Abstract
Deterministic particle transport codes usually take into account scattered photons with correct attenuation laws and application of buildup factor to incident beam. Transmission buildup factors for adipose, bone, muscle, and skin human tissues, as well as for various combinations of these media for point isotropic photon source with energies of .15, 1.5 and 15 MeV, for different thickness of layers, were carried out using Geant4 (version 10.5) simulation toolkit. Also, we performed the analysis of existing multilayered shield fitting models (Lin and Jiang, Kalos, Burke and Beck) of buildup factor and the proposition of a new model. We found that the model combining those of Burke and Beck, for low atomic number (Z) followed by high Z materials and Kalos 1 for high Z followed by low Z materials, accurately reproduces simulation results with approximated deviation of 3 ± 3%, 2 ± 2%, and 3 ± 2% for 2, 3, and 4 layers, respectively. Since buildup factors are the key parameter for point kernel calculations, a correct study can be of great interest to the large community of radiation physicists, in general, and to medical imaging and radiotreatment physicists, especially.
Introduction
Nowadays, the most commonly used cancer therapy is the radiotherapy modality, including many forms such as brachytherapy and gamma knife, with almost half of all patients receiving radiation as part of their treatment.1,2 From the many existing computational tools used for treatment protocol selection, we cite the Monte Carlo simulation technique3-8 and point-kernel method.9-11 The main difference between both techniques concerns the manner of particles transport handling, as 1 of them microscopically resolve the problem, whereas the second has a macroscopic approach. The point-kernel (PK) technique is based on the gamma radiation propagation assumed as beam-like and thus gaining time for decision-taking. From the important parameters forming a PK core, we refer to the buildup factor (BUF) quantifying the scattered to unscattered beam during particle–medium interaction.
Many existing tools have been used for BUF computations, such as PALLAS 12 and ASFIT, 13 other than Monte Carlo simulation programs (EGS4, 14 Geant4,15,16 and MCNPX 17 ). Moreover, from the many available modeling of BUF such as the invariant embedding method,18,19 the moments method, 20 and the iterative method, 21 we found the more sophistical one called the geometric progression (GP) fitting method by Harima et al.22-24 Also, as GP fitting methods, we found those provided by Kalos 25 and by Burke and Beck 26 used for monodirectional parallel-plane beam and stratified shields and the more recent one by Lin and Jiang 27 considering the case of isotropic point source and spherical layers. However, the last work does not investigate materials having close densities or atomic number, especially for human tissues and organs (as we can assume water medium for approximating the majority of them). Other fitting methods, which do not take into account the orientation of consecutive layers in terms of atomic number, 28 were not included in this work. It is the aim of this paper to propose a new empirical formula to reproduce buildup factors of stratified spherical shields for a point isotropic source so that it can be built into the presently widely applied point-kernel codes to resolve their deficiency in handling the buildup effect for stratified shields. Simple and easy application are the major concerns of the new empirical formula. Buildup factors of stratified shields are synthesized from those of the existing single-layered shields in a conventional form.
The main goal of this work was to propose a new modified model able to reproduce directly Geant4-based simulations of BUF for multilayered gamma-ray attenuators. Thus, we focused on the following 3 items: (i) Geant4 simulations; (ii) comparison against Lin and Jiang; Kalos and Burke and Beck models; and (iii) proposition of a modified fitting model. For that, 4 typically selected human tissues representing high, medium, and low atomic number (adipose, bone, muscle, and skin),were simulated for different configurations of isotropic point source with energies .15, 1.5, and 15MeV and 2, 3, and 4 layers as concentric spheres around the gamma-ray source. Moreover, an optimization procedure of the GP fitting parameters to calculate multilayer BUF from individual material BUF was proposed at the end of this work. However, our study was limited to small thickness (up to 8 mfp); the current work can be considered as a continuation on the attention made by radiation physicists, especially in terms of point kernel–based treatment planning and diagnosis imaging purposes.
Materials and Methods
Three isotropic point source energies were considered: .15, 1.5, and 15 MeV. Also, the same procedure for Zeq calculation, described in our previous work, 16 has been followed.
Briefly, such procedure was based on Log-Log interpolation in terms of elemental atomic number and Compton to total attenuation coefficient ratio search (μC/μT), for a given photon energy. Here, we will describe the Geant4-based simulation procedure carried out and enumerate the proposed parameterization (fitting) models of BUF for n concentric spherical layers including an isotropic point source at the center.
Simulation Procedure
Geant4 version 10.5 was used to mimic the transport of gamma rays isotropically from a point source through a given simple or group of media. The geometry of the problem, for monolayer cases, consists of a monoenergetic point source located at the center of ten concentric spheres, separated by 1 mfp for each step. For multilayer arrangements, we superposed consecutively spherical layers with specific thickness for each medium. We tracked the full history of gamma rays through the setup in order to compute the photon flux crossing each shell/surface of the medium. Then, each crossed photon energy has been internally (within SteppingAction class) converted into exposure. However, the BUF calculation needs the knowledge of transmission flux in the presence and in absence of the medium; we repeated computations for the air medium in place of each medium keeping the same setup characteristics (geometrical thickness). In this study, the buildup factor was computed for 109 gamma rays per run. We activated all the physical processes for electrons and photons. We used the G4EmStandardPhysics_option3 built-in physics library with a cutoff of 1 keV for electrons and photons. The overall statistical uncertainty does not exceed 1% in any case.
Proposed Parameterization Models
The general BUF formula for multilayers can be written in the following form: 1. Kalos
25
: 2. Burke and Beck
26
: 3. Lin and Jiang
27
: 4. M1: 5. M2: 6. M3: 7. M4: 8. M5: 9. M6:




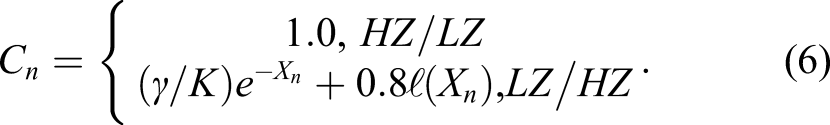



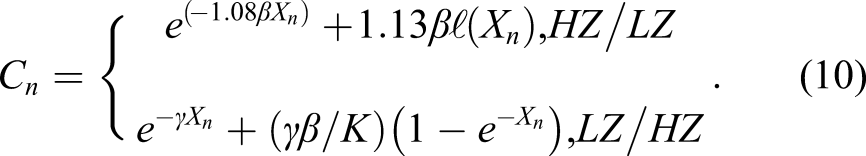



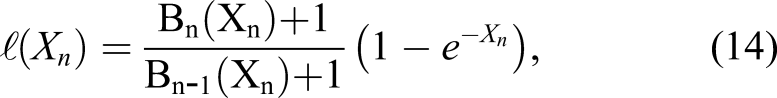
Results and Discussion
Monolayers and Bilayers Simulation
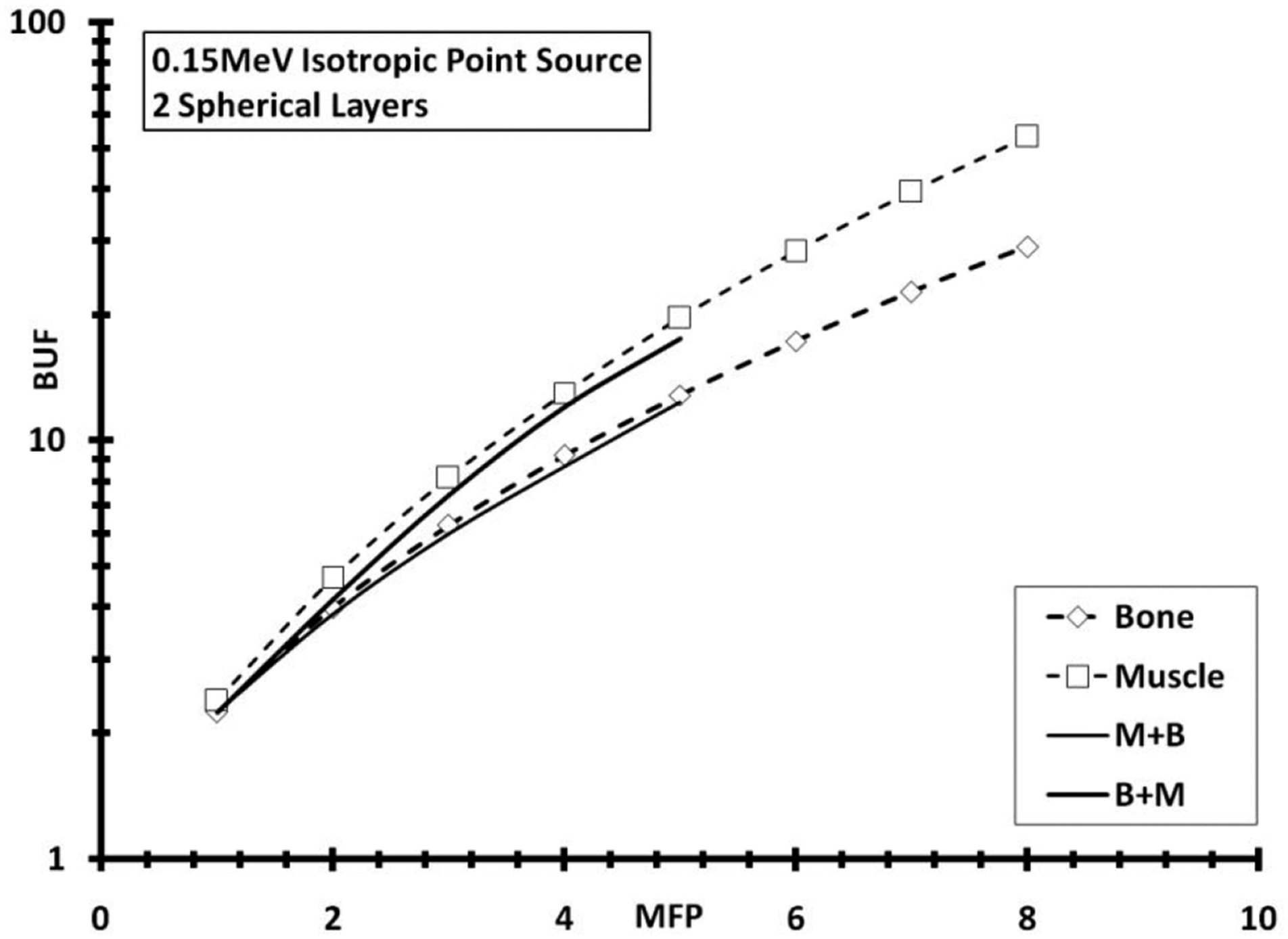
Geant4 simulated BUF for 2 spherical layers’ function of thickness (in mfp) for .15 MeV isotropic point source energy. Monolayer BUF was also plotted for bone (B) and muscle (M).

Geant4 simulated BUF for 2 spherical layers’ function of thickness (in mfp) for 1.5 MeV isotropic point source energy. Monolayer BUF was also plotted for bone (B) and muscle (M).

Geant4 simulated BUF for 2 spherical layers’ function of thickness (in mfp) for 15 MeV isotropic point source energy. Monolayer BUF was also plotted for bone (B) and muscle (M).
We confirmed previous explanation presented by Mann,28,31 illustrating that BUF for muscle was higher than that for bone tissue, due to the higher value of Compton mass attenuation coefficient μC and the lower value of Photoelectric and Pair Production mass attenuation coefficient μPP of muscle, as given in Table 1. This is due to the fact that Compton effect increases the scattering phenomena (described by BUF), whereas the absorption effect (Photoelectric + Pair Production) decreases it, which is clearly visible when going through the above cited figures. Moreover, combining muscle followed by bone tissues, in Figure 1, leads to lower BUF than for the reverse order of combination. This can be explained by the fact that a large number of scattered photons were produced within the first medium; however, they will be eliminated and absorbed by the second medium. On the other hand, we can see that BUF decreased, for a given thickness, when energy increased, as the ratio of Zeq (bone to muscle), as given in Table 1, decreased from 1.503 to 1.312 to 1.237, producing more and more closer BUF values. Also, BUF for 2 layers was found between both limits corresponding to BUF for single layers. Moreover, we clearly see a trend similarity of BUF for muscle followed by bone and adipose followed by skin, especially for deep locations where BUF values become larger.
Trilayer Simulation
Comparison of BUF Simulated Using Geant4 Against Other Model Data for 3 Layered Shields and 3 Photon Energies. LJ: Lin and Jiang, 27 K: Kalos, 25 BB: Burke and Beck, 26 M 1 -M 6 : 6 proposed models (A: Adipose, B: Bone, M: Muscle, S: Skin, 1B+1M+2A means 1 mfp B followed by 1 mfp M followed by 2 mfp A).


Geant4 simulated BUF for 3 spherical layers’ function of thickness (in mfp) for .15 MeV isotropic point source energy. Monolayer BUF was also plotted for adipose (A), bone (B), and muscle (M).
On the other hand, for .15 MeV photon energy, the arrangement of bone (Zeq = 11.530) followed by muscle (Zeq = 7.673) followed by adipose (Zeq = 6.486), we observed higher BUF values and rapid growth for deep penetrations than for the inverse order of arrangement, allowing us to reconfirm the hypothesis of applying the BUF function of bilayers to that of trilayers. Also, from Tables 2 and 3, for example, we can consider B+M+A arrangement as 2 layers with HZ followed by LZ, that is, B+M followed by A or B followed by M+A.
Multilayer Simulation
Also, Figure 5 and Table 4 show the middle location of global BUF between those for adipose and bone tissues. Moreover, previous interpretation (trilayer case) can be applied for actual situation. In order to optimize a best fitting model to reproduce data simulated with Geant4, the statistical analysis, in terms of average and standard deviation, of different models for 2, 3, and 4 layers in spherical geometry and isotropic point source, has been reported in Table 5. Geant4 simulated BUF for 4 spherical layers’ function of thickness (in mfp) for .15 MeV isotropic point source energy. Monolayer BUF was also plotted for adipose (A), bone (B), muscle (M), and skin (S). Comparison of BUF Simulated Using Geant4 Against Other Model Data for 4 Layered Shields and 3 Photon Energies: LJ: Lin and Jiang,
27
K: Kalos,
25
BB: Burke and Beck,
26
M
1
-M
6
: 6 Proposed Models (A: Adipose, B: Bone, M: Muscle, S: Skin, 1B+1M+2A+2S Means 1 mfp B Followed by 1 mfp M Followed by 2 mfp A Followed by 2 mfp S). Global Statistical Analysis (Average and Standard Deviation) of Different Models for 2, 3, and 4 Layers in Spherical Geometry and Isotropic Point Source.
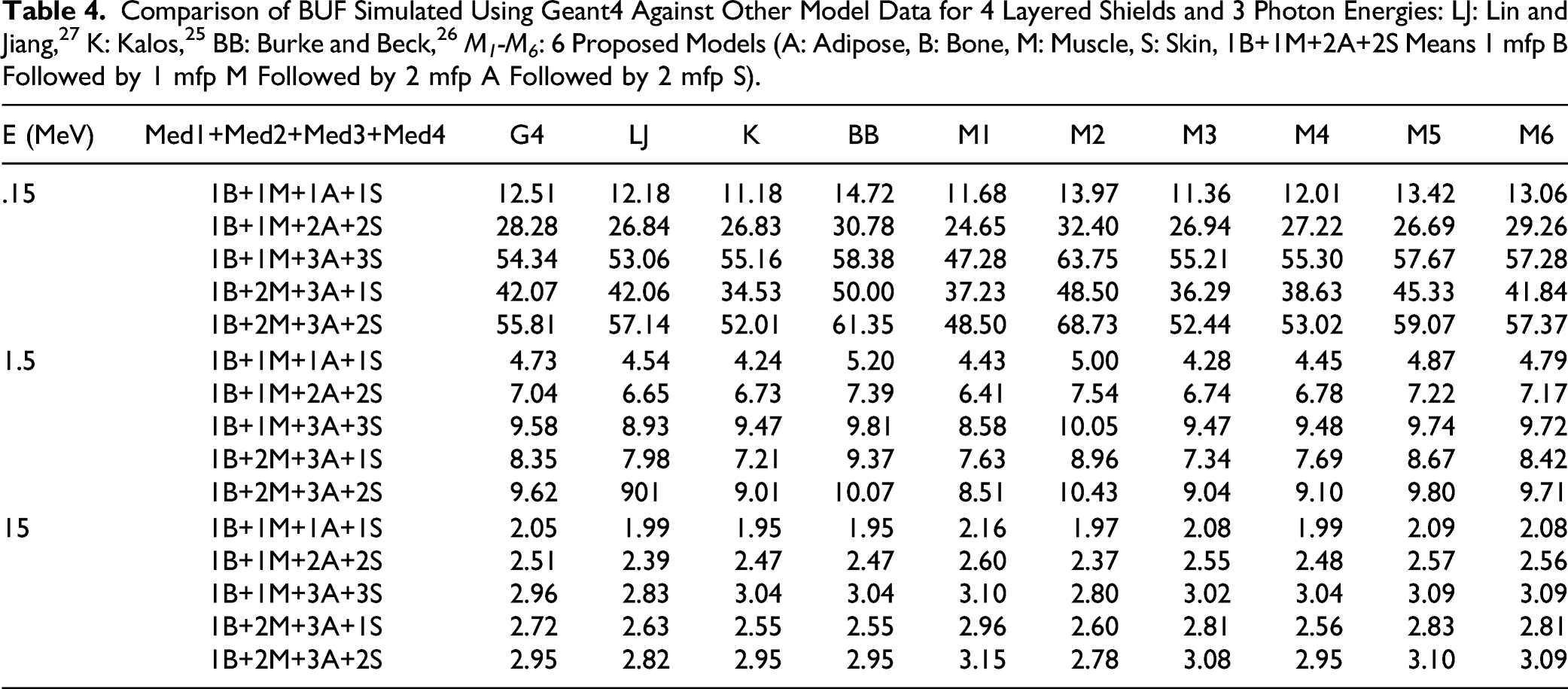

We can see that M 6 model has the best statistical analysis parameters, compared to those for Kalos model, which fails for the number of layers more than 3, and the worst one, for our situations of energy and thickness ranges, was for the Burke and Beck model. The main limitation of this work concerns the study of short penetration depths, so a straightforward extension to higher thickness could be conducted to be more consistent and justify our conclusions.
Conclusion
Buildup factor of a material for gamma-ray attenuation is an important parameter to describe scattered beam. During this work, the evaluation of gamma-ray buildup factors of stratified human tissue layers for a point isotropic source has been carried out using Geant4. Moreover, we proposed a fitting method able to accurately reproduce Geant4-based simulation results better than existing ones of Li and Jiang, Kalos, and Burcke and Beck models. The proposed fitting model M 6 , leading to an average deviation (according to direct simulation outputs) not exceeding 3 ± 3% for all studied cases, can be considered the better one of the 9 models. However, the reproducibility procedure should be extended to thickness and photon energies higher than 8 mfp and 15 MeV, adopted here, respectively. Nevertheless, we can consider that proposed results are important for medical imaging and radiation treatment planning for the most existing examination scenario.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This work was supported by the College of Applied Medical Sciences Research Center and the Deanship of Scientific Research at King Saud University, Saudi Arabia.


