Abstract
Extensive studies on evaluation of effectiveness/toxicity of different oral doses of iodine have not been explored yet. An open-labeled phase I clinical studies were conducted using iodine complex based research compound called Renessans. Study groups were observed for development of any adverse/serious adverse events and alteration in laboratory values of vital organs, TSH and T4 hormones before and after the administration of the products. Out of 31 consented individuals, 24 healthy individuals participated in the study. Rate of occurrence of mild Adverse Events (AEs) in group A was 8.3% while in Group B it was 33.33% but these Adverse Events were self-resolving. After completion of study treatment blood serum iodine was reported to 3522.88 µg/l while mean urine iodine concentration (MUIC) was greater than 2000 µg/l. Hormonal and vital organ’s testing revealed that all parameters of TSH and T4, LFT, CBC, RFT remained unaltered except from ALT-SGPT (P-value = .006) and AST-SGOT (P-value = .02). From all of these findings, it can be inferred that the use of Renessans formulations did not pose any sort of risk to human body and can be considered safe through this pilot study.

Introduction
Iodine is vital for thyroid hormones synthesis. These hormones play important role in metabolism, growth and different body functions. 1 Deficiency of these hormones may cause goiter, hypothyroidism, abortion, stillbirth, congenital abnormalities, prenatal mortalities of fetus, and impaired mental health. 2 Whereas, its abundance can also lead to goiter or increase in thyroid volume due to failure of homeostatic function of body, an overt hypothyroidism, thyroid autoimmunity, iodine-induced hyperthyroidism, and Grave’s disease. 3
In human beings, it is present in quantity of 15–50 mg of which almost 80% resides in thyroid gland. The total daily requirement for iodine intake in adults is about 150 µg/day, 4 whereas upper limit for its use is 1100 µg/day according to US Food and Nutrition Board of Institute of Medicine. The upper limit is defined as the quantity of iodine which does not pose any risk or damage to human body. However, doses above upper threshold are also well tolerated in body. 2 The main source of iodine comes from diet rich in iodine like seaweed, seafood, milk, and iodine salts. 5
The ingested iodine is mainly absorbed in the intestine 1 where more than 90% of the absorbed iodine is excreted from the body through urine within 12–24 hours. 5 Usually, the iodine required for removal of iodine deficiency is taken in the form of sodium or potassium salts of iodine or iodates. When used within limits, the use of iodine in these forms of salts is considered safe and has no negative effect on human body. 6 However, when an excess amount of the iodine is consumed, different auto-regulated compensatory mechanisms are switched on inside human body to keep balance of the iodine for proper functioning of thyroid. The amount of iodine taken by thyroid gland not only depends upon iodine intake but also on the intra-thyroid iodine concentration. At a normal or low dose of iodine, the production of T3 and T4 corresponds to the intake of the iodine. However, at dose above the normal level, thyroid intake of iodine may decrease but, the process of organification may increase due to presence of abundant intra-thyroid iodine. 7 At very large doses, excess iodine may stop production of thyroid hormones and this phenomenon is called as “Wolff-Chaikoff Effect.” 8 In addition to this, Eftychia et al., 7 (2017) narrated that thyroid may adopt different protective mechanism in presence of excessive iodine in which “thyroid auto-regulation” is one of them. 7 In this process, thyroid gland regulates iodine metabolism independent of TSH and other hormonal or physiological factors. This help to maintain normal thyroid function without being affected by fluctuation of daily iodine intake.
Iodine and its complexes with different salts like potassium iodide, sodium iodide, and povidone-Iodide are well known for their antibacterial and antiviral effects.9-11 Micro Technologies of Iodine Medical Private Limited (MTI Medical Pvt. Ltd) organization has developed a research product named as Renessans having iodine complex comprising iodine (100 mg), potassium iodide (143 mg), and ascorbic acid (5 mg). Although, the Renessans product in varying strengths has tested and found effective previously 12 ; however, structured safety evaluation is yet to be performed. The aim of the current study was to analyze the safety profile of 2 dosage forms of Renessans in healthy individuals by observing adverse event and impairment/malfunctioning of human body organs. If the safety is established, these formulations could become a readily available rich source of micronutrients particularly for iodine deficient patients. Because iodine has antimicrobial properties, 4 these products may pave the path for oral pharmacotherapy for bacterial, viral, or other types of infections, as iodine acts generically against such infections. The significance of this discovery gains importance as iodine in these formulations is widely available and inexpensive, whereas the production of such medicaments is also cost-effective.
Research Methodology
The study was conducted in accordance with the International Council for Harmonization of Technical Requirements of Pharmaceuticals for Human Use (ICH) guidelines for Good Clinical Practice [GCP-E6 (R2)], local and ethical guidelines of respective institutes for the research in human beings.
The protocol and other essential documents were submitted and approved by Ethical Committee of University of Lahore (Letter ref. no. IREC-2021-12 H dated: 30Aug2021), Lahore General Hospital (Letter ref. no. 00-118-20, dated 11Sep2020), National Bioethical Committee (NBC) of Pakistan (Letter ref. no.4-87/COVID-34/NBC/20/297, dated: 10 Sep 2020) and Drug Regulatory Authority of Pakistan (DRAP) (Letter ref. no. F. No.03-47/2020-DD (PS), dated: 19Oct2020).
Clinical Setting
Research was conducted at Lahore General Hospital, Post Graduate Medical Institute Lahore, Punjab, Pakistan, which is located at Ferozepur Road, Ismail Nagar, Lahore, Punjab, 54000
Study Design
The study design was an open labeled phase I clinical study in which healthy volunteers coming to Lahore General Hospital were included in the study. There were 24 healthy volunteers enrolled in the study comprising of 12 males and 12 females, who were divided randomly into two groups, that is, group A having 12 participants (6 males and 6 females) and group B having 12 participants (6 males and 6 females). The study participants were enrolled in almost 2 months of time and this safety study was completed in 3 months including their all follow-up visits. The participants were included in the study with the following inclusion and exclusion criteria as shown in Figure 1: Flow Chart of Inclusion/Exclusion Criteria and study procedures.
Inclusion Criteria
The volunteers aged 18 years or above years, willing to give consent for participation in the study at their own will and follow study procedures throughout experiment, and were enrolled in the study. Participants showing absence of any health issues during clinical examination and through medical history were selected for inclusion in the study. Females with childbearing potential were included in the study only on the basis of negative pregnancy test and they agreed to use contraceptive methods during entire duration of the study.
Exclusion Criteria
All those participants who had co-morbidities such as any pre-existing cardiac or pulmonary disease, diabetes, arrhythmias, pre-existing hepatic or renal disease, or thyroid dysfunctions were not included in the study. Also, those female who were either pregnant or planned to become pregnant in near future were not included in the study.
Informed Written Consent
The participants provided their free informed consent to participate in the studies as per inclusion and exclusion criteria. The document has complete information about the study, participant’s role and responsibility, risk associated with participation, confidentiality, and participant’s right to withdraw from the study. It was clearly stated that participants had right to withdraw their consent at any time during the study.
Data Collection
A comprehensive case report (CRF) form was developed for the collection of essential data in an organized manner. This CRF was approved by the concerned ethical committees; National Bioethical Committee (NBC) and the DRAP. Patient progress notes, lab reports, and case reports forms served as primary source data for the study. On CRF, questions related to demographics of the patients, vital sign measurements, medial history, physical examination, electrocardiogram, and other information related to adverse event and serious adverse events were collected on different timelines as per studies plan.
Procedure
Timelines of Biochemical Test in Phase I Clinical Study.

AbbreviationCBC: Complete Blood Count, LFTs: Liver Function Test, RFTs: Renal Function Test, ECG: Electrocardiogram, TSH: Thyroid Stimulating Hormone, MUIC: Mean Urinary Iodine Concentration.
Safety Assessment
Safety assessment was done by evaluating the development of adverse event or serious adverse event and observing laboratory tests conducted on the enrolled subjects.
Development of Adverse Event or Serious Adverse Event
One of the primary focus of safety assessment was to evaluate the occurrence of adverse event or serious adverse after the administration of the product. It was done by examining the participating subjects on every visit and reviewing their diary cards provided to them to note any health illness on it when they are at home.
Laboratory Test of Specific Interest
Laboratory Tests to evaluate vital function of body
Statistical Analysis
R software (version 4.1.2/1) was used for statistical analysis. Data being represented here is given in terms of mean ± SD (Standard Deviation). Laboratory finding before and after the administration of the drug was done using pair t-test through R by considering 95% confidence interval. Whereas significance of serum TSH and free T4 was evaluated through analysis of variance (ANOVA) where these values were compared at the start of medication, during the medication and after the medication. Adverse event and serious adverse events were estimated in percentage of occurrence in enrolled population using Microsoft excel 2016.
Results
Demographics and Clinical presentation
Demographic Details of participants of Group A and Group B

Serum Iodine
Serum Iodine Test Results.

Urinary Iodine Test
MUIC Test Results in Healthy Volunteers.
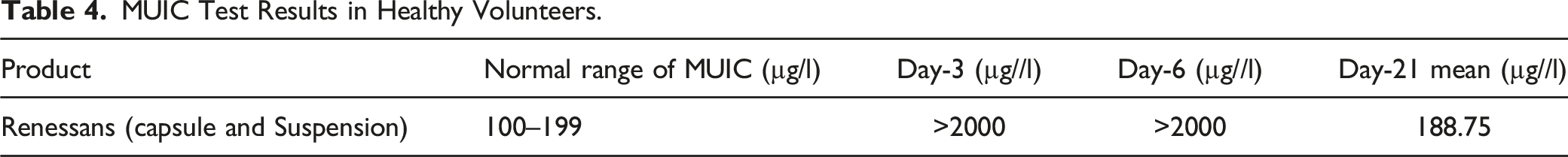
MUIC Test Results on Day-21.

Occurrence of Adverse Event and Serious Adverse Events
Summary of Adverse Events reported in Phase-I study.

Assessment of safety of organs through laboratory tests.
Hematological assessment through Complete Blood Count.
Complete Blood Count Test Results.

RBC=Red Blood Cells, HCT= Hematocrit, MCV= Mean Corpuscular Volume, MCH= Mean Corpuscular Hemoglobin, MCHC= Mean Corpuscular Hemoglobin Concentration, WBC= White Blood Cells.
Liver Function Test of Heathy Volunteers
Liver Function Test Results.

ALT-SGPT= Serum Glutamic-pyruvic transaminase, AS-SGOT= Serum Glutamic-oxaloacetic transaminase, Gamma GT= Gamma-Glutamyl Transpeptidase.
Renal Function Test of healthy Volunteers
Renal Function Tests Results.

BUN= Blood Urea Nitrogen.
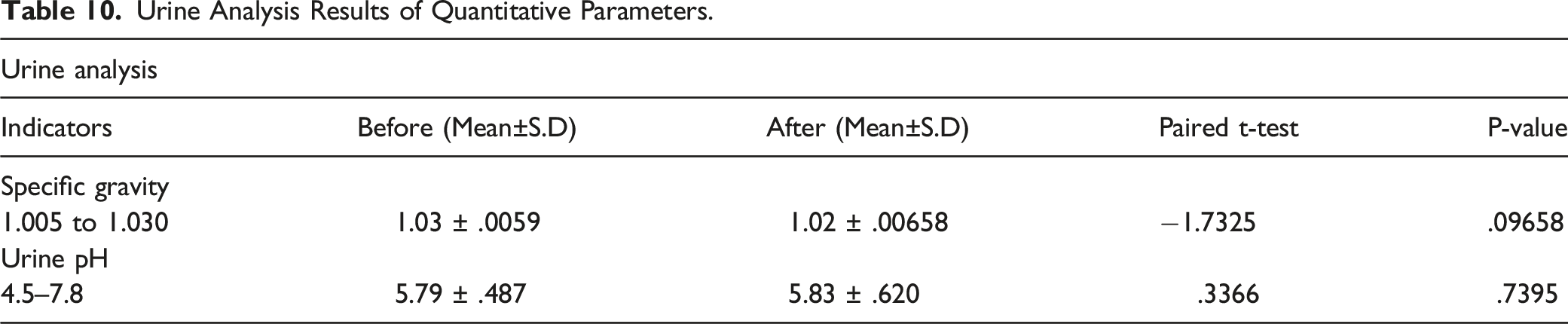
Urine Analysis
Urine Analysis Results of Quantitative Parameters.
Urine Analysis Results Qualitative Parameters.
Because iodine is primarily excreted through urine, urine analysis was performed before and after the use of study drugs in both groups of the study. Quantifiable parameters such as specific gravity and urine pH were assessed and reported in Table 10, whereas qualitative parameters such as color, turbidity, the presence of glucose, ketones, proteins, hemoglobin, urobilinogen, bilirubin, nitrates, leukocytes, pus cells, red blood cells, epithelial cells, amorphous cells, organisms, yeasts, and crystals were also measured to see if there was any change that could indicate the effect of Renessans on urine composition. The qualitative parameters obtained before and after the use of study drugs are reported in Table 11 with their reference characteristics and ranges. However, we did not find any difference in either quantifiable or qualitative elements of urine analysis after study drug administration.
Effect of Iodine Concentration on Serum TSH and T4 Test
Serum TSH Test Results.

Serum T4 Test Results.

Discussion
In human beings, iodine and its other salts remain in quantity of .02 mg/kg of the body weight or roughly it can be said 15–20 mg 6 (7). Except from the radioactive form of iodine which is used for diagnosis purposes, different studies have reported that iodine and its salts are safe for human use even in high doses. However, there is not much literature available on its safety evaluation.3,4 Burgi et al., (2001) conducted a study in mice to check lethal dose of iodine and from that he referred that adverse drug reaction due to iodine can occur in human beings at dose of 100 mg/kg of iodate. 5 Mao et al., (2011) wrote that mean lethal dose in human beings maybe 2–4 g in a day; however, toxicity due to iodine in human is very infrequently reported till date. 18 The investigational product Renessans (equivalent to 200 mg of iodine) was administered in healthy volunteers three times a day for five days in two dosage forms i.e. suspension and capsule separately. So a total 600 mg/day was given to the individuals in this study. The average serum concentration in 24 healthy volunteers was evaluated to be 3522.88 µg/l after Renessans use in the current study, whereas, normal reference range in literature was found to be 30.48–80.01 µg/l in human beings. 18 In this study, high serum concentration of iodine drug in human serum was well tolerated by the volunteers as there was no significant change observed at metabolic or physical level in all the individuals. High dose of iodine has strong influence on hormones like TSH and T4; 3 however, in the current studies, the serum TSH and T4 remained within reference normal ranges of TSH (age 21–54 years) .4–4.2 µlU/mL and T4 (age 20 and above years) .8–2.7 ng/dL before and after the use of Renessans (Table 12 and 13). There had been no significant change (P = .372) observed in TSH values at day-21 (mean value 1.46 µlU/mL) and 6th week (mean value 1.53 µlU/mL), compared to day-0 values of TSH (mean value 1.53 µlU/mL) when measured statistically. Similarly, T4 values at day-21 (.91 ng/dL) and 6th week (.93 ng/dL) were not statistically significantly different (P = .455) from the Day-0 (.93 ng/dL) when Renessans products were not taken by any group. This non-significant change in TSH and T4 functioning may be due to “Wolff-Chaikoff Effect” 4 where the synthesis of thyroid hormones are inhibited due to presence/exposure to high dose of iodine. Also, potassium iodide might have played its role in regulating iodine organification by influencing Na+-I symporters where K+ acts to regulate Na+ import and export through Na+-I-K+ATPase for iodine transport into the cells. 6 In addition to that thyroid auto-regulation might have been an another reason as Eftychia et al., (2017) stated that thyroid gland may adopt this procedure as one of the other protective mechanisms when thyroid senses high iodine in the body. 7 This auto-regulation of thyroid gland is independent of TSH functioning and thus it keeps balance of hormone production irrespective of fluctuation in iodine and TSH levels.
Urine iodine concentration test is one of most cost effective, well-accepted, and easy to perform test which is used as a biomarker to check the iodine level in body.3,19 In a healthy person, almost all of orally taken iodine is absorbed from intestine and more than 90% of it is excreted in urine within 12–24 hours.3,4,20 For accurate measurement of iodine excretion, 24-hour measurement is required which is not a practical approach so instead a spot urine test from a given population is performed and expressed as median urinary iodine concentration.4,19,20 According to WHO-Iodine Nutrition Epidemiologic Criteria, population mean urinary iodine concentration is considered adequate in the range of 100–199 µg/l for children and adults. Whereas, 200–299 µg/l and 300 µg/l or above is considered as above the requirement and excessive iodine intake, respectively. 19 In the current study, patients were given approximately 600 mg/day of iodine for five days and their mean serum iodine concentration was 3522.88 µg/l on sixth day whereas, the median urinary iodine concentration (MUIC) on day-3 and day-5 was observed to be greater than 2000 µg/l of iodine for both groups of the study. However, when a washout period of almost 14 days was passed, the mean concentration was found to be 188.75 µg/l on 21st day. Our findings are very much aligned with the available literature that high iodine urinary concentration was noticed within 12–24 hours of iodine intake and after a washout period of 21 days, individuals depicted normal iodine excretion. This means, that almost all of the excessive administered iodine had been excreted out of body and a normal range of iodine was attained in urine of healthy individuals on 21st day and there had been no side effects noticed in the healthy individuals.
The data obtained from our Phase-I study suggested that Iodine Complex based Renessans product had a fairly safe profile. Healthy volunteers after using Renessans formulations (suspension and capsule) three times a day for five days did not develop any Serious Adverse Event (SAE). Whereas, only 5 out of 24 subjects experienced Adverse Event (AE) which were self-resolving and related to change in taste or burning sensation in mouth, throat or stomach category. There is scarcity of literature where iodine safety is evaluated in human beings when it is used orally, however, Frank et al., (2020) also narrated safe nature of iodine–povidone complex when used in nasal and oral cavities for COVID-19 patients. 21 It is noteworthy that in our study, the occurrence of Adverse Events remained considerable high in Suspension Group (Group B), that is, 33.33% in comparison of Capsule group (Group A), that is, 8.33%. This may be because of the fact that in suspension dosage form, the iodine was directly in contact with mucosal membrane when it was administered in the patient whereas in capsule dosage form, the iodine would be released after the capsule shell had undergone disintegration in gut. Also, the fact that the drug in oral liquid dosage forms (syrup or suspension) is readily available and lesser taste masked due to which characteristic taste of iodine (metallic to acidic)22,23 was observed in Group B.
When iodine is ingested, human cells are exposed to the iodine and its salts which are developed in body fluids. These iodine salts attain oxidizing power due to which they may penetrate cell membrane and destroy cells of host resulting in human intravascular hemolysis. Mao et al., (2011) narrated incidence of 30-year healthy male in which hemolysis occurred after his suicidal attempt with the ingestion of 200 ml iodine tincture where the blood serum iodine concentration was found to be 1,155,901 µg/l. 18 Whereas, in the present study, mean serum iodine concentration was observed 3522.88 µg/l and mean urinary iodine concentration was above 2000 µg/l but still no change in the blood chemistry observed for RBC, HCT, MCV, MCH, platelets, WBC, Neutrophils, Lymphocytes and Monocytes. The “p” value greater than .05 in Table 7 for complete blood count showed insignificant change in the above-mentioned blood components before and after the administration of the Renessans product. This is a clear indication that blood cells had well tolerated high concentration of iodine in blood serum which is aligned with the literature.3,18,24
Liver Function Test is an effective screening tool providing early information about liver health but it does not provide gold-standard diagnosis of liver diseases. 15 Effect of Renessans on liver function was assessed through this test in which bilirubin, conjugated bilirubin, unconjugated bilirubin, ALT-SGPT, AST-SGOT, Gamma GT, protein, albumin, and Ggobulin were tested before and after the administration of the product. Except for ALT-SGPT and AST-SGOT, all of the parameters observed in the test remained within their referenced normal ranges (Table 8). The “p” value of these biomarkers were also greater than .05 which indicated that there was no significant change occurred in these after the use of study medication. In literature, there is no reference data found which can tell effect of iodine or its complexes at the experimental dose in human beings; however, Sani et al., (2021) narrated that they also did not observed significant change in liver biochemistry when they used 2000 mg/kg povidone iodine in cockerels. 25 There is also a similarity between Sani et al., (2021) and the current study in that both studies observed an increase in the values of the studied parameters of this test when iodine was used, despite the fact that they remained within their normal reference ranges. On contrary to all other parameters, serum ALT-SGPT and AST-SGOT had significant changes where the “p” value of ALT-SGPT was .006 and ALT-SGOT was .024. It happened because there were five volunteers who had ALT-SGPT and AST-SGOT values out of range at the time of study induction; however, these values did not impose any health issue and the volunteers were evaluated and considered healthy for study participation as per investigational team opinion. Interestingly, when their test was repeated on day-21, two subject’s ALT-SGPT and AST-SGOT values came in normal ranges. So the significant result of these two indicators were considered as an independent event because the subjects had out of range values at the start of the study and it was not related with the use of investigational product as evaluated by the investigators. Mao et al., (2011) mentioned that after poisoning with iodine tincture 200 ml (serum iodine concentration 1,155,901 µg/l), 18 the subject showed higher bilirubin, conjugated bilirubin, ALT and AST values, but this might be due to injury associated with alcohol as there was no relationship established by author in that case study. Also, Burgi et al., (2001) mentioned that very high dose of iodine may result in hepatic injury in animal 6 but there had not been any details for this finding. Whereas, the current study revealed that a dose up to serum concentration of 3522.88 u µg/l did not produce effect on liver functioning. This is in alignment with the results of a study in which “p” value remained insignificant for ALT, AST, total bilirubin, and direct bilirubin when 150 µg/day iodine supplement was given orally for 90 days. The metabolism of iodine takes place in thyroid gland in human beings and the fluctuation of iodine in blood modulate the release and functioning of thyroid hormone (T3 and T4) in connection with TSH hormone whereas the metabolism of T3 and T4 take place in liver. 26 In the current study, an insignificant change was evaluated in levels of TSH (P = .372) and T4 (P = .455), so there was diminutive chances that liver functions are effected by the Renessans product.
On an average, a human body excretes 480 µg of iodine in urine and almost 20 µg in stools. 26 Whereas, 90% of ingested iodine is excreted through urine within 12–24 hours.3,4,19 In the instant study, it has also been observed that median urinary iodine concentration became normal when it was checked on day 21 (mean 188.75 µg/l) whereas it was greater than 2000 µg/l on day-06 which meant that iodine was largely excreted via urine and achieved its normal state in the body without disturbing any kidney function measured through renal function test. In this test serum urea, serum BUN and serum creatinine were evaluated and found within normal reference ranges before and after the use of Renessans (Table 9). The “p” values of serum urea (.3748), serum BUN (.5402) and serum creatinine (.6637) also remained greater than .05 which meant that there had been no change observed in these kidney bio-indicators. This is in contrary to a case reported by Kim et al., (2017) in which a 47-year-old person had poisoning of 10% povidone–iodine and his serum BUN reached and creatinine reached up to level of 124 mg/dL and 6.3 mg/dL, respectively. 27 However, our results are aligned with Daei et al., (2018) who used a different strength of iodine (150 µg/day) for a period of 90 days, but he also reported that there were no changes occurred in serum urea, creatinine, and other biochemical elements of his study. 28 Urine analysis was also performed to further evaluate the effect of iodine on urine composition. This test had quantitative component in which urine specific gravity and pH were evaluated (Table 10) whereas qualitative component compared the urine color, turbidity, glucose in urine, ketones, proteins, hemoglobin, bilirubin, nitrates, leukocytes, pus cells, red blood cells, epithelial cells, amorphous cells, organisms, yeast, and crystal with their reference ranges/characteristics (Table 11). Like renal function test, the mean value of urine specific gravity and pH remained within their normal reference ranges (Table-10) and their “p” values of .096 and .739 respectively, also remained insignificant which assured that there had been no deleterious effect of iodine on these elements of the urine analysis. There is scarcity of literature to compare effect of iodine on both quantitative and qualitative parameters of urine analysis; however, we did not observe any significant change in these parameters when Renessans capsule and suspensions were used in healthy volunteers.
Conclusion
The Renessans product is evaluated to be safe in healthy human beings. The product having iodine strength of almost 200 mg after being administered three times a day for five days, achieved fairly high concentration of serum iodine in body, that is, 3522.88 µg/l and urinary iodine excretion greater than 2000 µg/l. At this concentration, no significant change in the body thyroid functioning was observed through serum TSH and T4 biomarkers. Also, the vital organ functionality tests like liver function test, renal function test, blood chemistry, and urine analysis remained stable when checked before and after the administration of Renessans product in healthy volunteers. With the exception of self-resolving adverse events which occurred in only five participants, other physical and clinical conditions of all participants remained stable. All these findings led to a conclusion that Renessans product would be safe to be used in healthy humans.
Footnotes
Acknowledgments
We want to thank all clinical staff including technical and paramedic staff of Lahore General Hospital, Post Graduate Medical Institute, Lahore, Pakistan who participated in the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded by MTI Medical (Pvt.) Ltd, Lahore, Pakistan.
Ethical Approvals
The clinical study was approved by: Institutional Research Ethics Committee of Faculty of Pharmacy, University of Lahore: Letter No. IREC-2021-12 H dated: 30Aug2021. Ethical Review Committee of Lahore General Hospital Lahore: Letter No. 00-118-20, dated 11Sep2020. National Bioethics Committee (NBC) of Pakistan: Letter No. 4087/COVID-34/NBC/20/297 dated: 10Sep2020. Drug Regulatory Authority of Pakistan (DRAP): Letter No. F. No. 03-47/2020-DD (PS), dated: 19Oct2020
