Abstract
Radiation therapy has been a critical and effective treatment for cancer. However, not all cells are destroyed by radiation due to the presence of tumor cell radioresistance. In the current study, we investigated the effect of low-dose radiation (LDR) on the tumor suppressive effect of high-dose radiation (HDR) and its mechanism from the perspective of tumor cell death mode and DNA damage repair, aiming to provide a foundation for improving the efficacy of clinical tumor radiotherapy. We found that LDR pre-irradiation strengthened the HDR-inhibited A549 cell proliferation, HDR-induced apoptosis, and G2 phase cell cycle arrest under co-culture conditions. RNA-sequencing showed that differentially expressed genes after irradiation contained pyroptosis-related genes and DNA damage repair related genes. By detecting pyroptosis-related proteins, we found that LDR could enhance HDR-induced pyroptosis. Furthermore, under co-culture conditions, LDR pre-irradiation enhances the HDR-induced DNA damage and further suppresses the DNA damage-repairing process, which eventually leads to cell death. Lastly, we established a tumor-bearing mouse model and further demonstrated that LDR local pre-irradiation could enhance the cancer suppressive effect of HDR. To summarize, our study proved that LDR pre-irradiation enhances the tumor-killing function of HDR when cancer cells and immune cells were coexisting.
Keywords
Introduction
Lung cancer, the most prevalent and lethal form of cancer, is the primary reason for cancer-related deaths in both men and women around the world.1,2 A total of 19.3 million new instances of cancer and about 10 million cancer-related deaths globally are reported by GLOBOCAN 2020. Lung cancer accounts for 14% of all new cases and 18% of all cancer deaths worldwide, making it the malignancy with the greatest fatality rate. 3 Non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC) have historically been considered the 2 main kinds of lung cancer. 4 Currently, there are various treatments for lung cancer, including surgery, chemotherapy, immunotherapy and radiotherapy. 5 Radiotherapy has advanced significantly in recent years and played a crucial part in the treatment of lung cancer, which has made it a very important treatment for lung cancer. 6 Radiotherapy is available for all stages of lung cancer treatment, and more than half of patients require at least 1 radiotherapy session for cure or remission. Tumor radiotherapy has advanced greatly, yet the rate of 5-year survival for cancer patients continues to be below average, and a major reason for this is the radioresistance of cancer cells. 7 The mechanisms of tumor cell radioresistance involve many aspects, including DNA damage repair 8 and pyroptosis. 9 Therefore, it is significant to find ways to reduce tumor cell radioresistance from the mechanism of tumor cell radioresistance to improve the effectiveness of clinical tumor radiotherapy.
At present, the role of low-dose radiation in tumor radiotherapy has attracted extensive attention from scholars. Low-dose radiation (LDR) is referred to as a dose below which harmful health consequences cannot be produced. It was originally established by the UN Scientific Committee on Atomic Radiation as less than 200 mGy for low linear energy transfer (LET) radiation or 50 mGy for high LET radiation. 10 LDR can be found everywhere in our lives, such as natural radiation in our life like food and soil, as well as in clinical treatment equipment. 11 There is still much controversy in the study of low doses of radiation. The “linear no-threshold” model reckons that radiation is harmful to the body at any dose. However, many studies of LDR in recent years have shown that LDR can produce biological effects distinct from those caused by high-dose radiation (HDR), and that LDR may produce beneficial effects on the organism. According to earlier research, LDR can also cause adaptive responses to chemotherapeutic agents in normal cells. 12 It was demonstrated that LDR can protect normal tissues from the harmful effects of radiation and drugs. Another study discovered that LDR controlled the production of plasminogen activator inhibitor-1 (PAI-1) to decrease radioresistance in NSCLC, 13 further demonstrating the potential value of LDR in tumor therapy.
The primary way that radiation destroys tumor cells is by causing DNA double-strand breaks (DSBs). 14 However, tumor cells have some DNA damage repair ability and can repair damaged DNA, which leads to tumor recurrence and metastasis. There are 2 major pathways for DSB repair: nonhomologous end-joining (NHEJ) and homologous recombination (HR). 15 Research has proven that silencing of XRCC4 (a critical factor in the repair pathway for NHEJ) increases the radiosensitivity of triple-negative breast cancer cells. 16 Targeted suppression of DNA-PKcs can cause an accumulation of DNA damage, impairing NHEJ, and rendering cells hypersensitive to radiation. 17 Presently, with the intensive research on DNA damage repair pathways, increasing studies have found that targeted inhibition of DNA damage repair in cancer cells can improve the radiosensitivity of cancer cells and enhance the efficacy of tumor radiotherapy. 18
Pyroptosis is a form of programmed cell death that combines inflammatory and cell death features. 19 There are various pathways of pyroptosis however, the most common pathway is induced by NLRP3, caspase-1, and GSDMD.19,20 Inflammasomes are composed of sensor proteins, inflammatory caspase, and adapter proteins that connect the two. 21 Inflammasome sensors include the NLR family such as NLRP1, NLRP3, and NLRP4. 22 Caspases-1 can be activated by the inflammasome and once triggered, they can activate precursors of IL-1 and IL-18 and also cleave GSDMD, causing cell pore formation and eventually leading to cell death.23,24 In recent years, many studies have proved that pyroptosis can inhibit tumorigenesis and progression. Treatment of A549 and H1299 cells with polyphyllin VI (PPVI), both of which showed reduced proliferative capacity and increased dead cells, as well as increased NLRP3, GSDMD, caspase-1, IL-1β, and IL-18 expression in both. 25 Another study showed that targeting NLRP3-mediated pyroptosis could modulate the radioresistance of triple-negative breast cancer, and increasing NLRP3 expression could enhance its radiosensitivity. 9 Research on pyroptosis suggests that the combination of targeted pyroptosis with other cancer therapies could provide new ideas for cancer treatment.
The most common cancer that leads to death is lung cancer. Radiation resistance of lung cancer cells reduces the efficacy of radiotherapy and prevents complete eradication of lung cancer. A key contributor to tumor cells' radioresistance has been identified as DNA damage repair. And pyroptosis has been found to influence tumor development in recent years. Previous studies on the combined exposure of LDR and HDR mostly focused on adaptive responses. Up till now, no study has been conducted to explore the effect and mechanism of LDR in enhancing HDR killing tumor cells. In the present study, we found that LDR enhanced the tumor inhibition of HDR when tumor cells and immune cells coexisted and illustrated its mechanism from the perspectives of pyroptosis and DNA damage repair. These findings may provide new insights and targets for improving the efficacy of clinical cancer treatment.
Materials and Methods
Cells and Culture
A549 cells, H1299 cells, Hela cells, MCF-7, and Jurkat cells were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). RPMI-1640 media or DMEM medium (Gibco, USA), 10% FBS (HyClone, USA), and 1% penicillin/streptomycin (HyClone, USA) were used to cultivate the cells. All cells were grown at 37°C in a 5% CO2 environment in a humidified incubator.
Experiments on Animals
Female BALB/c nude mice aged 6 weeks were purchased from Beijing Huafukang under animal license number SYXK (Ji) 2021-20003. PBS was used to make a cell suspension of A549 cells. The concentration was 1 × 107 cells/mL. 100 μL of the cell suspension was injected into the leg of each mouse. When the tumor volume reached about 200 mm3, radiation therapy was applied to the tumor. Following radiotherapy, the tumor volume was assessed every 2 days, the mice were executed after 21 d of irradiation, and the tumors were resected. Tumor volume(mm3) = length2 (mm) × width (mm)/2. The animal experiments were approved by the Medical Ethics Committee of the School of Public Health, Jilin University (2021-12-20).
Irradiation
An X-ray generator (model X-RAD320iX) was used to irradiate both cells and animals at room temperature. For cell irradiation, the LDR dose rate was .0134 Gy/min with an irradiation dose of 100 mGy, the HDR dose rate was 1.02 Gy/min with an irradiation dose of 20 Gy, and the interval between LDR and HDR was 6 h for cell lines. For animal irradiation, the LDR dose rate was .0134 Gy/min with an irradiation dose of 100 mGy, the HDR dose rate was 1.98 Gy/min with an irradiation dose of 20 Gy, and the interval between LDR and HDR was 24 h for animals. In mice irradiation, both LDR and HDR were localized. All irradiations of cells and animals were single irradiations, not fractionated irradiations.
Reagents and Antibodies
Cell Counting Kit-8 (CCK8) (MCE, USA), Annexin V-FITC/PI Apoptosis Detection Kit (Meilunbio, China), Elisa Kits (Mlbio, China), TaKaRa Reverse Transcription Kit (TaKaRa, Japan), Fluorescence Quantitation Kit (TaKaRa, Japan), Pierce ECL Kit (Sigma, USA), TRIzol (Invitrogen, USA), BCA Protein Quantification Kit (Beyotime, China), MCC950 (KKL MED, USA), The VND3207 was kindly provided by Prof. Pingkun Zhou (Beijing Key Laboratory for Radiobiology, Beijing Institute of Radiation Medicine, Beijing, China), Primary antibodies against GAPDH, Cleaved-caspase-1, GSDMD, DNA-PKcs and Ku80 (Abcam, USA), Primary antibodies against NLRP3, XRCC4, BRCA1 and Rad51 (Bioworld, China), Secondary antibodies and fluorescent secondary antibodies (Bioworld, China).
Co-culture Model
A co-culture model was created by adding an equivalent number of logarithmic growth stage Jurkat cells after the logarithmic growth stage A549 cells had adhered to the wall and extended in the cell culture dishes (plates).26,27
Cell Viability Assay
Before receiving radiation treatment, 96-well plates containing 4000 A549 cells were cultivated for 12 hours. After 24 h of irradiation, a mixture of medium and CCK8 reagent was prepared in a 10:1 ratio, and 100 mL of the mixture was added to each well for 2 h of reaction. The absorbance was measured using a microplate spectrophotometer (Tecan, Switzerland) according to the instructions.
Apoptosis Assay
After 24 h of cell irradiation, cells were collected; washed with cold PBS; 1x Binding Buffer, Annexin V-FITC, and PI were added according to the instructions; left for 15 min protected from light; and then 200 μL 1x Binding Buffer was added and assayed by using flow cytometry (BD, USA).
Cell Cycle Assay
After 24 h of cell irradiation, the cells were collected; washed with cold PBS; 150 μL of cold PBS and 300 μL of anhydrous ethanol were added; fixed for 2 h; washed with cold PBS; PI was added according to the instructions; and placed for 30 min away from light and detected on the machine.
Enzyme-Linked Immunosorbent Assay
After irradiation of the co-culture model, the supernatants were collected and the levels of IL-18, IL-1β, IL-32, and TNF-α protein in the supernatants were assayed by using ELISA kits according to the manufacturer’s instructions. In each assay, the absorbance was detected at 450 nm using a microplate spectrophotometer (Tecan, Switzerland). Protein concentrations in the supernatants were determined based on standard curves for each plate.
Small-Interfering RNA and Transfection
A small-interfering RNA (siRNA) of human NLRP3 gene was designed and synthesized. The sequence is as follows: sense (5'-3'): GUGCGUUAGAAACACUUCATT; antisense (5'-3'): UGAAGUGUUUCUAACGCACTT (GenePharma, China). Cells were transfected using GP-transfect-Mate transfection reagent according to the instructions.
Western Blotting
RIPA lysate was used to extract the total protein from cells and tissues, and the protein content was assessed using the BCA test. Adequate amounts of protein were detached by using SDS-PAGE, conveyed by PVDF membrane, closed with 5% non-fat milk, washed the membrane, incubated with primary antibodies overnight at 4°C, washed the membrane, incubated with secondary antibodies for 2 hours, washed the membrane, and then detected using an ECL kit.
Immunofluorescence
Cells were washed, fixed for 15 min, the membrane was ruptured, closed for 1 h, antibody was added and incubated overnight, the cells were washed again, fluorescent secondary antibodies were added and placed them for 2 h away from light, the cells were washed, sealing solution containing DAPI was added, and photos were obtained using fluorescent microscopy.
Quantitative Real-Time PCR
Sequences of the qRT-PCR Primers.

RNA-Sequencing Analysis
RNA was extracted using the TRIzol isolation method. For RNA sequencing, samples were delivered to GeneChem. Data analysis reports were provided by the company. Data analysis reports were provided by the company. Heat map was redrawn by using the Hiplot analysis website, and GO and KEGG analyses were redone by using the Hiplot analysis website and Sangerbox 3.0.
Statistical Analysis
The GraphPad Prism 8 program was used to statistically evaluate the experimental data for this study, and the results were expressed as means ± standard deviation. GraphPad Prism 8 is used for graphing. The outcomes of the experiment were statistically analyzed using Student's t-test. Differences were considered statistically significant at
Results
There is No Effect of LDR on the Tumor Suppression Effect of HDR When Irradiating Tumor Cells Alone
In the initial study, we evaluated the viability of A549 cells, Hela cells, MCF-7 cells, and H1299 cells after irradiation via the CCK8 kit. There was no discernible difference in the 4 types of cells' viability between the LDR+HDR group and the HDR group ( There is no effect of LDR on the tumor suppression effect of HDR when irradiating tumor cells alone. (A) Cell viability of A549 cells after irradiation was evaluated by using CCK8 assay, n = 4 per group. (B–D) Cell viability of tumor cells after irradiation was evaluated by using CCK8 assay, n = 5 per group. (E–G) The apoptosis rate of tumor cells after irradiation was detected by using flow cytometry, n = 3 per group. All data are presented as the mean ± SD.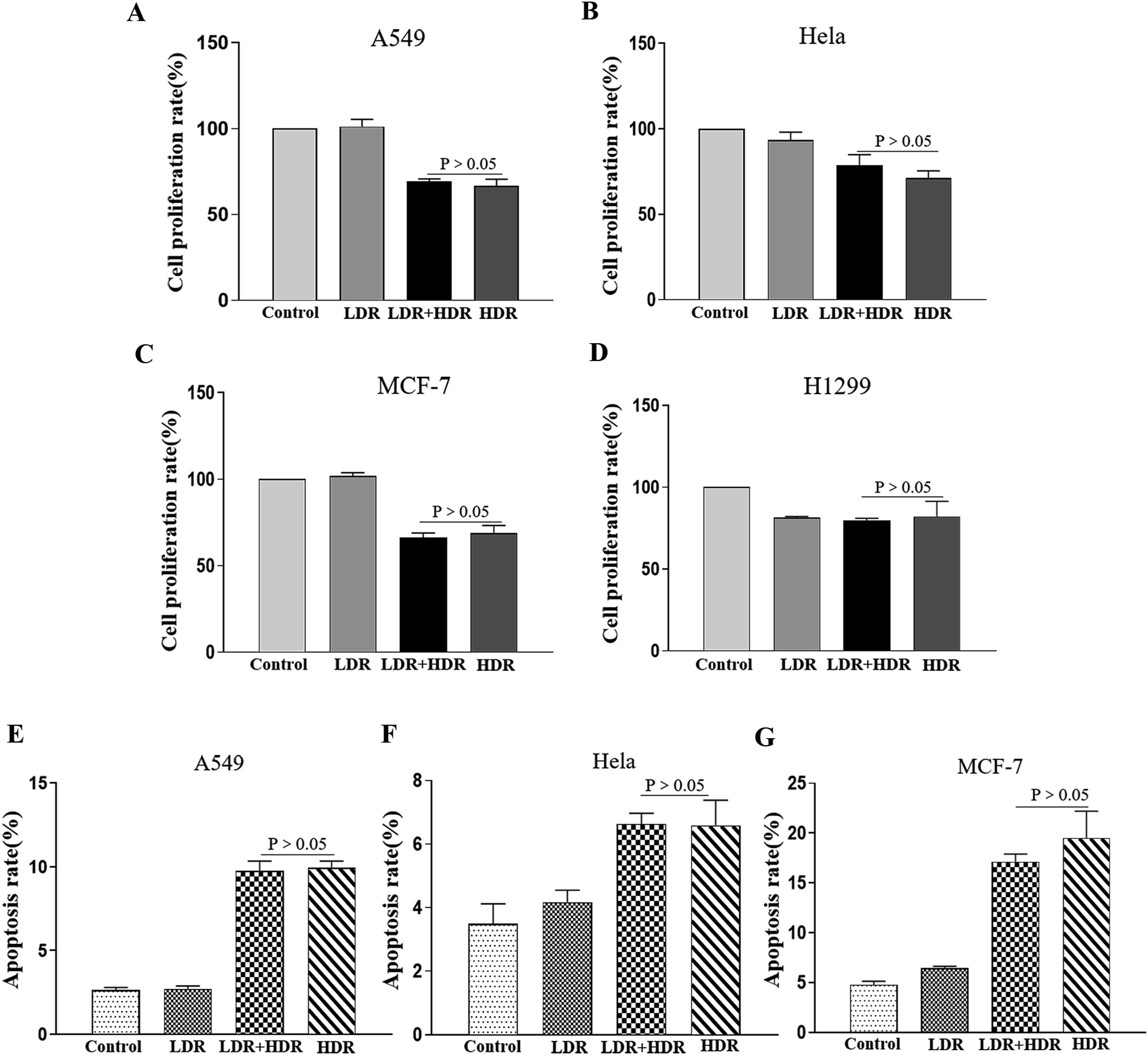
LDR Could Enhance the Inhibitory Effect of HDR on Tumor Cells Under Co-culture Conditions
Considering the theory of the immune excitatory effect of LDR
28
and the role of the tumor microenvironment after irradiation,
29
we designed the experiment of co-culture of tumor cells with immune cells (Figure 2A). Subsequent experiments will be conducted under co-culture conditions. Subsequently, we assayed the viability of A549 cells after irradiation under co-culture conditions. We found that the survival rate of A549 cells was decreased after irradiation, and the LDR+HDR group had a lower cell survival rate than the HDR group ( LDR could enhance the inhibitory effect of HDR on tumor cells under co-culture conditions. (A) Co-culture model was established by using A549 cells and Jurkat cells. (B) Cell viability of A549 cells after co-culture cell irradiation was evaluated by using CCK8 assay, n = 5 per group. (C, D) The apoptosis rate of A549 cells after co-culture cell irradiation was detected by using flow cytometry, n = 3 per group. (E, F) Flow cytometry analysis of the proportion of the different cell cycle phases in A549 cells after co-culture irradiation, n = 3 per group. All data are presented as the mean ± SD. *
Pyroptosis and DNA Damage Repair May be Potential Mechanisms
We used RNA sequencing to determine the mechanism by which LDR pre-irradiation enhanced the inhibitory effect of HDR on tumor cells under co-culture conditions. According to the RNA sequence data, there were 692 differently expressed genes in the LDR group, 4854 in the LDR+HDR group, and 5999 in the HDR group as compared to the Control group (Figure 3A-3D). Heat map results suggest that pyroptosis-related genes, such as GSDMD and IL-18, had higher expression levels and the expressions of DNA damage-repair-related genes XRCC4, BRCA2, and XRCC2 were downregulated in the LDR+HDR group compared with the HDR group (Figure 3E). GO enrichment analysis revealed that the differential genes in the LDR group were enriched in biological processes such as activation of immune response, double-strand break repair, and regulation of cell cycle G2/M phase transition compared to the Control group (Figure 3F). The LDR+HDR and HDR groups’ differential genes were enriched in biological processes such as regulation of inflammatory response, cell cycle checkpoint, activation of the immune response, and double-strand break repair (Figure 3G and 3H). KEGG pathway analysis suggested that the LDR group was enriched for differential genes in pathways containing PI3K-Akt signaling pathway, pathways in cancer, and cell cycle (Figure 3I). And the pathways of differential gene enrichment in the LDR+HDR group and HDR group included human T-cell leukemia virus 1 infection, DNA damage repair pathway, cell cycle, and TNF signaling pathway (Figure 3J and 3K). The results of RNA sequence gave us insight that DNA damage repair and pyroptosis may be the mechanisms by which LDR pre-irradiation enhanced the inhibitory effect of HDR on tumor cells. Pyroptosis and DNA damage repair may be potential mechanisms. (A) The Venn diagram shows the number of differential genes. (B–D) The volcano plot shows the number of genes whose expression was upregulated or downregulated in the LDR group, LDR+HDR group, and HDR group compared to the Control group. (E) Clustering of genes altered in the Control group, LDR group, LDR+HDR group, and HDR group. (F–H) Gene ontology analysis of differentially expressed genes in the LDR group, LDR+HDR group, and HDR group compared to the Control group. (I–K) KEGG was used for pathway analysis of differential genes.
LDR Pre-irradiation Could Enhance HDR-induced Pyroptosis Under Co-culture Conditions
Radiation can stimulate the secretion of pro-inflammatory cytokines,
30
and pyroptosis is known as inflammatory death. Heat map analysis of RNA-sequencing data indicated that the LDR+HDR group had higher expression levels of the pyroptosis-related genes GSDMD and IL-18 than the HDR group (Figure 3E). Therefore, after co-culture cell irradiation, we assayed protein expressions related to pyroptosis in A549 cells. We found that NLRP3, GSDMD, and cleaved-caspase-1 expressions were increased in the groups of LDR+HDR and HDR after irradiation. Significantly, the LDR + HDR group showed higher expression than the HDR group (Figure 4A). Then, we examined the expressions of IL-18, IL-1β, TNF-α, and IL-32 in the supernatant. We found that the LDR+HDR group was all higher than the HDR group (Figure 4B-4E). LDR pre-irradiation could enhance HDR-induced pyroptosis under co-culture conditions. (A) NLRP3, GSDMD, and cleaved-caspase-1 protein levels in A549 cells were determined after 48 h of irradiation. (B-E) Expressions of IL-18, IL-1β, TNF-α, and IL-32 in the supernatant after irradiation, n = 3 per group. (F) The expression of NLRP3 after transfection with siRNA targeting NLRP3 in A549 cells was assayed by using Western blot technique. (G) The expression of NLRP3 was assayed after treating A549 cells with 10 μM NLRP3 inhibitor-MCC950 by using Western blot technique. (H) A549 cells were transfected with si-NLRP3 and then co-cultured with Jurkat cells, cell survival was detected by using CCK8 assay after irradiation, n = 4 per group. (I) A549 cells were treated with MCC950 and then co-cultured with Jurkat cells, and cell survival was detected with CCK8 after irradiation, n = 4 per group. (J) A549 cells were transfected with si-NLRP3 and then co-cultured with Jurkat cells, and pyroptosis-related proteins were assayed by using Western blot technique after irradiation. (K) A549 cells were treated with MCC950 and then co-cultured with Jurkat cells, and pyroptosis-related proteins were assayed by using Western blot technique after irradiation. All data are presented as the mean ± SD. *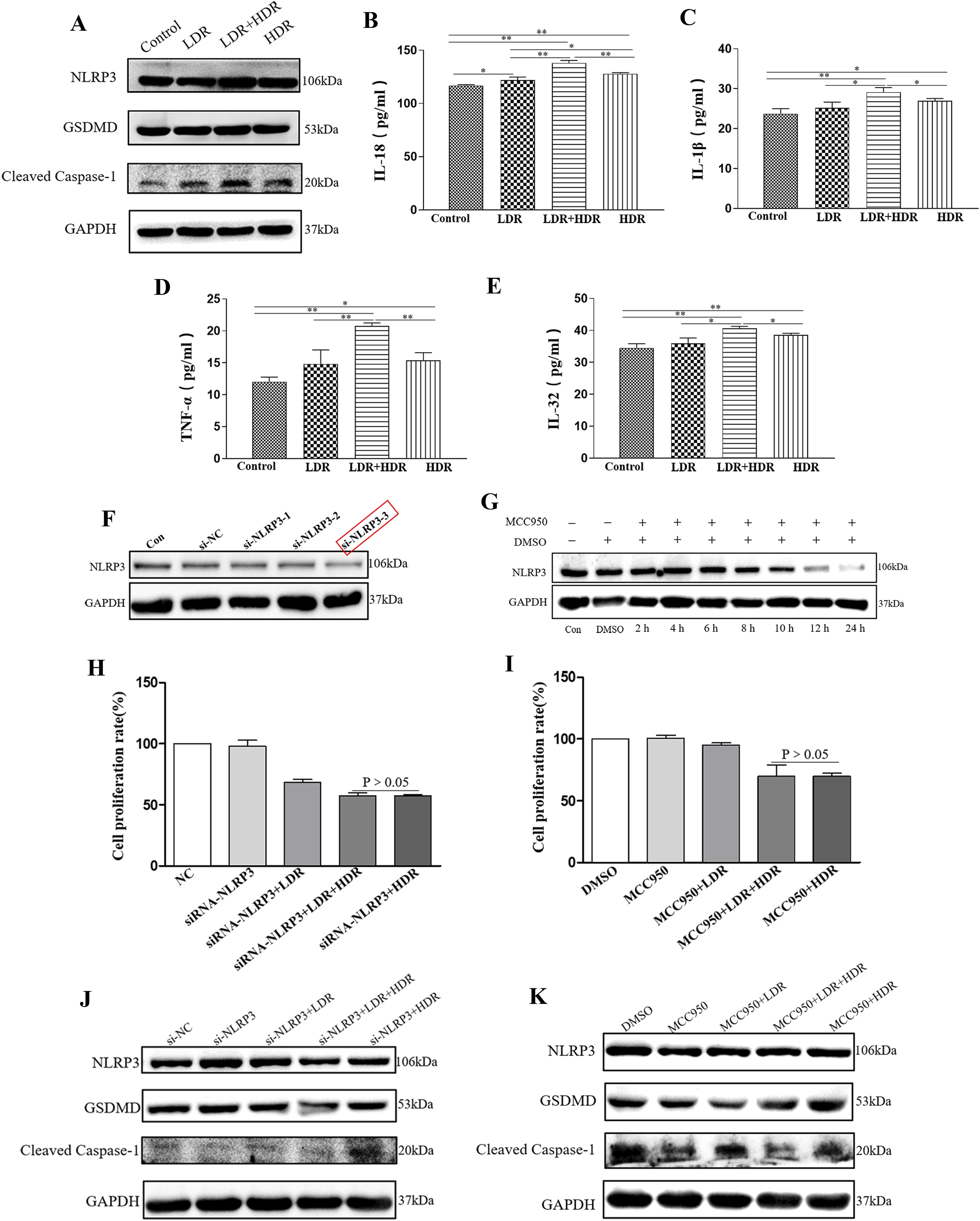
To confirm the role of pyroptosis even more, we constructed an NLRP3 low-expression model in A549 cells using si-NLRP3 and the inhibitor. First, the effect of NLRP3 low expression was tested by using Western blot; results indicated that both si-NLRP3 and inhibitor could reduce the expression of NLRP3 (Figure 4F and 4G). Subsequently, the NLRP3 low-expressing A549 cells were co-cultured with Jurkat cells, and we assayed cell survival after irradiation and found that the LDR + HDR group and the HDR group had no significant difference in cell survival (
LDR Pre-irradiation Enhances DNA Damage Caused by HDR Irradiation and Inhibits DNA Damage Repair
Radiation kills tumor cells mainly by causing DSBs.
14
The organism has 2 main repair pathways, HR and NHEJ, to repair the damaged cells.
15
Therefore, we first assayed the expression of γH2AX, a double-strand breakage marker, by using immunofluorescence, and found that under co-culture conditions, the focus of γH2AX was dramatically increased in the LDR+HDR and HDR groups after irradiation, and the LDR+HDR group had more focus than the HDR group ( LDR pre-irradiation enhances DNA damage caused by HDR irradiation and inhibits DNA damage repair. (A) Immunofluorescence staining for γH2AX in A549 cells after 3 h of irradiation. Scale bars: 40 μm. (B) The number of focal points of γH2AX, n = 25 per group. (C) Immunofluorescence staining for 53BP1 in A549 cells after 3 h of irradiation. Scale bars: 50 μm. (D) The number of focal points of 53BP1, n = 20 per group. (E-I) Relative expressions of Rad51, BRCA1, Ku80, DNA-PKcs, and XRCC4 mRNA after 48 h of irradiation, n = 3 per group. (J) Rad51, BRCA1, Ku80, DNA-PKcs, and XRCC4 protein levels in A549 cells were determined after 72 h of irradiation. All data are presented as the mean ± SD. *
LDR No Longer Enhances the Inhibitory Effect of HDR on Tumor Cells After the Treatment of A549 Cells With DNA-PKcs Agonist
VND3207 was known to increase the expression of DNA-PKcs.
31
To further determine DNA damage repair as the mechanism by which LDR enhances the tumor suppressive effect of HDR, A549 cells were treated with 40 μM of VND3207 and then co-cultured with Jurkat cells. Cell viability was measured after irradiation, and the results showed that cell viability was higher in the LDR+HDR group compared to the HDR group ( LDR no longer enhances the inhibitory effect of HDR on tumor cells after treatment of VND3207. (A) A549 cells were treated with VND3207(40 μM) for 2 h and co-cultured with Jurkat cells, and after irradiation the survival rate of A549 cells was assessed by using CCK8 assay, n = 3 per group. (B, C) A549 cells were co-cultured with Jurkat cells after VND3207 treatment, and the apoptosis rate of A549 cells was detected by using flow cytometry after irradiation, n = 3 per group. (D–H) Relative expressions of Rad51, BRCA1, Ku80, DNA-PKcs, and XRCC4 mRNA after 48 h of irradiation, n = 3 per group. (I) Ku80, DNA-PKcs, and XRCC4 protein levels in A549 cells were determined after 72 h of irradiation. All data are presented as the mean ± SD. *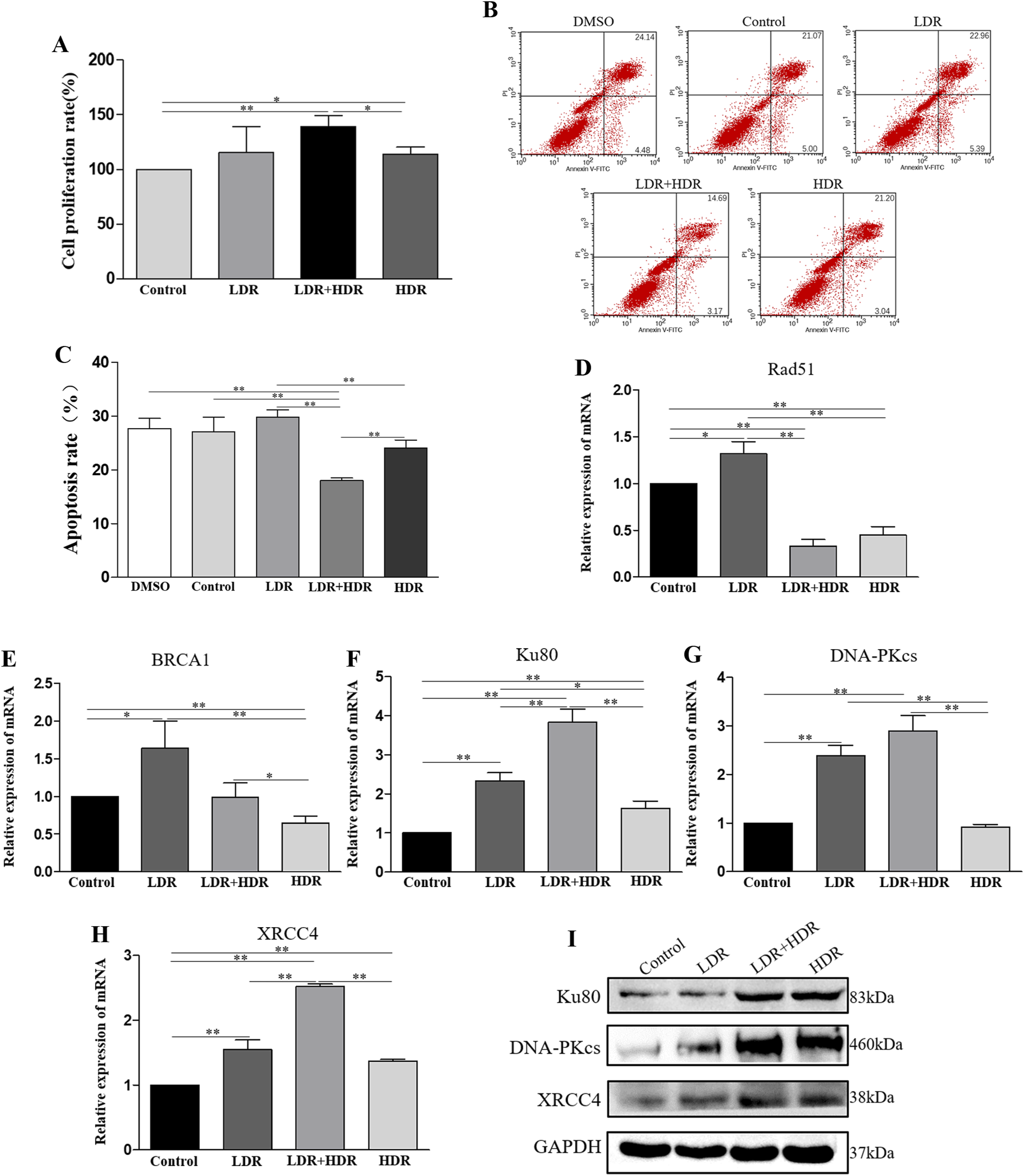
LDR Local Irradiation Can Enhance the Tumor Inhibition Effect of HDR In Vivo
After that, we established a tumor-bearing nude mouse model to verify the results of in vitro experiments. Different doses of irradiation were delivered to mice. After irradiation, the tumor size was assessed every 3 days. After 21 days of radiation treatment, the mice were killed and their tumors were removed. The size of the tumor is shown in Figure 7A and 7B. Tumors were the smallest in the LDR+HDR group, followed by the HDR group. The same is true for tumor volume results (Figure 7C). Subsequently, we detected mRNA and protein expression of DNA damage-repair-related molecules in tumor tissues, as well as the expression of pyroptosis-related molecular proteins, and the results were similar to those from in vitro experiments (Figure 7D-7I). Through these results, we are more certain that LDR can enhance the tumor suppressive effect of HDR and that DNA damage repair plays an important role. LDR local irradiation enhances the tumor suppressive effect of HDR in tumor-bearing mice. (A) Tumor location in the tumor-bearing mouse. (B) Cells were injected subcutaneously into the right leg of nude mice. When the tumor volume reached approximately 200 mm3, the mice were treated with irradiation. The tumors were excised 21 days after irradiation. (C) The average volume of the tumors was measured every 3 days after irradiation. (D-H) Relative expressions of Rad51, BRCA1, Ku80, DNA-PKcs, and XRCC4 mRNA in tumor tissues. (I) The protein levels of NLRP3, GSDMD, cleaved-caspase-1, Rad51 and XRCC4 were measured in tumor tissues. All data are presented as the mean ± SD, n = 4. *
Discussion
Radiotherapy is a part of the standard therapies for clinical cancer treatment, but the influence of radioresistance on tumor cells makes radiotherapy not achieve the expected effect. Therefore, research for ways to improve the effectiveness of radiotherapy has been a hot topic. Previous studies on LDR have focused on hormesis, adaptive responses, and bystander effects. 32 However, no report has been conducted on the tumor suppressive effect of HDR enhanced by LDR. Our current study demonstrates that when tumor cells and immune cells coexist, LDR could enhance the tumor inhibition of HDR and the mechanisms are related to DNA damage repair and pyroptosis. This study may provide a reference for improving the efficacy of tumor radiotherapy.
Previous studies have demonstrated that breast cancer MCF-7 cell growth was inhibited and radiosensitivity was enhanced in the presence of HSF fibroblasts. 33 Under the conditions of macrophage coexistence with colon cancer RKO cells, macrophages sensitize RKO to radiation-induced apoptosis. 34 In addition, another study has also demonstrated that LDR can stimulate the bone marrow-derived mesenchymal stromal cells (BM-MSCs) to secrete some cytokines that inhibit the proliferation of colorectal cancer (CRC) cells and induce their apoptosis. 26 Interestingly, in our earliest study, LDR had no effect on the tumor inhibition effect of HDR when irradiated with tumor cells alone. Considering the hormesis and the complex tumor microenvironment, we established a tumor cell and immune cell co-culture model to better simulate the environment of tumor radiotherapy. Subsequently, we found that under co-culture conditions, LDR enhanced the proliferation inhibition of HDR and increased its induction of apoptosis in tumor cells. Cells are most sensitive to radiation in the G2/M phase of the cell cycle. 35 Importantly, in our study, radiation also induced G2 phase cell cycle arrest, and LDR made the HDR-induced G2 phase arrest more pronounced.
The mechanism by which LDR pre-irradiation enhances the inhibitory effect of HDR on tumor cells under co-culture conditions has not been reported. Therefore, we carried out RNA sequencing to clarify the mechanism of this study. The results of the RNA sequence revealed that LDR pre-irradiation could increase HDR-induced expression of pyroptosis-related genes and decrease the expression of DNA damage repair-related genes (Figure 3E). By using GO analysis, we found that the biological processes of differential gene enrichment in the LDR+HDR and HDR groups contained double-strand break repair, activation of the immune response, and regulation of inflammatory response. HR 36 and NHEJ 37 are the main pathways for repairing DNA double-strand breaks. In addition, it has been documented that pyroptotic cells release pro-inflammatory factors, which cause inflammatory responses and immune responses. 38 Based on the RNA-sequence results, we speculated that pyroptosis and DNA damage repair were the mechanisms by which LDR pre-irradiation enhanced the tumor cell inhibitory effect of HDR.
Pyroptosis is a novel type of cell death. An increasing number of studies are proving the great potential of pyroptosis in cancer treatment.39-41 NLRP3-GSDMD-caspase-1 has been shown to be one of the pathways through which pyroptosis occurs and can release inflammatory factors such as IL-18 and IL-1β. 42 However, there are no reports that LDR can enhance radiation-induced pyroptosis. So, is LDR able to enhance HDR-induced pyroptosis? Is there an important role for pyroptosis in the enhanced tumor suppressive effect of HDR by LDR? With these questions in mind, we conducted this study. We found that HDR induced the onset of pyroptosis under co-culture conditions, as evidenced by protein expression increases of NLRP3, GSDMD, and cleaved-caspase-1 as well as IL-18 and IL-1 secretion. Subsequently, we demonstrate that LDR pre-irradiation can enhance HDR-induced pyroptosis. More intriguingly, HDR also induced the secretion of IL-32 and TNF-α, which can induce the onset of pyroptosis,43,44 and LDR pre-irradiation also enhanced the HDR-induced secretion of both. Most importantly, we proved that after NLRP3 expression was reduced by using NLRP3-siRNA or NLRP3 inhibitors, pyroptosis-related protein expression was no longer increased after irradiation, and LDR no longer enhanced the proliferation inhibition of tumor cells by HDR, which further confirmed that pyroptosis is the mechanism by which LDR enhances the tumor-suppressive effect of HDR.
DNA damage repair is a major reason why tumor cells are radioresistant. γH2AX is a marker of DSBs and can be used to monitor DNA damage after irradiation. 45 53BP1 can regulate the repair of DSBs. 46 DNA damage is mainly repaired through 2 pathways, HR and NHEJ. BRCA1 and Rad51 are important players in the HR repair pathway, and DNA-PKcs, Ku80, and XRCC4 play important roles in the NHEJ repair pathway.47,48 A previous study has shown that the sensitivity of NSCLC cells to X-rays was enhanced by eurycomalactone and can inhibit X-ray-induced DNA damage repair. 49 In the previous study, we demonstrated that under co-culture conditions, LDR can enhance the tumor inhibition effect of HDR. Based on these studies, we hypothesized that DNA damage repair has a mechanistic role in our study. As expected, our data showed that LDR pre-irradiation enhanced HDR-induced expression of γH2AX and reduced HDR-induced expressions of BRCA1, Rad51, Ku80, DNA-PKcs, and XRCC4. It was demonstrated that under co-culture conditions, LDR enhances HDR-induced DNA damage and inhibits DNA damage repair. In vivo experiments further demonstrated that LDR could enhance the tumor inhibition effect of HDR, and DNA damage repair was inhibited. VND3207 can enhance the expression of DNA-PKcs. 31 More importantly, our results showed that LDR no longer enhanced the inhibition of HDR on tumor cells after increasing the expression of DNA-PKcs by using VND3207. Therefore, these results suggest that DNA damage repair is the mechanism by which LDR enhances the tumor suppressive effect of HDR.
There are some limitations of our study that should be noted. No additional enhancement of the tumor-suppressive effect of HDR irradiation was observed following LDR pre-irradiation in single tumor cells (lung, breast, and cervical cancers) without co-culture. A549 cells were specifically chosen for co-culture modeling in subsequent studies due to their representation of a classical in vitro model for non-small cell lung cancer and their resistance to radiation as a type of lung adenocarcinoma cell. 50 Future research could focus on investigating more radiation-resistant tumor cell types.
Conclusion
Our current study demonstrates that when tumor cells coexist with immune cells, LDR pre-irradiation enhances HDR-induced G2 phase cell cycle arrest, increases HDR-induced DNA damage, and inhibits DNA damage repair after HDR irradiation, leading to an increase in HDR-induced apoptosis. On the other way, radiation promotes the secretion of some pro-inflammatory factors, causing the onset of pyroptosis, and LDR pre-irradiation upregulates the HDR-induced cell pyroptosis which ultimately results in the proliferation inhibition of tumor cells (Figure 8). Illustration of the mechanism by which LDR enhances the tumor-suppressive effect of HDR. When tumor cells coexist with immune cells, LDR pre-irradiation increases HDR-induced G2 phase cell cycle arrest, increases HDR-induced DNA damage, and inhibits DNA damage repair after HDR irradiation, causing an increase in HDR-induced apoptosis, meanwhile, radiation leads to the secretion of some pro-inflammatory factors, causing the onset of pyroptosis, and LDR pre-irradiation can enhance HDR-induced pyroptosis, ultimately leading to proliferation inhibition of tumor cells.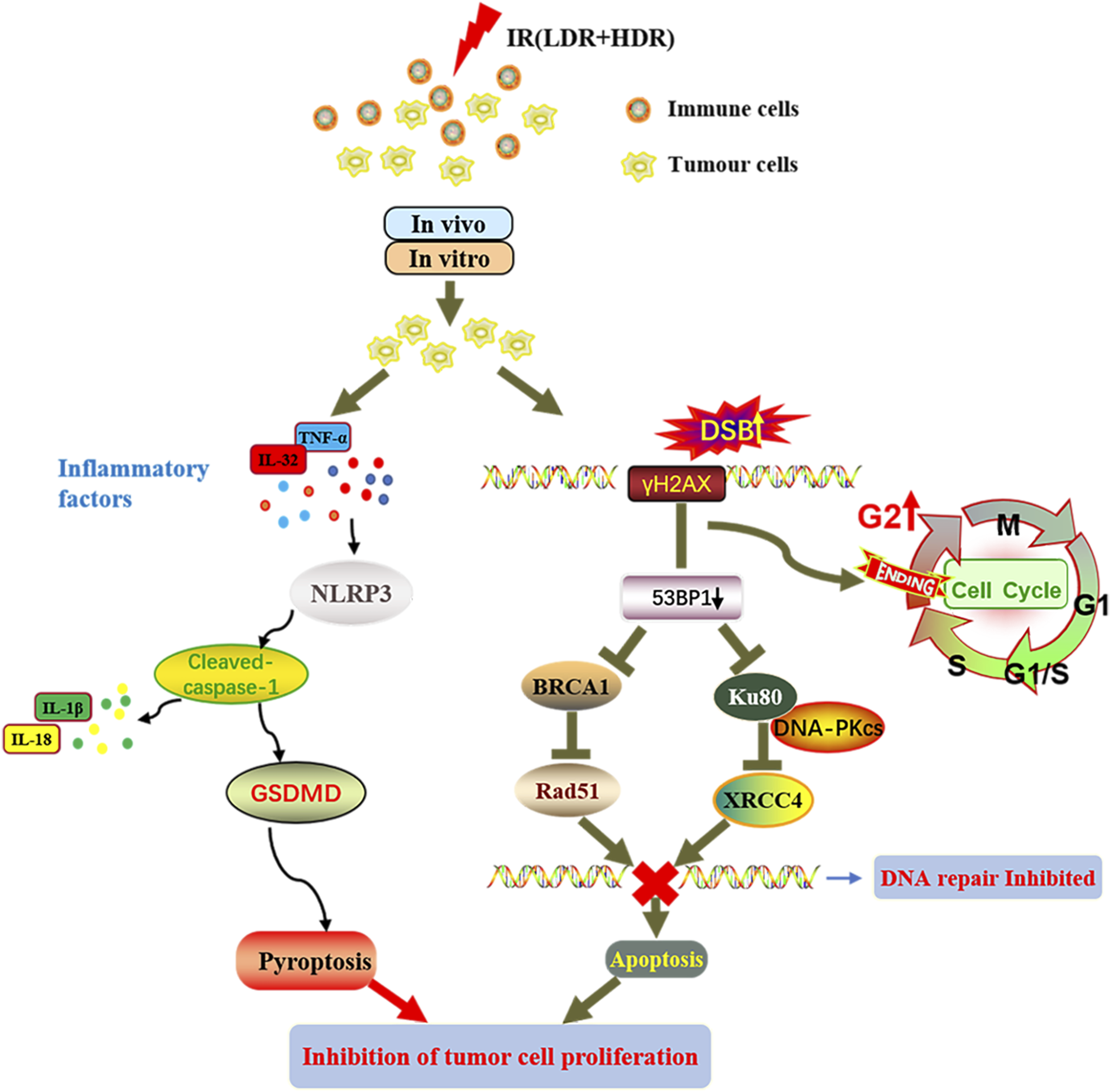
Footnotes
Acknowledgments
We thank Professor Zhou Pingkun for the kind gift of VND3207 to us. We thank the First Hospital of Jilin University Talent Reserve Program Fund for its financial support.
Author’ Contributions
Hui Gao provided research funding and participated in the design. Shunzi Jin directed the specific research. Xinfeng Wei designed and performed most of the experiments, and wrote and revised the manuscript. Junxuan Yi, Citong Zhang, and Teng Yang participated in the analysis of the data. Junxuan Yi, Mingwei Wang, and Rui Wang conducted part of the experiments. Weiqiang Xu, Mingqi Zhao, and Mengdie Zhao performed the data collection and analysis. Citong Zhang and Wei Wei reviewed and revised the paper. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the National Natural Science Foundation of China (Nos. 81872550 and 82173454).
Ethics Statement
Data Availability Statement
The data used to support the findings of this study are available from the corresponding authors upon request. The RNA-seq raw data has been submitted to the NCBI. The SRA accession is SRP414308. The Bioproject accession is PRJNA913831.
