Abstract
Purpose
Gamma knife radiosurgery (GKRS) delivers high-dose external radiation to a small intracranial lesion. However, scattering and leaked radiation can deposit a portion of the dose outside the radiation field, which may pose a risk to radiation-sensitive patients, such as pregnant women. Trigeminal Neuralgia (TN) is treated with one of the highest GKRS doses (80–90 Gy). This study aimed to estimate the risk of secondary cancer induction in the uterus, ovaries, thyroid gland, and eyes of TN patients undergoing GKRS.
Methods
Radiation doses to the uterus, ovary, eyes, and thyroid gland were measured for 25 female TN patients, with a mean age of 35 years, utilizing Thermo Luminescent Dosimeters (TLD).
Results
The mean absorbed dose for the uterus, ovary, thyroid gland, and eyes were .63 ± .24, .471 ± .2, 8.26 ± 1.01, and 10.64 ± 1.08 cGy, respectively. Lifetime Attributable Risk (LAR) has been calculated using BEIR VII (2006) method. LAR for the uterus, ovary, and thyroid gland was 1, 2, and 23, respectively.
Conclusion
The results of this study and its comparison with standard values demonstrate that on average, mean doses to mentioned organs were smaller than their tolerance doses, and there is no limitation to treating patients suffering from TN by GK.
Keywords
Introduction
Trigeminal neuralgia (TN) is a chronic neuropathic pain condition that affects the innervated regions of the face by the trigeminal nerve. Typical TN causes severe and sudden volleys of shock-like facial pain that lasts a few seconds to a few minutes in the distribution of one or more divisions of the trigeminal nerve. 1 Medication therapy is nearly always introduced as the primary treatment. However, they may be associated with side effects or unable to control the pain. Several invasive surgical procedures are used as TN treatment methods, including mechanical balloon compression, percutaneous radiofrequency rhizotomy, microvascular decompression, glycerol rhizotomy, and peripheral nerve section. However, these techniques have problems such as pain relapse, loss of facial sensation, and hospitalization. Stereotactic radiosurgery is a painless successful alternative treatment for TN patients.2-9 Sixty-nine percent of patients were reported to remain pain-free one year after GK surgery without additional medication. 10 GK has been the standard radiosurgery technique for trigeminal neuralgia treatments.11,12 Technically, the GK utilizes 201 60Co sources to irradiate the root entry zone of the trigeminal nerve. In comparison to LINAC-based radiosurgery, the GK system’s simultaneous activation of all radioactive sources with a single delivery setup provides a distinct advantage. This feature eliminates the necessity for multiple patient positioning and beam setups, thereby further reducing random setup errors commonly associated with LINAC-based radiosurgery. The GK’s streamlined approach contributes to enhanced precision and accuracy in treating trigeminal neuralgia patients. The fixed radiation source design of the GK provides advantages such as precise targeting, fixed positioning, and reduced treatment time, which contribute to minimizing the exposure of surrounding tissue in the low-dose range.13,14
It has been applied to intracranial targets such as primary or metastatic brain tumors, benign tumors, vascular malformations such as arteriovenous malformations (AVMs), and functional neurologic diseases like TN. The stereotactic radiosurgical method was initially introduced by Lars Leksell in 1951. From around 1953, he began treating trigeminal neuralgia patients using radiation sources such as X-rays. However, it was not until the late 1960s that Leksell and his team developed the GK system, which utilizes the cobalt radiation source. The introduction of the gamma knife marked a significant advancement in the field of SRS for destroying the particular focus in the brain to relieve pain instead of neurosurgery.
15
Achieving long-term tumor control vs saving neurovascular elements and maintaining cranial nerve function are the two important goals that can be reached by stereotactic radiosurgery alone or in combination with a more conservative surgery.
16
A reasonable concern regarding this method is that a specific portion of the dose is always deposited outside of the radiation field
17
because of (1) photon leakage through the treatment head of the machine, (2) radiation scattered from the collimators and beam modifiers, and (3) radiation scattered within the patient body from the treatment beams
18
which is schematically shown in Figure 1. Amount of this radiation is related to the treatment time
17
and average organ to isocenter distance. Sources of extracranial dose in GK radiosurgery.
This peripheral dose may not be an important issue under normal circumstances, but it must be ensured that there is no particular danger in long treatment time for radiation-sensitive patients. One of the highest prescribed doses in GK treatments belongs to TN patients (85–90 Gy). 19 So, this high-prescribed dose increases the treatment time and remarkably rises in peripheral doses. 20 The decay of the radiation sources significantly influences exposure times in GKRS, with shorter times possible for fresh sources. However, these factors do not notably impact the efficacy or safety of the treatment for trigeminal neuralgia pain management. The examined dose rate range (1.21–3.74 Gy/min) did not affect pain control or morbidity, and outcomes remained unaffected. 21 Estimating the peripheral dose is essential in patients with long-life expectancy after treatment, significantly when the dose affects healthy anatomical structures with low tolerance to radiation like the fetus and ovary. 17 Trigeminal neuralgia affects females slightly more often than males. The male-to-female prevalence ratio ranges from 1/1.5 to 1/1.7. Although the exact incidence is unknown, TN affects 4 to 13 per 100,000 people annually. 22 By the way, the likelihood of treating a pregnant woman with trigeminal neuralgia is low and hypothetical, as TN predominantly affects older women who are typically beyond child-bearing age. It is worth noting that radiation protection legislation varies across countries, and in some cases, the use of radiation therapy in pregnant women is entirely prohibited, despite the theoretical and practical adherence to dose limit values.
Regarding radiation treatments, knowing the doses to be deposited outside the treatment volume beforehand may allow a risk estimation of detrimental effects and, if possible, precautions to minimize the probability of developing them. 17 Because of the well-founded concern regarding any radiation exposure to the fetus and also the higher prevalence of TN in females, the question is whether GKRS is an appropriate treatment for TN pregnant patients or alternative treatment(s) in many cases fewer effective ones accompanied by side effects, must be employed? Moreover, it is crucial to assess radiation doses at critical, normal structures for patients undergoing GKRS. 23 This study is the first assessment of extracranial absorbed dose and resulting side effects in Iranian patients with TN. Radiation doses to the uterus, ovary, eyes, and thyroid gland were estimated in 25 female patients using TLDs. Based on the prior studies, phantom measurements show that the absorbed dose at different depths does not differ significantly (P-value <.05). This result was expected since, as shown in Figure 1, the patient’s head is a volume source of radiation, and doses to these extracranial sites are due to secondary radiation with nearly equal distance to the source of radiation. So, the surface dose is comparable to the depth dose, and we can place TLDs at the surface of the patients’ skin to estimate the depth (organ) dose. 23 The Lifetime Attributable Risk (LAR), or the probability of being clinically diseased by (or dying from) specific cancer after radiation exposure until the end of life, has been calculated for the received dose to the mentioned organs using BEIR VII (2006) method 24 and also doses have been compared to the maximum tolerable dose reported by International Commission on Radiological Protection (ICRP). 25
Materials and Methods
Phantom Modification and Calibration of the TLDs
A relative radiation dosimetry system is a system whose response to ionizing radiation must be calibrated in a known radiation field before its radiation-induced signal can be used to provide an absorbed dose or dose rate in the dosimeter chamber cavity. 26 TLD is a relative dosimeter, so to have a valid measurement, it is necessary to obtain its calibration curve by delivering specific multi-level doses to the dosimeter to derive the dosimeter’s response curve. In this study, the dose rate of GK was measured utilizing the Semiflex ionization chamber (PTW-TM31010 0.125 cm3), which is suitable for small-field dosimetry. 27 Background radiation in 10 consecutive time intervals has been measured to calculate the net dose rate.28,29 The ionization chamber was inserted into the Acrylonitrile Butadiene Styrene (ABS) spherical plastic phantom used for quality assurance in GK. 30
For this measurement, cubic chips (TLD-100), composed of lithium fluoride crystals doped with Magnesium and Titanium, with sizes of 3.2 × 3.2 × .9 mm3 and a usable energy range of 50 µGy to 500 Gy, were used. Annealing procedures for TLDs have been done based on the manufacturer’s instructions. TLDs were kept at 285°C for 30 min to remove residual effects of previous irradiations, followed by fast cooling. TLDs were numbered, and calibration was performed individually and in groups. The purpose of the individual calibration was to compensate for random inherent individual variation of TLD detectors. For this purpose, each TLD chip was placed in specific holes on a Perspex slab at the depth of the maximum dose irradiated by the Co-60 beam. A dose of 100 cGy was delivered to TLDs to gain the ECC, which is calculated using equation (1). 31
A correction factor for each detector, using the below equation, was calculated:
For group calibration, the standard spherical phantom was modified according to Figure 2A. This phantom is made of two hemispheres so that different Perspex layers dedicated to ion chamber, film, and TLD dosimetry can be inserted between them.33,34 (A) Sockets made by a laser in the center of a circular Plexiglas tissue-equivalent plate, 10 cm in diameter and 10 mm in thickness. (B) Plexiglas tissue-equivalent plate inside the standard spherical phantom. (C) Irradiation of TLDs inside the modified standard phantom in the Leksell GK system.
32

For group calibration, TLDs were placed in sockets made by a laser in the center of a circular Plexiglas tissue-equivalent plate, 10 cm in diameter and 10 mm in thickness (Figure 2A). Then, the entire plate was placed inside the gamma-knife’s spherical standard phantom (Figure 2B), centered at the device’s isocenter, and irradiated uniformly by 18 mm collimator size with different doses (Figure 2C).
The readout process has been done by Fimel LTM TLD Reader model HF15001 for all TLDs. To stabilize electrical circuits and reduce system noise, a 30-minute warm-up was performed, and before any readout, PMT (Photo Multiplier Tube) noise and reference light tests were made. 35 Then, the calibration curve was obtained by fitting the best curve to the points in MATLAB and Excel software.
Patient Information, Positioning, and Treatment Planning
In the first step, head immobilization to localize the target in stereotactic radiosurgery was performed by rectangular stereotactic frame under local anesthesia. After fixing the frame to the patient’s skull, the head coordinate was determined using a glass sphere to simulate the skull shape in the treatment planning system. Twenty-five female patients with normal weight and height were selected among TN patients referred to the Iran GK center. The specialist team did the treatment, including a neurosurgeon, radiation oncologist, and medical physicist.16,36 Then, patients were referred to a 1.5 T MRI unit to capture T1 and T2 images for treatment planning (Figure 3). For all patients, a 90 Gy dose was prescribed at the 100% isodose line to the trigeminal root entry zone of the nerve, and specific attention to confining the brain stem dose to 13 Gy.16,24 TN treatment was carried out with one single shot using the 4 mm collimator size and dose rate of around 70 cGy/min resulting in a typical treatment time of about 130 min. Illustration of the trigeminal nerve and brain stem as target and critical radiosensitive organ on MRI images.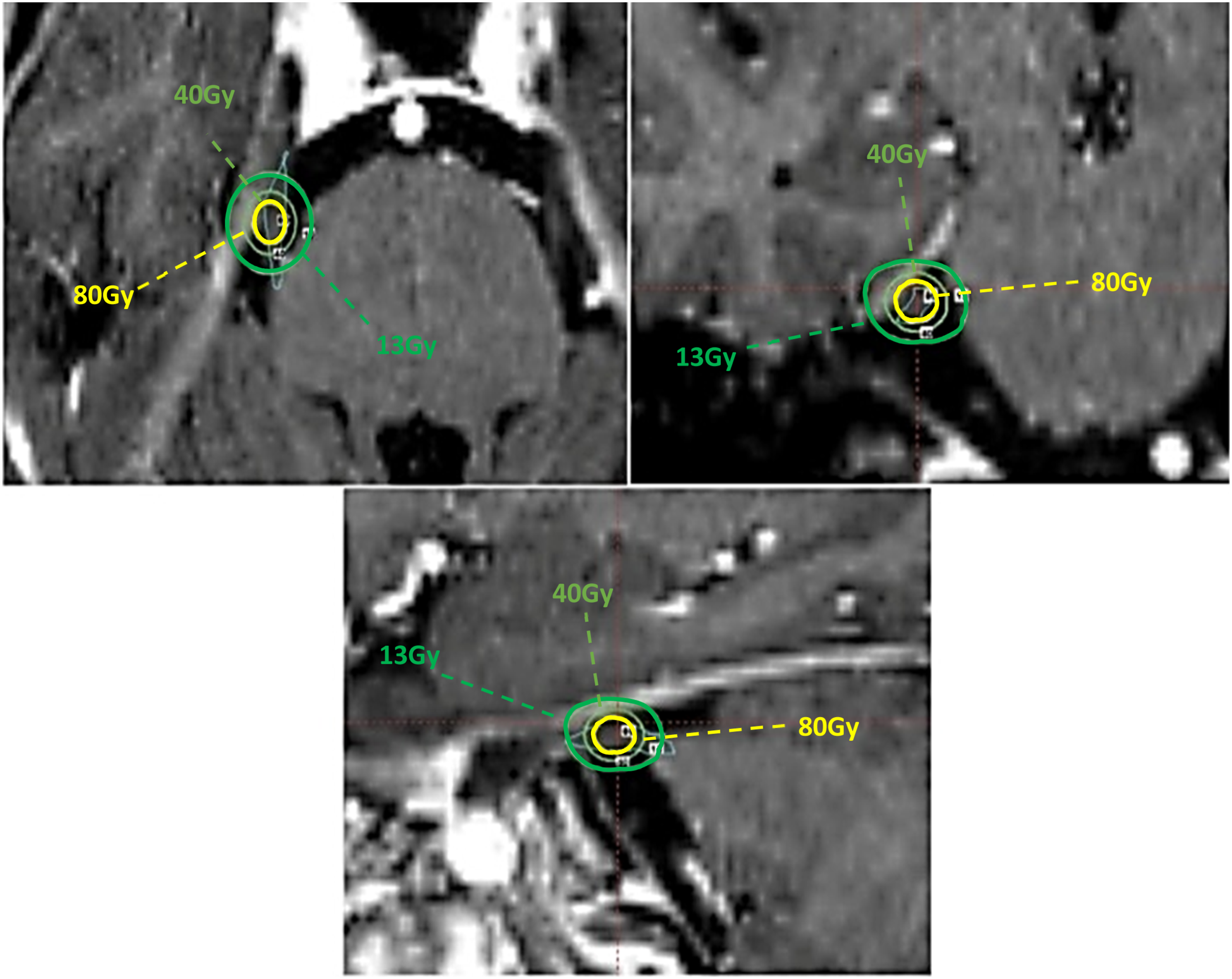
Patient Dosimetric Measurements
Three groups of TLDs were sealed in thin 1 × 1 cm2 plastic envelopes and then fixed on top of patients’ thyroid gland, eyes, uterus, and ovaries. Moreover, three separate dosimeters were used for background dose monitoring in each measurement, whose readings were subtracted from mean value readings reported in the main measurements.
Calculation of LAR
Lifetime Attributable Risk (LAR) is the probability of being clinically diseased by (or dying from) specific cancer after radiation exposure until the end of life and has been calculated for the received dose to the mentioned organs using BEIR VII (2006) method.
24
D represents dose, and S(a)/S(e) is the probability of surviving to the attained age (a) conditional on survival to exposed age (e) and was derived from the life span tables of the UK Office for National Statistics 2006–2008. L is the risk-free latent period (L = 5 for solid cancers).
Results
Calibration of TLDs
The actual output of TLDs during heating inside the TLD reader is light. The amount of this emitted light is directly related to the absorbed dose of radiation by the dosimeter, and the TLD reader convert emitted photons to an electrical current using a Photo Multiplier Tube (PMT). This current is collected by the TLD reader and reported in the nano-Coulomb (nC) unit as TLD response.
38
Calibration curves of TLDs at different dose levels are shown in Figure 4. The regression coefficient and matched line equation are specified too. Calibration curves of TLDs in low-level doses.
Extracranial Doses
Measured Dose (cGy) of Extracranial Organs for Treated Patients by Gamma Knife.

The LAR Values for Treated Patients With Trigeminal Neuralgia by GK.

Figure 5 gives the absorbed dose changes as a function of the average organs to isocenter distance. Regardless of the prescribed dose, the absorbed dose in various organs decreases by increasing the organs to isocenter distance. These results are in agreement with other studies by Di Beta,
17
Ioffe,
39
Novotny,
33
and Maarouf.
40
But in Berk’s study,
41
because of different head angles (125° vs 72° in the current study) (Figure 1), the thyroid dose was more than the eyes’ dose. The absorbed dose as a function of organs to isocenter distance.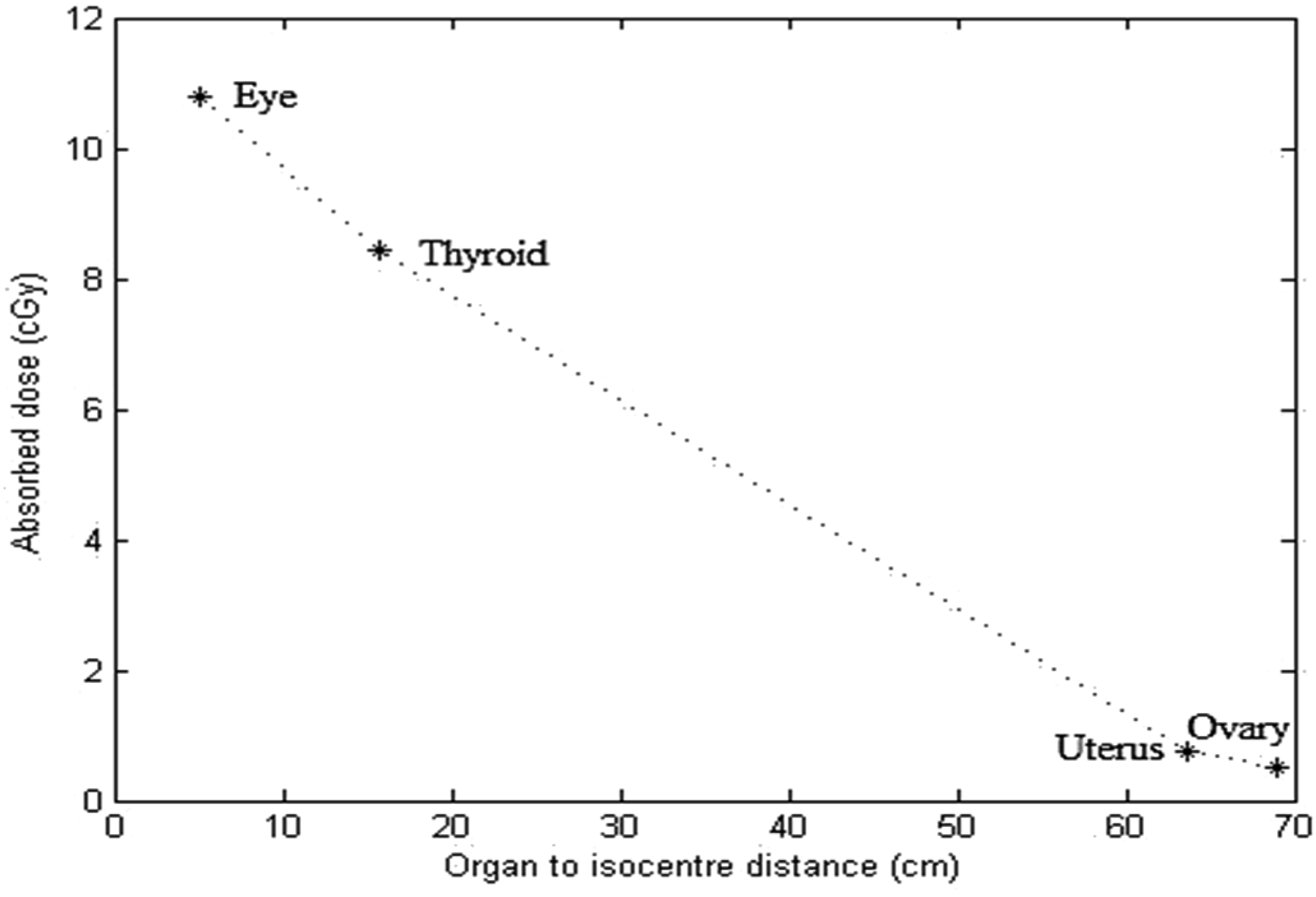
Discussion
Comparing the measured dose in studied organs with related tolerable doses reported by valid references demonstrates no serious concern regarding pregnant patients suffering from Trigeminal Neuralgia undergoing typical GK treatment. The duration of SRS treatment using a GK can vary significantly. For example, when a single isocenter is used for SRS in trigeminal neuralgia (TN) cases, the total treatment time shows relatively smaller variation, around a factor of ∼4. However, when multiple isocenters are employed, this variability can increase by up to a factor of 10. Several factors contribute to this variability. Traditionally, the decay of Cobalt-60 sources, with a half-life of 5.26 years, has been considered a significant factor. Over time, for comparable treatment plans, the beam-on time increases proportionally, doubling after one half-life. However, the introduction of progressive plugging/sector blocking can have a similar impact. Additionally, collimator factors and individual patient geometry also affect the beam-on time. While scheduled or unscheduled time gaps in treatment may occur, they are less likely in TN cases, where treatments are typically administered as a continuous exposure. It is suggested that prescribing a specific biologically effective dose (BED) rather than a physical dose may lead to optimal safety and efficacy in stereotactic radiosurgery (SRS) for trigeminal neuralgia (TN). The findings indicate that a therapeutic ratio for TN treatment could be achieved by targeting a BED range of 1820–1962.5 Gy. Within this range, a long-term pain-free incidence of 90% and a low risk (less than 10%) of developing hypoesthesia can be expected. However, it is important to note that higher BED values do not correspond to a higher probability of pain control, but rather an increased risk of complications. 42 The possibility of receiving the scattered and leaked radiation to tissues out of the radiation field will increase in longer treatments. Regarding radiation treatments, knowing the doses to be deposited outside the treatment volume beforehand may allow a risk estimation of detrimental effects and, if possible, precautions to minimize the probability of developing them. Absorbed doses in mentioned organs, reference values, and results of similar studies are discussed below in detail:
Eyes
Comparing Absorbed Dose of Organs With Previous Studies.

Thyroid
The thyroid equivalent dose was 8.27 ± 1.01 cGy, (.009% of the prescribed dose), and according to the calculated LAR value, the risk of secondary cancer is 23 per 100,000 people, which is the highest LAR between mentioned organs. In previous studies performed on thyroid dose measurement, higher thyroid doses of .4, .1, .2, .4, .5, and .13% of prescribed dose (Table 3) were seen owing to larger collimators size used (8, 14, 18, and 30 mm), multiple isocenters, multiple shots, and larger angle (125°) resulting in increased scattered radiation.17,20,23,33,40,41,45,46
Uterus and Ovaries
The measured doses of the uterus and ovaries were .63 ± .24 cGy and .471 ± .21 cGy, which are .006 and .005% of the prescribed dose, respectively (Table 1). These values are under the permissible dose threshold of the uterus and ovary.43,44 In other studies reported in Table 3, due to the previously mentioned reasons for eye and thyroid, the reported doses for the uterus and ovaries were higher than the corresponding dose in this study.20,33,39,40,47 Calculated LAR values for the uterus and ovaries were 1 and 2, respectively, meaning that if 100,000 patients are treated with this method, one or two people will probably develop secondary cancer.
TD5/5 and TD50/5
Comparing Absorbed Dose to Selected Organs With NCRP and ICRP.

Conclusion
In this study, the average absorbed dose of the eye, thyroid, ovaries, and uterus were 10.64 ± 1.08, 8.26 ± 1.01, .471 ± .21, and .63 ± .24 cGy, equivalent to .11, .09, .005, and .006% of the prescribed dose (90 Gy), respectively. Leakage radiation, collimator scattering, and internal scattering comprise the most important factors leading to extracranial organs’ receiving dose. The out-of-field organs’ absorbed dose is a function of the organs to isocenter distance, collimator size, and target location in the patient's head. On average, mean doses to mentioned organs were smaller than their tolerance doses, and it seems there is no significant limitation to treating TN pregnant patients by gamma knife. In the case of ovaries and fetuses, due to the fetus’s sensitivity to radiation and long-life expectancy, particular strategies (such as positioning) should be considered to reduce radiation as low as reasonably achievable (ALARA).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded and supported by Tehran University of Medical Sciences (TUMS), Tehran, Iran; Grant no 98-01-103-41980. This study was undertaken in Iran’s GK Center.


