Abstract
Objective
To evaluate the clinical efficacy and safety of baricitinib, a Janus kinase (JAK) inhibitor, in treating patient with progressing vitiligo, and to further explore the regulation of baricitinib on melanocytes (MCs) in vitro.
Methods
Four patients with progressing vitiligo were treated with oral baricitinib for a total of 12 weeks. MCs were cultured in vitro and irradiated by high-dose ultraviolet B (UVB, 150mJ/cm2) to make an MC damaged model (MC-Ds). Baricitinib was added at a final concentration of 25 μM. Dopamine staining and NaOH method were used to measure the tyrosinase activity and melanin level, respectively, real-time quantitative polymerase chain reaction (RT-qPCR) was used to measure the mRNA levels of tyrosinase (TYR), tyrosinase-related protein-1 (TRP-1).
Results
Significant re-pigmentation was observed in the week 12 without obvious side effects. Depigmentation occurred in 2 patients at the 3-month follow-up. Laboratory research found that higher doses of UVB irradiation (150mJ/cm2) could decrease melanin content of MCs, baricitinib (25 μM) could significantly promote tyrosinase activity, melanin content, and TYR, TRP-1 gene expression of MC-Ds.
Conclusion
Our preliminary study showed that baricitinib was effective and safe in treating progressing vitiligo. Baricitinib could promote tyrosinase activity, melanin content and TYR, TRP1 gene expression of MC-Ds in vitro.
Introduction
Vitiligo is an acquired, idiopathic autoimmune disorder characterized by depigmented patches in the skin, hair, or both. 1 The worldwide prevalence of vitiligo is approximately 0.5–2%, without obvious sex bias. 2 Although considered to be a benign disease that mainly affects the cosmetic appearance, vitiligo usually has a profound impact on patients’ quality of life and self-esteem, and may possibly predispose them to an increased risk of sunburn and skin cancer.3,4 The pathogenesis of vitiligo is multifactorial, combining genetic susceptibility, oxidative stress, metabolic disorder, and autoimmune response, among which the destruction of MCs via cell-mediated immunity plays a key role.5-8 Activated melanocyte-specific skin CXCR3+ CD8+ T-cells produce high levels of interferon gamma (INF-γ) and tumor necrosis factor alpha (TNF-α), which promote the detachment and apoptosis of MCs through E-cadherin disruption partly caused by increased matrix metalloproteinases 9 (MMP-9) produced by keratinocytes.9,10 Inhibition of MMP-9 or the IFN-γ signaling pathway mediated by the Janus kinase-signal transducer and activator of transcription (JAK-STAT) prevents MCs detaching from the basal layer in vitro and in vivo. 10
Studies showed that JAK inhibitors target the JAK/STAT signaling pathway and are now approved to treat many immune diseases. 11 In the treatment of vitiligo, a few published case reports, case series, and open-label studies have shown superior re-pigmentation rates in patients treated with JAK inhibitors, especially with concomitant ultraviolet or sun exposure.12-15 Given IFN-γ signaling is specially mediated by JAK1 and JAK2, baricitinib, a JAK1 and JAK2 inhibitor, has been supposed to be more effective than others in treating vitiligo. In a case report, a patient suffered from rheumatoid arthritis and vitiligo got much better re-pigmentation after substituting baricitinib for tofacitinib (a JAK1 and JAK3 inhibitor). 15
Vitiligo re-pigmentation is a complex process in which the MC damaged or depleted epidermis is repopulated by MC precursors which then proliferate, migrate and differentiate into mature MCs, which are the major source of melanin pigments. 16 Melanogenesis is modulated by a series of intracellular signaling pathways, the master regulator is microphthalmia-associated transcription factor (MITF), which controls three key melanogenesis genes expression of TYR, TRP-1, and tyrosinase-related protein-2 (TRP-2).17-19 TYR performs as the rate limiting activity in the melanin synthesis process. 17 The strongest stimulus for vitiligo re-pigmentation is UVB, several signal pathways and genes including TYR were significantly upregulated in the melanocyte samples of the UVB treated epidermis.16,18
In our study, we chose baricitinib as monotherapy and successfully treated 4 patients with progressing vitiligo without any severe side effects. As our previous study found that a high dose of UVB (150mJ/cm2) could cause damage to MCs with decreased melanin content 24 h after irradiation (data has not been published), we applied high dose UVB irradiation to make an MC damaged model (MC-Ds) to further explore the direct regulation effects of baricitinib on damaged MCs in vitro.
Materials and Methods
Clinical study of baricitinib for treatment of vitiligo
Four patients (1 female/3 males) with nonsegmental progressing vitiligo were treated by baricitinib from September 2020 to December 2021. They were all in good health otherwise, without any systemic infections (including viral, bacterial, fungal, and tuberculous), malignancies, or thrombosis. All of them had received systemic, topical, UV, or combined therapies before, such as systemic corticosteroid (TCS), traditional Chinese medicine, topical TCS, calcineurin inhibitor (TCI), or narrow band UVB exposure. None of them had favorable clinical results, and the disease continued progressing. They then commenced with oral baricitinib as monotherapy (4 mg daily during the first 4 weeks, then reduced to 2 mg daily for another 8 weeks for safety’s sake). Re-pigmentation, any uncomfortable feelings, and blood tests were recorded during the treatment periods. Before treatment, they were thoroughly informed about the benefits and possible side effects, such as infection, malignancies, cytopenia, dyslipidemia, thrombosis, and recurrence. Written informed consent was obtained from each patient. Patients’ age, disease duration, treatment histories, and lesions characteristics (including location, color, body surface area, vitiligo disease activity (VIDA) score, vitiligo area scoring index (VASI) 20 ) were recorded before treatment. Digital photographs were taken before treatment and at each subsequent follow-up visit (every 4 weeks at the first 3 months and then every 3 months for a total of 12 months) by the same camera and light source and from the same point of view. Blood tests were obtained before treatment, every 2 weeks in the first 4 weeks of treatment, every 4 weeks during the following 8 weeks, and 3 months after stopping treatment.
Baricitinib regulates MCs in vitro
Primary MC culture
Healthy adult male foreskin samples were soaked in 70% ethanol for disinfection, washed in phosphate-buffered saline (PBS) solution containing penicillin and streptomycin (Beyotime Biotechnology, China), and connective tissue and subcutaneous fat were removed. Skin samples were sterilized and rinsed in PBS, cut into 5 mm wide strips using a sharp scalpel, and treated with 0.25% trypsin (Gibco, USA) solution at 4°C overnight. The epidermis was gently separated from the dermis, and incubated in PBS solution by rotation and shaking on a shaker at room temperature to dissociate the cells. The cell suspension was washed with PBS and centrifuged. The collected cells were re-suspended with M254 medium (Gibco, USA) supplemented with 1% HMGS-2 (Gibco, USA) and 1% penicillin–streptomycin solution and then incubated at 37°C in 5% CO2; the medium was changed twice weekly. When cells were at 80% confluence, they were trypsinized for subculture.
L-DOPA staining for identification of MCs
M-254 medium was discarded, cells were washed 3 times with PBS for 5 min each time, and 4% paraformaldehyde was added to fix the cells for 30 min at about 25°C. The paraformaldehyde was then discarded. Cells were further washed 3 times with PBS for 5 min each, permeabilized with 0.3% TritonX-100 for 30 min, then washed 3 times with PBS for 5 min each time. Cells were then stained with 0.1% 3, 4-dihydroxy-L-phenylalanine (L-DOPA) buffer and incubated at 37°C for 4 h. After washing with PBS, MCs were identified under a microscope.
UV irradiation to make an MC-Ds model
MCs in the logarithmic growth phase were irradiated with UVB (311∼313 nm) lamp (model: SH4B-T UV, SIGMA, Shanghai, China), at a dose of 150 mJ/cm2 (according to our previous study, data has not been published). After UVB irradiation, MCs were cultured in an incubator at constant temperature of 37°C with 5% CO2 for 24 h to make an MC-Ds model.
Baricitinib treatment of MC-Ds
MC-Ds were treated with baricitinib at various final concentration of 0, 100 nM, or 25 μM for 48 h.
Measurement of tyrosinase activity
After treated with baricitinib, MC-Ds were seeded in a 96-well plate, 100 μL of 1%Triton X-100 solution was added to each well, and cells were shaken for 15 min. Then, 100 μL of 0.1% L-DOPA solution was added to each well, and the 96-well plates were incubated at 37°C for 2 h. Tyrosinase activity was evaluated by measuring the absorbance of L-DOPA at 492 nm (Multiskan MK3, Thermo Fisher Scientific, Waltham, MA).
Measurement of melanin content
After treated with baricitinib, MC-Ds were collected, washed twice with PBS, and mixed with 1 M NaOH. The mixture was seeded into 96-well plates at 100 μL per well and cells were incubated at 37°C for 1 h. The melanin content was evaluated by measuring the absorbance of the mixture at 492 nm (Multiskan MK3, Thermo Fisher Scientific). The inhibition rate of melanin synthesis was calculated as follows: [1-(absorbance value of drug well/cell density of drug well)/(absorbance value of control well/cell density of control well)] × 100%.
Real-time quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted with TRIzol reagent (Ambion, Austin, Tex) and cDNA was synthesized using PrimeScript II 1st Strand cDNA Synthesis Kit (TaKaRa, Kusatsu, Japan). RT-qPCR was performed using SYBR® Green Supermix (Bio-Rad Hercules, CA) in triplicate. All gene targets were normalized to β-actin (ACTB) mRNA by the 2–△△CT method. Primer sequences are as follows: Tyr, 5′-TTGTGAGCTTGCTGTGTCGT-3′ (forward), 5′-GTCAGGCTTTTTGGCCCTAC-3′ (reverse); TRP-1, 5′-GTCCCCCTGTTCCATTCAGG-3′ (forward), 5′-CCCACGGGAAAGTATGACCC-3′ (reverse); β-actin, 5′-ATCACCATTGGCAATGAGCG-3′ (forward), 5′-TTGAAGGTAGTTTCGTGGAT-3′ (reverse).
Statistical analysis
Statistical analysis and graphs were generated using GraphPad Prism 9.0 (GraphPad Software, San Diego, CA). Comparisons for two-group were carried out using Student’s t-test. Differences were considered statistically significant at P < 0.05.
Results
Baricitinib could promote re-pigmentation of vitiligo lesions
Patients’ demographic data and clinical outcomes.

VASI = ∑ Hand Units of all body sites × Residual Depigmentation

A 22-year-old young male with non-segmental progressing vitiligo on the trunk for 3 years. He was in good health otherwise. He had received systemic TCS (Compound betamethasone, intramuscular injection every 4 weeks) combined with topical chloroflumethasone twice daily for 3 months, without any re-pigmentation. He then commenced oral baricitinib as monotherapy. (A) Obvious re-pigmentation after 12 weeks of oral baricitinib application. (B) Quantitative analysis of melanin using Image Pro Plus 6 (**** means P < 0.0001).
MCs cultured and identified in vitro
Following culture, adherent MCs presented with two or more slender dendrites, with a fusiform shape or polygon shape. The cytoplasm and dendrite appeared brown or black (positive staining) with L-DOPA staining (see Figure 2). Primary MCs at (A) 7 d culture, (B) 14 d culture (magnification 100X), and (C)primary MCs identification: L-DOPA staining (magnification 200X).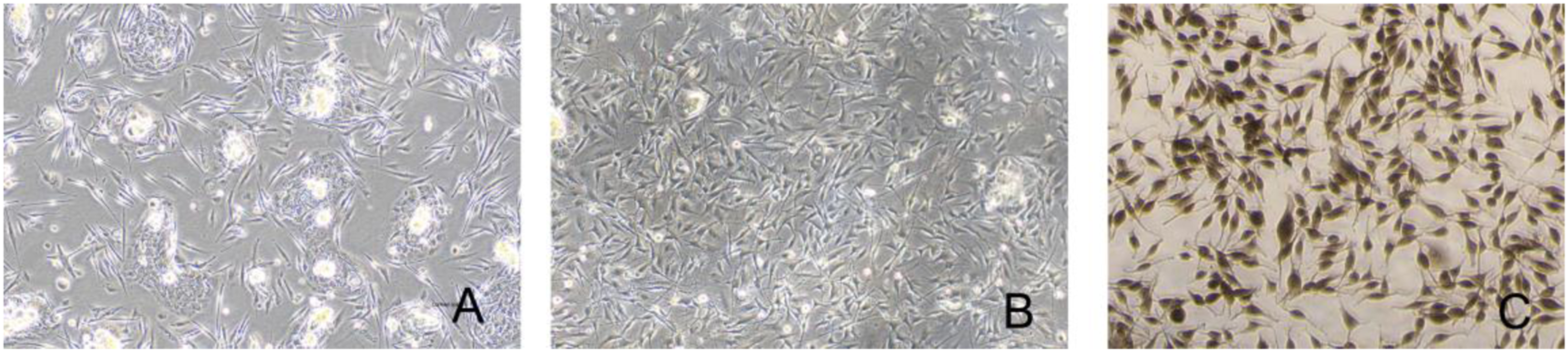
Baricitinib could promote tyrosinase activity and melanin content of MC-Ds
Our previous study showed that UVB irradiation at doses of 100, 150 and 200 mJ/cm2 could decrease melanin content of MCs (data has not been published). We chose 150 mJ/cm2 as the irradiation dose and 24 h as the irradiation time to make an MC-Ds model for the present study. We found that MC-Ds model had very weak L-DOPA staining. After adding baricitinib at a final concentration of 25 μM and incubated for 48 h, the color of the L-DOPA staining was much darker than that initially, indicating that baricitinib could significantly restore the tyrosinases activity of MC-Ds (P<0.05). Baricitinib concentrations of both 100 nM and 25 μM led to increases of melanin levels in MC-Ds by 1.3 and 1.7 times after 48 h, respectively. (P < 0.05, Figure 3) Baricitinib could promote tyrosinase activity and increase melanin content of MC-Ds (* means P<0.05 when compared with the untreated MC-Ds, # means P<0.05 when compared with 150mJ/cm2 UVB irradiation). (A) and (B) L-DOPA staining of MC-Ds and MC-Ds+b (baricitinib treated MC-Ds), respectively; (C) and (D) Tyrosinase activity and melanin content of MC-Ds and MC-Ds+b (baricitinib treated MC-Ds), respectively.
Baricitinib could upregulate TYR and TRP-1 gene expression in MC-Ds
Twenty-four hours after baricitinib treatment (25 μM), the expression levels of TYR and TRP-1 were significantly upregulated compared to untreated MC-Ds. (Figure 4) Baricitinib could upregulate TYR and TRP-1 gene expression of MC-Ds. (* means P < 0.05, **means P<0.01).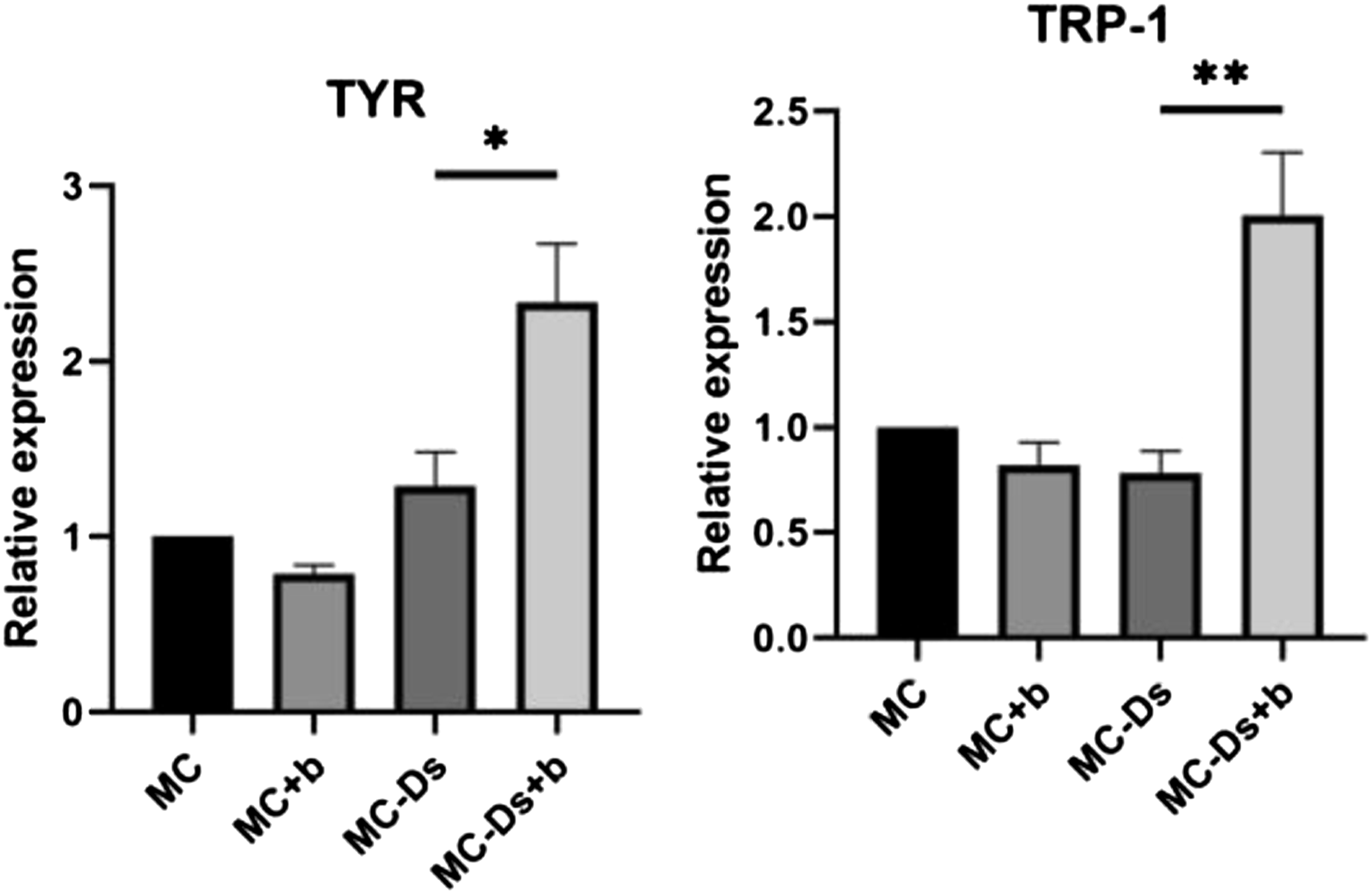
Discussion
Pathologic destruction of MCs and decrease of melanin synthesis are the major characteristic of vitiligo onset and development. Restoring the function of MCs has always been an emphasis of basic and clinical research of vitiligo to forestall and even reverse the course of the disease.8,21 Based on multiple studies conducted in vitiligo mouse models, the IFN--chemokine axis, together with its associated positive- feedback loop, has been identified as a potential pathway involved in the initiation and progression of vitiligo.22-24 A preliminary study on JAK inhibitors, which could block IFN- signaling, showed its effectiveness and safety in treating progressing vitiligo, especially when combined with low-dose UV therapy or sun-exposure.12,13,25 It was suggested that, wherein JAK inhibitors suppress T-cell mediators of vitiligo, light exposure is necessary to stimulate of MCs regeneration. However, there may be other mechanisms involved.
Baricitinib, a selective JAK1/2 inhibitor, is effective in treating patients with rheumatoid arthritis and is now widely used in dermatologic conditions such as alopecia areata, atopic dermatitis, and vitiligo.15,26-29 Small case studies have reported its effectiveness in treating vitiligo, and a new phase 2 study (NCT04822584) is currently being conducted to evaluate the safety and efficacy of the combination of baricitinib and phototherapy in vitiligo treatment.15,30 In this study, we treated 4 young patients with progressing vitiligo by using oral baricitinib monotherapy. Among them, 3 patients had vitiligous lesions on the face and neck, and 1 patient had vitiligous lesions on the trunk. All patients achieved favorable clinical results at the end of week 12, the VASI scores were significantly reduced, and the re-pigmentation rates were 59.26-74.17%. However, while statistical analysis was not performed in this patient group because of the small sample size, there was notable re-pigmentation potential with baricitinib, especially in the patients with vitiligous lesions on sun-exposure areas, which was consistent with the results of published literatures.15,31 All patients tolerated the treatment well without any severe side effects. The treatments stopped after 12 weeks because this was an off-label use of baricitinib.
We further explored the mechanism of baricitinib on the regulation of MC-Ds in vitro. As high-dose of UVB radiation reduces melanin synthesis in MCs and our previous study showed that the highest melanin content was observed at 24 h, we chose 150 mJ/cm2 and 24 h to make an MC-Ds model. By using this model, we observed the effect of baricitinib on tyrosinase activity and melanin content in vitro. We found that higher doses of UVB irradiation could decrease the melanin content of MCs. While baricitinib at 25 μM had no obvious effect on normal MCs (data not shown), it could significantly promote the tyrosinase activity and melanin content of MC-Ds, indicating that baricitinib could directly restore the tyrosinase activity and melanin synthesis ability of UV-damaged MCs. We also noted upregulation of TYR and TRP-1 in this process. As Tyrosinase is the key enzyme of melanin synthesis, and TRP-1 is an important antigen related to the cause of vitiligo, we thought their upregulation would be of great importance in the process of baricitinib regulating MC-Ds both in vivo and in vitro. However, the clinical efficacy and safety of baricitinib in treating progressing vitiligo as well as the exact mechanisms require longer-term studies with larger samples and further laboratory investigations.
Supplemental Material
Supplemental Material - Baricitinib is Effective in Treating Progressing Vitiligo in vivo and in vitro
Supplemental Material for Baricitinib is Effective in Treating Progressing Vitiligo in vivo and in vitro by Jie Dong, Xuan Huang, Li-Ping Ma, Fei Qi, Si-Nian Wang, Zi-Qin Zhang, Shi-Nan Wei, Ling Gao, and Fang Liu in Dose-Response
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
