Abstract
Objectives
Adult T-cell leukemia/lymphoma (ATLL) is a blood neoplasm with specific geographic distribution. Although radiotherapy is a palliative treatment that provides long-term local control, single use of radiation leads to complications for patients. To introduce a novel multimodal approach against ATLL, we investigated combinatorial effects of 7-geranyloxycoumarin and radiation in vitro.
Methods
Viability of MT-2 cells was determined by resazurin assay upon administration of 7-geranyloxycoumarin alone and followed by radiation. Then, apoptosis was detected by annexin V and propidium iodide, and the expression of candidate genes was analyzed by qPCR.
Results
Findings revealed significant (P<.0001) improvement in radiation effects upon 7-geranyloxycoumarin pretreatment, most notably when cells were pretreated with 5 µg/ml 7-geranyloxycoumarin for 96 h, exposed to 6 Gy radiation and recovered for 48 h. These results were confirmed by flow cytometry, as the percentage of early and late apoptotic cells was increased after combinatorial treatment. In addition, significant (P< .0001) changes in CD44, c-MYC, cFLIPL, BMI-1, NF-κB (Rel A), and P53 expression was induced by 7-geranyloxycoumarin and radiation.
Conclusions
Current research indicated, for the first time, that combinatorial use of 7-geranyloxycoumarin and ionizing radiation could be considered as an effective therapeutic modality for ATLL.
Introduction
Adult T-cell leukemia/lymphoma (ATLL) is a rare lymphoproliferative disorder of mature CD4+ T cells that is caused by human T-lymphotropic virus type 1 (HTLV-1). 1 ATLL requires a long latent period before onset, demonstrating that viral genes, as well as modifications of the host genome (genetic and epigenetic), are crucial in leukemogenesis. 2 HTLV-1 has specific geographical distribution, as Japan, South America, West Africa, the Caribbean and Melanesian islands, and the Middle East are main endemic regions for this virus.3-5 Aside from these areas, HTLV-1 is known to be widespread in Northeast Iran, particularly in Mashhad and Neyshabour.6,7 HTLV-1 incidence varies amongst different populations and increases with age, and the virus is known to be more prevalent amongst females.8,9 Approximately 3–5% of HTLV-1 infected patients develop ATLL, which can be classified as acute (60%), lymphomatous (20%), chronic (15%), and smoldering (5%).10,11 Despite advancements in chemotherapy and supportive care, the median survival time for ATLL is still less than a year. 12 The introduction of antiviral agents, monoclonal antibodies, and allogeneic hematopoietic stem cell transplantation seems to be promising; however, these methods are insufficient to ensure long-term survival for most ATLL patients. 13 Radiotherapy is an excellent palliative treatment option that provides long-term local control and clinical benefit, even in advanced-stage lymphomas. Nevertheless, the use of radiotherapy as a single modality requires wide extension of the radiation field, as well as raising the dose to normal tissue tolerance levels, which lead to the late development of complications for patients. 14
7-geranyloxycoumarin, also known as auraptene, is a natural monoterpene coumarin found in Rutaceae and Umbelliferae plants. This simple prenylcoumarin owns various pharmaceutical properties, such as antibacterial, antigenotoxic, antioxidative, and anti-inflammatory activities. 15 Studies have also reported cancer chemopreventive effects of 7-geranyloxycoumarin, in addition to its combinatorial effects with anticancer drugs and ionizing radiation.16-22
Numerous laboratory investigations are currently focused on the administration of agents that could boost the sensitivity of cancer cells to conventional therapies, while having minimal negative effects on non-neoplastic cells. Since radiotherapy is used for palliation of ATLL patients, improving the radio response of cancer cells would result in better clinical outcomes. Our research team has demonstrated radiosensitizing effects of 7-geranyloxycoumarin on colon carcinoma, gastric adenocarcinoma and esophageal carcinoma cells.19-22 Herein, we applied a combined therapeutic strategy against ATLL based on 7-geranyloxycoumarin and radiation. To do so, the viability of ATLL cells was determined by resazurin assay upon administration of 7-geranyloxycoumarin alone and followed by radiation exposure. Then, apoptosis was detected using fluorescein isothiocyanate (FITC)-annexin V and propidium iodide (PI), and the expression of CD44, c-MYC, cFLIPL, BMI-1, NF-κB (Rel A), and P53 was analyzed by quantitative polymerase chain reaction (qPCR).
Materials and Methods
Synthesis of 7-Geranyloxycoumarin
Under standard laboratory conditions, 7-geranyloxycoumarin (C19H22O3, MW: 298.4 g/mol) was sensitized through a reaction between 7- hydroxycoumarin (1 M) and transgeranyl bromide (1.5 M) in the presence of 1,8-diazabicyclo [5.4.0]undec-7-ene (2 M) as previously described. 23 Once the mixture was concentrated at low pressure, column chromatography (petroleum ether/ethyl acetate 9:1 v/v) was carried out to purify 7-geranyloxycoumarin (mp = 62.7°C–63.4°C). Then, 1H and 13C NMR were utilized to validate the structure of 7-geranyloxycoumarin. To prepare various concentration of 7-geranyloxycoumarin, dimethyl sulfoxide (DMSO) was used as solvent, and thus, equal amount of DMSO in all 7-geranyloxycoumarin concentrations (.4% v/v) was considered as control.
Treatment and Viability Assessment of Cells
MT-2 cells, lymphoma cells infected with HTLV-1, were derived from normal human cord leukocytes of a healthy donor by co-cultivation with leukemic cells from an adult T-cell leukemia (ATL) patient. 24 In the present study, MT-2 cells (donated by Inflammation and Inflammatory Diseases Research Center, Faculty of Medicine, Mashhad University of Medical Sciences) were cultured in Roswell Park Memorial Institute-1640 supplemented with 10% fetal bovine serum, 1% (W/V) penicillin/streptomycin and 1% L-glutamine, and incubated at 37°C with 5% CO2 in air.
To determine the toxicity of 7-geranyloxycoumarin, cells were seeded (5×104 cells/well in 96-well plates) and treated with 5, 10, 20, and 40 µg/ml 7-geranyloxycoumarin for 24, 48, 72, 96, 120, and 144 h. For combinational treatment, cells were pretreated with 5 and 10 µg/ml 7-geranyloxycoumarin for 24, 48, 72, and 96 h, followed by radiation exposure using Elekta Compact™ linear accelerator (Crawley) at three different doses (2, 4, and 6 Gy). Finally, cells were recovered for 48 and 72 h and assessed for viability.
Resazurin (7-Hydroxy-3H-phenoxazin-3-one 10-oxide) was used to assess cell viability upon treatment with 7-geranyloxycoumarin, alone or followed by radiotherapy. In this regard, 20 µl resazurin (.1 mg/ml, Sigma) was added to each well, and cells were incubated for 2 h at 37°C. Finally, the absorbance of wells was measured at 600 nm using a microplate reader (Epoch), and cell viability (%) was calculated using the following formula: (100-(AT-AU/AB-AU))×100, in which AT and AU represent the absorbance of treated and untreated cells, respectively, and AB was the absorbance of blank control.
Detection of Apoptosis
Cell apoptosis was measured by FITC-annexin V and PI. In summary, MT-2 cells (5×105 cells/well in 6-well plates) were incubated with 7-geranyloxycoumarin (5 µg/ml) for 96°h and irradiated at 6 Gy. After 48°h recovery, cells were collected and stained with FITC-annexin V and PI (MabTag) according to the manufacturer’s protocol. Finally, the degree of apoptosis was analyzed by flow cytometry (BD FACS) using FL1 and FL2 filters.
Gene Expression Analysis
List of primers and probes used for qPCR analysis in current study.

Statistical Analyses
All results were plotted in the form of bar graphs using GraphPad Prism. Dunnet and Bonfferroni statistical analyses were used to determine the significant difference between groups. Results were expressed as mean ± standard deviation (SD), and P values less than .05, .01, .001, and .0001 (shown by *, **, ***, and ****, respectively) were considered to be statistically significant.
Results
Resazurin assay demonstrated that 7-geranyloxycoumarin reduced the viability of MT-2 cells in a dose- and time-dependent manner. As presented in Figure 1, upon 24, 48, 72, 96, 120, and 144 h treatment with 5 µg/ml 7-geranyloxycoumarin, viability was calculated as 98%, 96%, 84%, 79%, 83%, and 81%, respectively. However, upon treatment with the highest concentration (40 µg/ml) during the same consecutive time periods, cell viability was determined as 92%, 65%, 52%, 42%, 37%, and 30%, respectively. Accordingly, 5 and 10 µg/ml 7-geranyloxycoumarin were used as optimum concentrations of this agent for combinatorial experiments. Time-based dose response analysis of MT-2 cells after treatment with 5, 10, 20 and 40 µg/ml 7-geranyloxycoumarin for 24, 48, 72 h, 96, 120, and 144°h. Resazurin assay was carried out for at least three times and results were presented as mean ± SD. *P < .05, **P < .01 and ****P < .0001 indicate significant difference with DMSO control.
To investigate radiosensitizing effects of 7-geranyloxycoumarin, MT-2 cells were pretreated with 5 and 10 µg/ml 7-geranyloxycoumarin for 24, 48, 72, and 96°h, then exposed to 2, 4, and 6 Gy X-radiation and recovered for 48 and 72°h. As shown in Figure 2, assessment of cell viability revealed significant (P < .0001) increase in the toxicity of applied radiation, almost in all combinatorial treatments, in comparison with relevant controls (.4% DMSO +2, 4, and 6 Gy). With regard to 2 Gy radiation, the lowest viability was 60.8% when cells were recovered for 48°h, and 49.5% when cells were recovered for 72°h (Figure 2-A and B). In addition, when 4 Gy radiation was applied, viability decreased down to 47.6% and 26.9% upon 48 and 72°h recovery, respectively (Figure 2-C and D). More interestingly, after radiation exposure at 6 Gy, viability was determined as .5% and 1.8% when cells were recovered for 48 and 72°h, respectively (Figure 2-E and F). Viability of MT-2 cells after combinatorial treatment with 7-geranyloxycoumarin and radiation. After pretreatment of cells with 5 and 10 µg/ml 7-geranyloxycoumarin for 24, 48, 72, and 96°h, radiation was applied at 2 Gy (A and B), 4 Gy (C and D), and 6 Gy (E and F), and cells were recovered for 48 h (A, C, and E) and 72 h (B, D, and F). Resazurin assay was carried out for at least three times and results were presented as mean ± SD. *P < .05, **P < .01, ***P < .001, and ****P < .0001 indicate significant difference with radiation alone.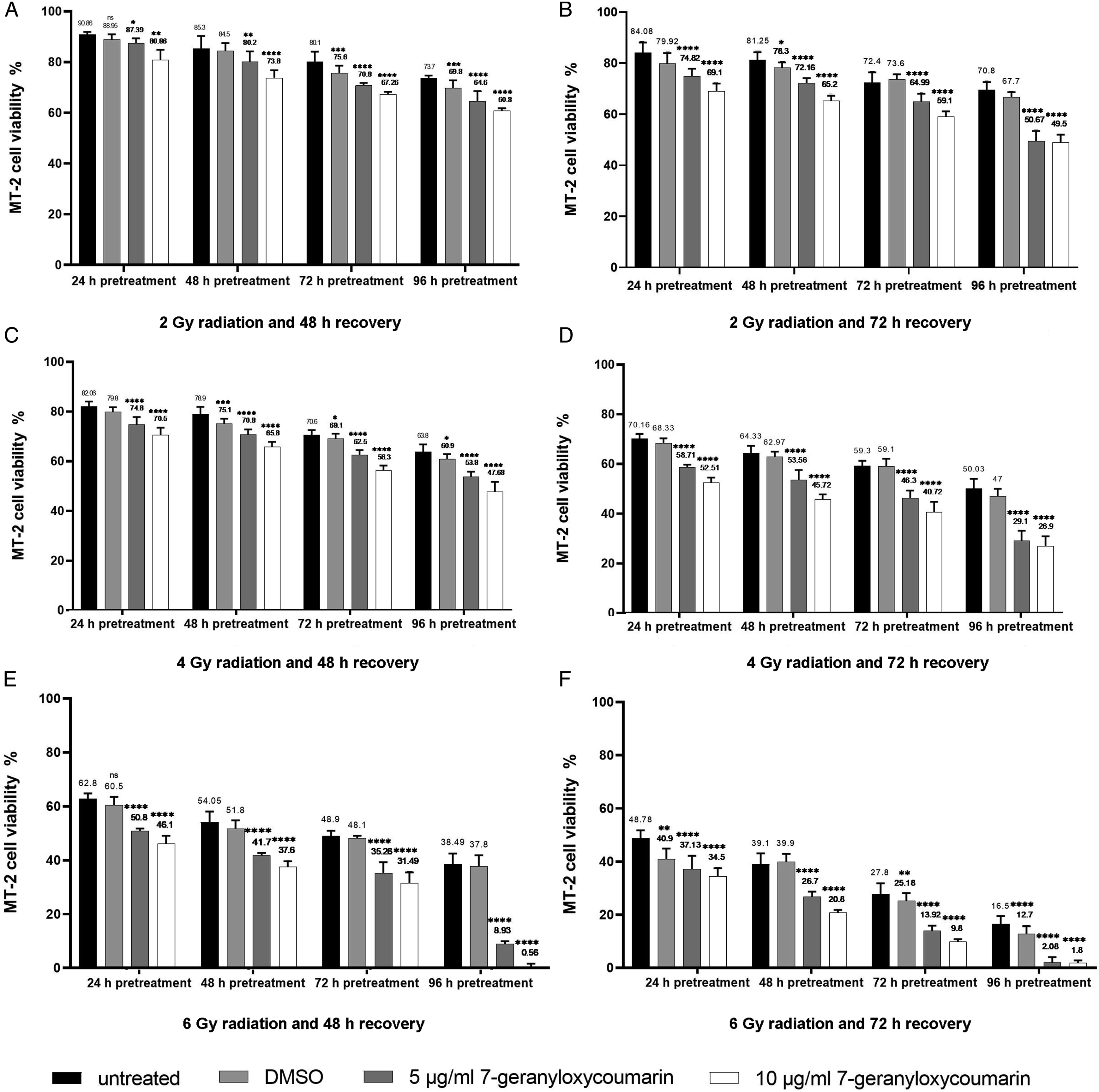
Figure 3 demonstrates flow cytometry analysis after FITC-annexin V and PI staining. As shown, upon 144 h treatment with 5 µg/ml 7-geranyloxycoumarin, the percentage of alive cells was 64.2%, lower than that for untreated and DMSO treated cells (97.4% and 70.4%, respectively). When 6 Gy radiation was applied and cells were recovered for 48°h, 64.1%, 18.8%, and 16.9% of cells were alive, early apoptotic and late apoptotic, respectively, which was similar to the percentage of cells detected after DMSO and radiation treatment. Interestingly, 96 h pretreatment of cells with 5 µg/ml 7-geranyloxycoumarin followed by 6 Gy radiation and 48 h recovery induced considerable changes in the cell population, as 45.1%, 24%, and 30.9% of cells were alive, early apoptotic and late apoptotic, respectively. Flow cytometry analysis after single and combinatorial treatments to differentiate alive cells from early and late apoptotic cells.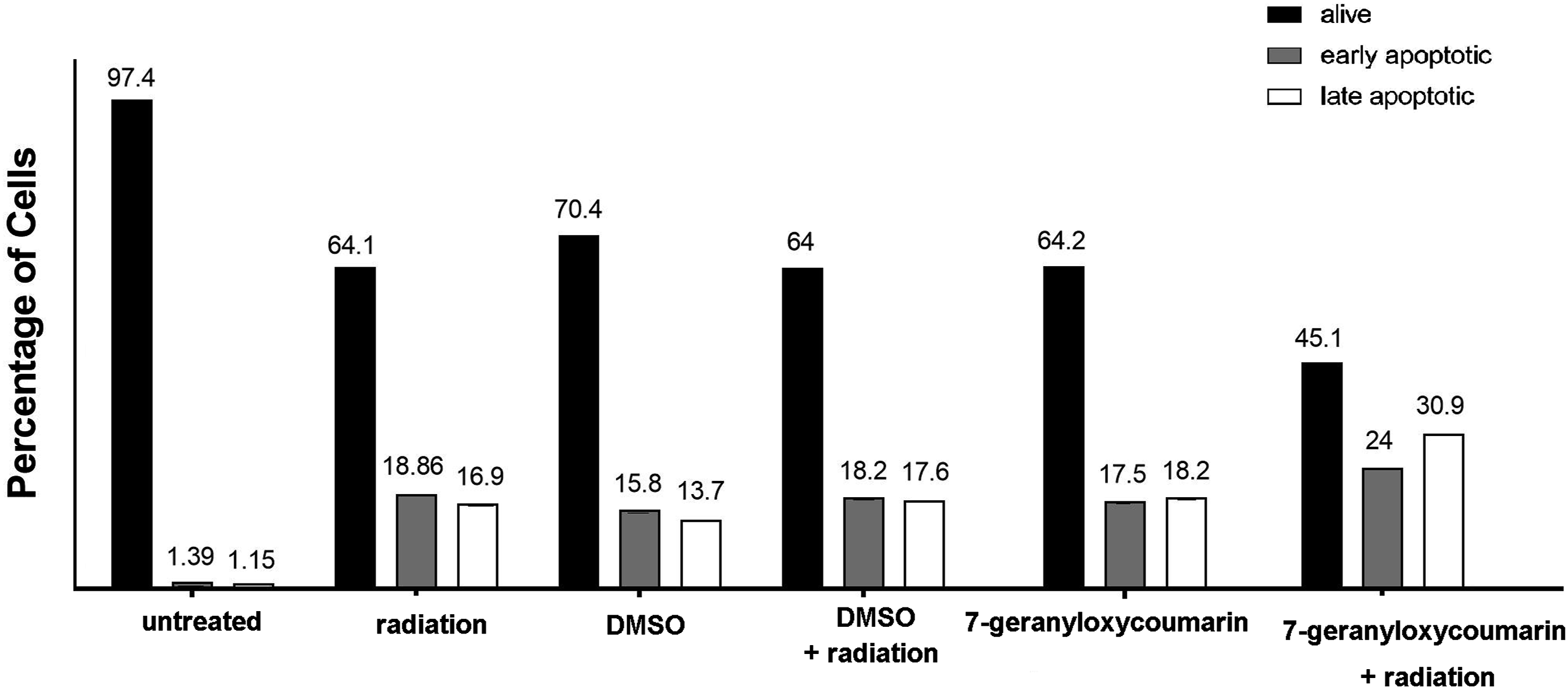
In order to evaluate alterations induced by our combined approach in the expression of CD44, c-MYC, cFLIPL, BMI-1, NF-κB (Rel A), and P53, qPCR was carried out and results were presented in Figure 4. As illustrated, 5 µg/ml 7-geranyloxycoumarin alone and followed by 6 Gy radiation significantly (P< .0001) reduced the expression of CD44, c-MYC, and cFLIPL (Figure 4-A-C). With regard to BMI-1 expression, significant (P< .0001) downregulation was detected after single and combinatorial administration of 7-geranyloxycoumarin in comparison with their relevant DMSO controls (Figure 4-D). On the other hand, 7-geranyloxycoumarin alone and in combination with radiation significantly (P< .0001) upregulated the expression of NF-κB (REL-A), while significant (P< .0001) induction in P53 expression was only detected upon 7-geranyloxycoumarin and radiation treatment (Figure 4E and F). Analysis of gene expression by qPCR. After 96 h pretreatment of cells with 7-geranyloxycoumarin (5 μg/ml), radiation (6 Gy) was applied, cells were recovered for 48 h and the relative expression of CD44, c-MYC, cFLIPL, BMI-1, NF-κB (Rel A) and P53 was evaluated. Normalized values were plotted over untreated cells. **P < .01, ***P < .001 and ****P < .0001 indicate significant difference with control.
Discussion
ATLL is a public health concern in specific geographic areas including our country Iran.6,7 Although radiotherapy is an effective modality for the treatment of most lymphomas, single-use of radiation requires raising the dose to normal tissue tolerance levels, which is associated with late development of complications for patients. 14 Hence, introduction of novel multimodal therapies that improve clinical outcomes is a critical demand. In the present study, we designed a new approach against ATLL cells and investigated combinatorial effects of 7-geranyloxycoumarin, a valuable pharmaceutical agent, and radiation in vitro. Our findings indicated improvement in radiation effects upon pretreatment with 7-geranyloxycoumarin, most notably when MT-2 cells were pretreated with 5 µg/ml 7-geranyloxycoumarin for 96°h, then exposed to 6 Gy radiation and recovered for 48°h. Results of cell viability assay were confirmed by flow cytometry, since the percentage of early and late apoptotic cells was increased after 7-geranyloxycoumarin and radiation treatment.
Studying the mechanisms involved in our combinatorial approach revealed significant downregulation in CD44, c-MYC, and cFLIPL expression, as well as significant overexpression of NF-κB (REL-A) and P53. CD44 is a transmembrane glycoprotein involved in drug resistance and anti-apoptosis mechanisms. 25 Upregulation of CD44 after high-dose irradiation contributes to longer-term cell survival through maintaining extracellular signal-regulated kinase (Erk) phosphorylation. 26 Induction of CD44 by radiation has been reported in glioblastoma, pancreas, bladder, colon, and prostate carcinoma cells,26-30 similar to our results that revealed upregulation of CD44 expression in ATLL cells upon single use of 6 Gy radiation. Interestingly, 7-geranyloxycoumarin downregulated CD44 expression, and when used in combination with radiation, expression of this gene was reduced down to lower levels. In line with this finding, negative regulatory effect of 7-geranyloxycoumarin on CD44 expression has been previously reported in esophageal carcinoma, colorectal carcinoma, and ATLL cells.20,21,31 c-MYC is an oncogene that controls cell proliferation, differentiation, and apoptosis. The radioresistant phenotype of embryonal rhabdomyosarcoma, glioblastoma, osteosarcoma, and lung adenocarcinoma cells has been linked to c-MYC expression, and radiosensitizing effects of c-MYC knockdown has been reported recently.32-35 cFLIPL prevents caspase-8 activation and cell apoptosis mediated by death receptors. It has been reported that cFLIPL negatively modulated radiosensitivity of lung cancer cells, and knockdown of this gene sensitizes cervical adenocarcinoma cells to radiotherapy.36,37 Present results demonstrated significant decrease in c-MYC and cFLIPL expression upon single and combinatorial use of 7-geranyloxycoumarin, which are in consistence with other studies.31,38 Accordingly, negative regulation of CD44, c-MYC, and cFLIPL explains, to some extent, the observed effects of our novel combinatorial approach.
The overexpression of BMI-1, an oncogene connected to cell cycle progression and immortalization, has been linked to disease progression and poor clinical outcomes in various human cancers.39-41 BMI-1 induced radioprotective effects via reducing the genotoxicity of radiation, and therefore, reduction of BMI-1 protein in cancer cells elevates susceptibility to radiotherapy. 42 Previous reports on radiation-induced BMI-1 expression in glioblastoma, breast, and esophageal carcinoma cells43-45 confirm our results on significant up-regulation of this gene in ATLL cells by 6 Gy radiation. Intriguingly, 7-geranyloxycoumarin counteracts radiation effects on BMI-1 expression, as significant downregulation of this gene was detected upon treatment with 7-geranyloxycoumarin alone and in combination with radiation. In agreement with current findings, negative regulatory effects of 7-geranyloxycoumarin on BMI-1 expression has been reported on other cancer cell types.20,31 NF-kB controls radioresistance through the regulation of DNA double-strand break repair, cell cycle arrest and apoptosis. It has been shown that activating NF-kB confers resistance of hepatocellular, oropharyngeal and esophageal carcinoma cells to radiation.46-48 In addition, it has been demonstrated that NF-kB stimulates the expression of death receptors (DR) 4 and 5, Fas and Fas ligand (FasL), which are associated with induction of apoptosis. 49 Results of present study are supported by above mentioned reports, as single and combinatorial use of 7-geranyloxycoumarin and radiation induced the expression of NF-kB in ATLL cells. P53 is a tumor suppressor gene with low expression level in non-transformed cells. Several studies have defined P53 as a key molecule involved in radioresponse of cells, since it is linked to radiotherapy efficacy,50-52 and is considered as a biomarker for clinical radiosensitivity. 53 Radiation-induced expression of P53 has been demonstrated in glioblastoma and hepatocellular carcinoma cells,54,55 similar to our results that indicated upregulation of P53 by 7-geranyloxycoumarin and radiation. Hence, observed combinatorial effects in present study could also be explained by changes induced in the expression of BMI-1, NF-κB (REL-A), and P53.
Beside modulation of CD44, c-MYC, cFLIPL, BMI-1, NF-κB (Rel A), and P53 expression, other possible mechanisms (Figure 5) could be associated with observed effects of 7-geranyloxycoumarin and radiation, although more research is required to confirm their involvement. For instance, aldehyde dehydrogenase 1 (ALDH1), which oxidizes intracellular aldehydes to carboxylic acids, reduces the level of reactive oxygen species (ROS) and oxidative stress induced by ionizing radiation.56,57 In this regard, negative regulatory effect of 7-geranyloxycoumarin on ALDH1 expression has been demonstrated previously.
19
Moreover, inhibition of poly (ADP-ribose) polymerase1 (PARP1), an essential enzyme involved in the repair of radiation-induced DNA damage, has been reported upon 7-geranyloxycoumarin treatment.38,58 Accordingly, it is possible that combinatorial effects of 7-geranyloxycoumarin and radiation were somehow due to the inhibition of ALDH1 and PARP1 expression. Schematic representation of changes induced by 7-geranyloxycoumarin and radiation on the expression of CD44, c-MYC, cFLIPL, BMI-1, NF-κB (Rel A), and P53 in ATLL cells, and possible involvement of ALDH1, COX-2, PARP1, ERK 1/2, MCL-1, CD44, and PCNA in observed effects of our combinatorial approach.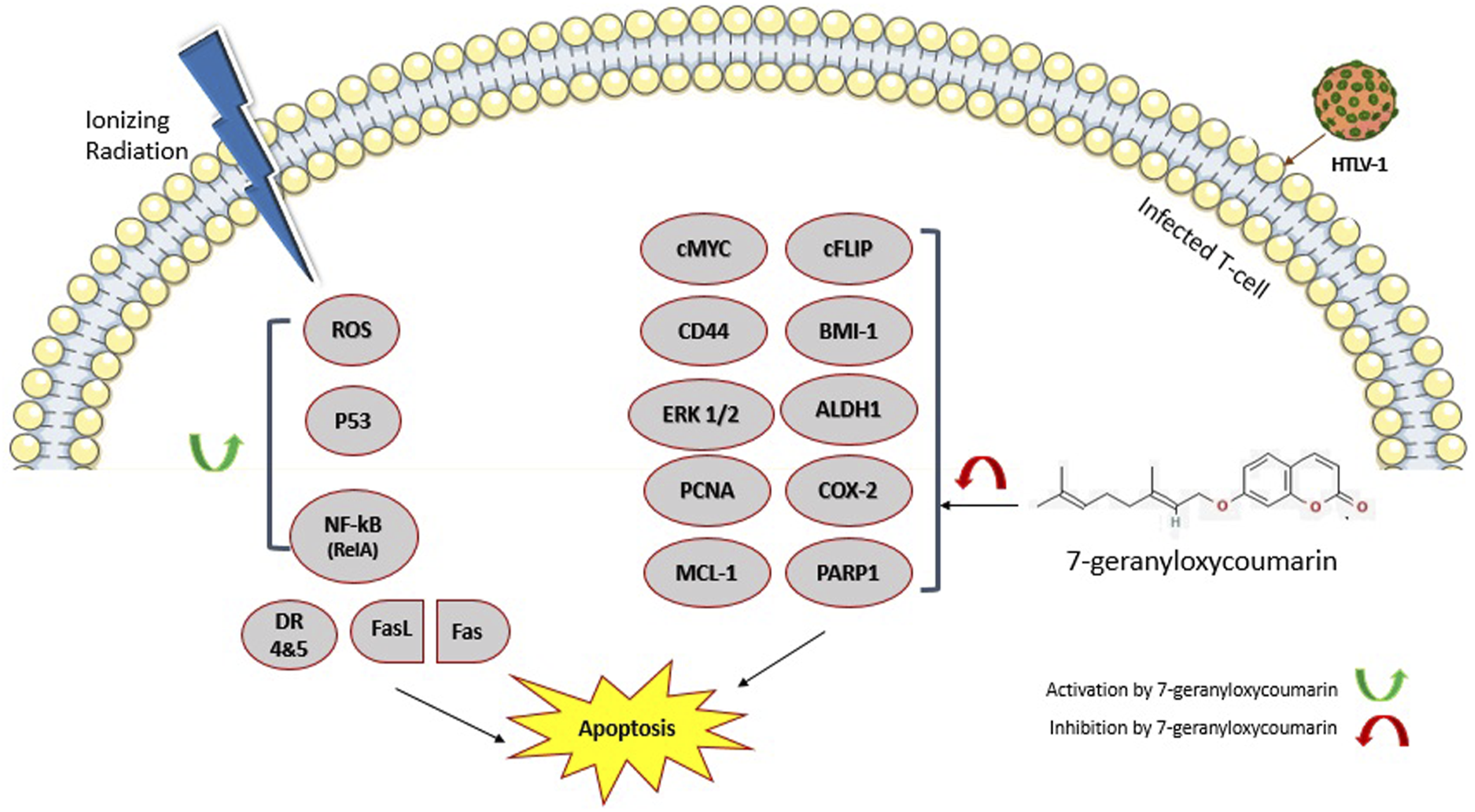
Proliferating cell nuclear antigen (PCNA) is an important protein involved in DNA replication and repair, as well as cell cycle regulation. 59 Cyclooxygenase‐2 (COX‐2) promotes proliferation, angiogenesis, inflammation, invasion, and apoptotic resistance in cancer cells. 60 As previously reported, 7-geranyloxycoumarin downregulated PCNA and COX-2 expression, 17 therefore, it is presumable that 7-geranyloxycoumarin improved radioresponse of ATLL cells via negative regulation of PCNA and COX‐2.
As a mitogen-activated protein kinase family member, Ras-dependent extracellular signal-regulated kinase (ERK) 1/2 regulates cell cycle and proliferation, and inactivates apoptotic genes such as BAX. 61 Myeloid leukemia 1 (MCL-1) is a member of BCL-2 family involved in apoptosis resistance of cancer cells.62,63 Since previous studies indicated inhibitory effects of 7-geranyloxycoumarin on ERK 1/2 and MCL-1,64,65 downregulation of both genes might also be involved in observed effects of 7-geranyloxycoumarin and radiation.
In conclusion, we reported for the first time, that 7-geranyloxycoumarin improved radioresponse of human ATLL cells. Complementary studies on other ATLL cell lines, such as Hut-102, SP, and MT-4, are recommended for comprehensive evaluation of 7-geranyloxycoumarin and radiation effects. In addition, future in vivo studies and clinical trials are necessary before introducing 7-geranyloxycoumarin as a safe and effective radiosensitizer agent for ATLL treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was financially supported by a grant from Mashhad University of Medical Sciences (No. 970298).
Ethical Approval
This study does not involve human participants and/or animals.
Data Availability
The data that support the findings of this study are included in the manuscript and available from the corresponding author upon reasonable request.
