Abstract
The reproductive system is vulnerable to ionizing radiation, which is a hot research topic at present. We tested the effect of polydatin on spermatocytes(GC-1 cells) after X-ray irradiation. The reproductive damage model of C.elegans was established by 60Coγ-ray, and the protective effect of polydatin on reproductive damage caused by ionizing radiation was evaluated. We quantified the ROS levels of GC-1 cells and C.elegans after irradiation with polydatin and evaluated the anti-apoptosis effect of polydatin at proper concentration. Differential genes of C.elegans reproductive damage were screened out from transcriptome sequencing results and comparable GEO datasets. It was proved that 100μM polydatin significantly reduced the apoptosis of GC-1 cells induced by 2 Gy X-ray. In addition, the longevity, reproductive capacity, germ cell apoptosis and spawning and hatching capacity of polydatin were tested. The results showed that 100 μM polydatin content significantly increased the influence of 50 Gy 60Coγ-ray on reproductive capacity of C.elegans. Quantitative analysis of mRNA and protein levels of apoptosis-related genes and reproductive-related genes by qRT-PCR and Western blotcon firmed that polydatin with appropriate dosage had good protective effects on reproductive damage caused by radiation, which laid a foundation for the application research of polydatin in radiation protection.
Introduction
Ionizing radiation technology has been widely used in daily life, but the accompanying risk of radiation damage is also increasing. The reproductive system is a sensitive target organ of ionizing radiation. Excessive radiation will affect reproductive function, resulting in infertility, exposure during pregnancy will also cause embryo deformity, abortion, and other adverse outcomes.1,2 The epidemiological results of the Chernobyl accident found that long-term low-dose radiation increased the risk of infertility or postpartum anemia among women, 3 cohort study in high background radiation area found long-term low dose exposure to reduce male sperm motility. 4 These radiation–induced reproductive health problems plague scientific research workers, and it is urgent to find appropriate radiation protection agents and explore its biological mechanism.
Because of the reproducibility of sperm and spermatogonia, spermatogonia are usually used by researchers in germ cell selection experiments or directly detecting sperm motility. 5 Among them, GC-1 cells of mouse spermatogonia played an important role in the study of reproductive damage mechanism caused by different types of radiation.6,7 In addition, due to the particularity of the reproductive system, it is particularly important to choose the appropriate model organism to study reproductive damage. In recent years, researchers have found that Caenorhabditis elegans(C.elegans) is a reliable model organism for studying reproductive damage, aging, and nervous system damage.8-10 In the study of radiation–induced reproductive damage, ionizing radiation can inhibit C.elegans reproductive ability, increase the level of ROS in vivo, and cause apoptosis of germ cells, which is a good biological model to study radiation-induced reproductive damage.11-13 And provides a scientific reference basis for researchers.
Currently, there are several anti-radiation drugs available in clinics with definite efficacy, such as amifostine, glycoprotein granulocyte colony-stimulating factor (G-CSF), and nitroxide. 14 However, these drugs have adverse effects and low safety profiles, and their effects on the reproductive system are elusive.15-17 Therefore, there is an urgent need to identify new and effective agents that can protect against radiation-induced damage to the reproductive system. Polydatin, the main active ingredient of Polygonum cuspidatum, is a natural precursor of resveratrol and has similar pharmacological effects as resveratrol. 18 However, polydatin has a stronger antioxidant activity and higher stability than resveratrol. 19 Studies have shown that polydatin can significantly inhibit the production of excessive oxygen free radicals and reactive oxygen species in vivo. 20 Ma et al. 21 results showed that polydatin inhibited the apoptosis of germ cells and reduced the damage of ionizing radiation to testis by scavenging ROS in C57BL/6 mice. Moreover, polydatin can improve the damage of hematopoietic system caused by neutron radiation by inhibiting cell apoptosis. 22 The above evidence shows that polydatin has a good prospect in radiation protection, and its reliability needs more experimental verification.
Based on the above reasons, this study used X-ray to irradiate GC-1 cells, and 60Coγ-ray was used to establish the radiation reproductive damage model of C.elegans and tested the influence of polydatin on various indicators of C.elegans reproductive ability after irradiation. The protective effect of polydatin on radiation–induced reproductive injury was verified by detecting apoptosis-related genes and reproduction-related genes. It is hoped that our research will provide scientific basis for exploring the feasibility of polydatin as a potential new radioprotective agent against reproductive injury.
Materials and Methods
Reagents and Instruments
Motic SMZ-168 body microscope: MacDiary Industrial Group Co., Ltd.; SHP-150 biochemical incubator: Shanghai Jinghong Experimental Equipment Co., Ltd.; Fluorescence microscope: Shenzhen Mingmei Microscope Co., Ltd; FV1200 scanning laser confocal microscope: Olympus, Japan; Gradient PCR instrument: Eppendorf, Germany; PRISM 7300 fluorescent quantitative PCR instrument; Meilincell; ROS reactive oxygen species detection kit (Tiandz); Acridine orange (AO) fluorescence staining kit (BBI); Cell Counting Kit-8 (CCK-8)(Meilun Cell); Reactive oxygen species Assay Kit(Nanjing Jiancheng Biological Engineering Research Institute Co., Ltd.); R333-01 Reverse Transcription Kit, Q711-02 SYBR green PCR Kit (Vazyme Biotech Co., Ltd); Annex V/FITC Cell Apoptosis Kit, Trypsin (Meilun Cell); Primary anti-β-Tubulin antibody (Beyotime); Primary anti-Actin antibody (Beyotime); these two proteins was used as an internal control. Primary anti-Trp53 antibody (Abcam); Primary anti-BH3(Noxa) antibody (Abcam); Primary anti-Ppp1ca antibody (Abcam); Primary anti-Tnp2 antibody (Affinity); Preparation of related solutions: 50mg/mL polydatin storage working solution: sterilized double distilled water and 37°C water bath in advance, 50mg polydatin was weighed and dissolved in 1 mL of 37°C ddH2O, and stored at 4°C in the dark.
Cell Culture
BALB/c mouse sperm cell-derived cell line (GC-1) and cell-specific medium CM-0600 were purchased from Pro cell Life Technology Co., Ltd. of Wuhan, China. Culture at 37°C and 5% CO2. For the purpose of each experiment, the number of cells was determined.
C.elegans Culture and Grouping
The wild-type N2 C.elegans worms and Escherichia coli OP50 (E. coli OP50) were kindly provided by Prof. Huimin Zhang, from the School of Basic Medicine and Life Sciences, Soochow University. The worms were cultured on NGM agar medium containing E.coli OP50 at 20°C. 23 According to the irradiation dose, C.elegans were divided into two groups: 0Gy (control group) and 50Gy group. Twenty C.elegans were observed and recorded in each group.
Synchronization of the Worms
Adult C.elegans worms were collected using M9 buffer and incubated in a bleach solution to synchronize them at the L1 stage as described by Shen et al. 24 After 28 h of culture at 20°C, these worms entered the L4 stage. For the synchronization of the descendant worms, the female worms in the oviposition period were transferred onto new NGM culture dishes for 2 h after oviposition, and then cultured at 20°C for 37 h to enter the L4 phase.
Irradiation Conditions
L4-stage C.elegans worms were irradiated with 50 Gy of radiation at a rate of 25 Gy/min at 20°C using a 60Co-γ irradiation device from the State Key Laboratory of Radiation Medicine and Protection and School of Radiation Medicine and Protection, Soochow University. When the cells reached the logarithmic phase, GC-1 cells were treated with 1, 2, 3, and 4 Gy biological X-ray machines (Rad source, Inc.). Under sterile conditions, the appropriate dose was screened at the dose rate of 1 Gy/min.
Drug Intervention
GC-1 cells and C.elegans intervention of resveratrol glycoside is to add sterile resveratrol glycoside to the culture medium in the pre-irradiation stage. 25 The drug concentrations were set to 0 μM (control group), 50 μM, 100 μM, and 200 μM, respectively. The control group was replaced by normal saline with the same dose.
Assessment of C.elegans Life Expectancy
Measurement life reference 26. After the intervention of resveratrol glucoside, they were transferred to new plates, each of which was 10. The target C.elegans will be picked in new incubators every day to prevent the influence of newborn larvae, until all C.elegans are observed to die based on no response to contacting C.elegans with the picker. 50 C.elegans were observed in each group.
Determination of the Number of Offspring
The worms were collected and washed with M9 buffer after treatment with polydatin. Each worm was transferred onto a new OMG plate containing OP50 and cultured at 20°C, renewing the plates every 24 h. All eggs and larvae on the plates were counted. The number of unhatched eggs was counted on the next day. After oviposition, the numbers of total and hatched eggs were counted to determine the hatching rate as calculated by the ratio of the number of live eggs to the total number of the eggs laid by each worm. Thirty worms per group were evaluated.
ROS Level Detection
After the cells in each group were treated and cultured to about 80% of the cell density, DCFH-DA staining solution was added according to the instructions of ROS detection kit, and incubated for 30 minutes in an incubator at 37°C. Five 40-fold fields of vision were randomly photographed and recorded under the fluorescence microscope. Fluorescence intensity was quantitatively analyzed by Image J. The experiment was repeated three times.
After irradiation, the warms were washed with M9 buffer and placed in a 1.5-mL Eppendorf tube. They were stained with 10 μM H2DCFDA in 1 mL of M9 buffer for 30 min at 20°C. Subsequently, they were washed 2–3 times with M9 buffer to reduce the background in fluorescence microscopy. Next, the worms were placed onto a glass slide with 2% agarose gel, anesthetized with 30 μL of levamisole, and covered with a cover glass. Images were captured with a fluorescence microscope at an excitation wavelength of 488 nm and a blocking wavelength of 515 nm. Thirty worms per group were examined.
Determination of Cells Apoptosis
GC-1 cells in logarithmic phase were inoculated into 6-well plates at 2 mL per well (2 × 106 cells/mL). The AnnexinV-FITC kit was used to detect apoptosis. Single staining of PI and accessories was carried out in V-FITC and incubated for 5 minutes in the dark at room temperature with PI and accessories in V-FITC as controls. Within 1 hour, the fluorescence intensity was detected by flow cytometry, and the experiment was repeated three times to analyze the apoptosis.
The method of Kelly et al. 27 was used to measure the apoptosis of germ cells in C.elegans. Briefly, irradiated worms were washed with M9 buffer, placed in a 1.5-mL Eppendorf tube, and centrifuged at 1500 rpm for 2 min, and the supernatant was discarded. The worms were stained with a 100-μL staining solution containing 25 μg/mL acridine orange and incubated with 5 μL of OP50 for 1 h in the dark. They were then transferred to NGM medium without food for 2 h. Next, the worms were placed onto a glass slide and examined under a fluorescent microscope as indicated above. Thirty worms per group were evaluated.
Transcriptomics Sequencing and Bioinformatics Analysis
We selected C.elegans with radiation-induced reproductive defects and C.elegans in Adult phase control group of the same age after 60Go-γ irradiation with 100Gy for RNA-seq analysis by Shanghai Europe Easy Co., Ltd. to obtain a transcriptomics sequencing. In addition, the gene expression profile of C.elegans GSE122996 after 200Gy irradiation was extracted from the GEO database, and the differentially expressed genes were analyzed by online GEO2R. The screening criteria of fold change>2.0 and P-value<.05 were adopted for both transcriptome and GEO datasets. Using David online analysis tool (https://David.ncifcrf.gov/home.jsp) for KEGG analysis and GO analysis of Differentialy Expressed Genes(DEGs) appeared in the two groups of data were conducted.
Screening of Hub Gene
A online database STRING (https://string-db.org/) was selected to analyze protein–protein interaction (PPI) in DEGs, visualize PPI networks and identify hub genes in differential DEGs. The MCC method in CytoHubba, a plug-in Cytoscape, was used to screen hub genes in TOP 40, and the important gene modules were classified according to the MCODE plug-in. According to the default parameters,28,29 the pivotal genes in modules with high scores were screened out. The combination of genes obtained by the two methods laid a foundation for further experimental verification.
Quantitative Real-Time Polymerase Chain Reaction (qRT-PCR)
The total RNA in GC-1 cells and C.elegans was extracted by Trizol, and the cDNA was obtained by reverse transcription. After the Real-time PCR reaction system was prepared, the amplification reaction was performed on a PCR instrument. The relative multiple ratio of mRNA expression in the experimental group and the control group was expressed as Folds=2-△△Ct. The experiment was repeated three times, and the average value was calculated. See Supplement Table S1 for primers.
Western Blot
Cells in each group were collected after treatment under different conditions. Subsequently, the cells were lysed on ice with RIPA lysis buffer for 30min. The lysates were centrifuged at 1200r/min for 5 min at 4°C, the supernatant was collected, and protein was quantified by BCA method. Then denatured with loading buffer at 100°C, for 10 min with 10μg per lane for SDS-PAGE. The isolated protein was transferred to PVDF membrane within 120 min at a constant voltage of 300mA. 5% skim milk was used to block non-specific binding at room temperature. After 1.5 h, the membranes were sequentially exposed to primary (4°C, overnight) and secondary (37°C, 1.5 h). ECL solutions were prepared to form protein bands and the chemiluminescent imaging system was operated to capture images. The differences of protein expression were analyzed by image J.
Statistical Analysis
Measurement data in mean ± standard deviation were analyzed on SPSS 20.0 statistical software(IBM). For statistical analysis between two independent samples, independent sample t test(t′ test used in heterogeneity of variance), one-way ANOVA test for multi-group results and LSD-t test for inter-group significance analysis are used. P < .05 indicated that the difference was statistically significant. Each experiment was repeated at least three times.
Results
Effect of different radiation doses and concentrations of polydatin on proliferation of GC-1 cells
From the results, we can see that when the dose of X-ray reaches 2Gy, the proliferation of GC-1 cells is significantly affected, and it is statistically significant. In later experiments, 2Gy was selected as the irradiation dose of GC-1 cells, as shown in Figure 1A. Different concentrations of polydatin enhanced the proliferation of GC-1 cells. When the concentration needed to reach 100μM, the cell proliferation was statistically significant with the control group. In the later experiments, 100μM polydatin was used as the interference concentration of GC-1 cells, as shown in Figure 1B. Screening of radiation dose and resveratrol glycoside concentration. A: The Effects of different doses of X-ray on the proliferation of GC-1 cells; B: Effects of different concentrations of polydatin on GC-1 cells proliferation. * indicates that the data were statistically significant (P < .05) compared with the control group.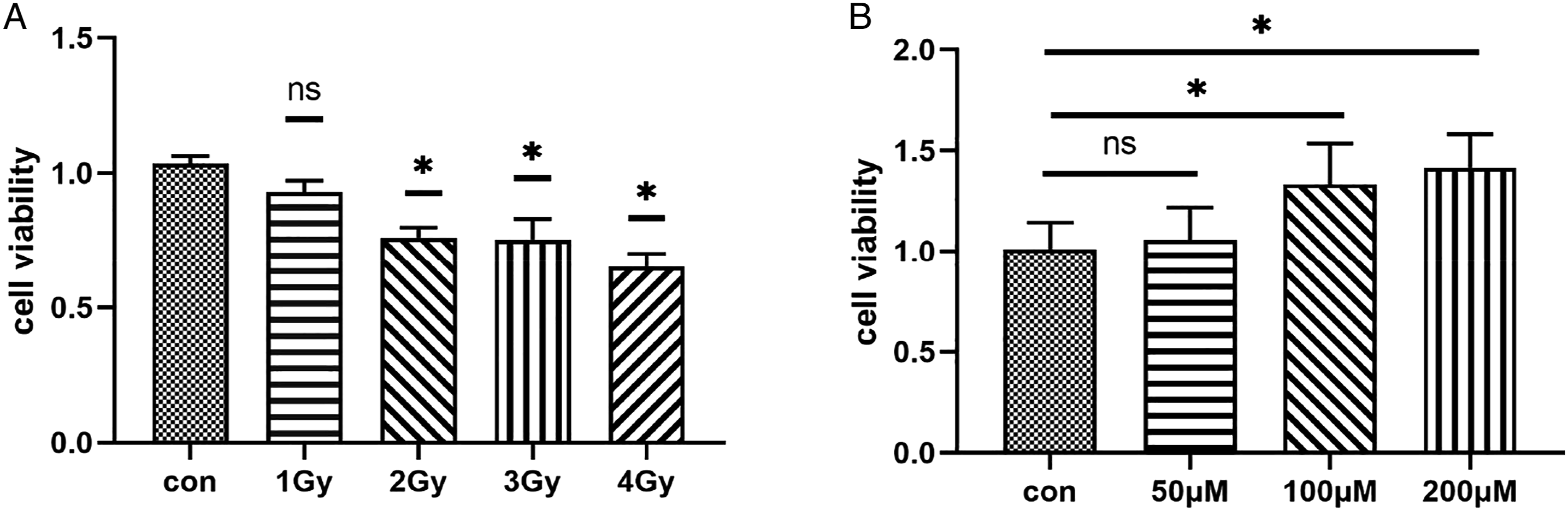
Effects of polydatin on ROS levels in GC-1 cells induced by ionizing radiation.
Under the fluorescence microscope, it was observed that the control group had the lowest ROS level of GC-1 cells. The fluorescence of irradiated cells treated with 100 μ M polydatin was not significantly different from that in the control group, but the fluorescence of cells in the simple irradiation group was stronger, shown in Figure 2A. Quantitative analysis of the fluorescence value of the photos by Image J revealed that there was no statistical difference in fluorescence intensity between the polydatin intervention group and the control group. The fluorescence intensity of pure irradiation group was higher than that of the control group, and the difference was statistically significant. Compared with the pure irradiation group, the fluorescence intensity of polydatin intervention group decreased, and the difference was statistically significant. The results showed that 100μM polydatin could significantly reduce the level of ROS in GC-1 cells induced by X-ray, shown in Figure 2C. Effects of polydatin on the level of ROS and apoptosis of GC-1 cells. A: Different groups of ROS fluorescence detection; B: Detection of apoptosis in different groups; C: Quantitative analysis of ROS level in each group; D: Quantitative analysis of apoptosis level in each group. *indicated that the difference was statistically significant (P < .05).
Effects of Polydatin on GC-1 Cells Apoptosis Induced by Ionizing Radiation
Flow cytometry was used to detect GC-1 cells apoptosis in each group. The apoptosis rate of GC-1 cells in the control group was the lowest and that in the irradiation group was the highest. The apoptosis rate of GC-1 cells in the intervention group of 100μM polydatin was between the two groups, shown in Figure 2B. Quantitative analysis of apoptosis rate by flow cytometry showed that the apoptosis rate of polydatin intervention group was significantly different from that of control group (P < .05). The apoptosis rate of irradiated group was higher than that of control group, and the difference was statistically significant (P < .05). There was a statistically significant difference in the apoptosis rate between the resveratrol intervention group and the radiation group (P <.05). The results showed that 100μM polydatin could significantly reduce X-ray–induced GC-1 cells apoptosis, shown in Figure 2D.
Effects of Different Concentrations of Polydatin on the Life Span of C.elegans
The lifespan of C.elegans is an important indicator for toxicity assessment.
30
As shown in Figure 1, polydatin exerted no suppressive effect on the lifespan of C.elegans. In contrast, 100 μM of polydatin significantly prolonged the lifespan compared with the control (P < .05). No significant difference was observed between the other groups and the control. Therefore, our results showed that 100 μM of polydatin could prolong the lifespan of C.elegans without any marked toxic effect, shown in Figure 3A. Intervention results of polydatin. A: Effect of different concentrations of polydatin on the lifespan of C.elegans. B: Effect of different conc entrations of polydatin on the C.elegans brood size at different concentrations. C: The ratio of incubation at different concentrations. D: Effect of polydatin on the brood size of C.elegans exposed to 60Co γ radiation. E: The ratio of the egg incubation periods. *indicated that the difference was statistically significant, P<.05.
Effects of Different Concentrations of Polydatin on the Reproductive Ability of C.elegans
As shown in Figure 3B, polydatin at 50 μM or 100 μM had no effect on the number of eggs laid compared with that in the control group. However, at the high dose of 200 μM, polydatin significantly decreased the number of eggs laid compared with the control (P < .05). As shown in Figure 3C, the hatching rate was unaffected when the worms were exposed to polydatin even at 200 μM, suggesting that although 200 μM polydatin decreased the oviposition ability, it had no effect on the survival rate of the embryos. Polydatin at 100 μM had no effect on C.elegans reproductive capacity, including oviposition and embryonic survival.
Polydatin Improves 60Coγ-Induced Reproductive Damage of C.elegans
Since polydatin prolonged C.elegans lifespan with no toxic or suppressive effect at 100 μM, 100 μM of polydatin was used for subsequent experiments. The effects of polydatin on the reproductive ability of C.elegans after irradiation are shown in Fig3. D,E. Compared with the control, 60Co γ irradiation significantly reduced the oviposition and hatching rates of C.elegans (P<.05). However, polydatin treatment significantly increased the post-irradiation oviposition and hatching rates of C.elegans (P<.05).
Polydatin significantly reduced the 60Coγ-induced elevation of ROS levels.
Quantitative analysis of the fluorescence intensity using Image J showed that there was no statistically significant difference in the fluorescence intensity between the 60Co γ-irradiated group treated with 100 μM polydatin and the control group (P > .05). The fluorescence intensity of the irradiated worms was significantly higher than that of the control group (P < .05). However, polydatin treatment significantly decreased the fluorescence intensity of the irradiated worms (P < .05). These results suggested that 100 μM of polydatin significantly reduced 60Co γ-induced elevation of ROS levels in C.elegans, as shown in Figure 4A, B. Effect of polydatin on 60Co γ-induced elevation of ROS levels in C.elegans. A: Fluorescent images show the ROS level. B: Fluorescence intensit y of cells was determined via ROS assay. Effect of polydatin on 60Co γ radiation-induced C.elegans germ cell apoptosis. C:Green fluorescent staining. Arrows show the green pellets of apoptotic cells. D:The number of apoptotic cells. *indicated that the difference was statistically significant, P < .05.
Polydatin Significantly Reduces the Number of Radiation-Induced Apoptosis of Germ Cells in C.elegans
C.elegans germ cell apoptosis following 60Co γ irradiation was evaluated through laser confocal microscopy. As shown in Figure 2C, green cells indicated by the arrow are apoptotic cells. There was no significant difference in the number of apoptotic germ cells between 100 μM polydatin-treated and control groups(P > .05) The apoptosis of germ cells was significantly induced by 60Co γ irradiation compared with the control (P < .05). However, polydatin treatment reduced the number of radiation-induced apoptotic cells (P < .05). These results suggested that 100 μM of polydatin significantly reduced the 60Co γ-induced germ cell apoptosis in C.elegans. as shown in Figure 4C, D.
Bioinformatics Analysis Results
The results of transcriptomics sequencing and the GSE122996 differential gene by combining 60Co γ-ray induced reproductive damage in C.elegans are presented in Figure 5A, B. A total of 258 common differential genes were obtained, as shown in Figure 5C. To analyze the biological action and potential function of DEGs, GO analysis including biological process (BP), molecular function (MF), and cellular component (CC) and KEGG signaling pathway enrichment were performed, as shown in Figure 5D. For MF, DEGs was mainly enriched in Regulation of oligodendrocyte Proliferative Growth, tissue morphogenesis, killing of cells of other organism, metal ion transport, and positive regulation of adenosine receptor signaling pathway. For MF, DEGs is mainly enriched in the Thiamine Phosphate Phosphatase Activity, Lysophosphatidic Acid Phosphate Activity, delta-catenin binding, Sodium: Potassium-Exchanging ATPase Activity, and gamma-catenin binding. For CC, DEGs were mainly enriched in perineuronal net, pseudopodium, collagen type VI trimer, endopodestase Clp complex, and Mitochondrial endopodestase Clp complex; Enrichment analysis of KEGG signaling pathway showed that DEGs was mainly enriched in non-homologous end-joining, protein export, homologous reconstitution, oxidative phosphorylation, and metabolism of xenobiotics by cytochrome P450, as shown in Figure 5E. Analysis of all the statistically different apoptosis-related pathways shows that the pathway of apoptosis process, apoptotic mitochondrial changes and oviposition, and actin filament depolymerizationis were closely related to egl-1. cep-1 that participates in the intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator. These two genes were associated with ionizing radiation-induced apoptosis, egl-1 was homologous to the BH3(Noxa) gene in human and mouse, cep-1 to human TP53 and mouse Trp53. The pathways involved by these two genes are shown in Supplement Table S2. Bioinformatics Analysis Results.A: Volcano map of transcriptomics results; B: Volcano map of GSE122996; C: venn diagram of two group; D: GO analysis; E: KEGG pathway analysis; F: The MCODE plug-in analysis; G: The cytohubba plug-in analysis.
Screening of Hub Gene for Reproductive Damage Induced by Ionizing Radiation
The first 40 hub genes were obtained by using cytohHubba plug-in cytoscape3.8.2 software, and the functional modules were screened by MCODE plug-in to select the first module with the highest score. A total of 38 genes were detected by these two methods. Screening of these genes shows that 10 genes belong to MSP sperm major protein family, which includes sperm formation related genes gsp-3 and glf-1, sperm specificity family ssh-1, and cell cycle related gene cyc-2.2, as shown in Figure 5F, G. In order to screen out the hub genes that can be used as biomarkers, in this study, we selected 38 genes with high scores in transcriptome and significant multiple changes. We screened ssh-1, a sperm-specific family gene, and gsp-3, a gene associated with spermatogenesis and meiosis. ssq-1 is closely related to the genes of heat shock protein family A, of which heat shock protein HSPA2 (a gene homologous to mouse Hspa2) is an important biomarker for the detection of male infertility. 31 gsp-3 is homologous to human and mouse PPP1CA(Ppp1ca), and serine/threonine protein phosphatase (PPPs) is involved in the regulation of sperm overactivity. Therefore, these two hub genes were selected as biomarkers to detect reproductive damage induced by ionizing radiation. Pathways for msp-10 and gsp-3 are shown in Supplement Table S3.
Effects of Polydatin on Expression of Regulatory Genes Related to Apoptosis and Reproduction by Ionizing Radiation
The effects of Polygonum cuspidatum on apoptosis regulation and reproductive regulation genes of GC-1 cells after 2 Gy X-ray irradiation are as follows. In the mRNA level analysis results of apoptosis genes BH3 and Trp53, these two apoptosis genes were up-regulated in the radiation group, which was statistically different from the control group. Compared with the control group, the two genes in resveratrol intervention group were up-regulated, but down-regulated compared with the irradiation group alone, with statistical significance, as shown in Fig. A, B. The mRNA level analysis of reproductive related genes Ppp1ca and Tnp2 showed that the expression of Ppp1ca was down-regulated and that of Tnp2 was up-regulated in the irradiation group alone and polydatin intervention group could obviously reverse this phenomenon, and there was no significant difference compared with the control group. The results showed that 100μM polydatin could reduce the damage of X-ray to GC-1 cells, as shown in Figure 6C, D. Expression of regulatory genes related to apoptosis regulation and reproduction. A: Expression of BH3. B: Expression of Trp53. C: Expression of Ppp1ca. D: Expression of Tnp2. E: Expression of egl-1. F: Expression of cep-1.G: Expression of gsp-3. H: Expression of msp-10. *P < .05 indicated statistically significant between-group comparisons.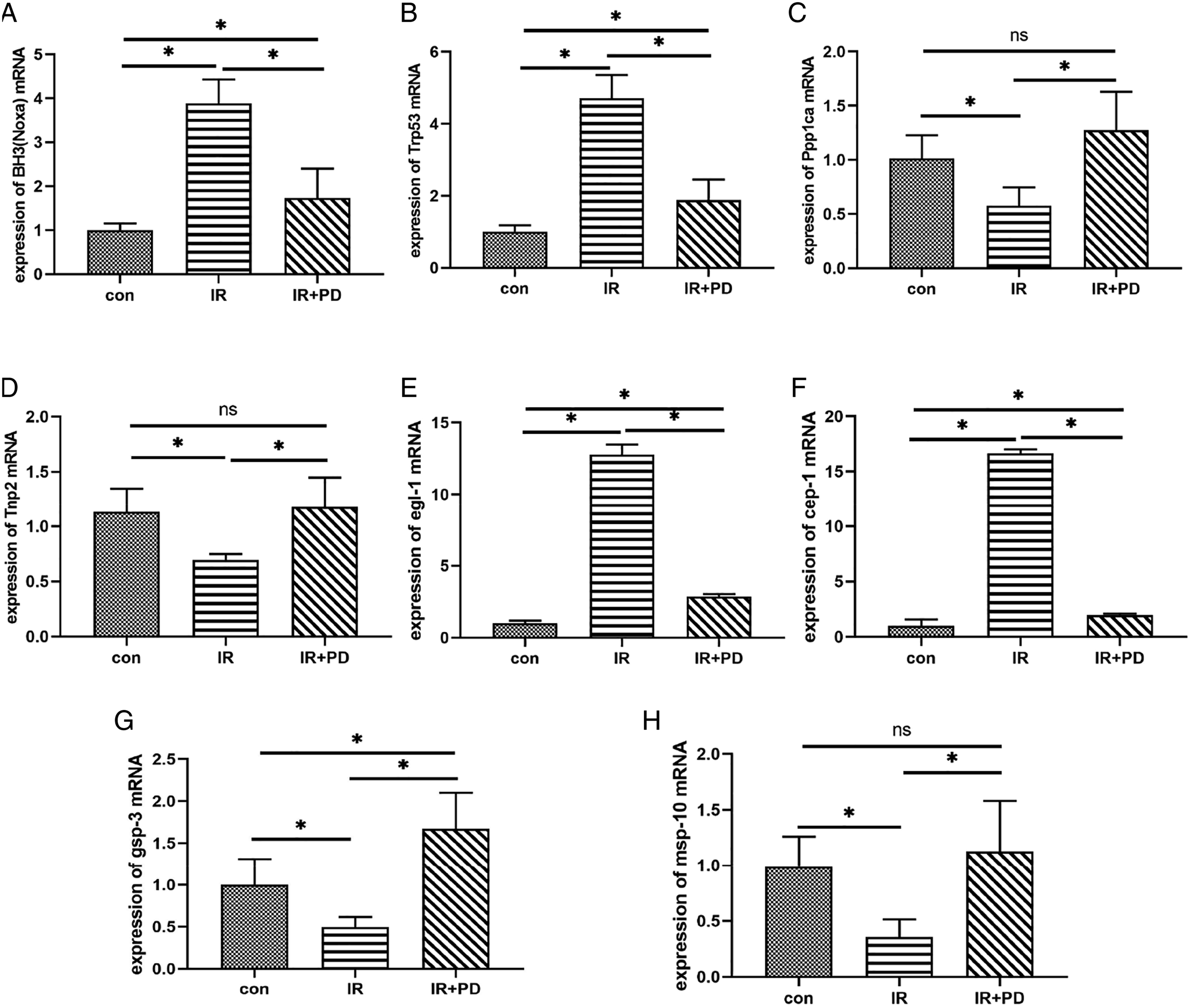
As shown in Figure 6E and F, in the influence of polydatin on apoptosis regulation and reproductive related regulation genes of C.elegans induced by 60Co γ-ray, the mRNA expressions of apoptosis regulation genes egl-1 and cep-1 in the irradiation group were significantly higher than that in the control group. The mRNA levels of reproduction-related genes gsp-3 and msp-10 were analyzed and the results were consistent with those of X-ray irradiated GC-1 cells. Resveratrol intervention group could significantly down-regulate the mRNA levels of the reproductive related genes gsp-3 and msp-10 induced by ionizing radiation. These differences are statistically significant (Figure 6G, H). The results suggested that 100 μM polydatin could significantly improve the 60Co γ -ray induced apoptosis regulation of C.elegans and the expression of reproductive-related regulatory genes and reduce the apoptosis of C.elegans germ cells.
Effect of Polydatin on Apoptosis of GC-1 Induced by Ionizing Radiation and Expression of Reproduction-Related Proteins
In the detection of protein expression level of Ppp1ca, a reproductive-related gene after polydatin interference, the protein level showed an upward trend compared with the control group and a downward trend compared with the X-ray irradiation group, with statistical differences. All other results are consistent with the effect of polydatin on the apoptosis and reproductive regulation genes of GC-1 after 2 Gy X-ray treatment. It was verified from the protein level that 100μm polydatin could reduce the damage of GC-1 induced by X-ray as shown in Figure 7C, D. Expression of regulatory proteins related to apoptosis regulation and reproduction. A: Expression of different proteins in GC-1 cells in different groups by western blot analysis. B: Comparison of expression levels of different proteins in each group. *P < .05 indicated statistically significant between-group comparisons.
Discussion
The damage of ionizing radiation to the reproductive system has always been a matter of great concern to people, but the existing radiation protective agents also have some defects. In recent years, researchers have been looking for new anti-radiation drugs, hoping to make up for the shortage of the current anti-radiation drugs. Polydatin is one of the first innovative antioxidant drugs submitted for clinical research in the United States and China, and it has great application potential in radiation-resistant injury and the treatment of such injury. 32 Study have shown that polydatin not only has the effect of lowering blood lipid, 33 lowering blood sugar, 34 and anti-inflammation, 35 but also can effectively prevent oxidative stress, reduce ROS level, increase SOD level, and prevent cell apoptosis caused by oxidative stress.36,37 Cao et al. 38 found that polydatin can inhibit epithelial–mesenchymal transition through TGF-β1/Smad3 signaling pathway and reduce radiation–induced pulmonary fibrosis. Li et al. 39 Study have shown that polydatin can increase the microvessel density of the small intestine after ionizing radiation, reduce the apoptosis of intestinal microvascular endothelial cells, and promote the growth and cell repair of intestinal crypts. In addition, polydatin was also effective in protecting the reproductive system from radiation-induced damage, such as infertility and sperm dysfunction, mediated by down-regulation of malondialdehyde and 8- hydroxydeoxyguanosine. 21 At the same time, polydatin can also reduce the damage to the testis of mice caused by ionizing radiation by inhibiting the ratio of apoptosis-related protein Bcl-2/Bax. 40 All the above studies have shown that polydatin has a significant radiation protection effect, but for reproductive damage induced by ionizing radiation, more experimental studies are still needed to demonstrate its effective dose and action mechanism.
GC-1 cells used in this study are currently an important cell for reproductive research and have a high sensitivity to ionizing radiation. 41 And is useful in the treatment of mouse germ cell apoptosis caused by heavy ion radiation induced DNA damage. 42 And hMSH5 C85T polymorphisms play an important role in differential studies of sperm production abnormalities induced by ionizing radiation. 43 The germ cells of C.elegans are also very sensitive to ionizing radiation and are widely used to study radiation-induced germ cell apoptosis. 44 We have previously demonstrated that 50 Gy of 60Coγradiation induces significant damage to the reproductive system of C.elegans, which is more pronounced at doses up to 100 Gy, 45 so in order to better study the genetic changes caused by ionizing radiation-inducedeproductive damage, we selected C.elegans that induced reproductive damage at the dose level of 100 Gy for transcriptomics sequencing and obtained the genes related to ionizing radiation–induced reproductive damage. At the same time, in order to verify the expression of our transcriptome sequencing results in C.elegans exposed to ionizing radiation without reproductive damage, we selected C.elegans with 200 Gy radiation for comparative analysis in GEO database, and found out the genes with common expression. Bioinformatics analysis was used to screen reproductive development-related pathways and apoptosis-related pathways, and hub gene is used to screen human and mouse genes for verification. It improves the scientificity and feasibility of using these genes to verify the protective effect of polydatin on the GC-1 cells damage and reproductive damage of C.elegans induced by ionizing radiation.
Polydatin can improve mouse follicular stability, telomerase activity and offspring survival rate. 46 In this study, we found that 100μM polydatin treatment greatly increased the hatch ability and embryo survival rate of C.elegans and improved the effect of 60Coγ-ray on their reproductive ability. These results preliminarily proved that polydatin had a protective effect on radiation–induced reproductive injury. Simultaneously, 100μM polydatin increased the lifespan of C.elegans. This result is consistent with the previous discovery that polydatin can prolong the life of mice and C.elegans.47,48 Ionizing radiation can affect the function of cells by increasing the level of ROS, and increasing the level of ROS will accelerate the apoptosis of cells, thus inducing diseases and even cancer.49-51 Polydatin can eliminate ROS in vivo, reduce insulin-like growth factor −1, increase insulin sensitivity and AMPK activity, increase the number of mitochondria, and improve motor function, with good effects. 52 In addition, many studies have shown that ionizing radiation can accelerate the production of ROS in germ cells, leading to the death and functional damage of germ cells.53,54 Our results demonstrated that polydatin significantly reduced the ROS levels in GC-1 cells and C.elegans induced by ionizing radiation, and decreased the rate of germ cell apoptosis. These results once again proved the protective effect of polydatin on radiation–induced reproductive damage.
It is well known that TP53 (p53) is a major gene regulating apoptosis, and BH3 is a priming factor for the apoptotic process. It has been found that in the irreversible DNA damage reaction, p53 promotes the BID nucleus output of BH 3-only to induce apoptosis, 55 and plays a key role in the selective attenuation of DNA damage–induced neuron apoptosis by mithramycin 56 and regulation of gingival protease–induced osteoblast apoptosis. 57 In addition, p53-BH3 pathway provides a new direction to promote apoptosis of ovarian cancer cells in gene therapy. 58 Horvitz 59 found that ionizing radiation induced apoptosis of C.elegans germ cells through the cep-1/egl-1 pathway, and these two genes were similar to p53 mediated BH3 domain protein found in mammals. 60 In addition, the bioinformatics analysis of the transcriptome sequencing results in this study also revealed that these two genes were involved in many ways of regulating apoptosis. The results of this experiment showed that polydatin could reduce the apoptosis of germ cells induced by ionizing radiation by regulating the expressions of Trp53 and BH3 in GC-1 cells and the expressions of cep-1 and egl-1 in C.elegans.
Msp-10, the major sperm protein gene of C.elegans, is a reproduction-related gene unique to this model organism, located on chromosome IV. It is found that sperm cells of nematodes lose actin and tubulin after meiosis, and then use the regulatory assembly and disassembly of the major sperm protein (MSP) to drive sperm movement, which plays an important role in inducing oocyte maturation. 61 And msp-10 may serve as a biomarker for detecting exposure to low dose ionizing radiation during nuclear waste disposal. 62 TNP2 is a unique gene of mammalian sperm development, located on chromosome 16. In the Tnp2-deficient mice, there were abnormalities in the sperm head, decreased motility, and affected the ability of the sperm to penetrate the zona pellucida of the egg cell in the acrosome reaction. 63 In addition, the mRNA level of TNP 2 in male smokers decreased, the motility of sperm decreased, and the morphology of sperm increased abnormally. 64
The gene gsp-3 related to spermatogenesis and meiosis is homologous to human PPP1CA (mice Ppp1ca), which is the catalytic subunit of phosphoprotein phosphatase 1(PP1) and has been proved to be a key regulator of sperm development and function. 65 Moreover, when the PPP1CC subunit is interfered with, the PPP1CA subunit can compensate for this effect, serving as an important pathway involved in sperm formation and a hot topic in the research on male contraceptive programs. 66 There are also PPP1CA, breast cancer, 67 lung cancer, 68 rectal cancer, 69 and others. At the same time, Wu et al. 70 proved that gsp-3 was necessary for the growth and sperm motility of C.elegans. In addition, gsp-3 is closely related to the meiosis of sperm, and can be combined with MEMI and participate in the fertilization process. 71 Our research shows that polydatin can reduce the damage of GC-1 cells by ionizing radiation by regulating the expressions of Tnp2 and Ppp1ca and reduce the damage of germ cells by ionizing radiation by regulating the expressions of gsp-3 and msp-10, thus playing an important role in sperm formation and germ cell development.
In conclusion, the radiation protection effects of 100μM polydatin on reproductive damage of GC-1 cells after irradiation with 2Gy X-ray and 50Gy 60Co γ-ray was confirmed in our research at the level of cell and model organism C.elegans. Reduce the apoptosis of germ cell by relieving the increase of ROS caused by ionizing radiation; The lifespan, reproductive ability and egg laying rate of C.elegans are detected, and the radiation protection effect of polydatin with the concentration is demonstrated. In addition, the expression levels of apoptosis-regulating genes BH3 (egl-1) and Trp53 (cep-1), the reproduction-related gene Ppp1ca (gsp-3), the mammalian spermatogenesis–specific gene Tnp2 and the C.elegans sperm major protein gene msp-10 were detected. The above results indicated that 100μM polydatin had a good protective effect on radiation-induced reproductive damage, which provided a scientific basis for future research. Therefore, research of polydatin as a new drug for resisting radiation–induced reproductive damage is still going on.
Supplemental Material
Supplemental Material - Protective Effects of Polydatin on Reproductive Injury Induced by Ionizing Radiation
Supplemental Material for Protective Effects of Polydatin on Reproductive Injury Induced by Ionizing Radiation by Jin Gao, Jincheng Qian, Nan Ma, Jianfang Han, Fengmei Cui, Na chen, and Yu Tu in Dose-Response
Supplemental Material
Supplemental Material - Protective Effects of Polydatin on Reproductive Injury Induced by Ionizing Radiation
Supplemental Material for Protective Effects of Polydatin on Reproductive Injury Induced by Ionizing Radiation by Jin Gao, Jincheng Qian, Nan Ma, Jianfang Han, Fengmei Cui, Na chen, and Yu Tu in Dose-Response
Supplemental Material
Supplemental Material - Protective Effects of Polydatin on Reproductive Injury Induced by Ionizing Radiation
Supplemental Material for Protective Effects of Polydatin on Reproductive Injury Induced by Ionizing Radiation by Jin Gao, Jincheng Qian, Nan Ma, Jianfang Han, Fengmei Cui, Na chen, and Yu Tu in Dose-Response
Footnotes
Author’s Note
Conceived and designed the experiment:Na Chen and Yu Tu. Performed the experiments: Fengmei Cui, Jincheng Qian, Nan Ma, Jianfang Han, Jin Gao. Analyed the data: Jianfang Han, Fengmei Cui, Jin Gao and Nan Ma. Wrote the paper: Jincheng Qian and Jin Gao.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (grant numbers 82003391, U186720), the Natural Science Research Projects of Colleges and Universities in Jiangsu Province (grant numbers 20KJB310007), the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), the Collaborative Innovation Center of Radiological Medicine of Jiangsu Higher Education Institutions, China and the Nuclear Energy Development Project, China (No. 2016-1295). We thanked Dr. Huimin Zhang for kindly donating the C.elegans and E. coli OP50. Thanks to my dear Duobao for her support.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
