Abstract
A dose–response study was performed in adult rats to select an optimal stable potassium iodide (KI) dose which could be implemented in repeated prophylaxis, in case of prolonged exposure to radioactive iodine. Increasing doses of KI were given orally to rats 1 hour before internal exposure simulated by I-125 injection. I-125 incorporation in the thyroid was measured by γ-spectrometry, and KI protection effect was modeled by pharmacological functions. The measurement method by inductively coupled plasma mass spectrometry previously developed for the quantification of stable iodine in urine was adapted to correlate KI effect with its distribution in the thyroid. More than 75% blockade of iodine I-125 incorporation in the thyroid was achieved for KI single doses above 0.5 to 0.7 mg/kg. Stable iodine content in the thyroid 24 hours after KI administration displayed a biphasic response, with a maximum level for a dose around 1 mg/kg. Besides, the urinary excretion of stable iodine is described by a sigmoid function. The change in the rate of iodine excretion for doses above 1 mg/kg KI suggests a body overload in iodine and corroborates a possible saturation of the thyroid. The results show that 1 mg/kg KI could be regarded as an optimal dose for thyroid protection.
Introduction
The consequence of a major nuclear or radiological accident can affect all levels of society including public health. Exposure of the population to radioactive iodine isotopes may occur as a result of uncontrolled nuclear accident, which might cause long-term consequences that can lead to an increased risk of developing thyroid cancers, particularly in infants and young children. 1 If inhaled or ingested, radioiodines are rapidly absorbed in blood from which they concentrate in the thyroid in the same way as stable isotope. Public protection actions must be taken rapidly to limit exposure to radioactive materials. Depending on the situation, such actions may imply sheltering in place and heeding orders, ingestion of stable iodine, evacuation of the public, and restricting activities and consumption of food and liquids. 2,3 Stable iodine intake aims to saturate the iodide transport mechanism of the thyroid by inhibiting the intrathyroid organification of radioactive iodide (also known as the acute Wolff-Chaikoff effect), 4 by diluting and promoting the excretion of radioactive iodine and thus reducing the amount of committed dose to the thyroid gland, its surrounding tissue, and the body. 5 -7 The ingestion of stable iodine, generally as potassium iodide (KI) tablets, should ideally occur 2 hours before exposure to the radioactive plume or at the latest within 2 to 8 hours after exposure. It is assumed that a single dose of KI provides adequate thyroid protection for 24 to 36 hours and that the protection effect decreases with time after the administration. 8 The benefit of iodine prophylaxis implemented in Poland after the Chernobyl accident has been highlighted in epidemiological studies, which have reported a lower thyroid cancer incidence in children of the age-group 1 to 18 years. 9 Many countries have adopted recommendations provided by international organizations regarding iodine prophylaxis. However, the recent Fukushima disaster raised questions about the conditions for the implementation of stable iodine prophylaxis in order to protect populations exposed to repeated releases of radioactive iodine. Although the official procedures in France envisage the possibility of a second intake of KI, particularly if rapid evacuation of populations is impossible, there are currently no indications regarding the conditions for the implementation of repeated stable iodine intakes: optimal dosage, dose regimen, or maximal duration of the treatment. 2
The currently recommended single dose of 100 mg of iodine (130 mg of KI), which corresponds to approximately 1.8 mg/kg of KI in adults weighing 70 kg, is considered sufficient to protect the thyroid. It is of the same order of magnitude as the dose range used for several years in the treatment of hyperthyroidism: 150 to 300 mg iodide daily administered up to 10 days. 10 It should be noted that this dose of 130 mg KI may be considered excessive, especially in adolescents and children, and it is thus recommended to use fractions of this dose in specific population groups (1 in adults and adolescents in addition to pregnant and lactating women, if necessary, 1 (65 mg) of 2 in children, 1 (32.5 mg) of 4 in infants, and 1 (16.25 mg) of 8 in newborns). 8,11,12 Besides, slight and nonspecific extra thyroidal adverse effects, such as headache, abdominal pain, sore throat, vomiting, diarrhea, eczema, and dyspnea, have been observed after a single administration of KI at doses ranging from 15 to 70 mg. 13,14 But it has been suggested that lower doses of KI ranging from 1.5 to 4.5 mg administered daily for 2 weeks to healthy humans could induce thyroidal effects such as a decrease in thyroxine release and an increase in thyroid-stimulating hormone release, 15 which could be potentially deleterious for vulnerable individuals, particularly for children.
To our knowledge, very few dose–response studies have been published after a single pharmacologic dose of stable iodide in healthy volunteers 7,16,17 or in rodents. 18,19 The results of the clinical studies showed that a single dose as low as 10 mg of stable iodide (corresponding to approximately 0.2 mg/kg KI in adults) administered 24 hours before exposure to I-131 is sufficient to protect the thyroid from radioactive iodine. In addition, the amount of stable iodine in thyroid insuring the blockade of the organ has been estimated to be lower than 1 mg. 17 Nonetheless, this value was extrapolated after computer simulations since the number of stable iodide doses which were actually tested in humans was limited (37, 100, 125, and 250 mg iodide). Alternatively, studies in animals usually offer the possibility to test a larger number and wider scale of doses. However, no pharmacological analysis of data and no mathematical model describing the effect of stable iodide on thyroid have been published so far.
Consequently, the purpose of the present work was to conduct a dose–response study in rats in order to model the efficacy of KI and to determine the optimal minimum dose of KI which could be implemented for a protracted prophylaxis of the population. Besides, it was assumed that this optimal KI dose should be lower than the current prescribed dose of 1.8 mg/kg KI and could be also selected on the basis of the distribution or excretion of stable iodine. Indeed, we hypothesized that the concentration of stable iodine in the thyroid could reflect the iodine saturation of the organ, and conversely, the excretion of excess iodine in the urines could be a result of the overload of the body, including the saturation of the thyroid, after a single administration of KI. Such new results on the efficacy of KI prophylaxis and on the distribution or the mode of action of stable iodine should constitute useful data for the international guidelines for iodine thyroid blocking, which are currently under revision. 20
Materials and Methods
Animals
Male Wistar rats (strain code: 003, 8-9 weeks age and weighing 280-360 g) were used in this study. The animals were purchased from Charles River Laboratories (Saint Germain sur l’Arbresle, France) and handled in compliance with the French Legislation (articles R214-87 to R214-137 of French rural code) and the European Directives (2010/63/EU) regarding the care and use of laboratory animals. Experimental protocols were reviewed and approved by the Institute for Radiation Protection and Nuclear Safety (IRSN) ethics committee. Six rats were used per treatment group. They had free access to water and food before and throughout the experimental period. The rats are fed with AO4C pellets (Scientific Animal Food and Engineering, Augy, France) daily during acclimation periods and experiments. The controlled iodine content of the diet meets rodent food requirements (0.3 mg/kg pellets). After exposure, the animals were anesthetized by isoflurane inhalation prior to being euthanized by intracardiac puncture.
Chemicals
Stable KI solutions at different concentrations in sterile water pH 7.4 were prepared and provided by the French Central Armed Forces Pharmacy (Orléans, France). Radioactive KI I-125 solution at a concentration of 3700 MBq/mL and with a specific activity of 643.8 MBq/pg was purchased from Biotrend (Köln, Germany).
Experiment Protocols
For the efficacy of thyroid blockade study, stable KI was administered at different doses orally 1 hour before the exposure of the rats to iodine I-125 intravenously to 7 groups of 6 rats. Potassium iodide doses were 0, 0.001, 0.01, 0.1, 1, 1.8, and 5 mg/kg. Oral administration was performed by esophageal gavage on nonanesthetized rats using a 1-mL syringe coupled with a disposable plastic cannula (Instech Laboratories, Plymouth Meeting, Pennsylvania). For the oral administration of KI, a total volume of 1 mL of each stable KI solution or control solution (sterile water) is administered to each animal. Intravenous administration of radioactive iodine I-125 was performed on the anesthetized rat (3% isoflurane, 1.5 L/min) using a 1-mL syringe coupled to a 25-G needle. The injection was carried out in the jugular vein after a 1-cm incision of the skin. The activity and volume of I-125 solution injected to each animal were adjusted according to the body weight recorded on the day of administration (1.11 MBq/kg in a volume of 1 mL/kg). The animals were euthanized 1 hour after exposure to I-125.
As for the evaluation of the distribution and excretion of stable iodine after KI administration, 7 other groups of 6 rats were administered exclusively at different doses of KI by oral gavage. The KI doses were 0, 0.03, 0.08, 0.3, 1, 3, and 8 mg/kg. The animals were then individually housed in plastic metabolism cage for the collection of the cumulated urines and euthanized 24 hours after the administration of KI.
Iodine Analysis in Biological Samples
After euthanasia of the animals, the thyroid is dissected, rinsed, dried on gauze, and weighed. The urines are centrifuged using an Eppendorf centrifuge 5810R (4000 rpm, for 20 minutes at 4°C). The samples of urines and thyroids are stored at −20°C prior to analysis. The total radioactivity of iodine-125 contained in the thyroids is determined by γ counting (γ multidetector RIASTAR A5410, Packard, Vernon Hills, Illinois). After slow thawing of samples at laboratory temperature (+21°C), the urines are diluted 1/10 000th with 2% ammonia solution (solution made from the Suprapur 25% ammonia solution, Merck, Molsheim, France) and then stabilized with a sodium thiosulfate solution at 184 mg/L in 2% ammonia. The total iodine content is determined by direct measurement of 3 aliquots of each sample by inductively coupled plasma mass spectrometry (ICP-MS, X Series II; Thermo Electron, Courtaboeuf, France) using the standard addition method. The internal calibration range consists of a standard solution of stable iodide at 1000 mg/L (Iodides AVS Titrinorm IC Standard, Prolabo, VWR, Fontenay-sous-Bois, France). Tellurium Te-125, used as the second internal standard, is added to the samples at a concentration of 1.25 mg/L from a standard 1000 mg/L solution (Tellurium ICP Standard Certipur; Merck). The internal calibration range is constituted in each sample by the following concentrations: 0, 2, 4, 6, 8, 10, and 12 μg/L of stable iodide. 21 The same method for determination of stable iodine by ICP-MS is applied to the thyroids after preliminary mineralization of the samples in a microwave oven (Ethos TC, Milestone, Italy) in 50 mL of ammonia 4 M for 30 minutes at 200°C. A volume of 1 mL of the mineralized solution is then diluted to 1/125th in 2% ammonia for iodine analysis by ICP-MS. The results of measurements of radioactive iodine I-125 expressed in number of counts per minute and per gram of tissue (cpm/g) are converted into kilo becquerels per gram of tissue (kBq/g) and then as a percentage of injected activity. The results of stable iodine measurements by ICP-MS in urine and thyroid samples expressed in counts per second are converted to μg/L (or ng/mL) of sample after correlation with calibration curves.
Statistical Analyses
All data are expressed as the mean (standard deviation, SD) of a minimum of 6 independent experiments. The comparisons of the results were performed using Student t test, and P values of less than .05 were considered significant. The theoretical pharmacological models are obtained by linear regression from the data using Prism software version 5.01 (GraphPad software, San Diego, California). The selection of the most accurate mathematical models is based on the comparison of the calculated statistical parameters of each model such as the coefficient of determination (R 2), the Akaike information criterion (AIC), or the Bayesian criterion of Schwarz (SBC): AIC = 2k − log(L) and SBC = −2log(L) + klog(n) (k is the number of estimated parameters of the model, L is the maximum value of the model probability function, and n is the number of observations). The most accurate mathematical model is the one with the highest coefficient of determination or the lowest AIC and SBC values.
Results
Efficacy of Thyroid Protection by Stable KI
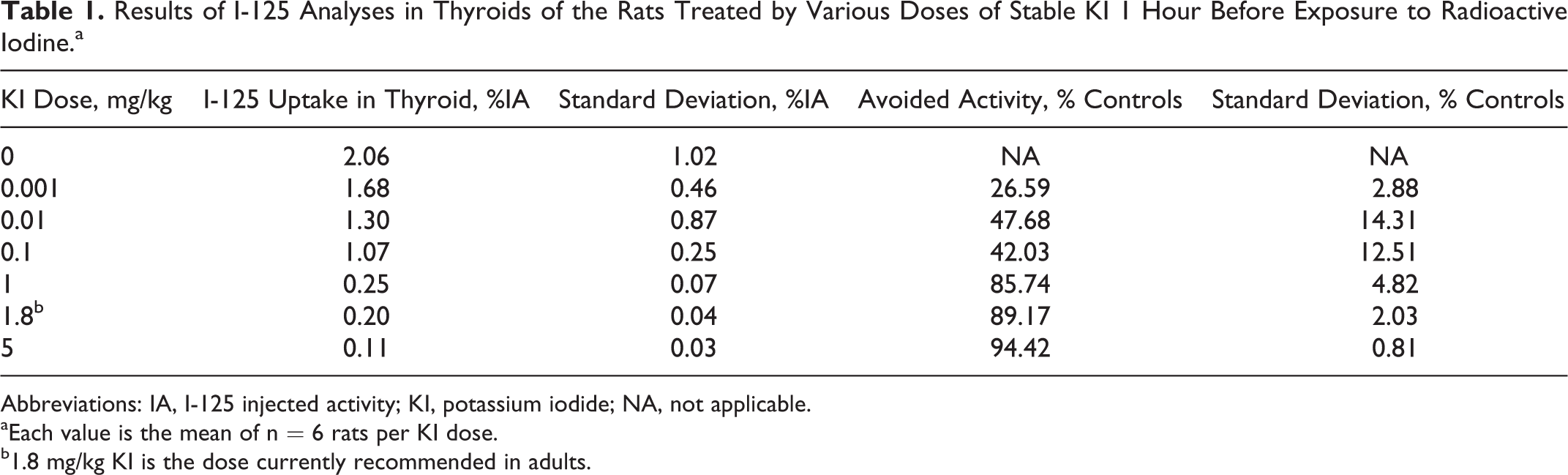
The first part of this work was to reassess the efficacy of stable KI and to determine the lowest dose which provides a satisfactory prevention of the incorporation of a radioactive isotope of iodine (I-125) into the thyroid of rats. The results of the iodine-125 count in the thyroid of rats pretreated with various doses of stable KI and then contaminated 1 hour later with iodine-125 show a significant reduction in the uptake of radioactive iodine (Equation 1) or an increase in avoided activity in the thyroid (Equation 2) as a function of the stable KI doses administered (Table 1).
Results of I-125 Analyses in Thyroids of the Rats Treated by Various Doses of Stable KI 1 Hour Before Exposure to Radioactive Iodine.a
Abbreviations: IA, I-125 injected activity; KI, potassium iodide; NA, not applicable.
aEach value is the mean of n = 6 rats per KI dose.
b1.8 mg/kg KI is the dose currently recommended in adults.
The uptake of iodine I-125 by the thyroid 1 hour after exposure and 2 hours after KI prophylaxis decreases from about 2% in control rats to 0.1% in rats treated with 5 mg/kg KI (Table 1). This decreased uptake as a function of stable KI dose can be modeled by an antagonist or inhibition-type pharmacological function (I) with a very good correlation (R 2 = .9939; Figure 1). The results of modeling by Prism software show that the logarithm of the inhibition function (I) can be related to the logarithm of the KI dose D according to Equation 3.
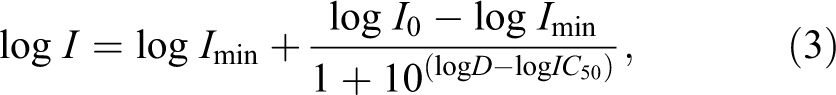
where I is inhibition of I-125 uptake in the thyroid by stable KI (%I-125 injected activity), D is KI dose (mg/kg), log IC50 = −0.1557, IC50 = 0.6987 mg/kg (half-effect dose), log I 0 = 0.1822, I 0 = 1.1512% (initial uptake), log I min = −1.1, and I min = 0.0794% (minimal uptake).

Injected activity (IA), the observed values are the mean of 6 rats ± standard deviation (SD) and represented by the circles, the inhibition function (I) is represented by the curve with an initial effect I 0 and minimal effect I min, graph (A) represents potassium iodide (KI) doses in linear scale, and graph (B) represents KI doses in log scale.
Since experimental data from the literature are variable depending on the experimental conditions and the animal species studied, there is no reference value to describe the effectiveness of current KI treatment in humans. We chose as an arbitrary value of minimum efficacy, the value of 75% based on the indication that the percentage of radiation dose avoided in the thyroid is usually comprised between 50% and 75% after the ingestion of 100 mg of stable iodide (130 mg KI in adults). 12 Thus, we looked for the stable KI dose value corresponding to 75% of the observed effect. The determination of the D75% dose corresponding to a 75% decrease in the initial uptake (I 0) therefore consists in calculating the dose leading to an uptake of 25% of the initial uptake I 0, that is, uptake I = 0.25 × I 0 = 0.29%. The resolution of Equation 3 gives log D75% = −0.2087, so the minimum KI dose necessary to inhibit 75% of the uptake of I-125 in the thyroid according to this pharmacological model is D75% = 0.619 mg/kg KI.
The second possible interpretation of the results of this dose–response study consists in expressing the avoided I-125 activity in the treated animals relative to the control animals as a function of the KI dose. Remarkably, the iodine I-125 avoided activity in the thyroid as a function of the KI dose was around 26% of the control rats with a dose as low as 0.001 mg/kg and increases to 94% with the highest dose tested of 5 mg/kg KI (Table 1). The pharmacological function that best fits the observed effect (E) is of agonist or stimulation-type with a correct correlation between the observed and theoretical values (R 2 = .9471; Figure 2).
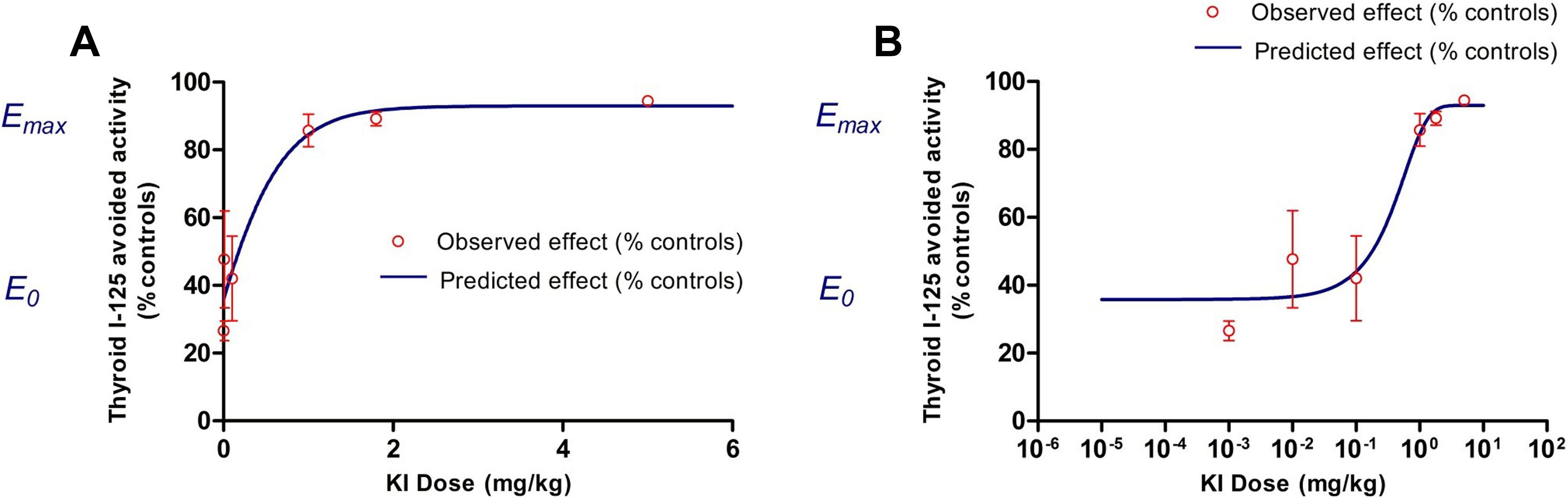
The observed values are the mean of 6 rats ± standard deviation (SD) and represented by the circles, the stimulation function (E) is represented by the curve with an initial effect E 0 and a maximal effect E max, graph (A) represents potassium iodide (KI) doses in linear scale, and graph (B) represents KI doses in log scale.
The results of modeling by Prism software show that the stimulation function (E) can be related to KI dose D according to the following equation (Equation 4):
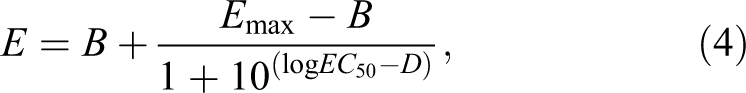
where E is stimulation of I-125 avoided uptake in the thyroid by stable KI (% controls), D is KI dose (mg/kg), log EC50 = −0.2565, EC50 = 0.5540 mg/kg (half-effect dose), B = −67.33%, and E max = 92.96% (maximal stimulation).
The determination of the D75% dose corresponding to a 75% increase in iodine I-125 avoided activity in the thyroid consists in calculating the dose inducing 75% of the maximum effect E max, that is, E = 0.75 × E max = 69.92%. The resolution of Equation 4 gives D75% = 0.518 mg/kg KI.
These 2 methods of interpreting the pharmacological effect of KI on the uptake of radioactive iodine in the thyroid allow us to approximate the minimum dose leading to 75% protection efficiency, in terms of avoided activity in the thyroid, which would therefore be between 0.5 and 0.7 mg/kg.
Distribution and Elimination of Stable Iodine
This part of the study aims to establish a possible correlation between the quantities of stable iodine administered and the distribution of iodine in the thyroid or its elimination in the urine. The calibration methods for the measurement of stable iodine in urinary matrix by ICP-MS have been recently validated by our group in order to quantify the total excretion of iodine in the urines. 21 This method was adapted in the present study for the measurement of stable iodine content in the thyroid of the rats.
The total stable iodine content in the thyroid (expressed in microgram iodine per thyroid or per gram of organ) of the animals only treated with increasing doses of KI from 0.03 to 8 mg/kg seems to display a biphasic response as a function of KI dose (Table 2 and Figure 3). Indeed, in our experiment conditions, the iodine content in thyroids sampled 24 hours after administration gradually rises for small increasing doses of KI to reach a maximum at a dose around 1 mg/kg with an iodine level (1163 ± 184 µg/g) statistically different from the thyroid content in control rats (830 ± 260 µg/g; P < .05). Then for higher KI doses, the total iodine level in the thyroid decreases to a significantly lower value after 8 mg/kg (746 ± 149 µg/g) than after 1 mg/kg (P < .05).
Content of Stable Iodine in the Thyroid and Total Excretion in the Urines 24 hours After Administration of Different Doses of KI.a

Abbreviations: KI, potassium iodide.
aEach value is the mean of n = 6 rats per KI dose.
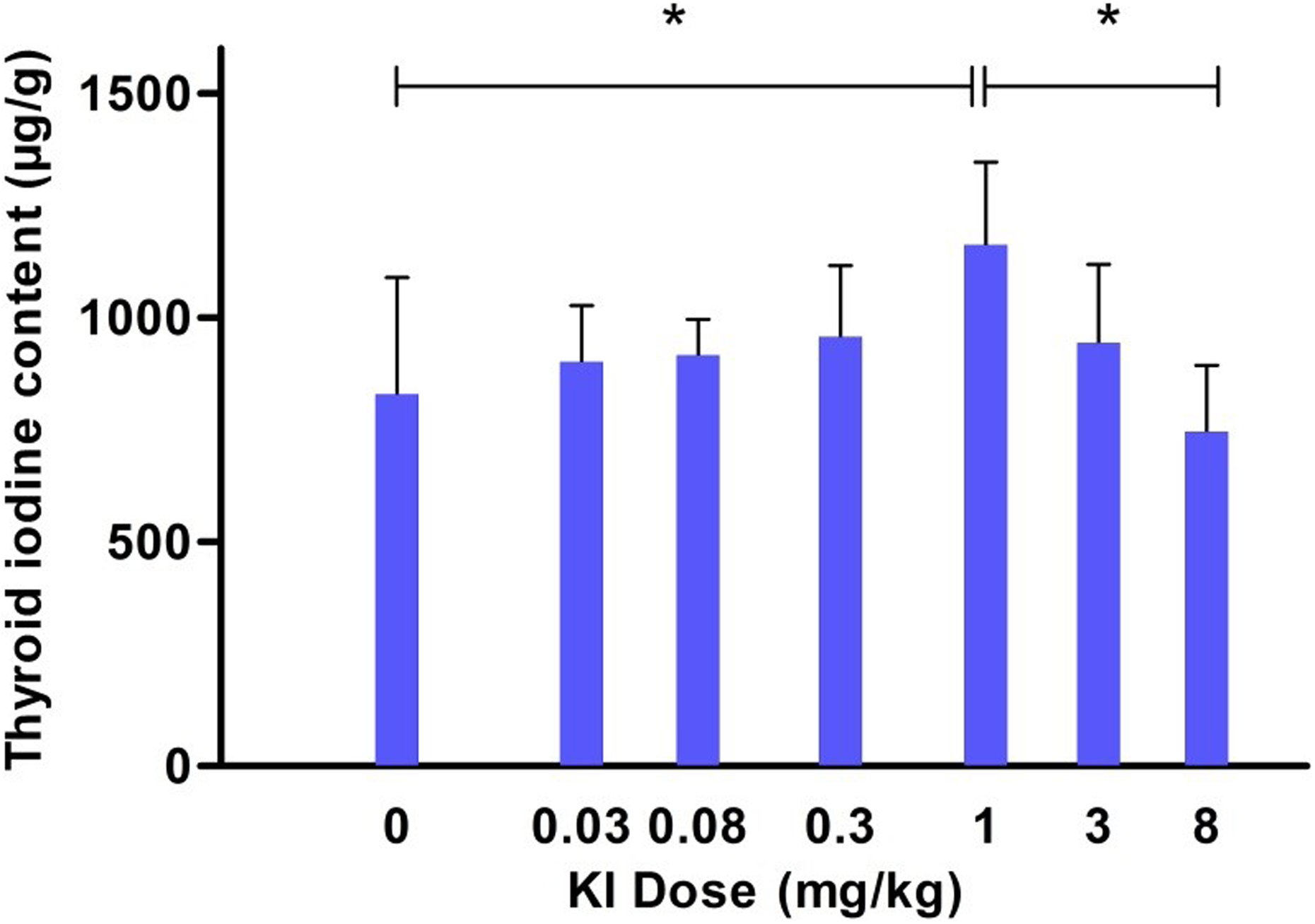
The observed values are the mean of 6 rats ± standard deviation (SD). *The difference between the 2 compared doses is significant with P < .05 (Student t test).
On the other hand, the analysis of the total cumulative amounts of stable iodine excreted in the urines 24 hours after treatment of the animals shows an increase in the iodine urinary excretion as a function of administered KI doses (Table 2). The urinary excretion of stable iodine can be modeled by a sigmoid-type pharmacological function with a better correlation compared to a linear function, according to the statistical parameters of the regressions (the AIC and SBC parameters of the sigmoid function are lower than those of the linear function; Figure 4). The sigmoid function parameters are given in Equation 5. The modeling of iodine urinary excretion by a sigmoid model suggests the existence of a saturation phenomenon of this function. In addition, a change in the slope of the urinary excretion profile observed on the semi-logarithmic scale representation occurs from administered doses above 1 mg/kg. This KI dose could be interpreted as a dose of body overload in iodine and, hence, could indirectly corroborate a possible saturation of the thyroid content in total iodine

where U is cumulated iodine excretion in 24-hour urines (mg) and D is KI dose (mg/kg).

The values observed for each rat are averages of 3 measurements ± standard deviation (SD) and represented by the circles, the linear function is represented by the dotted line and the sigmoid function by the solid curve, graph represents (A) potassium iodide (KI) doses in linear scale, and graph represents (B) KI doses in log scale.
Discussion
Evaluation of the efficacy of thyroid protection by stable KI in humans is rare, and contrary to animal studies, the clinical trials are often limited by the number of subjects and hence the dose of KI that could be tested. Nonetheless, it was suggested that the efficacy of KI depends on the diet status of the individuals and that KI doses as low as 0.2 and 0.4 mg/kg were efficient to block the uptake of iodine radioactive isotopes in thyroid, respectively, in adults with sufficient iodine intake and in individuals with inadequate iodine intake. 7,11,12 These data can be verified in the present study, since minimum KI doses of the same order of magnitude are efficient in adult rats with a sufficient iodine diet (KI doses about 0.5 to 0.7 mg/kg are associated with 75% thyroid protection 1 hour after exposure to I-125). Besides, a limited number of dose–response studies were conducted in rodents in order to reexamine the efficacy of KI 18 or to compare KI with other treatments such as perchlorate. 22 However, the doses of KI tested were higher than the currently administrated dose (from 2 to 22 mg/kg KI in mice and up to 30 mg/kg KI in rats) and no actual analyses of the dose–response relationship have been performed from a pharmacodynamic point of view. In the present work, the correlation between KI doses and its effect in thyroid in terms of protection of the organ from the incorporation of iodine radioisotope I-125 was best described by pharmacological functions. The inhibition-type model (Figure 1) or inversely the stimulation-type model (Figure 2) obtained suggests the existence of saturation phenomenon of physiological systems which imply iodine. Hence, those models are consistent with one of the assumed mechanisms of action of KI on thyroid such as a competition between stable iodide and radioiodine for their transport and distribution into the thyroid tissues.
The measurement of stable iodine content in thyroid by ICP-MS technique has never been described before in order to study the distribution of stable iodine after ingestion. Traditionally, the analysis of iodine in thyroid is performed by the Sandell-Kolthoff colorimetric method 23,24 or by molecular atomic spectrometry. 25 The results obtained by ICP-MS in the present study are in accordance with the other analytical methods, since the stable iodine contents in thyroid of control rats (18 ± 8 µg) are close to previously published values around 10 to 15 µg iodine per gland. 26,27 As a consequence, it is unlikely that ICP-MS may be not sensitive enough to discriminate small variation in thyroid iodine contents between control and treated animals. Besides, the observation time at 24 hours was selected on the basis of data from the literature. In subjects with normal thyroid function, iodine uptake by the thyroid is rapid and reaches a plateau at 24 to 48 hours, corresponding to a fraction of 10% to 40% of the total ingested iodine. 28 One would expect the total iodine content in the thyroid to reach a plateau and not to decrease as function of iodide doses in a dose–response study. However, the biphasic variation in total iodine contents in the thyroids observed in the present work (Figure 3) is consistent with the Wolff-Chaikoff effect, which can occur in less than 25 hours. 29 Such a variation in thyroid iodine is also consistent and comparable with previous studies in which increasing doses of KI labeled with I-131 were administered to rats. 19 In this latter study, the organification of stable iodine in the thyroid displayed a biphasic response with an increasing phase 4 hours after injection of graded KI doses lower than 0.3 mg/kg (50 µg iodide/rat) and a decreasing phase for higher KI doses up to 3 mg/kg (500 µg iodide/rat). 19 The KI dose corresponding to a maximal organification was however lower (0.3 mg/kg) than the saturation dose of the present study probably because the observation time was shorter (4 hours) and because the animals were kept under iodine-depleted diet before and during the experimental period. Thus, the concentration of stable iodine in the thyroid as a function of KI dose may probably be different in subjects with insufficient or low iodine intake in the diet. This hypothesis could be readily verified by measurements of iodine contents in the thyroids of animals kept under low iodine intake. Consequently, the decrease in the thyroid total iodine contents observed in our study 24 hours after the administration of KI doses higher than 1 mg/kg could be the result of 2 concurrent effects: first, the inhibition of the iodine incorporation and organification in the thyroid (Wolff-Chaikoff effect), and second, a continuous release of iodinated compounds such as the thyroid hormones thyroxine and triiodothyronine in order to maintain their physiological levels in the plasma. Further studies should be performed in order to verify the biochemical parameters and the thyroid hormone concentrations in the blood 24 hours after the administration of the same doses of KI to the rats. Additionally, the efficacy of thyroid protection by stable KI administration in subjects with sufficient iodine diet could be correlated with other parameters than iodine content in the thyroid. Urinary data, for instance, which are KI dose dependent in the present study, could be interpreted as indirect markers of thyroid saturation. Indeed, it is known that the fraction of iodine that is not up taken by the thyroid is completely excreted in urine from 24 to 48 hours following the incorporation, according to the current biokinetic models of iodine. 12,30 In our study, the pharmacological model that best fits the data of iodine urinary excretion as a function of KI dose is a sigmoid-type function (Figure 4A). Thus, a change in the slope of the curve could be interpreted as the consequence of a probable saturation of all physiological systems involved in the metabolism and transport of iodine in the organism, including the active transport of iodine in the thyroid by the sodium–iodine symporter for example. 31 -33 More particularly in the semi-log representation (Figure 4B), a change in the slope observed at a dose around 1 mg/kg KI and a subsequent increase in the excretion of iodine for higher doses could be interpreted as an overload or saturation dose of iodine in the body, and particularly in the thyroid. This result also suggests a possible saturation as a function of stable KI dose of the different mechanisms of transport and excretion of iodine in the kidney, such as the pendrin transporter present in the apical membrane of cortical ascending limbs and distal convoluted tubules 34,35 or the apical iodide transporter. 32 This dose of 1 mg/kg KI which is associated in our experiment conditions with more than 86% of thyroid protection 1 hour after exposure to iodine I-125, compared to untreated animals, could hence be selected as an optimal dose in order to propose adequate dose regimens for repeated prophylaxis. It should be noticed however that the application of conversion factors may be necessary for the transfer of the dose from the animals to the humans, although the dose range tested in the present study was derived from the dose currently used in human medicine.
Conclusion
In conclusion, the results of this dose–response study show that KI doses around 0.5 to 0.7 mg/kg provide at least 75% of thyroid protection in these experimental conditions. It was also developed in this work a new method based on ICP-MS measurement as an alternative to previous analytical methods or techniques requiring radiotracers to quantify stable iodine in complex biological matrix such as thyroid samples. The distribution of stable iodine in the thyroid 24 hours after KI administration was decreased for doses higher than 1 mg/kg. This observation could be correlated with the change in the urinary excretion profile of stable iodine, which suggests a possible saturation of the organism by KI doses higher than 1 mg/kg. The dose of 1 mg/kg could then be selected as an optimal dose in terms of saturation and protection of the thyroid. As perspectives, the pharmacokinetics of stable iodine after oral administration of this dose of 1 mg/kg KI should be determined in order to model adequate dose regimens for a protracted prophylaxis. In addition, the innocuousness of this dose of KI after repeated administrations should be assessed in different groups of rats representing the different groups of age of the population prior to considering the extension of the indication of KI in case of prolonged exposure to radioactive isotopes of iodine.
Footnotes
Acknowledgments
The authors are thankful to the Central Army Pharmacy for providing solutions of KI, Stéphane Grison and Philippe Lestaevel from the laboratory of radio toxicology of IRSN for their technical supports, Mélanie Berchet, Amandine Sache, Romain Granger, and Frédéric Voyer from the animal experiment support group of IRSN for animal care.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to research, authorship, and publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were supported by the research program in Nuclear Safety and Radiation Protection of the French National Research Agency (ANR; contract number ANR-11-RSNR-0019).


