Abstract
Introduction
Gastric cancer, often referred to as stomach cancer, is a malignant tumor that originates in the stomach lining. 1 It ranks as the fifth most common cancer globally and the third leading cause of cancer-related deaths. 2 Approximately one million new cases are diagnosed each year, and the worldwide impact of this disease continues to increase.3,4 Gastric cancer poses a significant public health challenge due to its aggressive nature, late diagnosis, and limited treatment options. 5 The prevalence of gastric cancer varies geographically, with the highest incidence rates found in Eastern Asia, Eastern Europe, and South America. 6 Several factors contribute to the development of gastric cancer, including Helicobacter pylori infection, high salt intake, smoking, and genetic predisposition.7-9 Early detection and prevention strategies are crucial in reducing the disease's burden.10,11
Despite advances in medical research, current therapeutic options for gastric cancer, such as surgery, chemotherapy, radiation therapy, and targeted therapies, remain insufficient. Many patients present with advanced-stage disease or develop resistance to existing treatments, resulting in poor prognosis and high mortality rates.12-14 The 5-year survival rate for gastric cancer remains disappointingly low, highlighting the urgent need for new and more effective treatment strategies. 14
In recent years, natural compounds have garnered attention for their potential role in cancer therapy. 15 Curcumin, the main bioactive component of turmeric (C longa), has been widely studied for its anti-inflammatory, antioxidant, and anti-cancer properties.16,17 Mounting evidence suggests that curcumin may modulate various cellular pathways involved in the initiation, progression, and metastasis of different cancer types, including gastric cancer.17-20 Curcumin's potential health benefits and clinical applications cover a broad range of medical conditions, such as Alzheimer's disease, cardiovascular diseases, and diabetes, among others.21,22 Its anti-cancer effects have been demonstrated in numerous in vitro and in vivo studies, as well as in preliminary clinical trials.7,23,24
Curcumin has been found to exert its anti-cancer activity through several mechanisms, including apoptosis induction, cell cycle arrest, angiogenesis inhibition, and suppression of invasion and metastasis.25-28 The phosphatidylinositol-3-kinase (PI3K) signaling pathway is a critical regulator of cell growth, survival, and metabolism in various cancers, including gastric cancer.29,30 Dysregulation of the PI3K pathway has been implicated in the development of drug resistance and poor prognosis in gastric cancer patients.30,31 Targeting this pathway may provide novel therapeutic opportunities for the management of gastric cancer. 31
Emerging evidence suggests that curcumin may inhibit gastric cancer cell progression by modulating the PI3K signaling pathway. 32 However, the dose-dependent effects of curcumin on gastric cancer cells and the precise mechanisms underlying its action on the PI3K pathway remain poorly understood. The aim of this study is to investigate the dose-dependent suppression of gastric cancer cell progression by curcumin through the inhibition of the PI3K signaling pathway. This study seeks to elucidate the molecular mechanisms underlying curcumin's anti-cancer effects and to evaluate its potential as a novel therapeutic agent for the treatment of gastric cancer. A better understanding of curcumin's role in gastric cancer therapy could contribute to the development of more effective treatment strategies and ultimately improve patient outcomes.
Methods and Materials
Ethics
The study has been approved by the Ethics/Review board of King Saud University under the reference number CT-2022-125, ensuring compliance with all relevant ethical guidelines for research involving human cell lines.
Cell Culture and Treatment
Human gastric cancer cell lines, AGS, were obtained from the American Type Culture Collection (ATCC) and maintained in their respective culture media: RPMI-1640 for AGS and MKN-45 cells and DMEM for SNU-1 cells. The media were supplemented with 10% fetal bovine serum (Gibco, Thermo Fisher Scientific, Waltham, MA, USA) and 1% penicillin–streptomycin (Gibco, Thermo Fisher Scientific, Waltham, MA, USA). The cells were incubated at 37°C in a humidified atmosphere containing 5% CO2. Cells were passaged regularly when they reached 80%–90% confluence to ensure continuous logarithmic growth.
Curcumin Treatment
Curcumin (purity ≥98%, Sigma-Aldrich, St. Louis, MO, USA) was dissolved in dimethyl sulfoxide (DMSO, Sigma-Aldrich, St. Louis, MO, USA) to create an mM stock solution. This stock solution was further diluted in culture media to achieve the desired concentrations for treatment (e.g., 0, 10, 20, 40, and 80 μM). Gastric cancer cell lines were exposed to various concentrations of curcumin for specific time intervals (e.g., 24, 48, and 72 hours) to assess the dose- and time-dependent effects of curcumin on the cells.
Cell Viability Assay
Cell viability was determined using the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) assay (Sigma-Aldrich, St. Louis, MO, USA). Gastric cancer cells were seeded in 96-well plates at a predetermined density and treated with different concentrations of curcumin. After incubation, the MTT reagent was added to each well, and the plates were incubated for an additional 4 hours. The formazan crystals formed were dissolved in DMSO, and absorbance was measured at 570 nm using a microplate reader (SpectraMax, BioTek, Thermo Fisher Scientific).
Cell Migration and Invasion Assay
Cell migration and invasion assays were performed using Transwell chambers (Corning, Corning Incorporated, Corning, NY, USA) with 8 μm pore size membranes. For the invasion assay, the upper surface of the membrane was coated with Matrigel (BD Biosciences, San Jose, CA, USA). Gastric cancer cells were suspended in serum-free media and added to the upper chamber, while media containing 10% FBS was placed in the lower chamber as a chemoattractant. After incubation, cells that migrated or invaded through the membrane were fixed, stained, and counted under a light microscope. Each experiment was performed with three independent replicates.
Reverse Transcription-Polymerase Chain Reaction for Phosphoinositide 3-Kinase Signaling Pathway Gene Expression
Total RNA was extracted from gastric cancer cells using TRIzol reagent (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer's instructions. Complementary DNA (cDNA) was synthesized using a reverse transcription kit (Thermo Fisher Scientific, Waltham, MA, USA), and quantitative real-time PCR (qRT-PCR) was performed using gene-specific primers and SYBR Green PCR Master Mix (TaqMan Universal PCR Master Mix, Applied Biosystems) in a real-time PCR system for PI3K, Akt, and mTOR. Gene expression was normalized to a housekeeping gene, and relative expression levels were calculated using the 2^(-ΔΔCt) method. Each experiment was performed with three independent replicates.
Western Blot Analysis for Phosphoinositide 3-Kinase Signaling Pathway Proteins
After curcumin treatment, gastric cancer cells were harvested and lysed using radioimmunoprecipitation assay (RIPA) buffer containing protease and phosphatase inhibitors to obtain total protein extracts. Protein concentrations were determined using the bicinchoninic acid (BCA) protein assay. Equal amounts of protein were separated on sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gels and transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with 5% non-fat milk or bovine serum albumin (BSA) in Tris-buffered saline containing .1% Tween-20 (TBST) for 1 hour at room temperature. Subsequently, the membranes were incubated with primary antibodies against PI3K (1:1000), AKT (1:2000), and mTOR (1:1000) (Sigma-Aldrich Corporation, St. Louis, MO, USA) overnight at 4°C. After washing, the membranes were incubated with appropriate horseradish peroxidase (HRP)-conjugated secondary antibodies. Each experiment was performed with three independent replicates.
Statistics
In the present study, GraphPad Prism 8 (GraphPad Software, Inc.) was utilized to perform statistical analysis on the collected data. The ANOVA test was chosen as the appropriate statistical method for comparing the study outcomes among the study groups.
Results
The study investigated the effects of curcumin on gastric cancer cells, assessing its impact on cell viability, migration, invasion, and the expression of genes associated with the PI3K signaling pathway.
Cell Viability
At 50 μM of curcumin, there was a significant decrease in cell viability at all time points (24 h, 48 h, and 72 h) compared to the 0 h control (P < .0001). The decrease in cell viability was more pronounced with higher curcumin doses and longer treatment durations. At 75 μM and 100 μM of curcumin, there was also a significant decrease in cell viability at all time points compared to the 0 h control (P < .05). At 100 μM of curcumin, cell viability decreased by 49% at 48 h and by 34% at 72 h compared to the 0 h control (Supplementary table 1) (Figure 1). Gastric cancer cell viability in response to various doses of curcumin (50, 75, and 100 μM) at different time points (0 h, 24 h, 48 h, and 72 h). A decrease in cell viability was observed with increasing curcumin dose and treatment duration. Each data point represents the mean of three independent experiments, each with three replicates (n = 3). At 50 μM of curcumin, there was a significant decrease in cell viability at all time points (24 h, 48 h, and 72 h) compared to the 0 h control (P < .0001). At 75 μM and 100 μM of curcumin, there was also a significant decrease in cell viability at all time points compared to the 0 h control (P < .05).
Cell Migration and Invasion
At 50 μM of curcumin, cell migration was significantly reduced compared to the control (P < .0001). Similarly, at 75 μM and 100 μM of curcumin, cell migration was significantly reduced compared to the control (P < .0001) (Supplementary table 2). Notably, the inhibitory effect of curcumin on cell migration increased with higher curcumin doses, with the greatest inhibition observed at 100 μM of curcumin (Figure 2). Effects of curcumin on gastric cancer cell migration and invasion at various doses (50, 75, and 100) compared to the control. A dose-dependent increase in cell migration and invasion inhibition was observed with increasing curcumin concentrations. Each data point represents the mean of three independent experiments, each with three replicates (n = 3). At 50 μM of curcumin, cell migration was significantly reduced compared to the control (P < .0001). At 75 μM and 100 μM of curcumin, cell migration was significantly reduced compared to the control (P < .0001). Similarly, curcumin significantly inhibited cell invasion in a dose-dependent manner. At 50 μM of curcumin, cell invasion was significantly reduced compared to the control (P = .0033). At 75 μM and 100 μM of curcumin, cell invasion was significantly reduced compared to the control (P < .001). Original magnification, ×100; bar = 200 μm.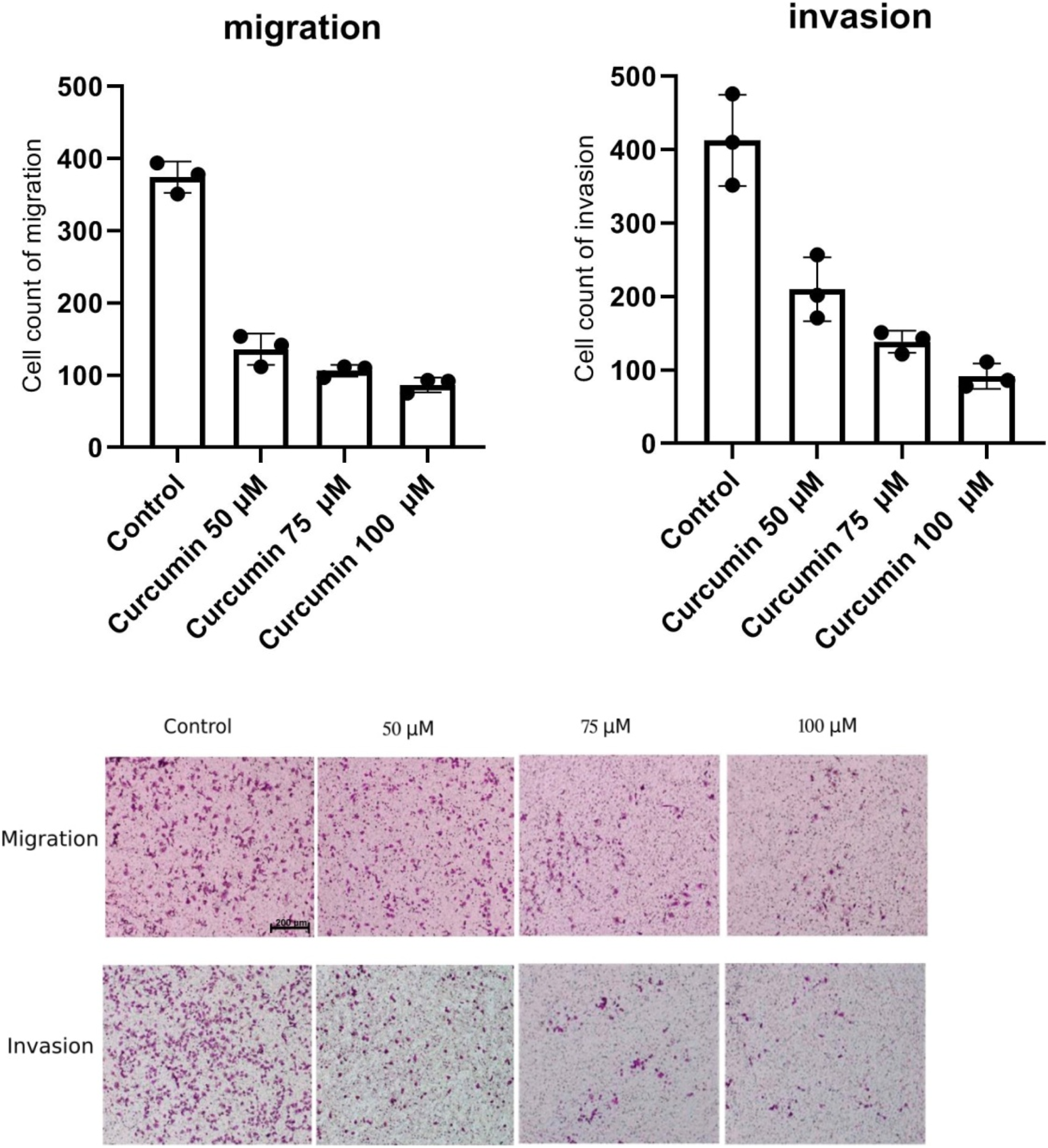
At 50 μM of curcumin, cell invasion was significantly reduced compared to the control (P = .0033) (Supplementary table 2). Similarly, at 75 μM and 100 μM of curcumin, cell invasion was significantly reduced compared to the control (P < .001). The inhibitory effect of curcumin on cell invasion also increased with higher curcumin doses, with the greatest inhibition observed at 100 μM of curcumin.
Expression of Phosphoinositide 3-Kinase Pathway Genes
At 50 μM of curcumin, the expression of PIK3CA, AKT1, and MTOR genes was significantly reduced compared to the control (P < .005). Similarly, at 75 μM and 100 μM of curcumin, the expression of these genes was significantly reduced compared to the control (P < .001). The inhibitory effect of curcumin on gene expression increased with higher curcumin doses, with the greatest inhibition observed at 100 μM of curcumin (Figure 3). Influence of curcumin on the expression of PIK3CA, AKT1, and MTOR genes at various doses (50, 75, and 100 μM) compared to the control. A dose-dependent decrease in gene expression was observed with increasing curcumin concentrations (P < .001). Each data point represents the mean of three independent experiments, each with three replicates (n = 3).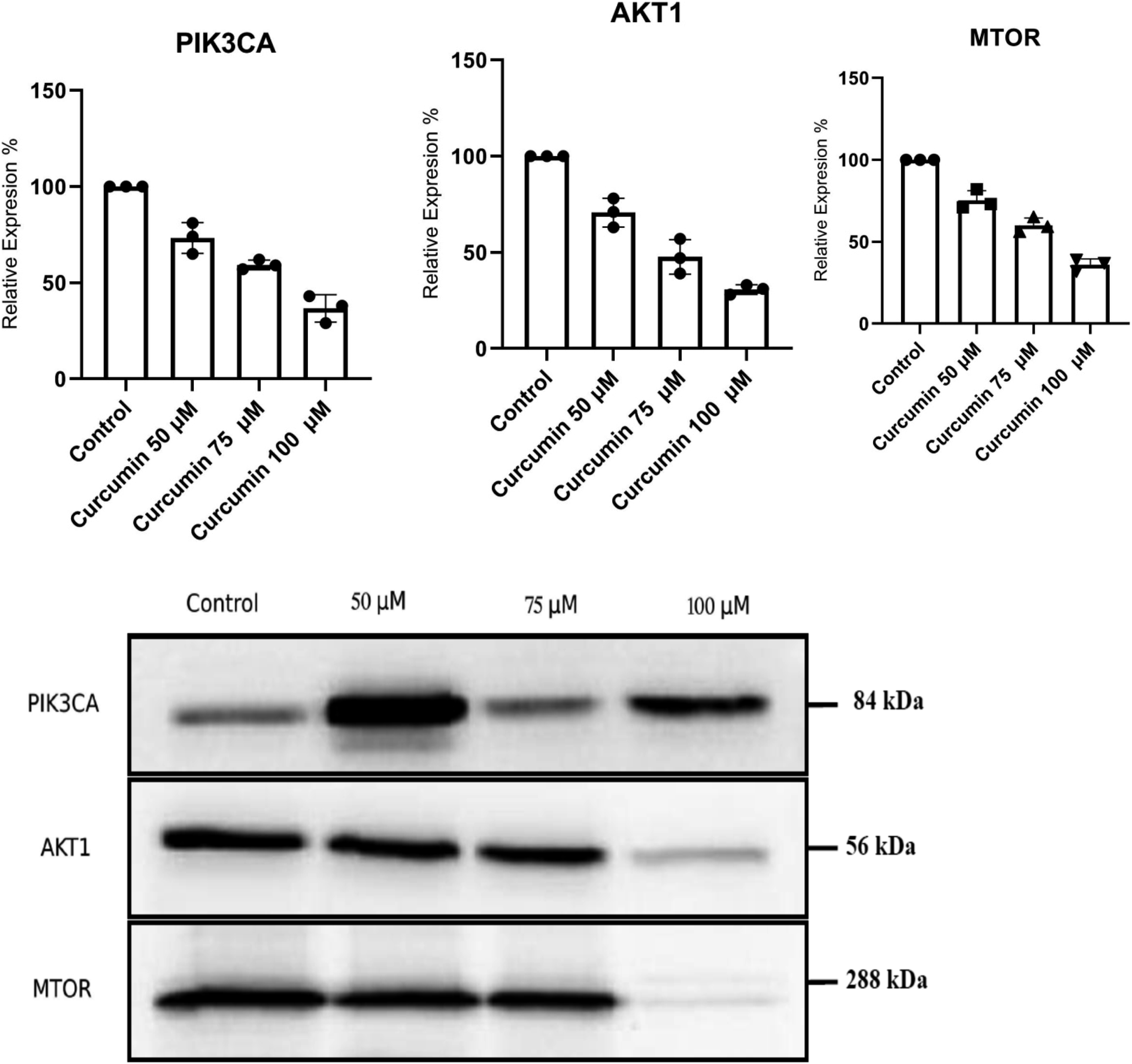
At 50 μM of curcumin, the expression of PIK3CA, AKT1, and MTOR genes was significantly reduced by 27%, 33%, and 28%, respectively, compared to the control (P < .005) (Supplementary table 3). Similarly, at 75 μM of curcumin, the expression of these genes was significantly reduced by 42%, 36%, and 38%, respectively, compared to the control (P < .001). The inhibitory effect of curcumin on gene expression increased with higher curcumin doses, with the greatest inhibition observed at 100 μM of curcumin. At this dose, the expression of PIK3CA, AKT1, and MTOR genes was reduced by 53%, 50%, and 52%, respectively, compared to the control (P < .001).
Western Blot of Phosphoinositide 3-Kinase Pathway Proteins
At 50 μM of curcumin, the expression of PI3K, Akt, and mTOR proteins was significantly reduced compared to the control (P < .005). Similarly, at 75 μM and 100 μM of curcumin, the expression of these proteins was significantly reduced compared to the control (P < .001). The inhibitory effect of curcumin on protein expression increased with higher curcumin doses, with the greatest inhibition observed at 100 μM of curcumin (Figure 4). The expression of Akt protein was reduced by 65% at 100 μM of curcumin compared to the control, indicating a strong inhibitory effect (Supplementary table 4). Western blot analysis of the effect of curcumin on PI3K, Akt, and mTOR protein levels at different doses (50, 75, and 100 μM) compared to the control. A dose-dependent decrease in protein expression was observed with increasing curcumin concentrations (P < .001). Each data point represents the mean of three independent experiments, each with three replicates (n = 3).
Discussion
The present study aimed to investigate the potential effects of curcumin on the progression of gastric cancer cells and its influence on the PI3K signaling pathway. The results revealed that curcumin inhibits gastric cancer cell proliferation, migration, and invasion in a dose-dependent manner, suggesting its potential as a therapeutic agent for gastric cancer treatment. This discussion will provide a comprehensive analysis of the study findings and their implications, as well as an evaluation of the underlying mechanisms and potential future research directions.
The study findings demonstrate that curcumin treatment leads to a significant reduction in gastric cancer cell proliferation, migration, and invasion. These results are consistent with previous research on curcumin's anti-cancer properties in various cancer types. 20 One of the critical mechanisms by which curcumin may exert its anti-cancer effects is through the inhibition of the PI3K signaling pathway, which plays a vital role in regulating cell growth and survival. 33
Our study represents a significant contribution to the existing body of research. While there have been several investigations exploring the impact of curcumin on the PI3K pathway in different cellular contexts, such as ovarian granulosa cells in polycystic ovary syndrome, 34 olfactory ensheathing cells in spinal cord injury, 35 human glioblastoma cells, 36 and exercise fatigue, 37 our study is the first to specifically evaluate the effects of curcumin on the PI3K pathway in gastric cancer cells.
Comparing our results with other studies that evaluated different signaling pathways in the context of curcumin's effects on gastric cancer, several interesting findings emerge. Zhang et al 38 utilized affibody-modified G-quadruplex DNA micelles for targeted delivery of curcumin in HER2-positive gastric cancer, enhancing synergetic therapy. Xu et al 39 investigated the inhibitory effects of the curcumin analog Da0324 on gastric cancer cells through the HOTAIRM1/miR-29b-1-5p/PHLPP1 axis. MaruYama et al explored the use of pyrolyzed deketene curcumin to control regulatory T cell generation and gastric cancer metabolism, cooperating with 2-deoxy-d-glucose. 40 Jabbari et al 41 studied the suppression of gastric cancer cell proliferation using Gemini curcumin and its modulation of lncRNA CCAT2 and c-Myc genes. Ham et al 42 investigated curcumin's inhibition of cancer-associated fibroblast-derived chemoresistance in gastric cancer through the suppression of the JAK/STAT3 signaling pathway.
Collectively, these studies demonstrate the diverse and multifaceted mechanisms through which curcumin exerts its effects on gastric cancer cells. Each study focuses on distinct signaling pathways, shedding light on different aspects of curcumin's therapeutic potential. Our study stands out as the first one specifically evaluating the effect of curcumin on the PI3K pathway in gastric cancer. By identifying the PI3K pathway as a key target of curcumin's action, we provide critical information for the development of targeted therapeutic approaches in gastric cancer treatment. The integration of findings from various studies, including ours, contributes to a comprehensive understanding of curcumin's potential in gastric cancer therapy, highlighting the importance of personalized and targeted treatment strategies for this complex malignancy.
The PCR and Western blot analyses conducted in this study indicate that curcumin can suppress the expression of genes and proteins involved in the PI3K signaling pathway. These results suggest that curcumin may target this pathway to inhibit the growth and progression of gastric cancer cells. It is essential to note that the PI3K signaling pathway is not the only pathway that curcumin may impact. Future research could explore other signaling pathways and molecular targets of curcumin to better understand its anti-cancer properties. 43
Another aspect to consider is the use of curcumin in combination with other anti-cancer agents or therapies. 44 Combining curcumin with other treatments may enhance its therapeutic effects and potentially overcome any drug resistance that may develop in cancer cells. 44 Further studies could investigate the efficacy of curcumin in combination with existing cancer therapies and its potential synergistic effects.
Curcumin, a polyphenolic compound derived from the turmeric plant (C longa), has been extensively studied for its health benefits, including anti-inflammatory, antioxidant, and anti-cancer activities. 21 The anti-cancer potential of curcumin has been demonstrated in various cancer types, including gastric cancer. Curcumin has been shown to inhibit cell proliferation, induce apoptosis, and suppress angiogenesis, metastasis, and invasion in different cancer models. 26
In gastric cancer, one study showed that curcumin might exert its anti-cancer effects, at least in part, through the inhibition of the PI3K signaling pathway. 45 This pathway plays a critical role in regulating cell survival, growth, proliferation, and motility in normal and cancer cells. 29 Aberrant activation of this pathway has been implicated in the development and progression of various cancers, including gastric cancer.
The anti-cancer effect of curcumin is attributed to its multifaceted and intricate molecular mechanisms. Curcumin exerts its anti-cancer properties by targeting various signaling pathways involved in cell growth, survival, invasion, and metastasis. 46 One of its central mechanisms is the inhibition of the PI3K/Akt/mTOR pathway, which is crucial for cell proliferation and survival. By blocking this pathway, curcumin suppresses cancer cell growth and induces apoptosis. Moreover, curcumin modulates the expression of key genes and proteins related to cell cycle regulation, such as p53 and cyclin-dependent kinases (CDKs), leading to cell cycle arrest and preventing uncontrolled cell division. 47 Curcumin also exhibits anti-angiogenic effects, inhibiting the formation of new blood vessels necessary for tumor growth and metastasis. Additionally, it interferes with various pro-survival and pro-metastatic molecules, including matrix metalloproteinases (MMPs), vascular endothelial growth factor (VEGF), and nuclear factor-kappa B (NF-κB), thus limiting cancer cell invasion and metastasis. 48 Furthermore, curcumin's antioxidant properties neutralize free radicals and reduce oxidative stress, which can contribute to cancer development. 46 Overall, the diverse and comprehensive mechanisms of curcumin's anti-cancer activity make it a promising candidate for further exploration and development in cancer therapy.
The dose-dependent action of curcumin observed in our results highlights the importance of considering its concentration in gastric cancer treatment. We found that increasing concentrations of curcumin led to a proportional decrease in cell viability, migration, and invasion in gastric cancer cells. This suggests that the efficacy of curcumin in inhibiting cancer cell progression is influenced by its dose, with higher concentrations resulting in more pronounced effects. The dose-dependent response may be attributed to curcumin's complex interactions with multiple cellular targets and signaling pathways. At lower concentrations, curcumin may predominantly target specific pathways, while higher concentrations may engage multiple pathways, leading to a more potent inhibitory effect.
Our study presents significant clinical relevance. Curcumin treatment led to a dose-dependent decrease in cell viability, migration, and invasion in gastric cancer cells, accompanied by downregulation of key PI3K signaling genes and proteins. This suggests curcumin's potential as a targeted therapeutic agent against gastric cancer, particularly when the PI3K pathway is dysregulated. The dose-dependent nature of curcumin's effects could allow for targeted inhibition of cancer cell progression while minimizing adverse effects on normal cells. Combining curcumin with existing treatments may enhance efficacy and overcome drug resistance, improving outcomes for gastric cancer patients. As a safe and readily available natural compound, curcumin holds promise for future clinical trials and may address the urgent need for effective and affordable treatment options for gastric cancer.
Despite the promising results, our study has several limitations. First, the research was conducted in vitro using gastric cancer cell lines, and thus, the findings may not fully reflect the complex tumor microenvironment and interactions with other cell types that occur in vivo. Therefore, further studies using animal models or patient-derived xenografts are necessary to validate the therapeutic potential of curcumin in gastric cancer treatment. Second, while we observed downregulation of key PI3K pathway genes and proteins, the precise molecular mechanisms underlying curcumin's actions on the PI3K pathway remain to be elucidated. Additional mechanistic studies are needed to unravel the specific molecular targets and signaling pathways involved. Furthermore, the effective concentration of curcumin required for optimal therapeutic outcomes may vary among different gastric cancer subtypes, and thus, the establishment of specific dosage regimens tailored to individual patient characteristics is warranted.
Despite the mentioned limitations, research on curcumin adds to the growing body of evidence supporting its anti-cancer properties in gastric cancer. More comprehensive understanding of its mechanisms of action would help optimize its therapeutic potential.
Conclusion
In conclusion, the findings from this study suggest that curcumin may be a promising therapeutic agent for gastric cancer treatment, partly due to its inhibitory effects on the PI3K signaling pathway. Future research should focus on exploring other molecular targets and signaling pathways impacted by curcumin, as well as the potential benefits of combining curcumin with other anti-cancer treatments. Additionally, in vivo studies and clinical trials could provide valuable information on the safety, efficacy, and optimal dosing of curcumin as a therapeutic agent for gastric cancer.
Supplemental Material
Supplemental Material - Curcumin's Dose-Dependent Attenuation of Gastric Cancer Cell Progression Via the PI3K Pathway Blockadesj
Supplemental Material for Curcumin's Dose-Dependent Attenuation of Gastric Cancer Cell Progression Via the PI3K Pathway Blockade by Gen Xi, Qingtao Dong, Bo Yang, Desheng Jiao, and Shahanavaj Khan in Dose-Response.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
