in a low-dose-radiation research program.
Needed new research related to Dt
New research related to cancer risk assessment based on population thresholds is needed. For threshold dose-response relationships for cancer induction, exceedance absorbed dose ∆D or exceedance RBE-weighted dose ∆Drbe could be used as the independent variable, but not exceedance equivalent dose “∆HT = HT – HT,t” whose use should be restricted to radiation exposure limitation; in which case “∆HT, = 0 mSv” (or related equivalent dose unit) should be a requirement. What threshold dose-response functions to use for cancer risk assessment need to be resolved. Possibilities are threshold-linear and threshold sigmoidal functions for doses below the high-dose population threshold for deterministic effects (e.g., hematopoietic syndrome). Ideally, the functions would be based on radiobiological mechanisms of cancer induction.
Addressing Dose Rate Influence
Dose rate has an important influence on Dt. For a fixed dose rate r for external radiation exposure, Dt can be replaced with Dt(r). For exposure-time (T)-dependent changing dose rate r(T) from internal radionuclides (e.g., ingested 137Cs),
89
Dt can be evaluated using a similar approach as is used for the median lethal dose D50 for radiation deterministic effects.116,117 One can first evaluate the radiation exposure duration (ed1) for achieving RD = 1, based on the dose-rate pattern over time for r(T). This is what the following equation relates to:
The value to use for Dt should be based on the solution to the following equation:
What would be a useful research project for a talented graduate student would be to develop a plausible mathematical expression for Dt(r) and use the expression to evaluate ed1 and Dt for different dose rate patterns (over time). The mathematical expression for Dt(r) derived may be similar to what is used 117 for evaluating median lethal dose D50(r) for radiation deterministic effects of low-LET radiation. In this case, Dt(r) would take on a fixed value for high dose rates where r > r* (some high dose rate) but progressively take on increasing values as r decreases below r*.
Now that the exceedance equivalent dose ∆HT has been introduced, it is possible to also use an exceedance effective dose ∆E where ∆E is obtained by applying tissue weighting factors (wT) to organ/tissue specific ∆HT values and adding the results. However, note that in this case ∆E will be zero (e.g., mSv) for many low dose radiation exposure scenarios. Thus, ∆E would be unlikely to be used in limiting radiation exposure but like for ∆HT could be used to indicate when risk of harm to health needs to be considered (i.e., when either ∆HT > 0 mSv or ∆E > 0 mSv). For health risk assessment, the most reliable variables to use are D or ∆D or Drbe or ∆Drbe or RD. In some cases, risk upper bounds may be the focus. As an example, I return to Figure 7.
Results in Figure 7 can be used to assign solid cancer mortality risk upper bounds for the Japanese population, for mortality from a specified gamma-ray-caused solid cancer type. The choice of which of the three lines (A, B, C) to use could be made by qualified experts, such as are on ICRP committees. Use of line C would be the least radiophobia promoting choice.
Analytical Solution for Dt and RBE (at Dt) for Mixed Radiations
Starting from equations provided for RD for mixed radiation exposures, one can derive analytical solutions for the mixed radiation absorbed dose threshold Dt. For mixed neutron and gamma-ray exposures at high dose rates as from a nuclear weapon, the solution for Dt (absorbed dose for mix) as a function of Dt,γ (for gamma rays only) and Dt,N (for neutrons only) is based on the following RD-linked relationship, where fγ is the fraction of the absorbed dose D due to gamma rays and “fN = 1 − fγ” is the fraction due to neutrons:
Note that multiplying both sides of equation (13) by the mixed radiation absorbed dose D yields the relationship for RD already introduced in equation (7). Equation (13) is similar to what is used for assigning the shape parameter
117
in the deterministic effects risk function for lethality risk assessment for mixed high- and low-LET irradiation. Note that because Dt,N < Dt,γ, more weight is assigned to the high-LET neutrons than for low-LET gamma rays, as has been done
117
for high- plus low-LET radiation caused deterministic effects. Since Dt,N = Dt,γ / RBEt,N, for threshold-specific neutron RBE (i.e., RBEt,N), the solution for Dt is as follows:
Note that for fγ = 1, “Dt = Dt,γ”, and for fN = 1, “Dt = Dt,γ / RBEt,N = Dt,N”. Note also that RBE when evaluated based on Dt for the mixed neutron and gamma-ray field and Dt,γ as reference is given by the following relationship:
For internal α + β + γ irradiation, the corresponding solution for Dt is as follows:
Here, fα and fβ are the alpha and beta fractions of the total dose. Note that RBE when evaluated based on Dt for the mixed alpha and beta and gamma-ray field and Dt,γ as reference is given by the following relationship:
The indicated beta and alpha RBEs (RBEt,β and RBEt,α) are evaluated relative to gamma rays. Similar equations for Dt and mixed field RBE would apply to the mixed radiations encountered by astronauts in space explorations.
The myeloid leukemia data of Ariyoshi et al, 3 if supplemented with data from a new study (to increase sample sizes), would allow for evaluating Dt,γ based on data for gamma rays. This would then allow for evaluating Dt,N based on the neutron exposures which involved gamma-ray contamination. The value to assign to fγ would depend on the gamma-ray contamination for the neutron irradiation. The value for Dt would be for the neutron gamma mix. Use of Bayesian inference implemented with Markov chain Monte Carlo137 would allow for assigning distributions for Dt,γ, Dt,N, and neutron RBEt,N (relative to gamma rays). Bayesian semiparametric modeling could also be used. 44
New radiation dose-response studies for cancer induction by low radiation doses using large numbers of small animals (e.g., mice) focused on the population dose threshold dependence on radiation type and on dose rate, for different cancer types, are needed and could be performed over several years at one or more facilities. Outcomes of the studies would hopefully include the following: RBEt, DREFt (i.e., dose rate effectiveness factor DREF evaluated at Dt) and resolved shape of the dose-response relationship for radiation-caused cancers.
In vitro neoplastic transformation studies could provide lower bound estimates of Dt for cancer induction as well as central estimates of RBEt,j for cancer induction for different radiation types j. Values for Dt for neoplastic transformation in vitro may be less than corresponding values based on cancer dose-response relationships because more protection (immune system related) acts against cancer in vivo than against in vitro neoplastic transformation.
Discussion
For addressing radiophobia, it is important to focus on what happens below the population threshold absorbed dose Dt for radiation-caused cancer where DPF(D) > 0 applies. The following two quotes are relevant to the subsections that follow that relate to D < Dt: “The stability of the genome is supported by an intricate machinery of repair, damage tolerance, and checkpoint pathways that counteract DNA damage” (Henry
54
). “… a certain amount of low-dose radiation exposure may induce anti-tumor immunity even in the absence of a tumor in the body” (Nakajima et al.
89
).
The > 15,000 Mice Study with Some Low Doses
Most epidemiological studies do not produce results for low radiation doses that permit reliable evaluation of the shape of the dose-response relationships for RR(D) and DPF(D), as they relate to cancer prevention. However, fortunately we can rely on small animal (e.g., mouse) studies that employ very large numbers of animals, although this is quite rare. Such studies usually have reliable characterization of both radiation dose and dose rate. One highly reliable study was conducted by Ullrich et al, 154 using >15,000 germ-free-derived, specific-pathogen-free, 12-week-old female RFMf/Un mice exposed to low-LET 2000-Ci 137Cs gamma rays or high-LET fission neutrons (with a small gamma-ray contribution).
Gamma-Ray study
For the gamma-ray experiment, mice were exposed at 0.45 Gy min−1 to 0, 0.1, 0.25, 0.5, 1.0,1.5, or 3.0 Gy. The lung adenoma incidence data of Ullrich et al 154 demonstrated a hormetic response to doses in the range 0 to 1.5 Gy. I have used the cancer incidence reported for each dose group as an estimate of AR(D) for lung adenoma occurrence. This allowed generating estimates of RR(D), thus allowing for estimating DPF(D). Uncertainties were based on 95% CI, obtained assuming a binominal distribution of lung adenoma cases. MC calculations were used to obtain the mean, median, standard deviation and 95% CI. For each dose group, 20,000 MC realizations were performed. MC error was <3.3 × 10−4 for each dose group. Medians for RR(D) for the MC analyses were essentially the same as averages calculated with the incidence data of Ullrich et al 154 For the 3.0 Gy group, the cancer AR estimate of 0.371 was significantly above (based on Ullrich et al 154 ) the baseline risk estimate of 0.302, thus, for this dose group DPF(D) = 0 and RR(D) > 1 (i.e., RR (3 Gy) = 1.228; 95% CI: 1.156, 1.308).
The dose-response relationship obtained for RR(D) is presented in Figure 9. Also presented are expected results based on the 3 Gy dose group when extrapolated to 0 mGy based on the LNT hypothesis. Note that the LNT-based extrapolation fails badly in predicting the results in the dose range 0 mGy to 1.5 mGy. The results in Figure 9 are suggestive of 1.5 Gy < Dt < 3.0 Gy, since RR(1.5 Gy) < 1 and RR(3 Gy) > 1. Hormetic relative risk RR(D) for lung adenoma occurrence after whole-body exposure of RFMf/Un mice to high dose-rate gamma rays (0.45 Gy min−1), based on cancer incidences published by Ullrich et al
154
Data points as well as the associated 95% CI values for each data point were joined via linear interpolation. Note that uncertainty associated with RR(D) = 1 is included. Also shown is the LNT-hypothesis-based extrapolation (solid straight line) from 3 Gy down to 0 Gy with 95% CI (dashed-dotted lines), based on linear interpolation between 0 Gy and 3 Gy.
The gamma-ray dose-response for DPF(D) based on the hormetic results in Figure 9, unadjusted for uncertainty in DPF(0 Gy) = 0, is presented in Figure 10. Results in Figure 10 were then adjusted for uncertainty in DPF(0 Gy) = 0, via subtracting the upper 95% CI value of 0.0641, leading to the results in Figure 11. Negative values for DPF(D) were set to 0. Note that the results in Figure 11 suggest that 0.35 is a credible upper bound for DPF(D) for preventing/removing spontaneous lung adenomas via high-dose-rate gamma irradiation of RFMf/Un mice. Unadjusted (for uncertainty in DPF (0 mGy) = 0) disease prevention function DPF(D), for preventing lung adenoma occurrence after whole-body exposure of RFMf/Un mice to high dose-rate gamma rays, based on RR(D) results in Figure 9. Data points as well as the associated 95% CI for each data point were joined via linear interpolation. Dashed lines relate to 95% CI values for the data points. Uncertainty for DPF (0 Gy) = 0 was assessed; however, the lower 95% CI value was negative and is not plotted. Adjusted (for uncertainty in DPF (0 Gy) = 0) disease prevention function DPF(D) for preventing lung adenoma occurrence after whole-body exposure of RFMf/Un mice to high dose-rate gamma rays (0.45 Gy min−1), based on unadjusted results in Figure 10. Data points as well as the associated 95% CI values for each data point were joined via linear interpolation. No evidence for gamma-ray-induced lung adenoma for doses up to 1.5 Gy (1500 mGy).
Neutron study
Figure 12 shows the dose-response relationship for RR(D) for lung adenoma occurrence after whole-body exposure of female RFMf/Un mice to fission neutrons (dose rate 50 mGy min−1), based on cancer incidences published by Ullrich et al
154
Doses are body midline doses. Note that like what was found for gamma rays in Figure 9, the dose-response relationship for the dose range presented is hormetic. For the dose range presented there were 1191 mice involved, which includes 648 mice (from 2 experiments) for the 0 mGy group and 335 mice for the dose range 48 mGy to 192 mGy. The ratio of the neutron dose component to the gamma-ray contamination component of the absorbed dose at the point of exposure of the mice was approximately 7:1.
154
Hormetic relative risk RR(D) for lung adenoma occurrence (solid points and 95% CI dashed lines) after whole-body exposure of RFMf/Un mice to fission neutrons (50 mGy min−1), based on cancer incidences published by Ullrich et al
154
The LNT-hypothesis-based results 95% CI (dashed-dotted straight lines) is based on extrapolating downward via linear interpolation to 0 mGy from the RR (470 mGy) = 1.68 (95% CI: 1.29, 2.10), for the 470 mGy group, that is above the dose range presented. Doses are body midline neutron doses, with the small gamma-ray contribution excluded.
154
.
Doses presented in Figure 12 are body midline neutron doses (gamma-contamination excluded), which were also used by Ullrich et al 154 in characterizing dose-response relationships. The results presented in Figure 12 suggest that based on the neutron fraction of the dose, 48 mGy < Dt < 470 mGy. This is because RR(48 mGy) < 1 and RR(470 mGy) > 1.
Also presented in Figure 12 are LNT-hypothesis-based RR(D) results (95% CI), that were obtained based on extrapolating downward from results for 470 mGy (112 mice used), to 0 mGy (648 mice used), based on linear interpolation. Note that the indicated LNT-based downward extrapolation fails in predicting the data in the dose range 0 mGy to 96 mGy. Thus, this is also strong evidence against LNT as applied to cancer induction by ionizing radiation.
Note that along with the Dt results for gamma rays, the Dt results for neutrons suggest that RBEt,N (for neutrons relative to gamma rays) is in the range 3000 mGy (gammas) / 470 mGy (neutrons) ≈ 6.4, to 1500 mGy (gammas) / 48 mGy (neutrons) ≈ 31. The indicated range 6.4 to 31 is consistent with wR = 20, as presented in Table 1 for neutrons with energies in the range 100 keV to 2 MeV. New research is needed to refine the RBEt,N estimate.
The results for DPF(D) based on Figure 12, unadjusted for uncertainty in DPF(0 mGy) = 0, are presented in Figure 13. Negative results were set to 0. The adjusted results are presented in Figure 14. The adjustment was carried out by subtracting the upper 95% CI value 0.162 from unadjusted results in Figure 13. Note that the results suggest 0.85 is a plausible upper bound for DPD(D). New research focused on low doses is needed to refine this result. Unadjusted (for uncertainty in estimate DPF (0 mGy) = 0) disease prevention function DPF(D), for preventing spontaneous lung adenoma occurrence using whole-body exposure of RFMf/Un mice to fission neutrons, based on RR(D) results in Figure 12. Data points as well as associated 95% CI for each data point were joined via linear interpolation. Dashed lines relate to 95% CI values for the data points. Uncertainty for DPF (0 Gy) = 0 was assessed; however, the lower 95% CI value was negative and is not plotted here. Adjusted disease prevention function DPF(D), for preventing spontaneous lung adenoma occurrence using whole-body exposure of RFMf/Un mice to fission neutrons, based on unadjusted DPF(D) results in Figure 13. Data points as well as associated 95% CI for each data point were joined via linear interpolation. Dashed lines relate to 95% CI values for the data points. Data were adjusted for uncertainty in DPF (0 mGy) = 0 by subtracting upper 95% CI value 0.162 from unadjusted results in Figure 13. No evidence for neutron-induced lung adenoma for doses up to 200 mGy.

Note also that for high-LET neutrons with gamma-ray contamination also present, the hormetic doses zone is much reduced when compared to the dose zone for gamma rays only. This may also be the case for other high-LET radiation types (e.g., alpha particles, heavy ions,) when dose rates are high. For X rays the width of the hormetic zone would be expected to be similar to that for gamma rays when dose rates are high, based on the associated LET. For low-LET beta radiation, dose rate can be low, in which case the width of the hormetic doses zone could be wider than for high dose rate gamma or X rays. This may also be the case for chronic, low-dose-rate, mixed alpha, beta, and gamma radiation exposure (e.g., as is associated with residential radon inhalation82,147,148).
RBE estimates based on invalid LNT models appear too small
Quite interesting, RBE estimates based on LNT applications are lower than results presented here based on Dt and inconsistent with wR values used in radiological protection. In a study by Ariyoshi et al, 3 the neutron RBE of 2.1 (95% CI: 1.1, 3.7; mean neutron energy 2.3 MeV) was reported, relative to 137Cs gamma rays, for induction of myeloid leukemia in male C3H/HeNrs mice irradiated at 1, 3, 8, or 35 weeks of age. RBE was independent of age at exposure, unlike the AR for leukemia induction, which was age-at-exposure dependent. Threshold dose-response relationships were, however, not considered, and if used it may lead to a much higher neutron RBE estimate.
An LNT-based RBE estimate for 3.1 MeV neutrons relative to X rays for inducing malignant tumors in C57BL mice was reported to be in the range 5 to 8 for mice irradiated at 1 or 3 weeks of age, without evidence of an age at exposure dependence3,83; however, the range of ages was small in the study than in the study of Ariyoshi et al 3 and possible threshold dose-response relationships were not considered.
Other studies of myeloid leukemia induction in mice employing LNT dose-response functions also reported small values for neutron RBE relative to low-LET X-ray or gamma-ray exposure: 0.7 for RF/Un mice (neutron energy, 1 and 5 MeV) 157 ; 2.8 for male RFMf/Un mice (fission neutrons) 153 ; and 2.3 for CBA/Cne mice (0.4 MeV neutrons). 34 In these studies, the mice were irradiated around 10 weeks of age.
Note that for the range of neutron energies discussed (i.e., 0.4 to 5 MeV), RBE values would be expected to be much larger based on the judgmental values for wR in Table 1 for neutron energies in the range 0.1 MeV to 20 MeV (wR = 20). This points to a potential problem with the use of RBE values derived from application of likely invalid LNT functions to cancer incidence data or to cancer mortality data.
What would perhaps be a nice assignment for a graduate student is to estimate Dt for different radiation types for high and low-LET radiation caused cancer and use results to derive RBEt,j (for radiation type j) estimates and related uncertainty. The RBEt,j estimates could be compared to those derived using non-threshold models and also to wR values in Table 1. The results obtained might be of interest to the radiological protection community, who may use the RBE findings for improving the assigned values for wR in Table 1.
As might be expected, the high-dose-rate gamma-ray data of Ullrich et al 154 suggests that Dt depends on the neoplasm type. For thymic lymphoma induction, the data suggest Dt > 100 mGy. For reticulum cell sarcoma induction, the data suggest Dt > 3000 mGy. A relevant research question is what features of gensadaptation help explain this implied large difference in Dt for thymus tissue compared to sites (lymph nodes, spleen, brain) where reticulum cell sarcoma occurs?
What would likely be informative would be for a graduate student to reanalyze the leukemia (myeloid leukemia and lymphoid neoplasms) data of Ariyoshi et al 3 for population dose thresholds, to see if they are both age and radiation-type-dependent. Threshold-linear or threshold-sigmoidal dose-response relationships for RR(D) may prove adequate for describing the data for doses > Dt. Below Dt, hormetic outcomes may be revealed; however, new experimental studies by Ariyoshi and colleagues at the Research Center for Radiation Protection, National Institute of Radiological Sciences, providing additional data to increase sample sizes would make the indicated analyses less challenging.
Health Benefits of Low Radiation Doses
The findings reported for DPFp > 0 in Table 6 and other data suggesting or clearly demonstrating a population RRp < 1 (references. 8,43,65,67,113,114,147,148) related to cancer prevention by low radiation doses, as well as the results in Figures 11 and 14 for DPF(D), are supportive of low-dose radiation health benefits for humans and other mammals. However, it is unlikely that these benefits are limited to mammals, as all living organisms today are products of evolution that occurred on our planet where natural background radiation exposure continues throughout life, for all generations.
It is now recognized that elevated natural background radiation is associated with increased lifespan.54,56 This is considered to relate to the enhancement of the body’s natural defenses against cancer and other diseases,54,56 which are gifts of evolution.
Chronic radiation exposure at somewhat above natural background radiation levels may suppress the rate of growth of lung tumors in humans as is suggested from findings in an A/J mouse study where low-dose-rate internal 137Cs gamma rays suppressed the rate of growth of injected-urethane-induced multiple lung adenomas per mouse. 89 Low dose (fractionated) high-dose-rate external 137Cs gamma rays suppressed the number of lung adenomas per A/J mouse induced by injected benzo[A]pyrene.15,123 Both the urethane and benzo[A]pyrene were injected in high doses so as to guarantee multiple lung adenomas per mouse. With this study design, relatively small numbers of animals per radiation exposure group can be used to study the impact of low-dose radiation on cancer suppression.
As you are likely aware, residing in homes with residential radon concentrations (in Bq m−3) at high levels has been found to be associated with lung cancer, which is the basis for the Environmental Protection Agency (EPA) regulating residential radon concentration levels. 107 However, the lung cancer risk vs exposure level dose-response relationship is hormetic27,107,113,114,120,122,135,147,148 implicating DPF(D) > 0 (i.e., health benefits) for the hormetic zone. This points to radiophobia related to residential radon as being unnecessarily harmful to society. It also points to the possibility of chronic low-level radon exposure stimulating the removal of lung cancer cells from the body and/or preventing smoking-related lung cancer occurrence.
A BEIR IV 90 LNT model was used by Cohen 27 to predict lung cancer rates as a function of mean residential radon level and found that the results failed badly in explaining the observed data which were hormetic. My recollection is that Dr Cohen was not expecting the cancer rate to decrease as the radon level increased and was heavily criticized by members of the radiophobia industry (those who intentionally profit from radiophobia) for his very important hormetic findings. Others147,148 have also found similar results for lung cancer prevention associated with radon in the home. My recollection is that they were also heavily criticized by members of the radiophobia industry for their important hormetic findings which support the view that residential radon at low levels can prevent smoking-related lung cancer.
The range of radon exposure levels associated with lung cancer prevention via inhaled radon is wide,27,147,148 unlike demonstrated in Figures 12 and 13 for high-dose-rate fission neutron exposure of mice. This points to the possibility of gensadaptation being much further along on the evolutionary scale related to chronic residential radon exposure (source of alpha, beta, and gamma radiation from 222Rn and 220Rn) than for fission neutrons, which very few are exposed to during a lifetime. Long ago, chronic (over lifetimes) radon exposure took place in residential caves where early humans resided.
Implications of “DPF(D) > 0” for disease therapy
Note that “DPF(D) > 0” has implications for possible use of low-dose radiation therapy for different diseases, as has been proposed by Cuttler, 31 Cuttler et al, 33 Kaul et al, 70 Dunlap et al, 36 Gao and Zhang, 46 and Kim et al, 72 as well as others. Diseases for which low-dose radiation therapy may prove to be quite beneficial include arthritis and other inflammatory diseases, cancer, Alzheimer’s disease, Parkinson’s disease, and COVID-19 pneumonia. Regarding Alzheimer’s disease, there is now evidence that it occurs in two distinct phases 45 : (1) an early phase involving a slow increase in pathology, the presence of inflammatory microglia and reactive astrocytes, the loss of somatostatin+ inhibitory neurons, and a remyelination response by oligodendrocyte precursor cells; (2) a later more serious phase with an exponential rate of increase in pathology and loss of both excitatory and inhibitory neuron subtypes. Possibly low dose radiation therapy may be more successful if applied during the early phase.
For external beam low-dose radiation therapy for cancer, high energy X rays are likely to be preferred over the use of gamma rays. The use of 6 MV X rays in the treatment of prostate cancer is being explored by Kennedy et al 71 Their view is that low-dose radiation therapy “has the potential to become an effective treatment option for managing recurrent prostate cancer and possibly other forms of malignant disease”. 71 Unfortunately, their recent study using high energy X rays used a total absorbed dose of 1500 mGy (from 10 exposures to 150 mGy) to a large part of the body, which caused undesirable hematopoietic damage. Reducing the total dose should be explored, as a much smaller total dose might be effective in stimulating anticancer immunity while not causing significant damage to the hematopoietic system.
Note that the wide hormetic radon-exposure-level zone27,147,148 related to lung cancer prevention/elimination is suggestive of possible chronic radon exposure therapy for smoking-related lung cancer. Radon therapy for some diseases is currently eagerly sought by many patients, but scorned or dismissed by physicians, likely related to radiophobia. 37 Dismissal is the case here in the USA where our EPA and some companies that profit from monitoring radon levels in the home regularly warn that even low-level radon inhalation supposedly poses a risk of radiation-induced lung cancer. However, away-from-home radon therapy has long been used to treat health problems that include back pain and high blood pressure.54,69,75,87,109,165 The therapy is being used not only in Japan but also in Europe. 166
Regarding inflammatory disease therapy, radon, when inhaled at therapeutic levels, is an alternative to conventional biomedical treatment in that the inhaled radon effectively relieves pain and other symptoms of arthritis and other inflammatory diseases. 37 Because the radon therapy benefits are long lasting, and because the therapy is relatively inexpensive, radon treatment allows many arthritis patients to discontinue using their non-radiogenic medications for months at a time, thus providing relief from medication side effects and financial relief at the same time. 37
Important Issues to Address Related to the Use of Low-Dose Radiation Therapy for Different Diseases Based on DPF(D).

Low-Dose Radiation Research Programs Are Needed
Low-dose radiation research programs that focus on needed new animal studies (genomic-stress related) for single and multiple generations, in vitro studies (genomic-stress related), epidemiological data analysis reliability/unreliability studies, theoretical modeling, and other studies that relate to helping to improve the SRP would be quite beneficial to society. Programs are initiated in Japan 164 and also in the USA, but the USA program is limited to National Laboratories with limited expertise in low-dose radiation biology, low-dose radiation chemistry, and low-dose radiation health effects (harm and benefits) assessment.
New low-dose radiation research is also needed related to possibly establishing low-dose radiation therapy for cancer, Alzheimer’s Disease, Parkinson’s Disease, COVID-19 pneumonia, and other diseases. Possibly countries like Japan, China, Canada, Poland, France, Brazil, and India could initiate such research programs. Products of these programs could be quite beneficial to the world community.
A major impediment for low-dose radiation therapy is the radiophobia industry. Members of this industry include research groups, journals (editorial staff), and others that profit from promoting radiophobia. Some journals related to radiation research mainly publish radiation health effects papers focused on high-dose radiation therapy or implied (but not proven in many cases) low-dose radiation harm to health.
Study Limitations
This study was not performed as part of a funded research project with a team of researchers. Thus, it has limitations. Ideally a team of researchers would have participated in the research, as was the case for many previous studies (with peer-reviewed publications) of the author prior to having retired in 2014. With a team of researchers and a funded project, mechanistic dose-response modeling could have been performed yielding a gensadaptation-based mechanistic model for the disease prevention function applicable to radiation doses less than the population threshold for radiation-caused cancer and a mechanistic model for radiation-caused cancer applicable to different radiation exposure scenarios. Radiation exposure scenarios of interest include nuclear accidents such as at Chernobyl and Fukushima, exposure during space exploration, and chronic exposure to natural background radiation, including residential radon exposure. Hopefully this publication will stimulate interest from others around the globe in the additional research needed.
Conclusions
Environmental carcinogens (multiple genomic stressors) exposure over many generations led to progressive gensadaptation for all mammals including humans. Without progressive gensadaptation, we humans and all other mammals would likely have vanished long ago. Also, without progressive gensadaptation, LNT risk models would likely apply to all environmental carcinogens. LNT risk modeling as it relates to cancer induction by ionizing radiation is the current basis for the SRP, which because of this, unintentionally promotes harmful radiophobia.
The ICRP needs to sever the SRP’s link to LNT risk models for harm to health and instead link the SRP to population thresholds for harm, thereby helping to prevent unnecessary radiophobia harm to society. Use of RD may facilitate making such a change, as RD can easily be linked to HT, allowing for radiation limits based on either RD or HT or both. Unfortunately, improving the SRP, so as not to promote radiophobia, will likely require not using effective dose limits. However, the exceedance equivalent dose ∆HT (> 0 mSv) and the exceedance effective dose ∆E (> 0 mSv) could be used as indicators for when health risks from radiation exposure are likely > 0.
Low-dose radiation research programs focused on needed new single and multigeneration animal studies, in vitro studies, theoretical modeling, testing of epidemiological studies reliability/unreliability, and other studies that relate to helping to improve the SRP would be quite beneficial to society. However, low-dose programs are also needed that focus on possible low-dose radiation therapy for different diseases. Findings from the low-dose programs would likely be beneficial to all nations.
Footnotes
Author’s Note
The views and conclusions contained herein are those of the author and should not be interpreted as necessarily representing policies or endorsement of his affiliated institution.
Acknowledgments
The author is grateful to the two reviewers for their comments on the paper and to “Sage Publishing” for publishing the “Special Collection” related to LNT-based radiophobia and for the APC waiver for this paper by an elderly retiree (Scientist Emeritus at LBRI). Comments from Reviewer #2 helped in improving the quality and comprehensibility of the publication.
Author Contributions
The author carried out the research presented and prepared the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


