Abstract
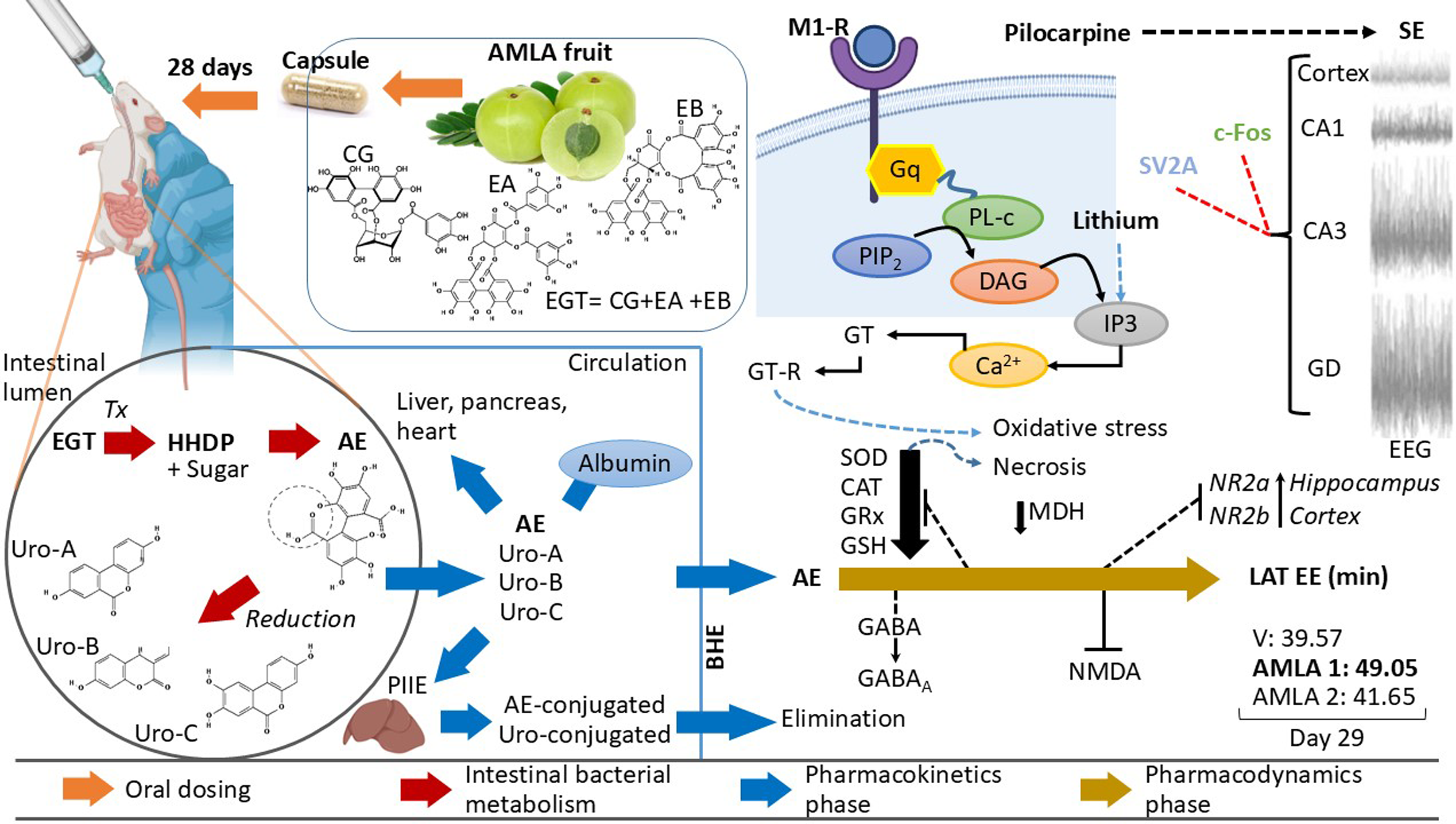
Introduction
Epilepsy is the fourth most common chronic neurological disorder, affecting approximately 65 million people worldwide. 1 It involves abnormal electrical activity that can lead to epileptic seizures and, in some cases, loss of consciousness, exacerbating cognitive and social impairments in the patient. 2 Among the most susceptible brain areas in epilepsy are the temporal lobe, superior frontal lobe, inferior occipital lobe, hippocampus, and amygdala. 3 Seizures can originate from a specific region (focal seizures) or have a point of origin that quickly spreads to the rest of the brain, resulting in generalized seizures. One of the main neurotransmitters involved in epileptogenesis is glutamic acid, which enhances and sustains neuronal excitability by binding to its receptors (i.e., AMPA, NMDA, and KAINATE). Conversely, the neurotransmitter γ-aminobutyric acid (GABA) exhibits reduced inhibitory activity on postsynaptic neurons, either due to decreased synthesis or diminished efficacy of postsynaptic receptors.4-6 This imbalance facilitates the depolarization of the neuronal membrane by lower-intensity stimuli, leading to the rapid propagation of action potentials.4-6 The brain is particularly susceptible to oxidative stress and is the most aerobically active organ in the body because of its high metabolic demands and/or inflammatory processes. 7 Evidence suggests that neuronal hyperexcitability and oxidative damage resulting from excessive free radical production may play a role in the onset and progression of epilepsy. 7 The primary treatment involves the chronic administration of antiepileptic drugs (AEDs) to prevent or reduce the severity of epileptic seizures and slow the progression of epilepsy. 8 However, their continuous use is associated with serious side effects, such as liver and/or renal damage, as well as psychiatric and cognitive disorders. 9 Neuroprotective or antioxidant compounds may have positive effects when administered concurrently with AEDs. 7 Their co-administration may contribute to safer and more effective treatments to mitigate the adverse effects of AEDs. This is exemplified by Emblica officinalis Gaertn (AMLA), which has anti-inflammatory, antidiabetic, hypolipidemic, hepatoprotective, cardioprotective, immunomodulatory, and neuroprotective properties in animal models. 10 Additionally, in vitro and in vivo studies indicated that AMLA restores enzymatic and non-enzymatic antioxidant levels, eliminates free radicals, and reduces lipid peroxidation. 11
Considering its antioxidant activity, the pharmacological prospects of AMLA suggest its therapeutic utility in diseases related to oxidative stress, such as neurological disorders and neurodegenerative diseases. 12 In Wistar rats with cognitive impairment induced by a high NaCl and cholesterol diet for eight weeks, oral treatment with 200 mg/kg/day AMLA extract enriched in tannins (70% w/w of emblicanin A/B) for seven weeks induced the expression of nuclear erythroid 2-related factor 2 (Nrf2) in the cortex and Cornus amon (CA1) area of the hippocampus, correlating with the neuroprotective effect of AMLA. 13 Preclinical studies have indicated an increase in the latency of epileptic seizures and suppression of generalized tonic-clonic seizures in experimental subjects treated with this plant.14,15 In the experimental pentylenetetrazol model, male Wistar rats treated intraperitoneally (i.p.) with 500 mg/kg and 700 mg/kg of AMLA hydroalcoholic extract, once daily for seven days, did not exhibit bilateral tonic-clonic seizures (previously referred to as “secondarily generalized partial seizures”). 12 Similarly, administration of 300, 500, and 700 mg/kg i.p. for seven days increased the latency to bilateral tonic-clonic seizures in adult Wistar rats treated with kainic acid (KA). 12 However, studies supporting the anticonvulsant effect of AMLA in other models of generalized seizures and considering the potential alterations in kidney and liver physiology and hematological parameters are needed. Therefore, the objective of this study was to evaluate the anticonvulsant effect of the chronic oral administration of Emblica officinalis by assessing the behavioral parameters of the status epilepticus (SE) induced by lithium-pilocarpine injections in male Wistar rats. Additionally, the effects on biochemical and hematological parameters were evaluated using blood chemistry, liver function tests, and a complete blood count in adult rats. The study of the effect of this plant on epileptic seizures could contribute to the development of complementary therapeutic strategies for epilepsy treatment and, in the future, improve the quality of life for patients with this neurological disorder.
Materials and Methods
Preparation and Administration of Emblica officinalis Doses
Doses used in present study were based on previous research indicating that 500 and 700 mg/kg of the plant extract suppress bilateral tonic-clonic seizures induced by pentylenetetrazole (PTZ) (60 mg/kg, i.p.) 14 and increase latency to the first generalized seizure induced by KA (10 mg/kg, i.p.),12,15 for this reason those doses were included in the experimental design. Regarding the biological plant material used in the present study, it was not collected in the wild, so it was not deposited in an herbarium or public collection, as Emblica officinalis Gaertn was purchased in a food supplement formulation under the trade name AMLA (Nature’s Way Organic India USA, Boulder 80301, CO, USA; UPC Code: 851469000717), each capsule containing 1000 mg from standardized dried extract of the plant. The manufacturer authenticated the contents of the container as Emblica officinalis Gaertn under batch number 801983. 16 Nature Mexico’s Way laboratories are regulated by COFEPRIS Federal Commission for the Protection against Sanitary Risks (Mexico) and have the corresponding Sanitary Permit. Therefore, a taxonomist did not authenticate it, and a voucher was not needed.
The AMLA capsules (1000 mg) were dissolved in purified water. For the preparation of the 700 mg/kg doses, the contents of the capsule were diluted with the vehicle, and its contents were brought to a volume of 1.43 mL (note: 1 mL of the working solution was used for 1 kg body weight of the animal, and a dose of 700 mg/kg was obtained). Next, 1 mL of the working solution was taken and, by rule of three, the volume required for a rat of 300 g body weight was obtained which is equivalent to 0.3 mL of the working solution. For the preparation of the 500 mg/kg doses, the contents of the capsule were diluted with the vehicle, and the contents were brought to a volume of 2 mL (note: 1 mL of the working solution was used for 1 kg body weight of the animal, and a dose of 500 mg/kg was obtained). In summary, all animals received the same volume of the treatment according to the body weight. The solution was shaken for 5 min until complete dissolution was achieved before each administration.
Experimental Animals
Twenty-eight male Wistar rats, weighing between 250 and 300 g, were housed in translucent acrylic cages (44 cm × 33 cm base, and × 25 cm height) in groups of 7 individuals and maintained at the Laboratory of Pharmaco-toxicology, Faculty of Pharmaceutical Chemical Biology, Xalapa region, maintaining an ambient temperature of 25 ± 2°C and a 12 × 12 hours light/dark cycle (lights on at 6:00 a.m.). The rats had ad libitum access to food and water.
Experimental Groups and Manipulations
For the selection of the number of animals per group, the 3 Rs of Russell and Burch, 2015 17 were taken into consideration (reduce, replace, and refine). In this sense, the number of animals per groups was based on previous studies in which it has been identified that several 6 to 7 rats per group allow the identification of behavioral and biochemical changes associated with convulsant and anticonvulsant treatments18-20 without compromising the power of statistical tests. A control group received purified water, two additional groups received AMLA 500 mg/kg and AMLA 700 mg/kg, and the last group (CBZ) received 300 mg/kg of carbamazepine as a pharmacological control of anticonvulsant drugs. For oral administration, a rounded-tip polyethylene intragastric cannula (4 cm in length, 1 mm in diameter; S-54-HLCole-Parmer, Vernon Hills, IL, USA) was used with an appropriate size and gauge, according to the animal’s size and weight. 16 The animal was securely held with its head and neck straight and its nose pointing upward. The cannula was passed between the incisors and premolars into the esophagus, where solutions of the extract and vehicle were deposited for the animal to ingest. At each dose, a maximum volume of 2 mL of purified water was used as the vehicle. 21 If resistance or choking occurred, the cannula was repositioned before continuing. 16
These treatments were administered every 24 h for 28 days. A dose of 300 mg/kg of carbamazepine (CBZ 300 mg/kg) was included on days 20, 22, 24, 26, and 28 as a pharmacological control for the anticonvulsant activity.22,23 On day 29, all experimental subjects underwent SE induction using the lithium-pilocarpine model, and behavioral assessments were performed.
Induction of the Status Epilepticus with Lithium-Pilocarpine
On the last day of administration in each experimental group, animals were treated intraperitoneally (i.p.) with lithium chloride 127.2 mg/kg (Meyer Reagents, Mexico City, Mexico) 20 h before the injection of pilocarpine hydrochloride (30 mg/kg, s.c.; Sigma-Aldrich, St. Louis, MO, USA). These drugs were administered using 1 mL syringes with 27G x 13 mm needles (Terumo Medical, Mexico City, Mexico). Behavioral manifestations of epileptic seizures were monitored for 60 minutes 24 and recorded after the administration of pilocarpine according to the scale reported by Racine (1972) 25 :
Phase 0, immobile and staring behaviors were defined as grade.
Phase I, facial myoclonus, winking ipsilateral to the site of stimulation and/or chewing.
Phase II, phase I behaviors and head myoclonus, repeated head downward tilting, nodding.
Phase III, Phase II behaviors and forelimb myoclonus
Phase IV, all the above behaviors and “kangaroo posture”.
Phase V, phase IV behaviors, loss of postural tone, and falling.
SE was considered when convulsive activity was continuous, reached at least stage IV or V severity, and was sustained for at least 30 min. The variables evaluated in the behavioral tests were the percentage of rats that developed SE, latency and duration to the first generalized seizure (stage IV or V), latency to SE, and the number of stage IV and V seizures.
Blood Sample Collection
Upon completion of the behavioral test, animals underwent deep anesthesia using a sodium pentobarbital overdose (120 mg/kg, i.p.; Laboratorios Aranda, S.A. de C.V., Queretaro, Mexico; Reg. SAGARPA Q-0449-124) until reaching an unconscious and pain-free state, with the absence of a palpebral reflex response or pinch response of the paw and tail pad. Once deep anesthesia was verified, a cardiac puncture was performed to obtain a blood sample using a 5 mL syringe with a 22 G × 32 mm needle. Blood was deposited in dry tubes (without anticoagulants) and tubes containing the anticoagulant ethylenediaminetetraacetic acid (EDTA; BD Vacutainer, Mexico City, Mexico). Subsequently, biochemical and hematological analyses were performed. Finally, rat death by pentobarbital overdose was confirmed, and the rat was disposed of according to the official guidelines for the management of biological and infectious waste (NOM-087-ECOL-SSA1-2002). 26
Processing of Blood Samples
Once the blood samples were obtained, they were deposited in dry tubes (without anticoagulants) to allow the blood to clot. Subsequently, the tubes were centrifuged at 3500 rpm for 5 min to obtain serum, which was immediately transferred with a Pasteur pipette to the corresponding containers for dry chemistry tests on Vitros 350 equipment (Johnson & Johnson, Ramsey, MN, USA). For a complete blood count, blood was transferred to tubes with EDTA and homogenized by immersion (8-10 times). All samples were then processed in closed automatic mode on a Cell-Dyn Emerald analyzer (Abbott Laboratories, Santa Clara, CA, USA). The test results were analyzed and compared with the corresponding biological reference intervals for Wistar rats to detect hepatic, renal, and hematological alterations.27–30
Statistical Analysis
The data were analyzed using the Normality Test and Equal Variance Test. Those that passed these tests were analyzed using one-way ANOVA, and those that failed were analyzed using the Kruskal-Wallis test, considering treatments as a single factor, using Sigma Stat software version 4.0. When significant differences were reported by ANOVA and Kruskal-Wallis tests, Tukey’s post-hoc test for multiple comparisons was performed. Statistical significance was set at P < 0.05. Results were expressed as the mean ± standard error of the mean (SEM).
Results
Behavioral Evaluation of the Status Epilepticus (SE)
The group treated with CBZ 300 mg/kg and three rats from the AMLA 500 mg/kg group did not develop generalized stage IV or V seizures or SE. However, a cut-off time of 60 min was used as the latency value for the first generalized seizure and SE in these cases. Regarding the duration of the first generalized seizure and the number of stage IV and V seizures, these subjects were assigned a value of 0. Regarding the latency to the first generalized seizure (phase IV or V) and SE induction, significant differences were observed among treatments [H = 25.74, 3; P < 0.001] and [H = 22.07, 3; P < 0.001], respectively. Post hoc testing revealed that AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg groups exhibited greater latency than the vehicle group did. Regarding the duration of the first generalized seizure (phase IV or V), differences were observed among the groups [F (3, 27) = 89.396, P < 0.001]. Post-hoc analysis revealed that the AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg groups exhibited shorter durations of the first generalized seizure than the vehicle group did. Moreover, examining the number of phase IV and phase V generalized seizures induced by pilocarpine administration up to one hour after SE onset, significant differences were identified among treatments [H = 14.33, 3; P < 0.002] and [H = 20.72, 3; P < 0.001], respectively). Specifically, the CBZ-treated group displayed a lower number of phase IV seizures at a dose of 300 mg/kg than the vehicle control group did. Additionally, the groups treated with 500 mg/kg AMLA and 300 mg/kg CBZ exhibited a lower number of phase V seizures than the vehicle control group did (Figure 1). Behavior of SE. (A) Percentage of experimental subjects that developed SE. Data are presented as the percentage of animals that exhibited SE in the different experimental groups. (B) Time to the onset of the first generalized seizure, comparison of groups: vehicle vs AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg through Kruskal-Wallis followed by Tukey’s post hoc test. (C) Time to SE induction, comparison of groups: vehicle vs AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg through Kruskal-Wallis followed by Tukey’s post hoc test. (D) Duration of the first generalized seizure: vehicle vs AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg using one-way ANOVA followed by Tukey’s post hoc test. (E) Number of stage IV and stage (F) V generalized seizures, comparison of groups: vehicle vs AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg through Kruskal-Wallis followed by Tukey’s post hoc test. Data are expressed as mean ± SEM. *P < 0.05, compared to the vehicle group.
Blood chemistry
Regarding the parameters included in this section, the values were within the biological reference interval. ANOVA showed significant differences among the AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg groups [F (3, 24) = 5.916, P = 0.004] for glucose. However, the post-hoc test did not reveal significant differences between CBZ group the study and the vehicle groups. For the remaining analytes, statistical analysis did not show significant differences among the groups: creatinine [F (3, 24) = 3.088, P = 0.046], urea [F (3, 24) = 3.199, P = 0.041], and blood ureic nitrogen (BUN) [F (3, 24) = 2.294, P = 0.103] (Table 1).
Liver Function Tests
Blood Chemistry Parameters in Wistar Rats After SE Induction.

Note: Data are expressed as mean ± SEM. *P > 0.05.
Liver Function Parameters in Wistar Rats After SE Induction.
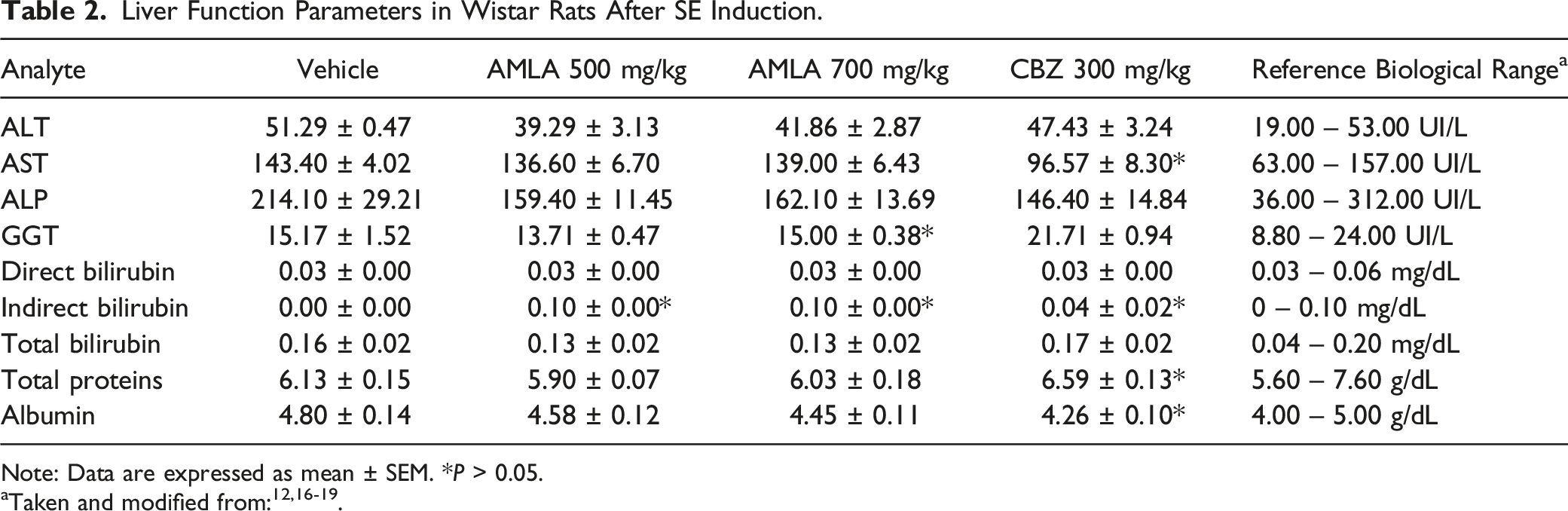
Note: Data are expressed as mean ± SEM. *P > 0.05.
Hematological and Leukocyte Differential Cytometry
Values of Hematological and Leukocyte Differential Cytometry in Wistar Rats After the Induction of SE.
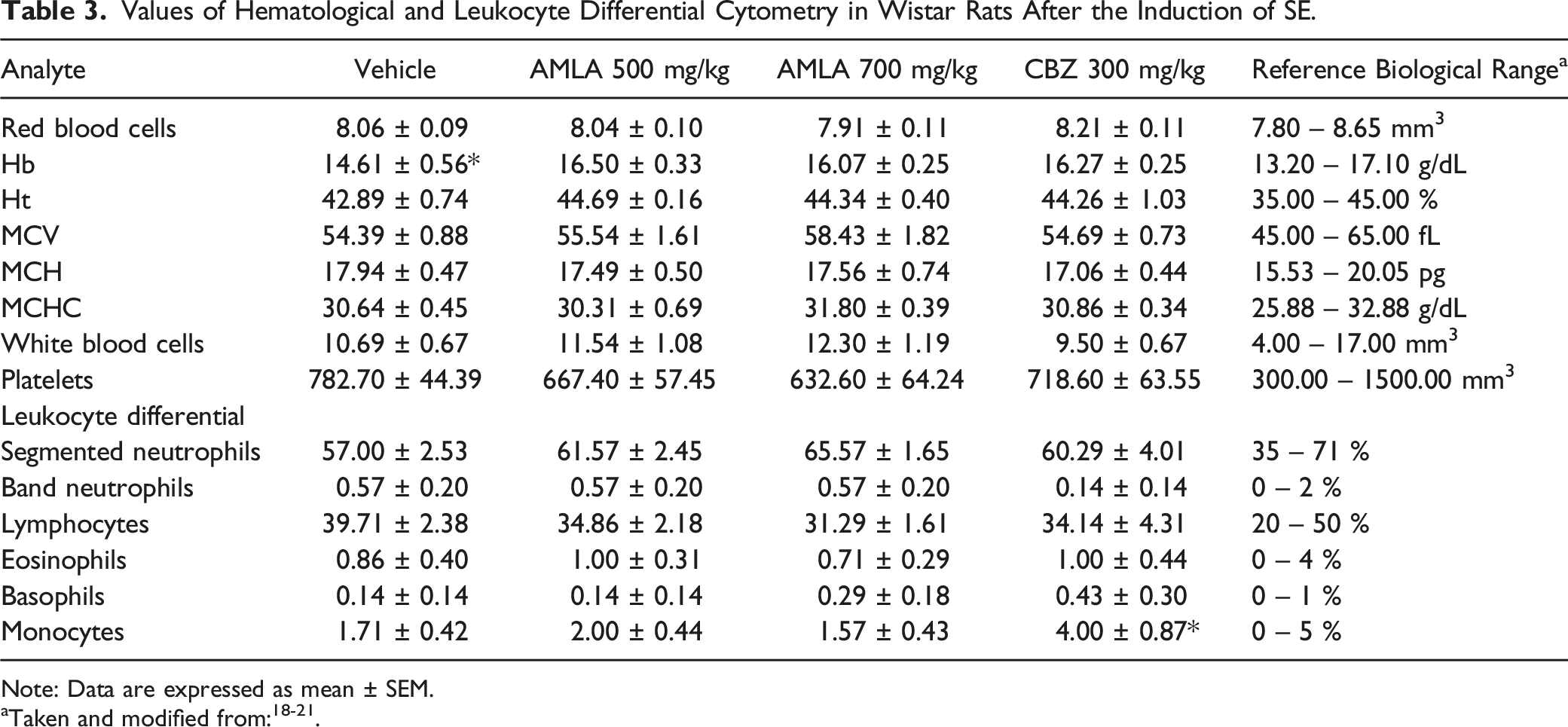
Note: Data are expressed as mean ± SEM.
The AMLA 500 mg/kg, AMLA 700 mg/kg, and CBZ 300 mg/kg groups showed higher hemoglobin (Hb) concentrations than the vehicle group did [F (3, 24) = 5.288, P = 0.006]. Finally, the CBZ 300 mg/kg group exhibited a higher number of monocytes than the AMLA 700 mg/kg and vehicle groups did [F (3, 24) = 3.912, P = 0.021].
Discussion
The objective of the present study was to evaluate the anticonvulsant effects of chronic oral administration of the standardized extract of Emblica officinalis and its effect on biochemical and hematological parameters in male Wistar rats with lithium pilocarpine-induced SE. The results demonstrated that chronic oral treatment with 500 mg/kg AMLA had an anticonvulsant effect, and nearly half of the animals in this group (42.86%) did not exhibit SE. Furthermore, the subjects in this group who experienced seizures showed a reduction in the number of generalized phase V seizures and an increase in the latency to SE. In addition, the AMLA 500 mg/kg and AMLA 700 mg/kg groups exhibited an increase in latency to the first generalized seizure and a shorter duration of generalized epileptic activity. These findings suggest that AMLA exerts anticonvulsant effects in the lithium-pilocarpine model, indicating its potential as an alternative for epilepsy treatment without adverse effects on hepatic and renal functions under the study conditions. Individuals in the CBZ 300 mg/kg group did not develop stage IV or V generalized seizures, or SE.
Carbamazepine is used for the treatment of focal-onset epilepsy and for controlling generalized tonic-clonic seizures. 33 In both lithium pilocarpine-induced SE and an experimental model of epileptogenesis with KA, carbamazepine blocked the development of motor seizures and reduced the frequency of generalized seizures secondary to SE. 34 This effect depends on the carbamazepine dosing regimen (dose and dosing interval) and protocol for the convulsive activity induction protocol. 33
A dose of 100 mg/kg (i.p.) of carbamazepine reduced the frequency of non-motor or focal seizures (Racine phases I and II) and motor seizures (Racine phases III to V), including generalized tonic-clonic seizures, resulting from the administration of multiple doses of KA in Sprague-Dawley rats. 34 Additionally, Osuntokun et al. 33 administered carbamazepine (25 mg/kg, i.p.), levetiracetam (50 mg/kg, i.p.), and carbamazepine-levetiracetam (12.5 and 25 mg/kg, respectively) 30 min before pilocarpine hydrochloride injection (300 mg/kg) in Wistar rats. In these groups, an increase in latency and a reduction in SE duration, along with a lower number of stage IV and V seizures (Racine scale), 25 were observed. 33 This effect was also reported in C57 mice treated with 10 mg/kg oral carbamazepine 60 min before pilocarpine hydrochloride injection (300 mg/kg, p.o.). 35
However, few studies have evaluated the anticonvulsant effects of AMLA in animal models. Administration of 500 and 700 mg/kg of the hydroalcoholic extract of AMLA once daily for 7 days (i.p.) significantly increased the latency to myoclonic seizures and blocked the development of clonic-tonic generalized seizures induced by PTZ (60 mg/kg, i.p.) in male Wistar rats. 10 Furthermore, treatment with 500 mg/kg and 700 mg/kg (i.p.) once daily for 7 days dose-dependently increased the latency to motor seizures in rats administered KA (10 mg/kg, i.p.). 15
The results of the present study showed that AMLA 500 mg/kg treatment modified a greater number of SE behavioral parameters than AMLA 700 mg/kg did. Generally, the intensity of a therapeutic effect is related to the drug concentration at the site of action; however, this effect does not surpass the maximum efficacy value, even at increased doses.35,36 This is known as the “ceiling” effect, which has not been reported in PTZ- and KA-induced convulsive activity models using doses of 500 and 700 mg/kg AMLA.14,15 In contrast, drug-receptor interactions can be modified by the internalization of GABAA receptors during SE generated with lithium-pilocarpine.37,38 Additionally, a decrease in the expression of GABAA receptors in hippocampal tissue was observed in this model. 37 This suggests an imbalance in inhibitory function due to endocytosis of GABAA receptors, a hypothesis that explains the ineffectiveness of GABAA agonists, such as benzodiazepines, in SE treatment.38,39
AMLA has been reported to contain ellagic acid and corilagin, which contribute to its anticonvulsant effect.13,40-43 Acute administration of 100 mg/kg ellagic acid (i.p.) increased the required dose of PTZ (i.p.) to promote the development of the first myoclonic and clonic seizures with the loss of the postural reflex. 42 In addition, oral treatment with 50 mg/kg ellagic acid in nanocapsules once a day for one week inhibited the development of clonic-tonic generalized seizures in mice treated with 37.5 mg/kg PTZ (i.p.). 43 Finally, administration of corilagin once daily at 15 mg/kg (i.p.) for 35 days reduced the frequency and duration of convulsive seizures in male Wistar rats 30 min before the injection of PTZ at 35 mg/kg (i.p.). 41 These compounds (ellagic acid and corilagin) may be responsible for the results of this study.
Effect of AMLA on Blood Chemistry
In this study, no alterations were found in creatinine, urea or BUN parameters, suggesting that there was no risk, injury, or impairment in renal function under these experimental conditions. This aligns with the findings of a study by Rahman et al., 40 in which AMLA administration in powdered food (2.5% w/w of the fruit) for two weeks did not alter the creatinine level in Long-Evans rats. Additionally, in 2K1C (two kidneys and one clip) hypertensive nephropathy rats with renal tissue fibrosis, creatinine levels remained within the reference biological range in the AMLA-treated group. 40 In another study, BUN and creatinine levels were within the reference values in 10-month-old male Wistar rats with tubular atrophy, glomerular sclerosis, or interstitial fibrosis treated with oral ethyl acetate and aqueous extracts of AMLA (10 and 40 mg/kg/day, respectively) for 100 days. 44 This result, along with a reduction in histological kidney damage, was reported in male Wistar rats with cisplatin-induced nephrotoxicity (12 mg/kg, i.p.), which were treated with AMLA at 200 and 400 mg/kg (p.o.) for 14 days. 45 Glucose allows the diagnosis of metabolic disorders such as diabetes mellitus. 46 In our study, the experimental subjects showed a decrease in this analyte (glucose), however, the concentrations were within the reference range. Thus, we can affirm that the treatments did not produce significant biological changes in glucose that could have a negative impact on the health of the animals. Ansari et al. 46 administered streptozotocin at 90 mg/kg (i.p.) in 48-hour Long-Evans rats to induce diabetes mellitus. After oral treatment for eight weeks with a hydroalcoholic extract of AMLA at 1.25 g/kg/day, 7-month-old rats showed basal blood glucose, creatinine, and ALT values within reference values 46 as in the present study.
Effect of AMLA on Liver Function Tests
In this study, no variations were found in the analytes comprising the liver function tests with prolonged AMLA consumption, suggesting its safety under the experimental conditions employed as it did not induce hepatotoxicity. This is supported by the findings of other research groups that have used animal models of chronic diseases or hepatotoxicity. In a study by Huang et al., 47 oral treatment with aqueous AMLA extract (250 and 500 mg/kg) and gallic acid (100 mg/kg) for eight weeks resulted in a reduction in transaminase levels in Sprague-Dawley rats that developed steatosis due to a high-fat diet (40% beef tallow). Similarly, oral administration of AMLA (125, 250, and 500 mg/kg) for 20 weeks reduced ALT and AST levels in C57BL/6 mice with non-alcoholic steatohepatitis induced by methionine and choline-deficient diet. 48 Furthermore, oral treatment with AMLA (1800 and 3600 mg/kg) once daily for 8 weeks decreased ALT and AST levels in Wistar rats that developed hepatic fibrosis following chronic administration of carbon tetrachloride (CCl4, 2 mL/kg, i.p.). 49
Effect of AMLA on Hematological Parameters
Regarding these parameters, the results in the experimental groups of this study slightly differ from the values reported by Anto et al. 50 : Hb (14.61 to 16.50 and 15.46 to 18.49 g/dL, respectively), HCT (42.89 to 44.69 and 55.65 to 58.94%, respectively), MCV (54.39 to 58.43 and 58.41 to 73.10 fL, respectively), and MCHC (30.31 to 31.80 and 24.30 to 26.70 g/dL, respectively). This variation is attributed to biological differences related to sex, age, strain, and altitude (meters above sea level). 31,32 in which the experiment was performed. Anto et al. 50 used male and female Sprague-Dawley rats treated with AMLA (2000 and 5000 mg/kg) for 12 days orally, and the study was conducted in Sumatera-Utara, Indonesia (1024 meters above sea level), with a lower altitude than that of Xalapa, Ver., Mexico (1400 meters above sea level). Meanwhile, Sarvaiya et al. 51 obtained lower values than those in the present study for HCT and MCV (42.35% and 53.50 fL, respectively) in male Wistar rats treated with AMLA (200 and 400 mg/kg, i.p.) for 28 days at an altitude of 39 m above sea level (Anand, India).
Regarding the white blood cell series, the number of leukocytes in the AMLA 500 and 700 mg/kg groups (11.54 12.30/mm3) was slightly higher than that reported by Sana and Sinha 52 and Patil et al. 53 (8.09 9.19 and 6.57 7.38/mm3, respectively). These differences may be due to transient increases in leukocyte percentages caused by infectious processes or immune system activation in laboratory animals during the study period.31,52 This is consistent with the maintenance of sanitary conditions in each laboratory or animal facility, such as water purification, cleaning and disinfection of cages and facilities, sanitation of drinkers, bed changes, and air filtration, among others. 32
Regarding the blood count, the percentages of leukocytes were within the biological reference intervals in the samples from the AMLA 500 and 700 mg/kg groups, indicating that AMLA did not generate morphological alterations in these cells. Sana and Sinha 52 administered 200 and 500 mg/kg aqueous AMLA fruit extract orally for seven days to male Sprague-Dawley rats. The blood count values reported by these authors for lymphocytes, monocytes, basophils, segmented neutrophils, and band neutrophils were similar to those obtained in the present study. However, they reported a higher proportion of eosinophils than we did (5.36%), which may be due to an allergic reaction or parasitosis in the experimental subjects. 52 In this study, the platelet count obtained was within the biological reference range (632.60 to 667.40/mm3). Although no alterations were detected, there was a variation compared to figures reported by Sarvaiya et al. 51 and Patil et al. 53 (1179.00 to 1081.00 and 810.00 to 970.00/mm3, respectively). These variations can be attributed to the strain and type of handling. 32
This study has some limitations. First, the mechanism of action by which Emblica officinalis Gaertn exerts its effect on status epilepticus seizures was not studied. Secondly, the CBZ group and 3 rats in the AMLA 500 mg/kg group did not develop generalized seizures or status epilepticus, so they were assigned a value equivalent to the maximum behavioral assessment time of SE. All this was done to standardize the observation period and to maintain the “n” of the experimental groups. Thirdly, the administration of AMLA 500 and AMLA 700 mg/kg as an adjuvant to the administration of CBZ 300 mg/kg was not carried out in the experimental groups, which represents an area of opportunity to be included in future works.
Conclusion
Oral administration of standardized extract of Emblica officinalis for 28 days exerted an anticonvulsant effect without altering renal and hepatic functions or hematological parameters. This suggests that Emblica officinalis could be an alternative for treating epilepsy. However, the findings of this study should be complemented by specific research before evaluating their efficacy in clinical trials.
Footnotes
Acknowledgments
Thanks to all the researchers who participated in this study.
Author Contributions
E.R.-D., J.F.R.-L., and I.Z.-B. conceived and designed the experiments; I.Z.-B., A.A.O.-G., and O.R-S. performed the experiments; J.F.R.-L., E.R.-D, I.Z.-B, and A.A.O.-G analyzed the data; E.R.-D. contributed to the reagents/materials/analysis tools; E.R.-D., J.F.R.-L., I.Z.-B., and A.A.O-G., wrote the paper. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was partially supported by financial resources from the Academic Group of Biology, Chemistry, and Molecular Functionality of Plan Metabolites (UV-CA-368) of Universidad Veracruzana and Sistema Nacional de Investigadores – CONAHCYT, Exp. 171150 (E.R.-D.) and Exp. 32753 (J.F.R.-L.).
