Abstract
The liver and kidneys are the vital organs of the body and perform important life-sustaining functions in the body. Synthetic drugs used in the treatment of liver and kidney diseases are sometimes inadequate and can lead to serious side effects. Medicinal herbs and plants were used to combat diseases for a long time and combination therapy is preferred over single plant therapy. In the current study, the Asparagus racemosus, Mucuna pruriens, Anacyclus pyrethrum, and Tribulus terrestris polyherbal preparation (PHP) was selected to evaluate its hepatoprotective, antioxidant, and anti-nephrotoxic potential. The methanolic extract of PHP was prepared following standard protocols. Fifty-six albino rats were divided into 7 groups (n = 8). The negative control (NC) having the healthy rats and the remaining 6 groups were induced liver toxicity by intraperitoneally injecting 0.5 mL/kg of 50% CCl4 in olive oil. Group 2 was positive control and group 3 and 4 received silymarin standard drug at the dose of 100 and 200 mg/kg body weight. Groups 5, 6, and 7 (PHP-1, PHP-2, PHP-3) were the liver-damaged rats receiving the PHP at a dose of 50, 100, and 150 mg/kg body weight. Blood samples were collected at 21 of the trial, to evaluate oxidative stress, hepatoprotective and anti-nephrotoxic potential. Results of liver function tests revealed significant (P < .05) hepatoprotective activities of PHP after intoxication with CCl4 of albino rats as compared to standard groups. Moreover, results of renal functions also showed that PHP has a significant (P < .05) restoring the capacity of blood urea, creatinine, and uric acid in intoxicated rats as compared with the control group. The PHP also reduced the oxidative stress in the treatment groups by increasing the total antioxidant capacity and reducing the total oxidative status. It can be concluded that selected medicinal plants have a potential role in the management of liver and kidney disorders. So, by running the clinical trial on a large scale and by isolating the phytochemical constituents responsible for hepatoprotective and nephroprotective activities, locally prepared drugs could be developed to manage liver and renal disorders.
Introduction
The liver plays an important role in detoxifying poisons from the blood, digesting food, processing medication, and restoring nutrients and energy. Genetic factors obesity, infections, liquor utilization and long-term use of medicines, for example, anti-depression medications, analgesics, anabolic steroids, and oral contraceptives are a portion of the normal elements causing liver ailments. Untreated liver sicknesses may likewise bring about Liver Cirrhosis which is a major cause of death.1,2 In the body, internal metabolism and toxicant exposure can be caused to produce reactive oxygen species in humans which lead to oxidative pressure.
The kidney is another vital organ of the body to play an important role in maintaining blood volume and electrolyte balance. Nephrotoxicity is one of the most overall kidney troubles that outcome from the introduction of an extrinsic or intrinsic toxicant. The medicinal plants extract has phenolic and triterpenoid compounds as well as vitamin C. The antioxidant potential of these extracts suppressed the CCl4-induced oxidative stress by scavenging the reactive oxygen species and increasing the cellular antioxidant index. These extracts also reduce the CCl4-induced inflammation by inhibiting the gene expression of NF-кB, and iNOS, and in rotating the level of nitric oxide. 3 In past, it has been seen that medicinal herb treatment has developed as another option and better methodology for the treatment of kidney stones and urolithiasis, as the greater part of the traditional treatments accessible to date are not 100% powerful. 4
Since long ago, herbal compounds are found to be used as traditional medicine for the treatment of several diseases. The manufacturer of traditional medicine had an edge as they do not have the requirement to prove claims about treating diseases are valid or not; in contrast, if they deal with a product which would be a drug, they need to provide several proofs. Some of those compounds from herbal products might be safe, while some are not. There are some assumptions for these reports with the possibility of the products being contaminated or being in contact with toxins, heavy metals, and drugs or possibly not containing the claimed/listed ingredients. There is also the possibility of some traditionally used herbal medicines interacting with drugs, resulting in serious side effects, or might be considered unsafe for numerous patients with specific conditions. 5 So, all the traditional and folk medicine with no research data must be scientifically approved for their efficacy and side effects. In the present study, we aim to evaluate the hepatoprotective and nephroprotective effects of a polyherbal preparation (PHP) comprising methanolic extracts of four plants, A racemosus, M pruriens, A pyrethrum, and T terrestris
Methods
Procurement of Plants
All four plants, A racemosus, M pruriens, A pyrethrum, and T terrestris were collected locally. The plant was identified by an expert Botanist and kept in the herbarium of the Department of Botany, Government College University Faisalabad, Pakistan.
Extract Preparation
After washing with distilled water, the plants were shade dried and ground into a fine powder and 50 g of each plant powder was mixed and soaked for 72 h in 250 mL of methanol with periodically stirring and mixing. The solution was subsequently sieved through Whatman® filter paper. The extract after filtration was evaporated and concentrated using a rotary evaporator (SCI100-Pro; SCILOGEX, USA) at 40°C and transferred into a Petri dish and kept in an incubator at 40°C until dried properly. The percentage yield was calculated as 15.7%. The extract was stored at 4°C till further analysis.
Qualitative Phytochemical Analysis
Phytochemical analysis of a methanolic extract of the polyherbal preparation was carried out qualitatively using standard methods as described by Mustafa et al. 6 to verify the presence or absence of potentially active phytochemicals.
Experimental Design
Following the approval of all animal protocols by the Animal Care and Ethical Committee, Government College University of Faisalabad, 56 albino (Wistar strain) rats of mixed gender, aged 8 ± 1 weeks, weighing 260 ± 10 g, were procured from the Department of Physiology Animal Experimental Station, Government College University, Faisalabad, Pakistan. The rats were housed in elevated wired cages with ad libitum access to food and drinking water, at standard conditions (temp = 26°C ± 2°C; light = 12 h light and dark cycle; ambient humidity = 40–60%). The rats were divided into 7 groups with an equal number of rats (n = 8). The negative control (NC) having the healthy rats and the remaining 6 groups were induced liver toxicity by intraperitoneally injecting 0.5 mL/kg of 50% CCl4 in olive oil.7,8 After the confirmation of liver damage, group 2 was named as a positive control with damaged liver receiving no drug. Groups 3 and 4 were the standard controls (SC-1 and SC-2) receiving silymarin standard drug at the dose of 100 and 200 mg/kg body weight, respectively. 7 Groups 5, 6 and 7 (PHP-1, PHP-2, PHP-3) were the liver-damaged rats receiving the PHP at a dose of 50, 100, and 150 mg/kg body weight, respectively. The dose of the PHP treatment was adjusted according to the previous literature.9–12 After the completion of 21 days of treatment, all the rats were sacrificed by cervical decapitation for the collection of blood and serum was separated by centrifugation at 2000 r/min for 10 min and stored at −20°C till further analysis.
Total Antioxidant Capacity, (TAC, mmolTroloxequiv./L)
The method for evaluating total antioxidant capacity (TAC) in serum samples was previously defined by Nisar et al. (2017). 13 In short, the presence of antioxidants in the sample bleaches the orthodianisidine color in the assay reagent. Increased antioxidant levels in the sample contribute to higher bleaching and reduced absorbance, suggesting an inverse standard curve. The Biolab® 310 semi-auto analyzer was used with biochromatic wavelength modification (660 and 870 nm) by calibrating vitamin C standards at 1, 3, 5, and 7 mmol/L concentrations. This assay had a minimum observable value of .18 mmol/L and linearity of up to 7 mmol vitamin C equivalent/L, with a coefficient of variance (CV) below 3% in the intraassay.
Total Oxidant Status (TOS; µmol H2O2 equiv./L)
The serum TOS was measured by using the technique previously adopted by Mustafa et al. 14 The standard curve was constructed from different hydrogen peroxide (H2O2) concentrations and the TOS was represented as μmol H2O2 equivalent/L. The assay’s detection range was <3% and the CV of the Intra assay was held below 10% and linearity was up to 200 μMol H2O2 equivalent/L. 14
Serum Liver Enzymes
Aspartate aminotransferase (AST), alanine aminotransferase (ALT), and alkaline phosphatase (ALP) levels were examined by commercially available kits from Sigma diagnostics Co. (Cat#: 112 024, Cat#: 112 003 respectively). Total protein and albumin were also measured by using colorimetric assay through commercially available kits by Sigma diagnostics (Cat#: 610, Cat#: 913, respectively).
Evaluation of Renal Function Tests
“Urease – GLDH”: enzymatic UV test described by Thomas, (1998) was utilized to evaluate the urea level in the serum samples. A kinetic test without deproteinization as demonstrated by the Jaffé system was used to measure the serum creatinine level (Newman, 1999). 15 Serum uric acid level was examined using the methods for changed trinder peroxidase test using 3,5-dichloro-2-hydroxybenzenesulfonic acid (DCHB; Fossati et al., 1980). 16
Statistical Analysis
The SPSS software version 23 was used for statistical analysis. All datasets were statistically demonstrated as mean +standard error. One-way Analysis of Variance (one-way ANOVA) was observed to seek the significance of differences among various groups followed by Turkey’s post hoc test. The level of significance was actively considered at P < .05.
Results
Oxidative Stress Markers
The results of TAC and TOS are shown in Figure 1. The data are represented in mean ± standard error of a total of 8 samples for each experimental group. The mean TAC was decreased significantly (P ≤ .05) in the PC group (.45 ± .04b mM/L) as compared with other experiment groups (Figure 1). There was an increase in the TAC value in all the silymarin- and PHP-treated groups as compared with the PC and NC groups. The polyherbal preparation treatment significantly (P ≤ .05) decreased the TOS in the PHP-1, PHP-2, and PHP-3 groups in a dose-dependent manner as compared with the PC and NC groups. The TOS levels were also decreased significantly (P ≤ .05) in the silymarin-treated groups SC-1 and SC-2 in a dose-dependent manner. Effect of Silymarin (standard drug) and polyherbal preparation (PHP) methanolic extract on TAC (A) and TOS (B) in the serum of rats of various groups of the study. NC is Negative control, PC is Positive control, SC-1 and SC-2 means standard control groups receiving silymarin at the dose of and 100 and 200 mg/kg body weight, respectively, PHP-1, PHP-2, and PHP-3 shows the liver-damaged rats receiving the PHP at dose of 50, 100, and 150 mg/kg body weight, respectively. Different alphabets (a–c) on the bars show significant difference among the groups.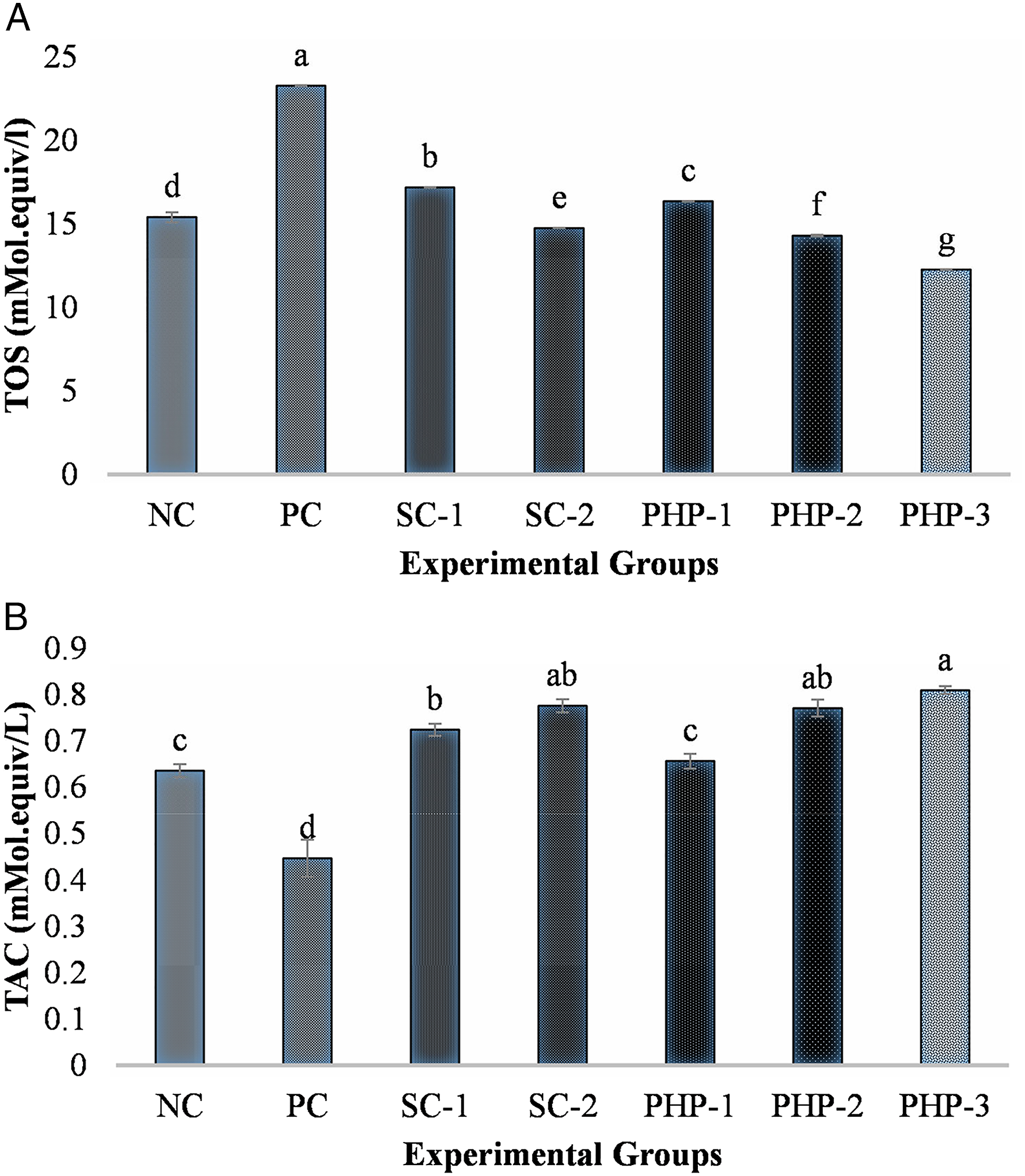
Liver Enzymes and Bilirubin
Effect of Silymarin (Standard Drug) and Polyherbal Preparation Methanolic Extract on Liver Enzymes and Total Bilirubin in the Serum of Rats of Various Groups of the Study.

NC is Negative control, PC is Positive control, SC-1 and SC-2 means standard control groups receiving silymarin at the dose of and 100 and 200 mg/kg body weight, respectively, PHP-1, PHP-2, and PHP-3 shows the liver-damaged rats receiving the PHP at dose of 50, 100, and 150 mg/kg body weight, respectively. Different alphabets (a–e) on the values in a row show significant difference among the groups.
Serum Proteins
The results of serum proteins, albumin, globulin, and total proteins are shown in Figure 2. The data are represented in mean ± standard error of a total of 8 samples. Serum albumin level was significantly increased in the PC group but was not significant in the silymarin- and PHP-treated groups. The serum globulin levels were increased significantly in the SC-2 and PHP-3 groups as compared with the PC group. While the total protein level was increased in all the silymarin- and PHP-treated groups as compared with the PC group. Effect of Silymarin (standard drug) and polyherbal preparation methanolic extract on albumin (A), globulin (B), and total proteins (C) in the serum of rats of various groups of the study. NC is negative control, PC is positive control, SC-1, and SC-2 means standard control groups receiving silymarin at the dose of and 100 and 200 mg/kg body weight respectively, PHP-1, PHP-2, and PHP-3 shows the liver-damaged rats receiving the PHP at dose of 50, 100, and 150 mg/kg body weight, respectively. Different alphabets (a–c) on the bars show significant difference among the groups.
Renal Function Tests
The results of serum levels of urea, uric acid, and creatinine are shown in Figure 3. The data are represented in mean ± standard error of a total of 8 samples. The results show that the serum urea level was significantly increased in the PC group as compared to the NC group. However, this rise was normalized by treatment with the silymarin and PHP in the SC-1, SC-2, PHP-1, PHP-2, and PHP-3 groups showing a significant (P ≤ .05) decrease in the serum urea levels as compared with the PC group. A similar trend was seen in the case of serum creatinine levels that were increased significantly (P ≤ .05) in the PC group as compared with all other experimental groups. While the treatment with the silymarin and PHP in the SC-1, SC-2, PHP-1, PHP-2, and PHP-3 groups significantly (P ≤ .05) decreased the serum urea level as compared with the PC group. Serum uric acid level was also increased significantly (P ≤ .05) in the PC group as compared with the NC group. Silymarin showed no effect on the serum uric acid level at the low dose of 100 mg/kg in the SC-1 group but its higher dose of 200 mg/kg was effective significantly (P ≤ .05) decreasing the serum uric acid level in the SC-2 group. Similarly, PHP treatment was effective in significantly (P ≤ .05) decreasing the serum uric acid levels at the dose of 100 and 150 mg/kg in PHP-2 and PHP-3 groups, respectively. Effect of silymarin (standard drug) and polyherbal preparation methanolic extract on urea (A), creatinine (B), and Uric acid (C) in the serum of rats of various groups of the study. NC is negative control, PC is positive control, SC-1 and SC-2 means standard control groups receiving silymarin at the dose of and 100 and 200 mg/kg body weight respectively, PHP-1, PHP-2, and PHP-3 shows the liver-damaged rats receiving the PHP at dose of 50, 100, and 150 mg/kg body weight, respectively. Different alphabets (a–c) on the bars show significant difference among the groups.
Discussion
Medicinal plants have various bioactive compounds, including terpenoids, flavonoids, glycosides, and alkaloids, having antifungal, antibacterial and antioxidant activities. Selected portions of plants in the current study were screened for the presence of phytochemical constituents. Results showed that phytochemicals such as alkaloids, flavonoids, tannins, saponins, glycosides, steroids, and triterpenoids were present in studied medicinal plants. Phenolic constituents are reported to be present in almost all types of plants. They comprise phenols, benzoic and cinnamic acid, coumarins, tannins, lignins, lignans, and flavonoids. Significant advancements in the research field concluded that phenolic compounds have been found to have therapeutic and nutritious value. Phenolic contents possess a significant role in reducing oxidative stress.6,17 In the current study, polyherbal preparation increased the TAC level and decreased the TOS level in the toxic rats. In an in vitro study, Karuna et al. 18 described the antioxidant activity of roots of A racemosus that is part of the current polyherbal preparation. Similarly, different studies also described the antioxidant activities of the other plants of polyherbal preparation. In another study, Tumbas-Saponjac et al., 19 demonstrated the M pruriens as a good antioxidant source. A pyrethrum and T terrestris plants also have demonstrated antioxidant effects.20,21 In our study, polyherbal preparation of these plants also shows the in vivo antioxidant activity by decreasing the TOS and increasing the TAC which might be due to the presence of phenolic and flavonoid constituents in the plants included in the polyherbal preparation. 22
In the current study, polyherbal preparation shows the hepatoprotective response by normalizing the ALT, AST and ALKP levels. ALT, AST, ALKP and bilirubin have great importance in the liver function test. Similar to our study, A racemosus decreased the ALT and AST levels in the t-BHP-treated rats. 23 In a study, T terrestris at the dose of 200 mg/kg protects the liver against cadmium toxicity. 24 Recently, Okorie et al. 25 demonstrated the M pruriens leaf extract effect on the liver. The result showed that ALP, AST, ALT, and albumin were found to be statistically significant among the treated groups when compared with the control group. 25 Various studies have reported the presence of phenolics, flavonoids, and anthraquinones in the A racemosus, M pruriens, A pyrethrum, and T terrestris used to prepare the PHP as a trweatment of liver injury in current study.26-31 The presence of these phytochemicals with proven antioxidant potential are responsible for the reversal of liver injury in current study. 32
Kidneys play a vital role in the excretion of toxins such as creatinine, uric acid, and urea. The nephron is the functional unit of the kidney and the renal function test is important to reveal the nephrotoxicity in the patients. In the current study, a protective response toward nephrotoxicity was revealed by performing the renal function test. Polyherbal preparation decreased the urea and creatinine levels in all the groups treated with PHP. However, PHP decreased the uric acid level in a dose-dependent manner in PHP-2 and PHP-3 groups. T terrestris showed a protective kidney response in nephrotoxic rats. 33 M pruriens at the dose of 700 mg/kg b.wt show a protective response against arsenic-induced liver and kidney toxicity by reduced levels of kidney and liver enzymes. 34 A racemosus showed the kidney protective effect against alloxan induced diabetes rats. 35 Similar to above-described findings, polyherbal preparation showed the nephroprotective response in the current study due to the presence of phytoconstituents in plants of PHP which reversed the oxidative stress that caused the nephrotoxicity induced by CCl4. 30
Conclusion
The current study concludes that the methanolic extract of the poly herbal preparation consists of the four plants named, A racemosus, M pruriens, A pyrethrum, and T terrestris contains antioxidant, hepatoprotective and nephroprotective activities. The active constituents of these plants can be separated to evaluate the above-mentioned activities of each constituent of each plant separately and a cheaper drug can be prepared from the most active constituents with maximum therapeutic efficacy and less side effects.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
