Abstract
Despite the great diagnostic improvements that CT has introduced, which have allowed it to spread widely and become one of the most important tools in medical imaging, this technique has always had to face two main inconveniences: the iodinated contrast media with its known adverse reactions and the exposure to ionizing radiation. The evaluation of patient safety is a fundamental parameter for the introduction of any new technology in clinical practice. In recent years, clinical applications of
Keywords
Introduction
Since its introduction in the 1970s, computed tomography (CT) has revolutionized the clinical practice, becoming one of the most essential tools in medical imaging for the diagnosis and assessment of diseases, traumas, or abnormalities. 1
The importance of CT does not reside only in its diagnostic capacity but also for its role in monitoring disease and cancer staging, for management of therapy, and for guiding minimally invasive interventional procedures. 2
There was a time when the excitement around the development of CT had peaked to the degree that clinics and medical imaging facilities promoted a new service:
The evaluation of patient safety is a fundamental parameter for the introduction of any new technology in clinical practice.
7
Two risks faced by patients who undergo a CT examination are the potential for adverse reactions after the administration of contrast
In recent years, the technological development of CT, the
This innovation led to the possibility of distinguishing tissue regions that had similar Hounsfield unit values with different atomic components. 2
For example, it allowed differentiating the calcific plaques of the arterial walls from the intraluminal iodinated contrast medium. 9 Another example was the DECT possibility to analyze kidney stones and easily characterize its molecular component (calcium, uric acid, struvite, cystine, etc.) despite the overlap in attenuation values. 10
There are different DECT acquisition technologies available from different vendors. These can be classified as prospective techniques (before the patient is scanned) and retrospective techniques (after the patient is scanned).
Prospective techniques include dual-source DECT (dsDECT), single source DECT (ssDECT) which can be further divided into ssDECT consecutive, ssDECT twin-beam, ssDECT sequential (or “rotate-rotate), and ssDECT fast kVp switching.
Retrospective techniques include dual-layer DECT (sandwich).
DECT has therefore introduced a new tool for the study of many types of disease, providing new information about them, and taking a role in a subfield of research studies, but it also has the properties of reducing the main CT risks due to the contrast medium administration and ionizing radiation exposure, which might be of interest in clinical practice. 8
This review aims to highlight how this innovative CT technique is allowing to reduce both the X-ray dose and the contrast medium amount, thus obtaining a better cost-benefit ratio for patients.
We will first review the basics of contrast media adverse reaction, X-ray damages, and DECT physical principles and then examine in detail how the use of DECT can change the dose of contrast media in routine examinations in clinical settings and how it can change dose administration.
CT contrast media adverse reaction: Contrast agents are a group of medical drugs used to improve the visibility of internal organs and structures. 11 As a result, iodinated contrast media are the most used agents in X-ray-based imaging techniques such as radiography and CT. Although iodinated contrast agents are safe, their administration could lead to serious adverse reactions, yet most of the time, these are mild and self-limiting. 12 The frequency of adverse events associated with iodinated contrast ranges between 2.2 and 12.7% when ionic medium is used and between .2 and 3.1% when non-ionic contrast is used.13,14
Iodinated contrast media adverse reactions are divided into •
Also called •
Also called
Anaphylactoid reactions can produce mild (such as skin rash, nasal congestion, nausea, and vomiting), moderate (e.g., the persistence and increased intensity of minor symptoms, bronchospasm, dyspnea, facial edema, glottis edema, tachycardia, or bradycardia), or severe symptoms (such as arrhythmias, marked hypotension, acute bronchospasm, severe glottis edema, pulmonary edema, convulsions, shock, coma, and death).15,18,19
CT ionizing radiation: a reported carcinogen. The exposure to ionizing radiation carries with it primarily stochastic risks, with reports of deterministic effects from accidental exposures. Ensuring that radiation doses are catered to the diagnostic task, interpreting physician preferences, and now to artificial intelligent diagnostic systems is an essential role for any medical provider.
Radiation-induced stochastic effects continue to concern the public and medical community. The national council on radiation protection (NCRP) showed in a 2009 report that since 1987, medical radiation exposure increased by nearly 6 times. 20
Improvements in imaging technology led to increased use, particularly in the usage of CT scanning. Due to several radiation accidents, and growth in reports highlighting the increased risk of developing cancer after exposure to ionizing radiation, particularly for children and from CT scans, several initiatives, such as image gently, were launched to help mitigate the usage of medical radiation.
A byproduct of the initiatives was new technology, such as DECT, tube current modulation, additional changes, size-specific specific scanning protocols, and improvements in image reconstruction software. As a result, the NCRP reported in NCRP 184, titled “Medical Radiation Exposure in Patients in the United States,” that from 2006 to 2016, there was a 15 to 20% reduction in nontherapeutic medical radiation dose to the US population. 21 The reduction was partly due to fewer medical imaging procedures. However, NCRP 184 shows that the use of CT increased by 20%, but the overall dose per CT exam decreased. While the radiation dose may have been reduced in the US, globally, the statistics are not as clear. 22 Within the European Union, between 2012 and 2017, increased usage of CT was observed in almost all of the Member States for which data were available.23,24
The evident advantages provided by CT scans compared to radiographic images, such as more detailed information, improvement of the diagnosis capacity of various diseases, and the possibility of monitoring cancer patients follow-up (evaluating the size of the primary tumor, the onset of metastasis, and the effectiveness of therapies), are responsible for improving the way medical care is delivered. However, national and international standards continue to highlight that the prudent approach is to continue to balance radiation dose to which the patient is subjected with the image quality that achieves the diagnostic task.22,24
Adverse effects of ionizing radiation exposure may be categorized as
The
Vaiserman et al. in 2018 published a review in which they analyzed the validity of the LNT model based on recent scientific studies. They show that the evidence in support of LNT generally lack statistical power and are not able to disprove the LNT model. The risk of cancer in patients undergoing medical imaging has been investigated by several studies, but no data were found that correlate with certainty the possibility of tumor induction to the exposure to radiation cumulative effective dose lower than 100 mSv, and there is currently an intense debate on this. 29
In the scientific papers examined by Vaiserman et al., they found no evidence that low dose levels (below 100 mSv) could cause health damage. They also showed that numerous studies suggested how this mild exposure might lead to
Several large cohort studies were conducted to verify cancer risk in subjects who had been exposed to CT examinations in childhood, but while in some of them they provided evidence in favor of the association between cancer risk and CT scans, others contrarily did not highlight it. 24
Despite this disagreement, the ionizing radiation damage derived from the exposure to radiological procedures must be considered as scientifically proved, and, consequently, also low levels of radiation can lead to an increased risk. Therefore, it is necessary to minimize the radiation doses administered to the patient consistent with the diagnostic request, according to the ALARA principle (As Low As Reasonably Achievable). 28
Commentary from the NCRP regarding implications of recent epidemiological studies for the LNT model highlights that due to a lack of statistical power, studies evaluating stochastic risks when for exposures <100 mGy are often not statistical signification. Further, literature reports of increased risk of stochastic effects from radiation exposure may have additional confounding factors that need to be accounted for, such as smoking or other lifestyle factors. 27 However, a limited number of studies with statistical power, as determined by the NCRP, demonstrate support for the LNT model assumptions that there may be cancer effects at low doses. 31 While the studies support LNT, they do not prove the applicability of the LNT model for radiation protection. 27 The NCRP, international commission on radiation protection (ICRP), and United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) collectively conclude that currently the LNT model is most prudent for radiation protection purposes. 27
Patient safety is of paramount importance for healthcare providers. Although accidents are rare, and the current evidence regarding stochastic risks statistically derived, a growing body of research efforts introduces new mechanisms to reduce iodine load and the ionizing radiation dose. Among the technological advances, DECT enables the reconstruction of multiple image types, such as quantitative iodine concentration or distribution maps, and virtual non-contrast images, which could provide further opportunity to manage iodine load and radiation dose.
DECT and contrast media reduction: The administration of contrast medium in CT examinations improves the visibility of internal organs and structures because the higher tissue iodine concentration causes an increase in the attenuation of the X-rays.
32
This parenchymal accumulation of iodine, therefore, leads to an increase in the
The main ones can be summarized as follows: (1) Scan parameters (low kVp or low keV, scan timing, and delay), (2) Contrast media factors (IDR, iodine concentration, volume, injection rate, and duration), and (3) Patient factors (cardiac output and BMI).
7
Shen et al. showed how the concept of low tube voltages to increase the enhancement could be combined with a reduction in the contrast medium load. They analyzed vascular studies performed with the so-called
However, although voltage reduction gives these great advantages, on the other hand, it causes the problem of noise growth for SECT images.
In a retrospective study, Corrias et al. highlighted the inverse correlation between aortic attenuation and BMI of patients. They also noted a direct proportionality between aortic enhancement and injection flow rate. 36
The
An important scan parameter is the peak X-ray tube voltage used to for imaging. Traditionally, CT scanning has been performed using a single tube potential:
To reconstruct a tomographic image for viewing, the polychromatic X-ray spectrum is reduced to an effective monochromatic energy expressed in kilo-electron volts (keV). This process requires several assumptions and could lead to an overlap in Hounsfield unit (HU) values. Since conventional SECT uses a single kVp for image acquisition, one cannot selectively or simultaneously display the attenuation of materials acquired at low and high energies. It has been seen that the attenuation of the X-rays at a given energy of the tube is the result of the summation between the attenuation due to the Compton effect and the attenuation due to the photoelectric effect. 39
At diagnostic energy ranges, typically between 20 keV and 140 keV, X-rays interact with biological tissues primarily via
The Compton effect is primarily observed when the photon energy is substantially greater than the binding energy of the electron. Compton interactions are characterized by the X-ray partially transferring some energy to the outer shell electron. The partial transfer causes the electron to be ejected while the X-ray is redirected from its incident direction, 9 Compton interaction depends on the electron density of a material, not the atomic number. 41
The photoelectric effect occurs when the incident X-ray energy is greater than, but closer to the energy that binds an electron to the innermost atomic orbital “K.” The incident X-ray transfers all its energy to the electron, which causes it to be expelled from the atom.
9
The probability of photoelectric absorption is proportional to the cube of the atomic number,
Therefore, through the application of different radiation energies, diverse attenuation values will be obtained, which will vary characteristically based on the atomic composition of the scanned tissues. 9 As a result, any material can be decomposed into a combination of two base materials, which differ in their photoelectric and Compton attenuations, and the relative contributions of these two basic constituents can also be determined. 2
DECT exploits the differential attenuation characteristics of materials by scanning the patient with a low and high energy tube voltage. In practice, the two tube voltages depend on the DECT vendor, but a common approach is to use to 80 kVp for the lower energy and 140 kVP for the higher energy. 9
With DECT, multiple image types can be reconstructed. The selection of image type is often determined by the diagnostic task, but common choices for the basic materials are iodine–water or calcium–water.
When iodine and water are used as the base materials, one can Material density image reconstructions, starting from a standard DECT acquisition. To the left iodine-density images, to the right, water-density images. From a single DECT acquisition we can obtain several material-specific densities reconstructions.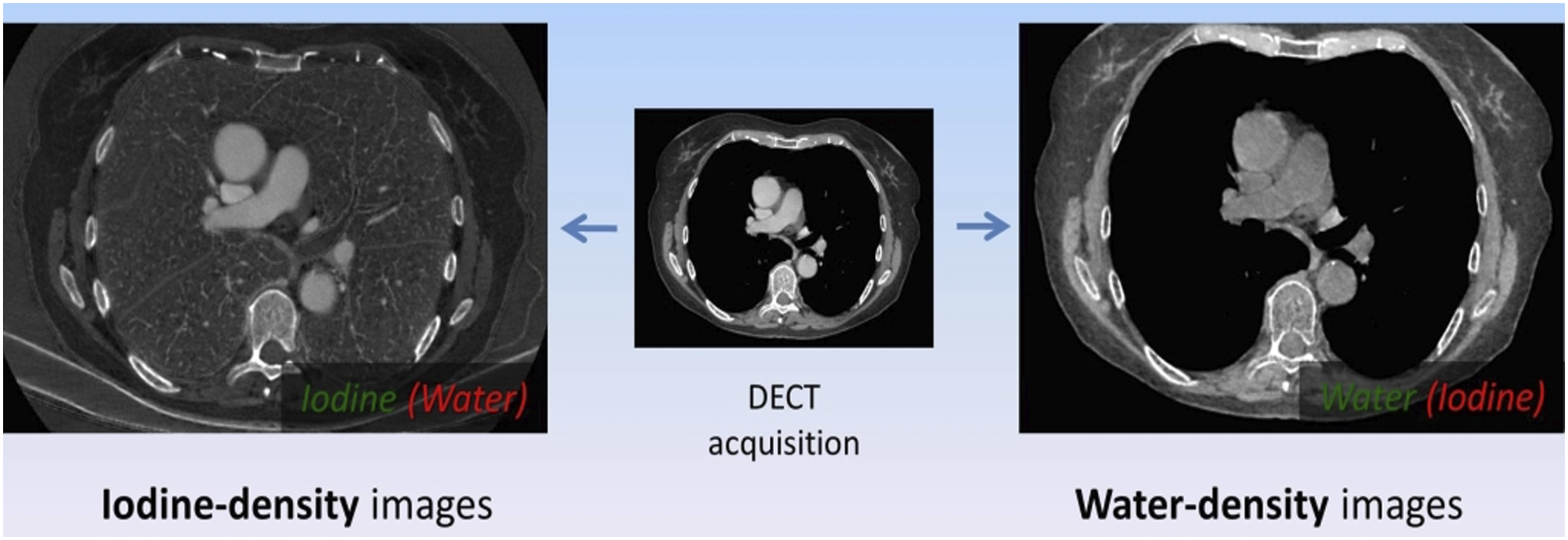
Materials that are neither iodine nor water, such as metallic clips or bone, are represented as mixtures of the two base materials and seen with non-zero densities in both MDI. 41 Depending on the manufacturer, the image intensity units for MDI are g/cm3 or 100 x g/cm3. This contrasts with the HU value typically seen with conventional CT.
An alternative to MDI is Virtual monochromatic images (VMI) at 65 keV obtained from DECT scans with 80/140 kVp acquisition parameters. To the top, a lesion seen with a VMI, to the bottom a specific detail of the lesion showing a perilesional halo, not seen on no. MDI (material density images) processed from the same dataset shown in Figure 2. We can observe that in the second image the lesion conspicuity is increased.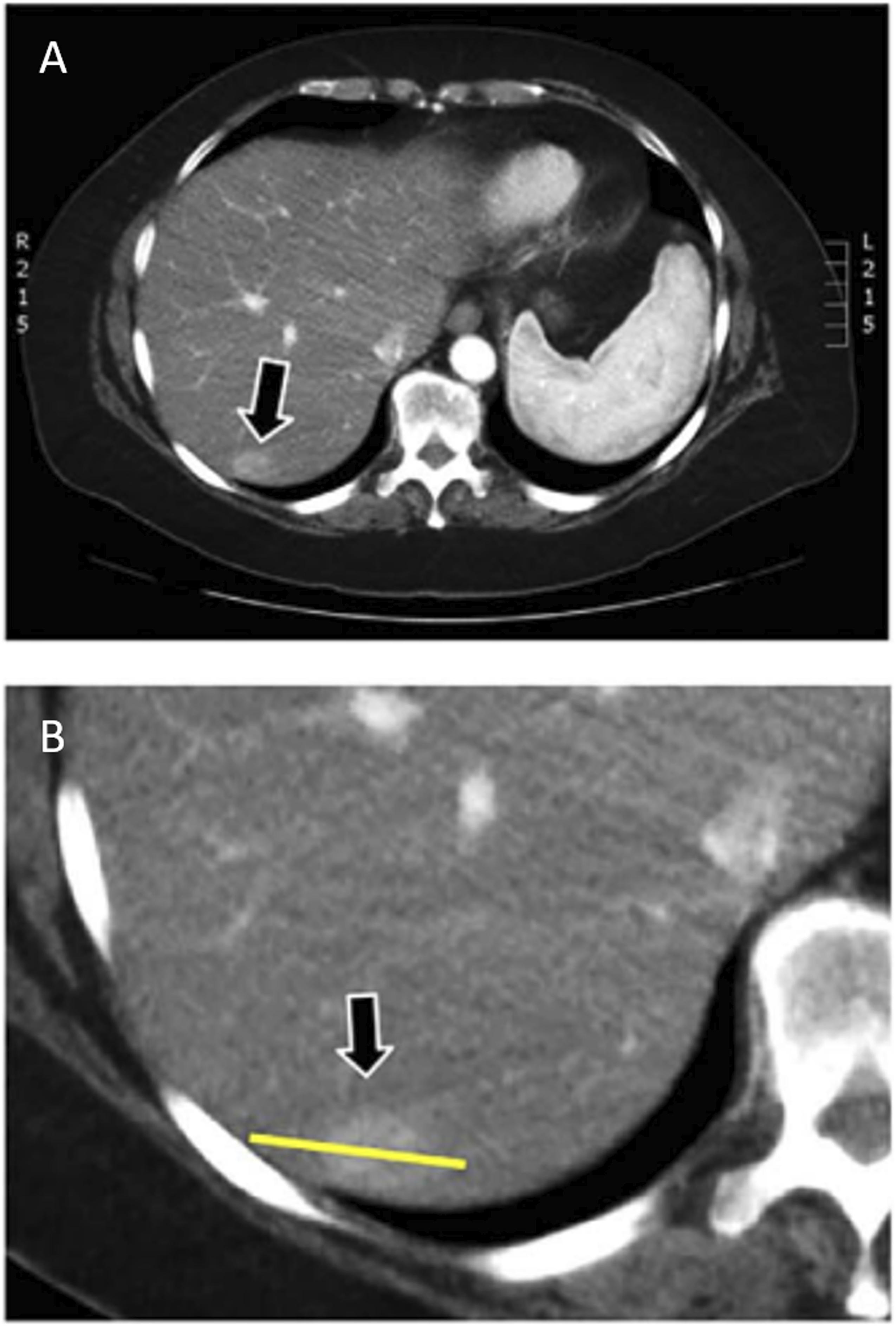

The units of intensity for each pixel displayed on VMI are the typical HU value. The VMI at about 70 keV, in addition to their function of increasing the contrast-to-noise ratio, contributes to improving the overall image quality because, by eliminating the very low energy photons that contribute only to the image noise, the image of beam-hardening artifact is reduced. 51 The selection of VMI keV is task-specific and best determined by the radiologist’s preferences.
Furthermore, DECT can provide maps of electron densities in a given tissue: the attenuation of X-ray, next to others, depends on the electron density and the effective atomic number. Both parameters are characteristic for different materials. For example, M ileto et al. showed that non-enhancing renal cysts, including hyperattenuating cysts, can be discriminated from enhancing masses on effective atomic number maps. 52
This property of being able to produce images at different energy levels is important because while low energy levels, with their lower contrast and noise, allow a better evaluation of soft tissues, high levels, with their higher contrast and noise, improve the study of vascular structures and reduce metal artifact, which could be produced in the presence of implants and stents. 7
However, if energy levels very close to the k edges of iodine are used for image generation, the image noise increases significantly. But technological progress has recently led to the creation of a monoenergetic image noise reduction algorithm, which combines the typical high iodine attenuation of low energy images with the noise characteristic of high energy images. 33
Albrecht et al. 48 showed that through the introduction of noise reduction algorithms, it is possible to reach energy levels of 40–50 keV to generate VMI. When lower energy VMI is used, the amount of contrast medium administered can also be reduced without affecting the overall image quality. 7 This is especially relevant when trying to avoid CI-AKI in patients with borderline glomerular filtration rate (GFR). 53 This has been confirmed by several scientific papers, most of which concerned the reduction of the contrast agent load in DECT angiographic studies.54-59
Shuman et al. showed that with DECT, the contrast medium load could be reduced by 49%, while the aortic attenuation of contrast on VMI reconstructed at 50 keV was significantly greater than the average value obtained through SECT scans at 120 kVp using standard doses of iodine (47 g). They also demonstrated how the contrast-to-noise ratio and the signal-to-noise ratio were comparable between the two techniques. 54 In another experiment, they administered the same iodine dosage and showed that image noise measured on 77 keV VMI was comparable to the noise measured on SECT scans at 120 kVp. 54
Agrawal and colleagues evaluated patients with abdominal aortic aneurysm using DECT angiography. They showed that it was possible to reduce the iodine administration of 28% (only 24 g per examination) using virtual monochromatic imaging set at low energy (from 40 to 60 keV), thus obtaining 185% higher attenuation and 25% higher contrast-to-noise ratio compared to the typical SECT angiography performed with a standard iodine dose of 33.3 g. 55
Shuman et al. performed a second DECT aortography study further reducing the amount of contrast medium. They found that in virtual monochromatic images, reconstructed at energy levels of 50 and 77 keV with a contrast medium dose reduced by 70%, the aortic attenuation, contrast-to-noise ratio, and signal-to-noise ratio (SNR) were comparable to those observed using the standard iodine dose in SECT scans. 56
A recent study compared CT pulmonary angiography of two groups of 41 patients. The first group had been given a reduced level of iodine (only 6 g) for performing DECT pulmonary angiography with monoenergetic reconstructions at 40 keV while the second had been examined with SECT scans at 100 kVp applying the standard contrast medium dosage (24 g of iodine). This study demonstrated how the acquisition of DECT data in combination with advanced reconstruction of monoenergetic images enabled the diagnosis and safe exclusion of central, lobar, and segmental pulmonary embolism, even using a very low dose of iodine. 57
Almutairi et al. 58 investigated the performance of DECT angiography in peripheral artery imaging by comparing the effectiveness of the low contrast medium volume with a routine iodine amount and showed that the image quality was clinically acceptable in both groups.
Another study found that DECT coronary angiography allows for >50% reduction in iodine administration while maintaining signal-to-noise ratio, contrast-to-noise ratio, and diagnostic interpretability with a slight impairment of image quality. 59
Contrast media dose reduction in DECT angiography.

While a fair number of scientific papers have investigated the possibility of reducing contrast agent doses for vascular DECT examinations, far fewer studies have been performed for non-vascular DECT assessments.
Clark et al. conducted a study in which they researched patients who had undergone abdominal DECT examination with reduced contrast protocol for suspected liver or pancreatic disease (19–46 g of iodine) and who had also performed a SECT scan with routine contrast protocol (29–70 g of iodine) for a matched anatomical coverage. They found a statistically significant increase in vascular and parenchymal enhancement and an improvement of the lesion-to-parenchyma contrast in reduced iodine dose DECT examination with comparable values of contrast-to-noise ratio and good image quality. This intra-patient study demonstrated that a 37% reduction of the contrast dose for abdominal CT could be possible using low energy DECT, thus providing a mechanism to minimize risks to patients with kidney dysfunction. 60
Tsang and colleagues did a study on 51 pediatric patients that had undergone contrast-enhanced DECT scans across all body sites to estimate the potential iodine dose reduction when interpreting a virtual monoenergetic image. They demonstrated that, with monoenergetic DECT reconstruction at 50 keV, it was possible to obtain a reduction of 50% iodine with similar or better contrast-to-noise ratio as compared with polychromatic 120 kVp SECT images. They found a 2.4-fold increase in iodine contrast enhancement in blood vessel images. 61
However, for many types of vascular studies, it is not important to see the lumen well opacified but it seems more important to study the vessel wall: that is, especially important in the atherosclerotic disease studies, where the neoangiogenesis of the vascular wall is recently been advocated as one of the most important risk factors, and it can be studied, to a certain extent, with angio-CT protocols62,63; intraplaque hemorrhage is another clinical setting in which it is of importance the analysis of vessel walls. 64 In this setting, a reduction of keV has no benefit since it could increase the noise surrounding the vessel lumen. 65
Therefore, even these few studies seem to suggest what has been previously highlighted in vascular studies. The new technological advances and the reconstruction capabilities of virtual monoenergetic images with DECT open new perspectives on the creation of reduced medium contrast protocols that will ensure diagnostic efficiency while maximizing patient safety and benefits.
DECT and ionizing radiation dose: DECT has not had a very wide diffusion in clinical practice.43,66 Some of the first studies evaluating DECT performance led to a limitation of its expansion.
For example, Ho et al. in 2009 published a scientific article comparing the radiation dose values of DECT and SECT using adult liver, renal, and aortic imaging protocols. They found that CTDIvol (volumetric computed tomography dose index) values were 49.4 mGy for DECT and 16.2 mGy for SECT examinations and that effective dose was 22.5–36.4 mSv for DECT and 9.4–13.8 mSv for SECT scans. Therefore, the doses provided by DECT examinations at 80–140 kVp were up to 3 times higher than those given by SECT scans at 120 kVp. 67
In the early development of DECT, its clinical value was still unclear, and the observation of these exposure values, according to the ALARA principle, did not justify its widespread clinical use. 68
However, later studies showed that by using DECT for the assessment of different body regions, the same diagnostic performance of SECT could be obtained with the administration of radiation doses lower than the published reference levels, and therefore acceptable for clinical use. 7
Kamiya and colleagues did a study on postoperative patients, who had undergone both DECT and SECT head scans within a short time for follow-up after brain surgery, to evaluate whether the image quality of virtual monochromatic reconstruction at 65 keV with DECT could be comparable to SECT images at 120 kVp without increasing the radiation dose. They found that the mean CTDIvol for DECT was 11% lower than SECT, with respective values of 70.2 mGy and 78.9 mGy, while the image quality of both techniques was similar and therefore clinically acceptable. 69
In a scientific article, Bauer et al. compared the dose and image quality of CT pulmonary angiographies obtained using SECT and DECT. They found that the latter technique allowed a significant dose reduction with similar image quality to that of standard SECT images. 70
Shuman and colleagues 71 evaluated the use of DECT for the study of hyper-enhancing focal lesions in patients with advanced cirrhosis and saw how there was no statistical difference in mean CTDIvol between DECT (12.8 mGy) and SECT (14.2 and 14.4 mGy).
Therefore, these studies showed that the use of DECT did not necessarily lead to an increase in the dose administered to patients. In general, the radiation doses administered with the new high-end DECT scanners currently available are lower or not more than 10% to 15% higher than SECT. 7
Moreover, they did not consider one of the most important DECT capabilities: the possibility of creating
As previously mentioned, DECT can characterize the material composition of the tissues. In the images obtained after administration of contrast medium, DECT can calculate the attenuation provided by the iodine for each voxel and then subtract it to reconstruct virtual non-enhanced images. 72
This ability offers DECT the opportunity to reduce the number of acquisitions performed in multiphasic examinations, and consequently to lower the administered dose levels.
73
In fact, virtual non-enhanced images can simulate
De Cecco et al. 74 compared the effective dose of a DECT triple-phase protocol (true non-enhanced, arterial, and portal phases) with that of a dual-phase protocol (arterial and portal phases) for liver lesion detection and found that the dose achieved by omitting the unenhanced acquisition was reduced with an average of 21.1%.
Graser and colleagues assessed the diagnostic accuracy of DECT in renal mass characterization as benign or malignant using a single nephrographic phase protocol. They showed how this could replace biphasic scanning and at the same time significantly reduce radiation exposure by almost 50%. 75
The DECT’s ability to reduce the number of acquisitions performed in multiphasic examinations by saving true non-enhancing images can also be increased with the use of the
The split bolus is an innovative technique that involves administering intravenous contrast medium in two or three boluses and combining post-contrast phase images in a single scan, and which can be used to reduce the radiation dose, especially in cancer patients. 76 This procedure is commonly used in various SECT protocols such as, for example, in SECT urography.
Split bolus allows reducing the classic three-phase scan to 2 acquisitions by combining the nephrographic and excretory phases, thus reducing the radiation dose. DECT further improves this ability because, being able to reconstruct virtual non-enhanced images, it does not require the execution of true non-enhanced scans. So, by combining the split bolus technique with DECT, a triple-phase protocol can be reduced to a single post-contrast acquisition, which results in a significant reduction in the radiation dose. 77
Javor et al. compared the performance of single-phase DECT angiography to the three standard acquisitions (not enhanced, arterial, and venous) for the detection of endoleak after repair of the endovascular aneurysm. They demonstrated how, using the split bolus technique in combination with the reconstruction of non-enhanced virtual images, there was a significant reduction in the radiation dose of up to 42% with comparable diagnostic accuracy and image quality. 78
Mahmood et al. 79 showed that radiation dose, particularly organ dose, is lower with rapid-switching DECT (rsDECT) than conventional SECT exams, in patients with liver cancer, especially in smaller patients.
Radiation dose reduction in DECT.

Limitations of Dual Energy Computed Tomography
We have shown clearly the benefits of DECT in terms of dose and contrast reduction, however due to various limitations, this technology, despite not being new, fails to fully penetrate clinical practice, especially in non-vascular exams. 80
The reasons for this lack of penetrance could be divided into two big different groups. First, the benefit for daily clinical practices and activities is not easily evaluable and it is not yet clear which areas could benefit the most of this technology, despite its higher costs than traditional SECT. 81 Material decomposition and analysis seemed to be the most promising field of DECT but some recent studies have shown how there are different limitations to this application,80-82 especially when more than 3 materials are present in a single voxel.
A second important limitation is due to the image quality of DECT exams, which is not appreciated by many radiologists. In particular, DECT images could appear more plastic or artificial to many, beam-hardening artifacts are more often present in material decomposition images, 83 and noise is particularly increased at low kV acquisitions.49,71
Conclusion
A potential improvement in patient safety, due to the ability to reduce both the amount of contrast medium and the dose of ionizing radiation administered, has been made possible by not only the constant perfecting in DECT technology but also by other co-existing technological advances, such as reconstruction algorithms, IA-based reconstruction, and injection protocols.
Besides, the creation of DECT-specific protocols and the new technological advances, such as the iterative reconstruction techniques that have significantly improved the image quality, are expanding the possibilities of clinical use of DECT. However, some of the many associated improvements (e.g., reconstruction algorithms, IA-based reconstruction, injection protocols, etc.) are not DECT technology per se.
Although more studies are needed to confirm what has been said, DECT, combining its double-reduction ability with its multitude of capacities, including tissue characterization, reconstruction of virtual monochromatic images, generation of virtual non-enhanced images, calcium quantification, etc., is certainly one of the most promising techniques for the future.
Footnotes
Author Contributions
The authors were equally involved drafting of the manuscript, critical revision of the manuscript for important intellectual content, statistical analysis, technical, and material support of this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Grant support was provided by MSK Cancer Center Support Grant/Core Grant P30 CA008748.
