Abstract
Objectives
This study was conducted to determine the efficacy of mesenchymal stem cells (MSCs) or low-dose gamma radiation (LDR) on liver injury compared to the effect of
Methods
Rats were allocated into six groups; group I served as the negative control. Group II received 5% dextran sodium sulfate (DSS) in its drinking water for 1 week. Group III was injected with a single dose of 1 × 106 bone marrow-derived mesenchymal stem cells (BM-MSCs) intravenously. Group IV was treated as in group III after 5% DSS treatment. Group V was given 5% DSS, followed by
Results
Rats treated with OLE, BM-MSCs, or exposed to LDR exerted significant alleviation in all hepatic biomarkers, significant enhancements in oxidative stress parameters, and improvements in inflammatory biomarkers Interleukin-1 beta (IL-1β) and Interferon gamma (INF-γ) hepatic contents compared with those of the DSS group. Histological pictures emphasized the biochemical findings.
Conclusions
BM-MSCs might be a valuable therapeutic approach to overcome hepatic injury. Exposure to LDR provided protective mechanisms that allow the body to survive better.

Highlights
• Gut-liver axis often links liver abnormalities to inflammatory bowel illness. • There are numerous novel and beneficial ways for treatment of liver diseases. • OLE has a high antioxidant activity. • BM-MSCs are effective and safe in treatment of hepatic injury, improving liver function. • LDR enhances the body’s survival systems. • Histological images reinforced the biochemical findings.
Introduction
The liver is a multipurpose organ of the body that controls the internal chemical environment. 1 It handles the metabolism and excretion of drugs and other xenobiotics from the body, thereby providing protection against foreign substances by detoxifying and eliminating them. 2 The Liver purifies esthetic chemical molecules through oxidation, reduction, and/or conjugation. 3 It is certainly affected by free radicals and causes diseases hepatitis, cirrhosis, liver cancer, and other alcohol-related disorders. 4 Liver disease is the major cause of serious health problems leading to morbidity and mortality worldwide and the problem has increased in search for hepatotherapeutic agents.5,6
The olive tree has long been recognized as having antioxidant molecules, such as oleuropein, hydroxytyrosol, oleuropein aglycone, and tyrosol.
7
Olive leaves and fruits contain polyphenols, flavonoids, flavones, iridoids, and sugars. These compounds have a significant pharmacological action while posing low toxicity. Total
Studies on the beneficial effect of radiation suggested that biological systems can respond in a positive way to exposure to LDR.10,11 Some evidence showed that low doses of radiation may increase the physiological performance, immune competence, general health status, life span 12 and have potential applicability in the treatment of diseases such as cancer, diabetes, and tissue degeneration. 13 The positive impact of exposure to LDR on tumor regression and immune response has recently been emphasized. Enhancement of immune response with LDR treatment is 1 of the important factors responsible for improving therapeutic gain 14 ; this enhancement involves a number of mediators, including inflammatory cytokines produced by macrophages, epithelial cells, and fibroblasts.
BM-MSCs are pluripotent stem cells capable of differentiating into hepatocyte-like cells both in vivo and in vitro.15,16 MSCs are readily accessible from multiple potential sources, including adipose tissue, umbilical cord (UC), umbilical cord blood, peripheral blood, synovial membranes, muscle, dermis, and liver.17-19 Importantly, the harvested MSCs maintain their pluripotent potential, robust proliferative ability, and capacity for ex vivo expansion. 20 MSCs have the ability to migrate and engraft at sites of injured tissue. 21 MSCs have immunosuppressive properties that allow for allogeneic transplantation. The immunosuppressive ability of MSCs also includes anti-fibrotic and antioxidant effects, which can protect the liver from fibrosis and oxidative damage. 22 MSCs produce extracellular vesicles (EV) that contain growth factors and cytokines that promote regeneration of impaired tissue such as liver parenchyma. 22
The current study aims to explore the potential efficacy of BM-MSCs or LDR on liver injury induced by acute ulcerative colitis in male rats compared to
Material and Methods
Animals
Adult Wistar male albino rats (150-180 g) used in this study, were collected from the breeding unit of the National Center for Radiation Research and Technology (NCRRT), Cairo, Egypt. The animals were adapted in the animal facilities of the National Center for Radiation Research and Technology for 1 week before starting the experiments. The experimental animals were housed in a room with 65% humidity and a 12:12 h light- dark cycle at an ambient temperature of 20 ± 1°C. Standard diet, feed pellets, and tap water were freely accessible throughout the experimental stages.
Chemicals and Treatment
Isolation, Preparation, Culturing, and Labeling of BM‐MSCs
Six male albino rats (150-180 g) were euthanized with thiopental (50 mg/kg; i. p.), and femur bones were separated and used for the extraction of bone marrow samples. The bone marrow samples were obtained by flushing the bone marrow cavity with phosphate ‐ buffered saline (PBS), Sigma Chemical Co (United States). A preparation of 15 mL from flushed bone marrow cells was carefully layered for 35 min on 15 mL Ficoll Paque (Gibco Invitrogen). Then the preparation was centrifuged at 400 g. The mononuclear layer of cells was judiciously aspirated and washed twice in PBS containing 2 mM ethylene diamine tetraacetic acid (EDTA) and centrifuged for 10 min at 200 g at 5°C. On 25 mL flasks, the isolated BM- MSCs were stretched. Cells were refined in Roswell Park Memorial Institute (RPMI) −1640 medium containing 10% fetal bovine serum (FBS product of Sigma), 0.5% penicillin, and streptomycin. The prepared flasks were incubated at 37°C and 5% carbon dioxide (CO2) until they reached 80%–90% confluence within 7 days. The expansion of BM-MSCs was done until it reached the third passage. Rat BM-MSCs cells were harvested and labeled with PKH26 fluorescent linker dye (Sigma-Aldrich Co, USA) as previously reported.24,25 Transplanted stem cells were directly injected into rat tail veins of assigned groups at a dose of 1 × 106 cells for each rat. 26
Low Radiation Dose
Animals were exposed to low dose radiation (0.05 Gy) with a Caesium-137 irradiation source at a dose rate (2.21 mGy/h) and about (53.04 mGy/day) at NCRRT. An especially designed galvanized cylindrical cage was used in this experiment. The source was placed in the center of the metal cage. The distance between the rats and the source was 20 cm.
Sample Size Determination
Prior to the study, a power calculation based on the collected data was utilized to determine the required number of samples in each group.27,28 Utilizing G*Power software (Version 3.1.9, Düsseldorf, Germany), a sample size of 48 rats (n = 8/group); they were split evenly among six groups; was established to achieve 80% power with a 95% confidence interval and a 5% significance level.
29
The effect size (f) was calculated to be 0.99 at the 0.05α and 0.05β levels (Figure 1). A chart showing F-tests. 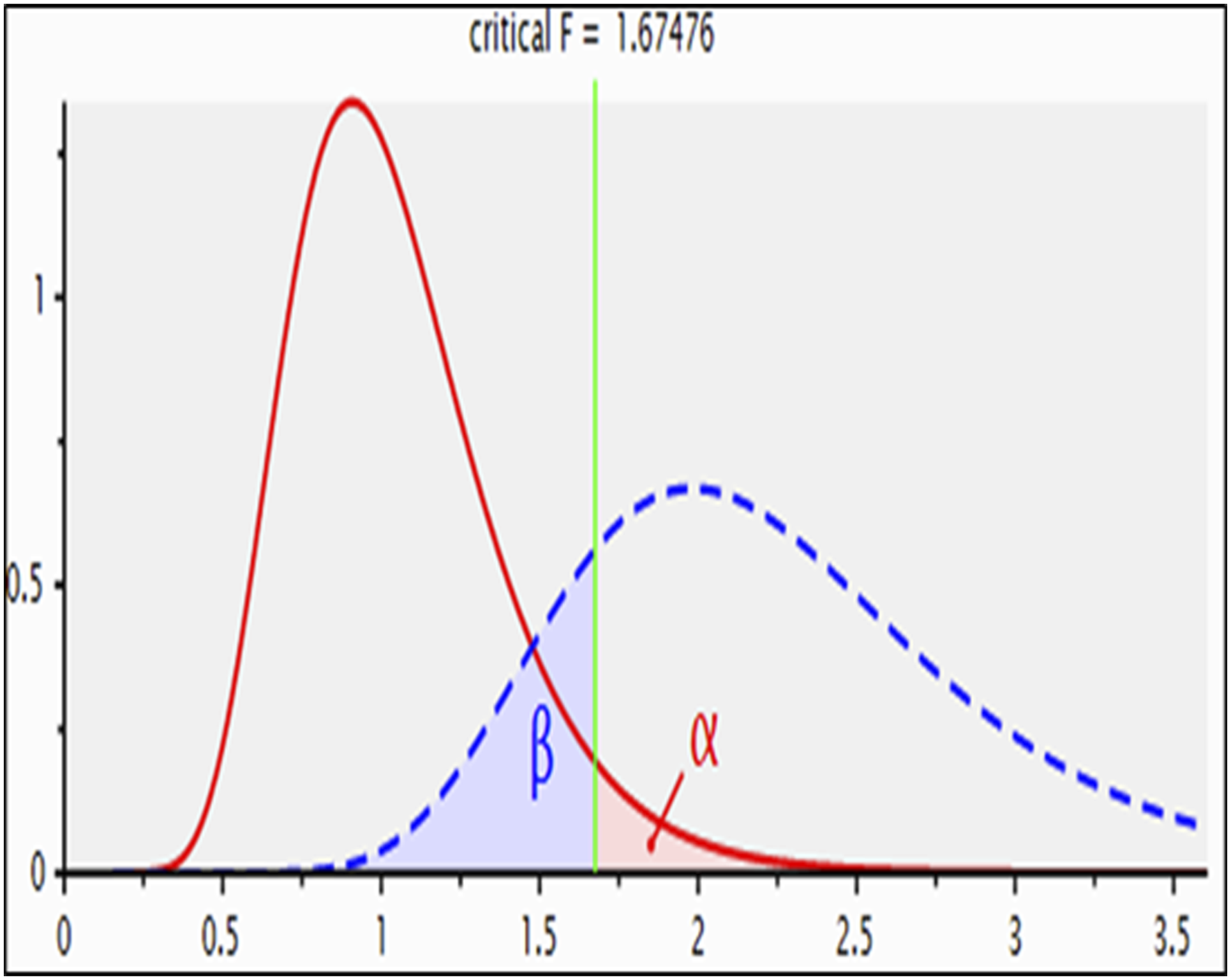
Experimental Study
A total of forty-eight (48) rats were randomly divided into six groups, (8 animals in each group). The experiment was carried out for 6 weeks.
After six weeks, rats were anesthetized under urethane anesthesia (1.2 mg/kg; intraperitoneal (i.p.). 30 Under anesthesia, the rats were then euthanized by cervical dislocation. Blood samples were collected by heart puncture for serum preparation by centrifugation at 3000 revolutions per minute (rpm) for 15 min and then stored at - 80°C for subsequent assays of alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, total protein, and gamma-glutamyl transferase (GGT) levels. Liver tissues were dissected, and washed with ice-cold isotonic saline. Part of the liver was fixed in 10 % formalin for the histopathological examination, and the second part was weighed and homogenized (10% w/v) in ice-cold phosphate buffer (0.05 M, pH 7.4), followed by centrifugation at 3000 rpm for 10 min at 4°C. The collected supernatant was stored at −80°C for analyzing oxidative stress parameters. The third part was stored at −80°C for enzyme-linked immunosorbent assay (ELISA) measurement of inflammatory biomarkers.
Biochemical Measurements
Serum Enzyme Biomarkers of Liver Injury
For assessing acute liver injury; ALT, AST, total protein, and albumin were measured according to the procedure,31,32 respectively, using enzymatic colorimetric kits (Biodiagnostics, Egypt). GGT serum level was assessed by kinetic method. 33
Oxidative Strees Biomarkers
Reduced glutathione (GSH), malondialdehyde (MDA), and nitric oxide (NOx) were assessed in liver homogenates. Reduced glutathione (GSH) was determined according to method. 34 The absorbance was read at 412 nm against a reagent blank within 5 min using a spectrophotometer, UNICAM 5625, UV/VIS, England. The results were stated as mmol of GSH/g wet tissue. The lipid peroxidation level in hepatic tissue was estimated relative to the formation of MDA following the standard protocol. 35 The NOx assay was measured for detection of the total nitrite level (intrinsic and nitrite obtained from reduction of nitrate). 36
ELISA Analysis of Inflammatory Markers
Levels of interleukin-1 beta (IL-1β), and interferon gamma (IFN-γ) were estimated in liver homogenates by using commercially available rat-specific ELISA kits (IL-1β, cat. No. E-EL-R0012, Elabscience, USA; IFN-γ, cat. no. E-EL-R0009, Elabscience, USA). The Rat IL-1β solid-phase sandwich ELISA is designed to measure the amount of the target bound between a matched antibody pair. A target-specific antibody has been pre-coated in the wells of the supplied microplate. Samples, standards, or controls are then added to these wells and bound to the immobilized (capture) antibody. The sandwich is formed by the addition of the second (detector) antibody, and a substrate solution is added that reacts with the enzyme-antibody-target complex to produce a measurable signal. The optical density (OD) is measured at wavelength 450 nm. ELISA plates are pre-coated with an antibody specific to Rat IFN-γ (Elabscience). After adding samples/standards, a biotinylated detection antibody specific for Rat IFN-γ and Avidin-Horseradish Peroxidase (HRP) conjugate are added successively to each well and incubated. The optical density of IFN- γ conjugated with the biotinylated detection antibody is measured spectrophotometrically at a wavelength of 450 nm using a plate reader (BMG Labtech, FLU Ostar Omega, Germany). The OD value is proportional to the concentration of Rat IFN-γ.
Histopathological Examination of Liver Tissues
Liver tissue specimens were fixed in 10% formol saline, then trimmed off, washed, and dehydrated in ascending grades of alcohol. The dehydrated specimens were then cleared in xylene, embedded in paraffin blocks, and sectioned at 4-6 μm thick. The obtained tissue sections were deparaffinized using xylol and stained using hematoxylin and eosin (H&E) for histopathological examination through the electric light microscope. 37 The frequency and severity of lesions in the liver were assessed semi-quantitatively as previously reported using a scale where, grade 0: No apparent injury, grade I: Swelling of hepatocytes, grade II: Ballooning of hepatocytes, grade III: Lipid droplets in hepatocytes and grade IV: Necrosis of hepatocytes. 38
Statistical Analysis
The whole data were checked for normality in addition to homogeneity of variance by means of the Kolmogorov-Smirnov test and Bartlett’s test, respectively. The data that agreed with the assumptions for parametric analysis were examined using one-way ANOVA followed by Tukey’s multiple comparisons test. The hepatic injury scores were analyzed by the Kruskal Wallis nonparametric one-way ANOVA and Dunn’s multiple-comparisons test and were expressed as the median (interquartile range). The results are presented as the mean ± Standard error of the mean (SEM). Statistical analysis was done by means of GraphPad Prism software. The level of significance was fixed at
Results
Impact of Olive l. Extract, BM-MSCs or LDR On Liver Function Tests in Rats With DSS - Induced Hepatic Injury
Impact of Olive l. Extract, BM-MSCs or LDR on Serum AST, ALT, Total Protein, Albumin and GGT Levels in Rats With DSS - Induced Hepatic Injury. Data are Presented as Mean ± SEM (n = 8). @ , * and # Indicate Significant Changes From Control , DSS and, DSS + Olive Leaf Extract Groups Respectively, at

Data are presented as mean ± SE (n = 7). @, * and # indicate significant changes from control , DSS and, DSS + OLE groups respectively, at
Influence of Olive l. Extract, BM-MSCs or LDR on Oxidative Strees Biomarkers in Rats with DSS - Induced Hepatic Injury
DSS administration caused a significant rise in MDA and NOx levels, whereas it showed a significant decrease in hepatic GSH content in comparison to normal levels. Treatment with olive leaf extract, BM Influence of olive l. Extract, BM-MSCs or LDR on oxidative strees biomarkers in rats with DSS - induced hepatic injury. (A) MDA; (B) GSH; (C) NOx. Data are presented as mean ± SEM (n = 8). @ and * indicate significant changes from control and DSS groups respectively, at 
Effect of Olive l. Extract, BM-MSCs or LDR on Inflammatory Biomarkers in Rats With DSS - Induced Hepatic Injury
Usage of 5 % DSS for a week caused significant elevations of inflammatory markers, IL-1β and INF- γ contents in hepatic tissues, indicating hepatic injury, as compared with normal levels. Moreover, rats treated with OLE, BM‐MSCs, or exposed to a low dose of radiation with DSS-induced hepatic injury exhibited significant improvements in IL-1β and INF- γ hepatic contents when compared to the DSS group ( Effect of olive l. Extract, BM-MSCs or LDR on inflammatory biomarkers in rats with DSS - induced hepatic injury. (A) IL-1β; (B) INF-γ. Data are presented as mean ± SEM (n = 8). @ ,* and US dollar indicate significant changes from control, DSS and DSS + Stem cell groups respectively, at 
Histopathological Results of Liver Tissues
The liver tissue section of the control group showed normal histological structure of hepatic lobules and organization of hepatic cords with a prominent central hepatic vein. Polygonal hepatic cells were joined to 1 another in anastomosing plates, with borders that face either the sinusoids or adjacent hepatocytes (Score 0).
Liver tissue sections of the DSS group revealed disorganization of hepatic cords and necrobiotic changes of hepatocytes, characterized by nuclear pyknosis and granularity of cytoplasm. Severe multifocal >¾ affected lobules showed necrosis. Apoptosis of some hepatocytes which appeared as eosinophilic bodies scattered all over the hepatic lobules. Numerous numbers of intracellular fat droplets were also noticed in score (4).
Liver tissue section of stem cell group revealed normal histological picture of control animals’ group without any significant pathological alterations score (0).
Liver tissue section of the DSS + BM-MSCs group showed mild swelling of hepatocytes and granularity of its cytoplasm with centrally situated vesiculated nuclei with peripheral condensation of nuclear chromatin. Minimal-mild focal, limited to centrilobular region, and less than ¼ of affected lobules revealed necrosis. Narrowing of hepatic sinusoids without significant necrosis or apoptosis was also noticed (score 1).
On the other side, a liver tissue section of DSS + OLE showed revealed pyknotic and lytic changes of hepatocytes nuclei. Mild to moderate focal and multifocal, central to midzonal lobular region ½ affected lobules are necrotic. Few numbers of apoptotic cells were scattered all over hepatic lobules. Dilatation of hepatic sinusoids and hyperplasia of Kupffer cells were also seen score (2).
Liver tissue section of DSS + LDR showed mild injury of hepatic lobes resembling the untreated group (DSS). Centrolobular zone showed fatty degeneration, which presented as fat globules occupying less of hepatocytes cytoplasm with peripheral eccentric nuclei score (1) Figure 4 & Table 2. Photomicrograph of hepatic tissue section showing: (A) normal histological structure of hepatic lobules and organization of hepatic cords Scores With Descriptions on Histopathological Changes in Liver Cells; Scores Range From 0 (Normal Hepatocytes) to 5 (Severe Hepatocytes Loss).

Discussion
It is generally recognized that a variety of liver illnesses are linked to addictive behaviors such as alcohol drinking, smoking, and, to a lesser extent, dietary imbalance and obesity, viral and parasite infections, and occupational chemical exposure. According to recent research, environmental exposures such as air pollution, pollutants, and physical stresses are significantly connected with liver disease. Furthermore, microbial metabolites and the “gut-liver” axis play an important role in liver illnesses. 39 In the current study, we investigate the possible therapeutic role of olive oil extract, BM-MSCs, or LDR on liver injury induced by acute ulcerative colitis in male rats. The beneficial effects of treatment with BM-MSCs, or LDR and the possible mechanistic pathways against DSS-induced hepatotoxicity were studied in this work. DSS brought pathological liver damage along with impairment of hepatic function tests, specified by increased serum levels of AST, ALT and GGT, besides a reduction in serum total protein and albumin. These observations are in agreement with other previous studies which reported that DSS causes adverse liver reactions accompanied by an increase in liver function tests,40-42 further supporting that the liver injury is the extra intestinal manifestation of colitis.
Conversely, treatment with OLE, BM-MSCs, or exposure to LDR utilizes significant alleviation in all hepatic biomarkers as compared to the DSS-treated group alone. Previous study demonstrated the efficacy of BM-MSCs in the amelioration of liver damage through reducing inflammatory cells in the hepatic tissue and decreasing the alanine aminotransferase level. 43 It has been accepted that the radiation hormesis phenomenon exists, which means that exposure to low dose ionizing radiation stimulates beneficial biological effects. 44 Therefore, it was reported previously that the changes in serum ALT and AST levels were mitigated by LDR exposure, presumably by maintaining hepatocyte membrane integrity. 45 Moreover, treating the hepatotoxic rats with olive oil reversed the activity of transaminases and restored them towards normal values, indicating maintenance of functional integrity of the hepatic cell membrane. 46
Regarding the oxidative stress caused by DSS, it was reported that after DSS administration, ROS levels in the colon mucosa were significantly increased. 47 In general, DSS increases oxidative stress and suppresses the antioxidant response in acute colitis models induced by 5% DSS. 48 Our findings showed that DSS administration caused a significant rise in MDA and NOx levels with a significant decrease in hepatic reduced GSH content in comparison to normal levels. This is consistent with the aforementioned studies.49,50 These results suggest that, in the DSS-induced ulcerative colitis model, oxidative stress and lipid peroxidation are increased in the colon and hepatic tissue due to the activation of neutrophils and macrophages, while the antioxidant defense system is greatly suppressed.48,51
Instead, treatment with olive leaf extract, BM
An earlier study showed that DSS-induced colitis can disrupt the gut-vascular barrier and aggravate hepatic inflammation and fibrosis. 69 In the current study, usage of 5% DSS for a week caused significant elevations of inflammatory markers, IL-1β and INF- γ contents in hepatic tissues, indicating hepatic injury, as compared with normal levels. These results were consistent with previous reports70-72; thus, we speculated that DSS alone mainly induced liver injury by mediating the inflammation. A considerable number of studies have reported that liver abnormalities, including inflammation and fibrosis, are typical extra-intestinal manifestations of colitis and are frequently observed.73,74 Additionally, a recent study has also reported that the administration of DSS changed the expression of pro-and anti-inflammatory cytokines such as IL-1β, IL-10, and transforming growth factor beta-2 (TGFb-2), suggesting that intestinal inflammation led to liver inflammation 75 Due to the altered intestinal permeability, intestinal inflammation caused by DSS led to the passage of bacterial products into the liver, and in turn, might promote the activation of Toll-like receptor 4 (TLR4) signaling and Hematopoietic stem cells (HSCs) to induce liver inflammation and fibrogenesis. During hepatic inflammation, hepatic macrophages are activated 76 and then promote the release of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β to induce liver injury. 77
Alternatively, rats treated with OLE, BM‐MSCs, or exposed to LDR with DSS-induced hepatic injury exhibited significant improvements in IL-1β and INF- γ hepatic contents when compared to the DSS group. Furthermore, the current results revealed that treatment with BM‐MSCs, or exposure to LDR amended hepatic injury induced by 5% DSS while equated to the group of rats treated with DSS + OLE. These results suggested that BM‐MSCs as well as low doses of radiation (LDR) could suppress the inflammation response to improve colitis-induced liver injury. These findings may be attributed to the anti-inflammatory effects exerted by both BM-MSCs and low dose irradiation via inactivation of the NF-κB pathway, which suppresses inflammatory cytokine release, thus ameliorating tissue damage.
As previously mentioned, BM-MSCs may suppress inflammation processes in different ways, through the upregulation of anti-inflammatory factors or the downregulation of proinflammatory factors. In addition, BM-MSCs may also suppress immune reactions via direct cell contact. 78 Besides, BM-MSCs can perform multiple functions, including immunomodulation, homing, and differentiation, to enable damaged tissues to form a balanced inflammatory and regenerative microenvironment under severe inflammatory conditions. 79 BM-MSCs have a favorable effect by releasing cytokines and chemokines that aid to reduce inflammation, fibrosis, and oxidative stress caused by the disease. 80 Co-culture with MSCs reduces pro-inflammatory cytokines like TNF-α, IFN-γ, IL-6, and IL-17 while increasing IL-10 and TGF-β levels, according to previous studies.81,82 Low dose radiation may have a modulatory effect on the endothelial cells, which are very sensitive to radiation, 83 which in turn modulates the pro-inflammatory mediator TNF-α, and consequently a wide variety of pro inflammatory cytokines, chemokines, and growth factors. 84 Previous research found that low dose γ-irradiation has a significant anti-inflammatory effect, reducing IL-1β levels in the colon to near-normal values.85,86 Former results strongly suggested that OLE contains polyphenols such as oleuropein and hydroxytyrosol reverses the chronic inflammation and oxidative stress.87-89
To confirm the aforementioned results, histological studies for liver tissues were performed. The histological abnormalities, including extracellular matrix (ECM) deposition, fibrogenesis, inflammatory cell infiltration, and the enhanced inflammatory response caused by colitis, were in line with further studies.40,90,91 In addition, according to the result from hepatic histopathology, we observed that administration with BM‐MSCs could effectively alleviate liver tissue injury and reduce inflammatory infiltration.
92
The damaged hepatocytes were repaired and fibrosis was resolved, resulting in an overall improvement in liver function.
93
Meanwhile, low dose gamma irradiation decreased the harmful effects on liver tissues,94,95 The ameliorative effects of LDR on structural changes might be attributed to its role in the modulation of essential cellular activities, including cell growth and differentiation.
45
This effect might be attributed to the reduction of DNA damage induced early after irradiation and activation of anti-oxidative and anti-apoptotic mechanisms via modulating the nuclear factor E2-related factor 2 (Nrf2)-mediated antioxidant response pathway, and mitochondria-mediated caspase activation and apoptosis. Moreover, when diabetic rats were treated with
In terms of BM-MSC therapeutic applications, stem cell treatment can temporarily improve liver function. Stem cell therapy can help patients with end-stage liver disease wait longer for liver transplants and serve as a bridge between the two. Repeated stem cell therapy could be a viable strategy for achieving long-term therapeutic effects. 97 LDR is currently used in cancer for inflammation-related disorders. LDR has lately been used in several trials to guard against lung inflammation following COVID-19 infection and to treat degenerative disorders such as Alzheimer’s disease. It was shown that a low dose can cause a biological effect greater than a higher dose. 98
More research is needed to identify the molecular processes of BM-MSCs or LDR as liver-protective therapies. Large-scale clinical trials are required to prove the therapeutic potential of MSCs, which are still in their early stages in treating liver illness, and to elucidate the role of stem cells in liver disease. Low dosage γ-radiation may have a preventive effect against liver injury. Yet, further research is needed to determine the long-term impacts of this low dose.
Conclusion
The findings suggest that both BM-MSCs and LDR can effectively treat liver impairment caused by acute ulcerative colitis. Both BM-MSCs and LDR showed greater improvement in terms of oxidative stress, inflammatory indicators, and hepatic biomarker management. As a result, it could be used as an effective treatment strategy to treat hepatic damage. These findings encourage us to develop an effective cell therapy for the treatment of liver disease. The hepatoprotective action of OLE can be linked to the presence of biologically active substances that have the ability to scavenge free radicals.
Footnotes
Acknowledgments
The authors are very grateful to the Egyptian Atomic Energy Authority for supporting the authors until their work has been carried out. The authors would like to acknowledge Prof. Dr Ahmed Othman; Professor of Histopathology, Faculty of Veterinary Medicine, Cairo University for his efforts in accomplishing the histopathological tissue examinations of the current study.
Authors’ Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
