Abstract
Background
Dexamethasone in conjunction with type 3 serotonin receptor antagonists are being used to the prevention and treatment of chemotherapy-induced nausea and vomiting and postoperative nausea and vomiting in clinic. The present study aimed to investigates the stability of ramosetron with dexamethasone in infusions, with the goal of enhancing the safety and clinical applicability of their combined use.
Methods
Ramosetron hydrochloride (3.0 μg/mL) combining with dexamethasone (0.05, 0.1, 0.2 mg/mL) were prepared with 0.9% sodium chloride injection and then packaged in polyolefin bags or glass bottles. The stability were investigated kept in the dark at refrigeration for 14 days and at room temperature for 48 h.
Results
The concentration of both drugs maintained at least 97% in the various solutions for both storage conditions with light protection. In the light exposure conditions, as the extension of storage time, the concentration of both drugs had declined. All antiemetic mixture solutions remained clear and no changes in color, turbidity, precipitation, and the pH remained stable. The insoluble particles were in line with Chinese Pharmacopoeia.
Conclusion
Our findings suggest that combinations of ramosetron hydrochloride with dexamethasone sodium phosphate in 0.9% sodium chloride injection remain stable for 14 days at 4°C and 48 h at 25°C when protected from light.
Keywords
Introduction
Surgery, chemotherapy and radiotherapy are one of the important methods for the treatment of malignant tumors, but nausea and vomiting caused by surgery and chemotherapy are common complications. 1 Chemotherapy-induced nausea and vomiting (CINV) is one of the most common side effects caused by chemotherapy for malignant tumors, and its incidence is 50%∼96% according to the patient’s gender, age, mental state, chemotherapy drugs and other influencing factors.2,3 Postoperative nausea and vomiting (PONV) is an inevitable complication that occurs within 24 h of the perioperative period, with an incidence ranging from 20 % to 80 %. 4 CINV and PONV generally do not pose a threat to patients’ lives, but they can result in decreased appetite, malnutrition, weight loss, electrolyte imbalance, aspiration pneumonia, postoperative wound rupture and other clinical adverse outcomes. Additionally, they significantly impact patients’ mood, social interaction, quality of life and treatment adherence. This has become a common concern for physicians, patients and pharmacists.5-8
The therapeutic drugs for CINV and PONV include histamine receptor blockers, dopamine receptor blockers, 5-HT3 receptor antagonists, neurokinin-1 receptor antagonists, glucocorticoids and traditional Chinese medicine.2-4 In recent years, clinical trials and evidence-based medical evidence have shown that the “antiemetic mixture” composed of 5-HT3 receptor antagonist such as ramosetron hydrochloride and dexamethasone is one of the most widely used and effective drug treatment regimens for the prevention and treatment of CINV and PONV.5-12 Currently, there is no commercially available finished infusion preparation of ramosetron hydrochloride and dexamethasone sodium phosphate injection mixture, which belongs to off-label drug use. These infusions must be mixed and prepared in centralized intravenous infusion centers (CIVAS) or hospital ward for clinical use. Limited research data on the compatibility and stability of these two drugs in infusion, as indicated by drug package insert and literature investigation, poses significant risks to patient infusion safety. Therefore, the purpose of the current study was to investigated the stability of ramosetron hydrochloride and dexamethasone sodium phosphate injection, at 3 different concentration combinations, diluted with 0.9% sodium chloride injection and packaged in polyolefin bags or glass bottles for 14 days at 4°C and 48 h at 25°C.
Methods and Materials
Drugs and Reagents
Ramosetron hydrochloride (purity 99.8%, lot# 100662-202003) and dexamethasone phosphate reference substance (purity 98.5 %, lot# 101116-202204) were purchased from the China Institute for Food and Drug Control (Beijing, China). Ramosetron hydrochloride injection (2 mL/0.3 mg, lot# 20220312, Chenxin Pharmaceutical Co, Ltd), dexamethasone sodium phosphate injection (1 mL/2 mg, lot# 210911001A, Shanghai Jindi Jiuzhou Pharmaceutical Co, Ltd), and 0.9 % Sodium chloride injection (NS, 500 mL/4.5 g, lot# 22062022, Dalian Dazhong Pharmaceutical Co, Ltd) were purchased from Sinopharm Holding Company Limited (Hubei, China). Potassium dihydrogen phosphate (KH2PO4, AR grade), phosphoric acid (AR grade), and acetonitrile (HPLC grade) were purchased from Tianjin Bondi Technology Development Co, Ltd.
Chromatographic Conditions
The HPLC system (LC-20AT, Shimadzu Instruments, Suzhou, China) consisted of a LC-20AT four-element low pressure gradient system, CTO-20A column temperature box, SIL-20A automatic sampler, SPD-M20 A diode array detector, CBM-20A system controller, and LC Solution data processing software. The chromatographic column was Intersil ODS-3 C18 (4.6 mm × 150 mm, 5 μm). The mobile phase was acetonitrile-0.05 mol/L KH2PO4 (pH was adjusted to 4.0 with phosphoric acid) (25/75, V/V). The flow rate was 1.0 mL/min and the detection wavelengths of ramosetron and dexamethasone were 308 nm and 241 nm, respectively. The column was thermostated at 30°C and injection volume was 20 μL during analysis.
Preparation of Solution
Preparation of Reference Solution
Accurately weigh 6.0 mg ramosetron hydrochloride and 20 mg dexamethasone phosphate working standard into 100 mL calibrated volumetric flask. 70 mL NS was added to the flask and sonicated to dissolve it completely, and then completed to volume with NS to get the primary standard stock solution containing 0.2 mg/mL of dexamethasone sodium phosphate and 60 μg/mL of ramosetron hydrochloride. An appropriate amount of the above reference solution was taken and placed in a 10 mL volumetric flask, and diluted with NS to a series of ramosetron hydrochloride reference solutions with concentrations of 0.3, 1.5, 3.0, 6.0, 15.0, and 30.0 μg/mL. In addition, a series of dexamethasone phosphate reference substance solutions with a concentration of 5.0, 10.0, 20.0, 50.0, 80.0, and 100.0 μg/mL were prepared in the same way.
Preparation of Sample Solution
The ramosetron hydrochloride injection (2 mL/0.3 mg) was transferred into a 100 mL bottle and diluted with NS to prepare a sample solution of ramosetron hydrochloride with a concentration of 3.0 μg/mL. Another dexamethasone sodium phosphate injection (1 mL/ 2 mg) were prepared using the same procedure to obtain a sample solution of dexamethasone sodium phosphate with a concentration of 100 μg/mL.
Methodology Validation
Specificity Test
The specificity of this method was confirmed through acid, alkaline, oxidation, and high temperature damage tests. Reference solutions of ramosetron hydrochloride and dexamethasone phosphate were placed in 0.1 mol/L hydrochloric acid solution, 0.1 mol/L sodium hydroxide solution, and 3% hydrogen peroxide solution, respectively, and destroyed at 60°C water bath for 5 h. After neutralization, the resulting destruction solutions were diluted with NS and filtered. Then, following the chromatographic conditions described above, a sample volume of 20 μL was injected into the system for analysis. The analysis involved comparing the chromatograms obtained before degradation with those obtained after degradation to identify any presence of degradation peaks and investigate changes in concentration, retention time, and peak shape for both drugs.
Linear Range
The reference solution of ramosetron hydrochloride and dexamethasone phosphate series under preparation of reference solution section was taken, and the sample was determined according to the above chromatographic conditions. The peak area (A) was used as the ordinate, and the drug injection concentration (C) was used as the abscissa. Linear regression was performed to obtain linear equations and correlation coefficients (r).
Precision
Three different concentrations of high, medium and low reference solution of ramosetron hydrochloride (3.0, 6.0, 15.0 μg/mL) and dexamethasone phosphate (20.0, 50.0, 80.0 μg/mL) were taken. The injection was repeated 6 times on the same day and analyzed for 6 consecutive days. The peak area was determined, the relative standard deviation (RSD) value was calculated, and the intra-day and inter-day precision of the instrument was investigated.
Repeatability and Stability
The reproducibility test was to prepare 6 samples of ramosetron hydrochloride and dexamethasone phosphate, inject the samples, record the peak area, and calculate the RSD value of each drug content. The stability test was to take and prepare ramosetron hydrochloride and dexamethasone phosphate test solutions, inject and analyze them at 0, 1, 2, 4, 6, 8 h, respectively, record the peak area of each chromatography, calculate the drug content and RSD value, and investigate the stability of the sample solution.
Recovery
By adding the known contents of ramosetron hydrochloride and dexamethasone phosphate reference products (high, medium and low concentration levels of 80%, 100% and 120% respectively) into the sample solution prepared under preparation of sample solution section with known content. The above ramosetron hydrochloride solution and dexamethasone phosphate solution diluted 5 times were taken respectively, and the samples were analyzed according to the above chromatographic conditions. The chromatographic peak area was recorded and the drug content was determined. The recovery rate and RSD value of the two drugs were calculated by subtracting the ratio of the known amount and the added amount from the measured amount.
Preparation of Dexamethasone-Ramosetron Mixture Solutions
Dexamethasone sodium phosphate injection, ramosetron hydrochloride injection and NS were extracted by disposable syringe and stored in 100 mL glass or polyolefin container, respectively. Three different concentrations of dexamethasone sodium phosphate (50, 100 and 200 μg/mL) and ramosetron hydrochloride (3.0 μg/mL) were prepared. The above different concentrations of the mixture solutions were placed at refrigerated conditions (4 ± 0.5°C) for 14 days under dark conditions, and at room temperature (25 ± 0.5°C) with exposure to natural light or protection from light for 48 h. The concentration of the mixture solution was determined according to the clinical CINV and PONV diagnosis and treatment guidelines, clinical research literature, as well as hospital clinical practice.5-14
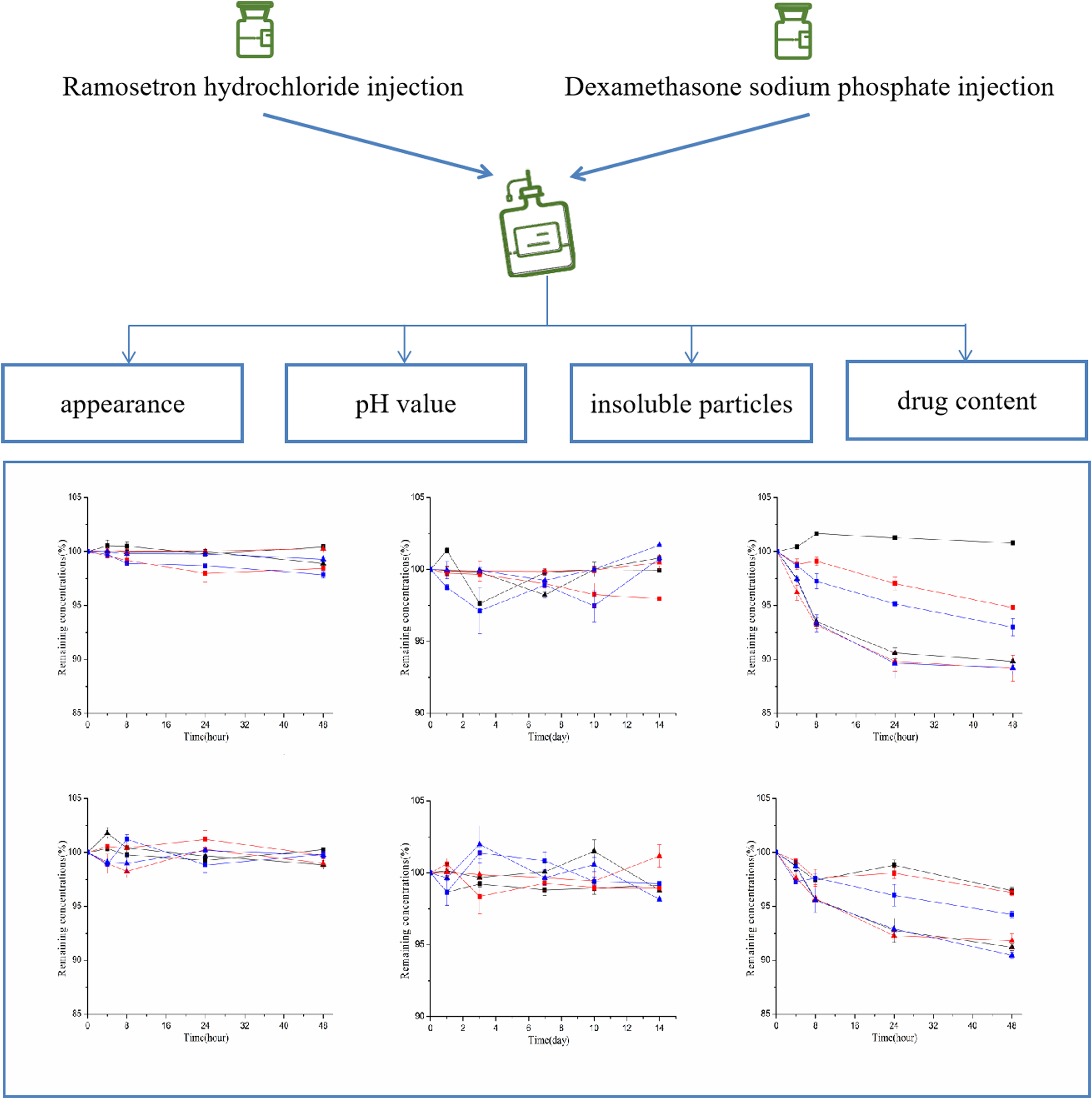
Stability of Dexamethasone-Ramosetron Mixture Solutions
The experimental protocol was approved by the ethical committee of Sinopharm Dongfeng General Hospital, Hubei University of Medicine, Shiyan, China. The reference number of the approval was LW/2023/039. The physico-chemical stability study was evaluated in triplicate for the appearance, pH, insoluble particles, and drug content. Samples were collected at various time points under refrigerated (0, 1, 3, 7, 10 and 14 days) and room temperature (0, 4, 8, 24, and 48 h). Initially, a volume of 5 mL of the solution was taken at each time points for assessment of appearance such as precipitation, turbidity, color change and gas generation using a clarity detector (YB-2A, Tianjin) with NS as blank according to the standards of Chinese Pharmacopoeia 2020 edition. Subsequently, the pH values were measured using a PHS-3C laboratory pH meter (Shanghai Instrument Electrical Science Instrument Co, Ltd). The insoluble particles were examined by the microscopic counting method in the Chinese Pharmacopoeia (2020 Edition) using CX41 PF microscope (Olympus). Finally, the solutions were filtered through a microporous filter membrane (0.45 μm), and 20 μL of the samples were injected and determined according to the above chromatographic conditions.
Each sample was tested three times, and the contents of the two drugs at different time points were calculated. The relative percentage content changes of the two drugs at different time points were calculated with the drug content at 0 hours being 100 %. The content results were expressed as mean ± SD. When the drug content was less than 90 %, it indicated that the drug in the mixture solution was unstable in the infusion. 12
Results
HPLC Method Validation
The typical chromatograms of ramosetron hydrochloride and dexamethasone sodium phosphate under undamaged and damaged conditions, such as exposure to acid, alkali, and oxygen are shown in Figures 1 and 2, respectively. It is evident that the average retention times of ramosetron hydrochloride and dexamethasone phosphate were 8.4 and 10.8 minutes, respectively. The chromatographic peaks of the two drugs were analyzed based on their chromatographic peak shape and 3D absorption spectrum. The results indicated that the separation between ramosetron hydrochloride, dexamethasone phosphate and their degradation products were greater than 1.5. Furthermore, the peak purity indexes for both compounds exceeded 0.999, and the decomposition products did not interfere with the content determination of the two components, which indicating that the method had good specificity. HPLC chromatograms of dexamethasone sodium phosphate from degradation testing. (A) fresh sample at time zero. (B) acid degradation. (C) alkaline degradation. (D) oxidized degradation. Retention times were 10.8 minutes for dexamethasone sodium phosphate (peak 1). The other peaks were for degradation products. HPLC chromatograms of ramosetron hydrochloride from degradation testing. (A) fresh sample at time zero. (B) acid degradation. (C) alkaline degradation. (D) oxidized degradation. Retention times were 8.2 min for ramosetron hydrochloride (peak 2). The other peaks were for degradation products.

The linear regression equations for ramosetron hydrochloride (0.3-30 μg/mL) and dexamethasone phosphate (5.0-100 μg/mL) were A = 36364C-1811 (r = 0.9999, n = 6) and A = 21592C-44347 (r = 0.9996, n = 6), respectively. The RSD values of intra -day and inter-day precision for both compounds were less than 2.0%, indicating excellent instrument precision. Repeatability test results showed that RSD of peak area for both two drugs were less than 2.0%. The RSD of the peak area of the two drugs were 1.1% and 1.5%, respectively, which showed that the samples remained stable within 8 hours. The results of sample recovery showed that the average recovery for both drugs were within the range of 100 ± 2%, and RSD less than 2%, indicating that the recovery rate of this method was good.
Stability Study of Dexamethasone and Ramosetron Mixture
No change in color or precipitation was observed in all of the three different concentrations of ramosetron hydrochloride and dexamethasone sodium phosphate infusion mixtures throughout the study. The pH value of all drug admixtures ranged from 6.7 to 7.2, and fluctuated within ±0.3 under refrigerated or room temperature conditions, which indicating that the pH value of the mixed solutions were relatively stable during the study. The number of particles with a particle size ≥10 μm in each 1 mL of all drug admixtures was ≤12, and the number with a size ≥25 μm was ≤2, which meeting the requirements of the Chinese Pharmacopoeia (2020 edition). The results of the content change of the different mixture solutions during storage are shown in Figures 3 and 4. There was no loss of ramosetron hydrochloride and dexamethasone sodium phosphate in three different concentration combinations stored in glass bottles and polyolefin bags for 14 days at 4°C and for 48 h at 25°C protection from light. However, under light exposure conditions, both drugs showed declining concentrations over extended storage time periods. Regarding dexamethasone sodium phosphate, the initial concentration remaining in the admixtures after 48 hours were only about 90 % in the three different concentrations, stored in both polyolefin bags and glass bottles. Drug concentrations of dexamethasone sodium phosphate and ramosetron hydrochloride in NS when packaged in glass bottles or polyolefin bags, and protection from light over 14 days at 4°C. (A) glass bottles; (B) polyolefin bags. Drug concentrations of dexamethasone sodium phosphate and ramosetron hydrochloride in NS when packaged in glass bottles or polyolefin bags, and protection from light or exposed to light over 48 h at 25°C. (A) protected from light in glass bottles; (B) exposed to light in glass bottles; (C) protected from light in polyolefin bags; (D) exposed to light in polyolefin bags.

Discussion
Combination therapy is a crucial approach for the clinical treatment of diseases. 13 Based on the drug package insert, diagnosis and treatment guidelines, clinical research, evidence-based medicine evidence and patient needs, it is common for clinicians to use ramosetron hydrochloride injection and dexamethasone sodium phosphate in combination.5-12 If the antiemetic mixture has long-term compatibility and stability, it can significantly reduce the workload of nurses, reduce the incidence of transfusion preparation and administration errors. However, incompatibility or instability of the antiemetic mixture may lead to unpredictable medical risks. Therefore, this study investigated the stability and compatibility of an antiemetic mixture composed of ramosetron hydrochloride (3 mg) and dexamethasone sodium phosphate injection (5∼20 mg) in the infusion based on clinical needs. Stability studies were conducted under various storage conditions and different storage containers at different time points. Meanwhile, the drug concentration of the two drugs in this study was based on clinical practice, clinical research and evidence-based medicine literature to improve the clinical practicability and suitability of the research results.5-12,14,15 The study results demonstrated that the mixture of ramostone hydrochloride and dexamethasone sodium phosphate injection was prepared in CIVAS, diluted in 100 mL NS infusion, and stored at room temperature for 48 h or refrigerated for 14 days were stable. However, under natural light conditions at room temperature, although the mixture solutions remained stable for 48 h, but the drug content of the two drugs gradually decreased over time.
Ramosetron hydrochloride injection is a weakly alkaline strong salt that remains stable under weak acid conditions, but its decomposition or solubility decreases in alkaline and light-exposed environments. Literature survey has revealed that few data are reported on the stability of ramosetron hydrochloride when used alone or in combination with other drugs in infusion solutions. Song 16 F. reported a 10-hour stability of ramosetron hydrochloride prepared in NS, 5% glucose injection, or glucose and sodium chloride injection at a concentration of 0.003 mg/mL when stored protected from light at 25°C. Current literature indicates that ramosetron hydrochloride injection can maintain stability when combined with dezocine injection, fentanyl injection, midazolam, and dexamethasone injection.17-21
Due to the widespread use of dexamethasone in clinical practice, there a substantial body of literature on the compatibility and stability of dexamethasone sodium phosphate injection, either alone or in combination with other medications.22-32 Among these, dexamethasone sodium phosphate injection has incompatibility contraindications with various injectables commonly used in clinical practice, such as amoxicillin sodium/clavulanic acid, cirofloxacin lactate, daunorubicin hydrochloride, diphenhydramine hydrochloride, doxapram hydrochloride, doxorubicin hydrochloride, fenoldopam mesylate, glycopyrronium bromide, haloperidol lactate, hydromorphone hydrochloride, imipenem- cilastatin/relebactam, isavuconazonium sulfate, ketamine hydrochloride, levomepromazine, methotrexate sodium, midazolam hydrochloride, morphine hydrochloride, pantoprazole sodium, rocuronium bromide, vancomycin, topotecan, telavancin hydrochloride, etc.25-32 Dexamethasone sodium phosphate is a partially alkaline water-soluble compound that remains stable under weak alkaline conditions and decompose easily under light exposure conditions. Upon compatibility testing with the aforementioned drugs, the pH value of the solution may change, resulting in decreased drug solubility and compromised stability leading to discoloration, turbidity, and precipitation.
A limitation of the study design relates to the potential of bacterial contamination during infusion preparation, which was not investigated in this study. In clinical practice, aseptic operation during infusion preparation should be adhered to in order to guarantee the safety of infusion. Additionally, the results of the in vitro compatibility and stability study of ramosetron and dexamethasone mixtrues do not automatically ensure that the pharmacokinetics and pharmacodynamics of the two drugs remain unaltered in vivo, and follow-up pharmacokinetics and pharmacodynamics studies are requisite.
Conclusion
Based on the findings of this experimental study, it is advisable for clinical practice to take into consideration the following points when preparing the drug mixture of ramosetron hydrochloride and dexamethasone sodium phosphate: (1) Enhance training and evaluation on aseptic and standardized operations during infusion preparation. (2) Dexamethasone sodium phosphate and ramosetron hydrochloride injection should be successively added to the NS injection bag during the process of infusion preparation, in order to avoid the turbidity or precipitation caused by direct mixing of the two drugs due to pH value and drug solubility factors. (3) Conduct routine visual inspection on the prepared infusion, with a focus on observing any occurrences such as discoloration, bubbles, turbidity, and crystallization. (4) The finish prepared infusion should be stored in a dark place to prevent drug decomposition from affecting infusion quality and medication safety. (5) The drug mixture of ramosetron hydrochloride and dexamethasone sodium phosphate prepared in CIVAS remains stable at room temperature for 48 h or refrigerated for 14 days with light protection.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Hubei Provincial Natural Science Foundation of China (number 2022CFB003) and the Technology Key Program of Shiyan, China (number 21Y75).
Data Availability Statement
The majority of the data used to support the findings of this study are included within the article. Other data are available from the corresponding author upon request.
