Abstract
The present work aimed to evaluate acute and sub-acute toxicity of cortex cassava crude extract containing scopoletin and cyanide. The limit test method of OECD at 2000 mg/kg was used to determine the LD50 of the crude extract in female Wistar rats, and the sub-acute toxicity was conducted in broilers by incorporating their feed with the cortex powder at 2 mg, 4 mg and 8 mg of powder per 100 g of standard food, corresponding to MIC, 2MIC and 4MIC: those concentrations, respectively, corresponded to .07 μg, .14 μg and .28 μg of scopoletin and 2.8 μg, 5.6 μg and 11.2 μg of cyanide. An oral LD50 greater than 2000 mg/kg was obtained, and the effective antifungal dose was non-toxic. No changes were observed in the rate of biochemical parameters and relative weight of the liver, kidneys and brain of broilers. Any damages were not observed after the histopathological exams of these organs regardless the diet considered, compared to the control group. All results ranging from efficacy and safety of cassava cortex extract containing scopoletin and cyanide allowed us to consider it as biopesticide for staple food preservation against post-harvest losses.
Background
Scopoletin is a kind of botanical natural phenolic coumarin and an important member of the group of phytoalexins isolated from many plants, such as Erycibe obtusifolia, Aster tataricus, Foeniculum vulgare, Artemisia annua, Sinomonium acutum and Melia azedarach.1,2 Studies have shown that scopoletin has a wide spectrum of biological activities, such as acaricidal, anti-inflammatory, antitumoral, antioxidative, hepatoprotective, insecticidal, antifungal and alleopathic properties.3–5 Despite its beneficial effects, scopoletin like all secondary metabolites has chemical functions which at a certain dose interact with vital molecules in the body and cause deleterious effects on organs such as, liver, kidneys and brain.
Indeed, the toxicities associated with cassava products are most often attributed to the presence of cyanide there in. Some of these toxicities, which are commonly seen in cassava consuming populations, include endemic cretinism characterized by very severe mental retardation and several neurological abnormalities. 6 Osuntokun 7 has also described a tropical ataxic neuropathy among Nigerians subsisting on cassava products. It is believed that the endemic cretinism associated with cassava-consuming populations is the result of more than a single factor. 6 Moreover, scopoletin is a compound also found in cassava products. 8 A previous study found that scopoletin exhibits excellent contact killing, as well as systemic, repellent and oviposition inhibition activities against Tetranychus cinnabarinus. 5 Hou et al 9 reported that after exposure to scopoletin, several typical neurotoxic symptoms, such as excitement and convulsions, were observed in mites, and the compound specifically inhibits the nervous system targets, Na+-K+-ATPase, Ca2+-Mg2+-ATPase and Ca2+-ATPase, which indicates that scopoletin is a neurotoxin, in which Ca2+ plays a key role as an intracellular second messenger. Furthermore, Ezeanyika et al 10 studied the effects of scopoletin on the rat brain and showed that the relative brain weights of the rats fed by scopoletin were significantly (P < 0.05) less than that of the control from the third month of feeding. There were no significant changes in the lipid peroxide levels of the rat brains in the various groups. This result suggested that scopoletin is involved in the pathogenesis of the neuropathy seen in cassava consuming populations. Likewise, Gloria et al 11 reported forty three deaths among pigs fed by boiled cassava meal at a private piggery over a period of 2 years. The signs of sudden death in some cases were blood exuding from the external nares, vomiting, muscular weakness and pain or reluctance to move, and stunted growth. The necropsy lesions included skeletal and cardiac muscle degeneration and necrosis, icterus, hepatic necrosis and oedema of the dependent parts. The deaths and clinical signs were not due to a cyanide toxic principle in cassava, possibly the coumarins (scopoletin) without precise dose, which is found in high levels in cassava diet even after heat treatment. Due to the human harmful effects of scopoletin and cyanide, it is becomes imperative to know the toxicity level of the cassava cortex crude extract.
However, recent studies were conducted by Njankouo et al; Njankouo et al8,12 and showed that Cameroonian-improved cassava varieties accumulated scopoletin more than locals. All parts (skin and parenchyma) of the cassava root contain scopoletin and cyanide. Traditional processing reduced cyanide content and modified scopoletin level in derived foods (chips having the highest scopoletin level, compared to the gari and fufu). Furthermore, extract from cassava cortex containing scopoletin showed a better mycelium growth inhibition. The minimal inhibitory concentration ranged between .07 ± .00 μg/ml and .15 ± .00 μg/ml. The dose of 8 mg per 100 g of maize corresponding to 4 MIC and containing .28 μg of scopoletin and 11.2 μg of HCN was more effective in preserving maize.
This work was therefore aimed to evaluate the oral acute and sub-acute toxicity of cassava cortex crude extract containing scopoletin and cyanide in the rats and the broilers, respectively.
Materials and Methods
Plant Material
The cassava roots of improved variety ‘0110’, which present the highest scopoletin content were considered as plant material. 8 They were harvested, washed, peeled, and then cortex was sundried and ground to obtain dry cortex powder.
Experimental Animals
Female albino rats of Wistar breed (60 days old, weighing approximately 100 ± 10 g) not genetically modified were purchased from the laboratory of animal physiology at the animal house of the Laboratory of Pharmacology and Toxicology, Department of Biochemistry, University of Yaoundé I. Those animals were kept at room temperature for a photoperiod cycle of 12 hours light/dark for the assessment of oral acute toxicity. After a 5-day acclimation period, the animals were kept in their cages and received standard diet and water ad libitum. In order to ensure good hygienic status of animals, the sawdust used as litter was renewed twice per week.
Food Composition of the Different Diets for 100g.
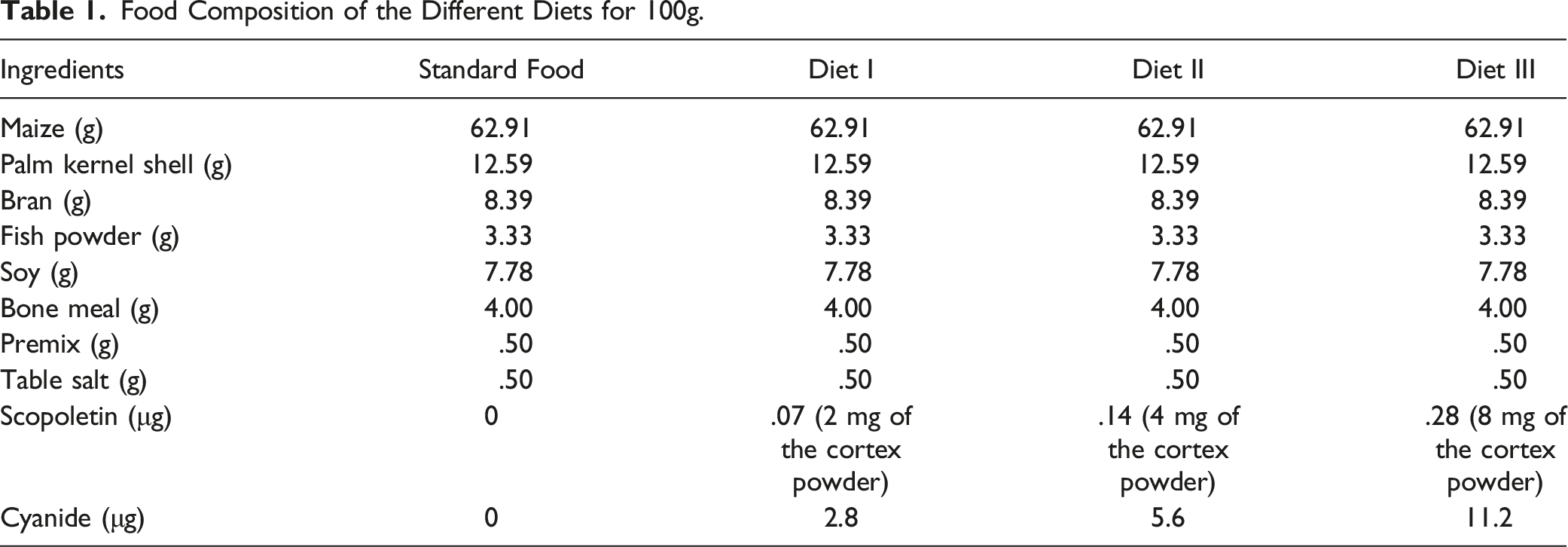
Preparation of Cassava Cortex Extracts
The cortex of improved cassava variety ‘0110’ that accumulated the highest of scopoletin, as shown by Njankouo et al 8 was used in the present study. The method described by Gnonlonfin et al 13 was performed for scopoletin extraction and quantification. Here, 2 g of this cortex powder in 10 mL of pure ethanol (Sigma-Aldrich) was macerated for 48 hr (in a porcelain crucible), filtered into small sterile flasks (using Whatman No. 1 fluted filter paper) and concentrated to obtain the crude extract.
Preparation of Cortex Crude Extracts Solutions and Different Diets for Broilers Feeding
A final concentration of 100 mg/mL was obtained by dissolving one hundred mg of crude extract containing scopoletin in 1 mL of sterile distilled water. The solutions were conserved in a freezer after homogenization using a shaker. The volume of solution corresponding to the doses of 2000 mg/kg containing 92.74 μg of scopoletin and cyanide traces to be administered to the female rat for the assessment of oral acute toxicity was calculated as follows
𝑉 = volume of extracted solution to be administered (mL), D = dose (mg/kg), 𝑊 = animal weight (kg), and 𝐶 = the concentration of extract solution to be administered (mg/mL).
The powder of cassava cortex ‘0110’ used for the sub-acute toxicity assessment was mixed with standard food of broilers (provender) at the doses of 2 mg/100g, 4 mg/100g and 8 mg/100g corresponding to MIC, 2 MIC and 4 MIC and containing .07 μg, .14 μg and .28 μg of scopoletin and 2.8 μg, 5.6 μg and 11.2 μg of cyanide, respectively. The used doses were informed by the previous study that showed an efficient antifungal activity on the moulds isolated from the maize. 8
Acute Toxicity Study
Acute toxicity of the cassava cortex extract containing scopoletin was studied in accordance with the OECD guideline 425. A limit test was performed using healthy female albino rats weighing 100 ± 20 g. 14 Prior to dosing, animals were fasted overnight, and the dose for each animal was determined based on body weight. Initially, a single dose of 2000 mg/kg of extract was administered to 1 animal by gavage using a stomach tube. After the administration, food was withheld for a further 3–4 hours. The animal was observed once during the first 30 minutes after dosing, then periodically, during the first 24 hours. As the animal was not dead, 4 additional animals were treated with the same dose and observed similarly. Then, all the survived animals were observed within 14 days. The LD50 is higher than 2000 mg/kg, if you have any animal tested dead; high or equal if you have 1 animal dead and lower than 2000 mg/kg if you have 2 or 3 dead.
Sub-Acute Toxicity Study
The Organization for Economic Cooperation and Development (OECD) guidelines relating to sub-acute toxicity was used to study sub-acute toxicity on the broilers with slight modifications. 15 Forty broilers of 21 days were weighed, marked orderly and divided into 4 batches of 10 broilers each. Batch 0 was taken as the control and received only standard diet (provender). Batches I, II and III were the test and received the diet prepared at the doses of 2 mg, 4 mg and 8 mg of powder of cassava cortex per 100g of standard food (provender) of broilers. These doses correspond to 1 MIC, 2 MIC and 4 MIC and containing .14 μg and .28 μg of scopoletin and 2.8 μg, 5.6 μg and 11.2 μg of cyanide, respectively, and fed during 30 days. These doses corresponded to the antifungal activity efficiency doses.
Biochemical Parameters Dosing
At the end of the 30th day of feeding, all broilers were fasted overnight (12 hrs) and then arterovenous blood samples collected during sacrifice in dry 5 mL tubes (non-heparinized) were centrifuged at 3000 r/min for 15 min. The serum obtained was served for the assessment of liver and kidney function parameters. The experiment was performed in accordance with protocols provided with commercial kits Biolabo. The rates of aspartate amino transferase (AST), alanine amino transferase (ALT), creatinine, LDL cholesterol, total cholestorol, triglyceride and malonic dialdehyde were assessed.
Histopathological Exams of Broilers Organ
All broilers in this study were subjected to general autopsy. They were pinned down in a dissection tray by placing them with ventral side up. The abdominal skin was left with forceps and cut through with scissors. The scissor blade was close and inserted under the skin and moved in the cephalic direction. The broilers were cut along the body midline, from the public region to the lower jaw. A lateral cut was made about half way down the ventral surface of each limb. Liver, kidney and brain were then cut, cleaned and kept in the fridge. The relative weight of the liver and kidney was determined by the formula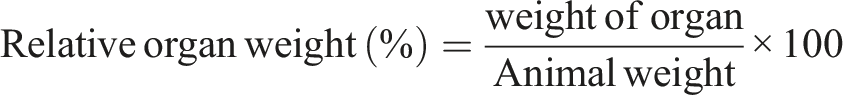
The liver, kidney and brain fixed in 10% formaldehyde for 3 weeks were cut into small pieces of 5–10 mm and then dehydrated in cassettes by immersion in alcohol and acetone successively. The latter was eliminated by xylol before being flowed in moulds containing paraffin melted by heating at 60°C. 16 After cooling the strong block of paraffin containing cloth, it was cut with a microtome to achieve cuts of 5 μm of thickness. The cuts were spread out and glued-dried on blades in steam for 1 night. They were coloured by a solution of Haematoxylin-Eosin. After colouration, the installation was made with Eukitt, and the sample was placed between slide and slide-covers. The preparation was air-dried, and the architectural aspects of the kidney, liver and brain were observed.
Statistical Analysis
Data were expressed as mean ± standard error (SE). All analyses were carried out using Graphpad prism 7. Statistical significance was determined by Student t test. Significant differences were considered at P < .05.
Results and Discussion
Acute Toxicity
Effects of the Extract on the General Appearance, Behaviour and Mortality of Rats
Observations of Rats After Administration of the Dose of 2000 mg/kg of Cassava Cortex Extract (0110) Containing Scopoletin in Acute Toxicity Test.

R1…R5: number of rats in the group; x: not determined.
Effects of the Extract on the Body Weight of Rats
The changes in body weights of the tested animals during the observation period had been shown in Figure 1. It can be seen that rats R1, R2, R4 and R5 of the test group and the 5 rats of the control group had normal growth. The weight increased gradually during the 14 days of observation. For rat R3 in the test group, the first 48 hours were marked by a sharp decrease in weight. Food intake had promoted a return to normal growth until the end of the 14 days observation period. Variations in body weights of rats in the acute toxicity test.
Indeed, in toxicity studies, growth retardation is used as an indicator of the adverse effects of chemical compounds. 17 It is correlated to the physiological state of the animal. This growth reduction can be explained by a reduction in food consumption, but also by the possibility of dose/absorption interactions and by the reduction in the quantity of food absorbed. 17 The information presented on the experimental data of acute toxicity in female Wistar Albinos rats suggests that the crude extract containing scopoletin from cassava cortex powder ‘0110’ should be classified as a low oral toxicity substance with an LD50 > 2000 mg/kg. 15 No deaths were recorded among the animals treated with the single dose of 2000 mg/kg of this extract after the 14-day experimental time.
Sub-Acute Toxicity
Effects of Different Diets on the Broilers Body Weight
During experimentation, no particular behaviour or deaths were reported. Broilers did not show any visual signs of toxicity due to feed. This feed had promoted rapid growth of broilers in the groups fed with diets II and III (i.e. 4 mg and 8 mg of cassava cortex powder/100 g of feed) during the third and fourth week compared to the control batch. The monitoring of the variation of broilers weight during sub-acute toxicity experimentation is represented in Figure 2. This figure highlighted normal growth in all groups (control diet and tests diets). This growth was more pronounced in diet I, that is, 2 mg powder/100g feed and control diet during the first and second week, with average growth differences of 202.14 g and 197.13 g for the first week, 364.57 g and 325.43 g for the second week, respectively, compared to 121.71 g and 182.86 g only for the first week, 252.00 g and 199.71 g for the second week in diet II, that is, 4 mg/100g of feed and diet III, that is, 8 mg/100g of feed, respectively. This difference in growth level became more significant in diets II and III (170.57 g and 247.14 g, 279.43 g and 279.43 g, respectively), compared to 88.00 g and 61.14 g, 233.57 g and 221.64 g for the control diet and diet I, respectively, during the third and fourth week of study. These differences in animal growth recorded during the study show that the diets enriched with cassava cortex powder have a positive impact on the growth of broilers. Change in body weight of broilers during the sub-acute toxicity test of diets enriched with cassava cortex powder containing scopoletin and cyanide. The results are mean ± SE, (n = 10). Control diet = 0 mg of powder per 100g of standard feed; diet I = 2 mg of powder per 100g of feed (MIC/100g); diet II = 4 mg of powder per 100g of feed (2 MIC/100g) and diet III = 8 mg of powder per 100g of standard food (provender) (4 MIC/100g).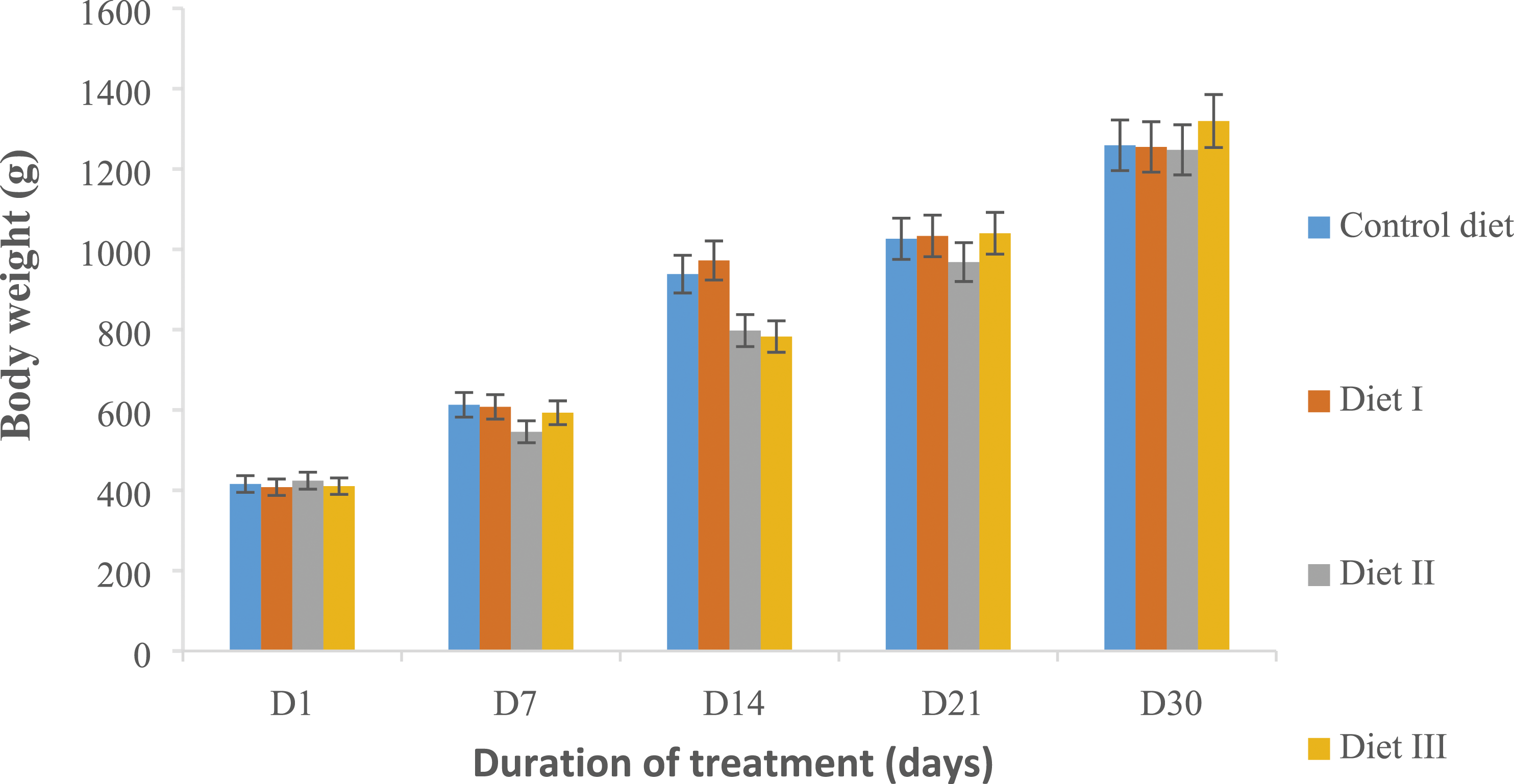
However, the follow-up of the weight evolution of the birds shows that during the first and second week, the growth was more important in the control diet, fed with standard feed and diet I, that is, 2 mg/100 of feed, while it was slowed down in diet II, that is, 4 mg/100g of feed and diet III, that is, 8 mg/100 g of feed, respectively. During the third and fourth week, the opposite effect was observed. The growth delay recorded in the first and second week in diets II and III would be considered as an adaptation period to the new diet incorporated with the cassava cortex powder. The rapid growth recorded in the same diets in the third and fourth week would be explained by the fact that the cassava root cortex would have increased the energetic potential of the feed, thus favouring a rapid weight gain. Fajemisin et al 18 reported that the energetic potential of cassava is comparable to that of cereals, especially maize. These results are similar to those of Olaifa et al 19 who showed that substitution of maize in the standard diet with 25% protein-enriched cassava cortex powder gives the similar results to those of poultry fed with standard diet. However, diets with 50% and 75% substitution rates were more likely to result in weight loss in poultry after 28 days of exposure. This suggests that in these substitution proportions, constituents other than cyanide exist in the fermented cassava cortex powder, resulting in adverse effects in broilers. Otherwise, Jerome and André, 20 showed that it is possible to replace maize in complete rations fed to pigs, poultry and rabbits with cassava chips flour at 20, 30 and 40%, respectively, without observing any negative effect on animal performance. However, the main disadvantage inherent in the use of cassava is the presence of cyanide. This is more or less important depending on the variety (sweet or bitter) and the preparation of the tubers (peeling, retting, cooking, drying or ensiling). Sulphur amino acids play a major role in the detoxification of ingested cyanide. Furthermore, the adverse chronic effects of cyanide on animal growth and reproduction can be limited by ensuring the protein balance of the rations distributed, especially with respect to sulfur amino acids. 20 These effects are relatively important in monogastric animals, whereas in ruminants, cassava is partially detoxified in the rumen.
Effects of Different Diets on Biochemical Parameters Dosed
Levels of Some Biochemical Parameters in the Serum of Broilers Fed With the Diets Enriched With Cassava Cortex Powder Containing Scopoletin and Cyanide.

The results are mean ± ES, (n = 10), ns (not significant) and P < .05).
Control diet: 0 mg of powder per 100g of standard feed; diet I: 2 mg of powder per 100g of feed (MIC/100g); diet II: 4 mg of powder per 100g of feed (2 MIC/100g) and diet III: 8 mg of powder per 100g of standard food (provender) (4 MIC/100g).
aAlanine amino transferase (U/L).
bAspartate amino transferase (U/L).
cTotal cholesterol (g/L).
dCreatinine (g/L).
eHDL-cholesterol (g/L).
fMalonic aldehyde (g/L).
gTriglyceride (g/L).
In contrast to our results, Ezeanyika et al 10 studied the comparative effects of scopoletin and cyanide on the brains of rats fed by a standard diet containing .07 μg of scopoletin and 1.8 mg of cyanide for 12 months and showed that this diet did not induce any lipid peroxidation and that only the clinical signs of neuropathy were observed. Hence, the implication of scopoletin in the cause of neuropathy in populations who regularly consume cassava. Furthermore, Gloria et al 11 showed that 43 pigs died after being fed with a diet of boiled cassava flour for 24 months. Signs leading to death were nasal discharge of blood, vomiting, muscle weakness, difficulty moving and stunted growth. These clinical signs were not caused by the toxic effect of cyanide, but probably by the toxic effect of coumarins (scopoletin), which is found at high rates in cassava processing products even after heat treatment.
Effects of Different Diets on Relative Weight and Histopathology of Some Vital Organs (Liver, Kidneys and Brain)
Relative Organ Weights of Broilers Fed With the Diets Enriched With Cassava Cortex Powder Containing Scopoletin and Cyanide.

The results are mean ± SE, (n = 10); *(Significance, P < .05); ns (not significant), control diet: 0 mg of powder per 100g of standard feed; diet I: 2 mg of powder per 100g of feed (MIC/100g); diet II: 4 mg of powder per 100g of feed (2 MIC/100g) and diet III: 8 mg of powder per 100g of standard food (4 MIC/100g).

Microphotographs of broilers livers (×100), kidneys (×200) and brains (×100). (Hematoxylen-Eosin (HE) stain). Control diet = 0 mg of powder per 100g of standard feed; diet I = 2 mg of powder per 100g of feed (MIC/100g); diet II = 4 mg of powder per 100g of feed (2 MIC/100g) and diet III = 8 mg of powder per 100g of standard food (4 MIC/100g). Liver: Vp = hepatic portal vein; He = hepatocyte; Cs = sinusoidal capillary; Ah = hepatic artery; Cb = bile duct; Kidney: Gl = glomerulus; Eu = urinary space; Tcd = distal convoluted tubule; Tcp = proximal convoluted tubule; Brain: Cn = neuronal cells.
Histopathological examination of organs (livers, kidneys and brains) of poultry showed no obvious signs of toxicity following exposure to a diet consisting of a mixture of standard feed and cassava cortex powder for 30 days. This confirms what was observed with the biochemical parameters measured. This result does not corroborate those of Chilaka et al 21 who showed that scopoletin after prolonged exposure induced toxic liver damage at very low concentrations causing hepatocyte necrosis resulting in jaundice in pigs. Histological examinations of organs showed necrosis of cardiac and skeletal muscles, hepatocyte necrosis and oedema of dependent organs. Similarly, Ojewole et al 22 presented scopoletin as a neuromuscular blocking agent, which would cause paralysis in pigs. Furthermore, the signs of cyanide toxicity being goiter, neuropathy and fatty liver, 23 Gloria et al 12 report that none of these clinical signs were observed, therefore, the toxicity seen was due to scopoletin.
Exposure of the broilers to the different diets (diet I, diet II and diet III) containing .07 μg and 2.8 μg (MIC); .14 μg and 5.6 μg (2 MIC); .28 μg and 11.2 μg (4 MIC) of scopoletin and cyanide, respectively, for 30 days showed no signs of toxicity in the weight evolution, biochemical parameters measured (AST/ALT, total cholesterol, triglycerides, creatinine, malonic aldehyde and HDL-cholesterol), relative weights and histological sections of organs (liver, kidneys and brain).
These results are due to the fact that the level of scopoletin in the food would not be sufficient to induce toxicity, since scopoletin is a compound whose content in cassava processing products is neither affected by solar drying, refrigeration, storage or preparation. 24 While cyanide is a highly volatile compound, the preparation of the feed would have allowed its escape, significantly reducing its content in the cassava cortex feed/powder mixture. 25 The observed difference may also be attributed to the prolonged and repeated exposure (12–24 months) of study animals to these diets by these authors. 26
Conclusion
This work was aimed to evaluate the oral acute and sub-acute toxicity of cassava cortex crude extract containing scopoletin and cyanide in the rats and the broilers, respectively. The information presented on the experimental data of acute toxicity in female Wistar Albinos rats allows us to conclude that the LD50 is higher than 2000 mg/kg, making this extract a low toxic substance, which can be classified in category 5 of the Globally Harmonized Classification System (GHS) of the OECD. In the sub-acute toxicity study, all the results obtained from broilers weights, relative organ weights, biochemical parameters and histological sections of organs showed no signs of toxicity of the standard food incorporated with powder of cassava cortex containing scopoletin and cyanide at the maximum effective antifungal dose of 8 mg of powder per 100g of standard food corresponding to 4 MIC and containing .28 μg of scopoletin and 11.2 μg of cyanide.
Footnotes
Acknowledgments
The authors are very grateful to the Institute of the Agricultural Research for Development (IRAD, Nkolbisson Yaoundé) and the University of Yaoundé I, Cameroon, who provided all the facilities for the study.
Author Contributions
Njankouo Ndam Youchahou: formal analysis, investigation and writing original draft. Njingou Ibrahim: formal analysis and investigation. Mounjouenpou Pauline: conceptualization, methodology, writing-review and editing, supervision. Fewou Moundipa Paul: conceptualization and methodology. Nyegue Maximilienne Ascenssion: writing-review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
