Abstract
Arsenic has been reported to cause damaging effects on different body organs. This study was designed to evaluate the protective effect of resveratrol (RSV) against arsenic trioxide (ATO)–induced intoxication in experimental animals. Twenty-four Wistar rats were allocated in 4 groups: group 1: control group, received normal diet; group 2: received ATO (3 mg/kg); group 3: received RSV (8 mg/kg) 30 minutes before administration of ATO; and group 4: received ascorbic acid (25 mg/kg) 30 minutes before administration of ATO. Treatments were given to experimental rats daily for consecutive 8 days. At the end of experimental period, bioaccumulation of arsenic in liver and kidney was assessed by hydride generation-atomic absorption spectrophotometer to investigate the association of arsenic accumulation with histological aberrations. Following parameters were also investigated: serum biochemical profile (alanine aminotransferase, aspartate transaminase, alkaline phosphatase, blood urea nitrogen, and creatinine) for evaluation of liver and kidney functions and lipid peroxidation and oxidative stress (malondialdehyde, glutathione, superoxide dismutase, catalase, and glutathione peroxidase) in tissue homogenates of liver and kidney for estimation of oxidative status. The findings of this study indicate that RSV remarkably ameliorated the hepatic and renal toxicity in arsenic-exposed rat model due to its strong antioxidant potential.
Introduction
Arsenic is a naturally occurring chemical metalloid of earth’s crust and widely distributed in environment in the form of inorganic arsenic (iAs) from soil to the groundwater. 1 Arsenic and compounds containing different species of arsenic such as oxidized arsenate (iAsV) and trivalent arsenic (iAsIII) are highly poisonous in nature. 2 Contamination of drinking water with arsenic has increased due to anthropogenic and human activities such as process of extraction of minerals, utilization of pesticides and fertilizers, and processing of industrial products. 3 Inorganic species of arsenic are more potent to induce toxicity as compared to organic species. 4 Chronic exposure of arsenic is becoming a major issue related to public health all over the world. Arsenic exposure to human has been linked with many diseases like cardiovascular disease, cancer, and diabetes mellitus. 5 iAsIII binds to thiols or biological ligands having sulfur group leading to increase in the level of reactive oxygen species (ROS) and suppresses antioxidant defense system resulting in disruption in the activities of multiple organs. 6
In 1970s, arsenic trioxide (ATO) had been used for the treatment of acute promyelocytic leukemia (APL)7,8 and promyelocytic leukemia (PML). 8 But it has been observed that patients of APL treated with ATO suffer from nephrotoxicity, mainly due to the generation of oxidative stress. 9 Hence, the dose-dependent toxicity of ATO has limited its clinical uses. 10 The symptoms of acute arsenic poisoning are vomiting, gastrointestinal cramps, diarrhea, disturbance in the liver function, and changes in electrocardiogram. 11 ATO can cause cardiotoxicity by alteration in action potential for long period and unfortunately lead to death. 12
The oxidative methylation of arsenic depends upon the metabolism of arsenic. 13 The pentavalent arsenate (As+5) upon absorption in the body rapidly undergoes the reduction and forms trivalent arsenite (As+3), especially in liver and blood. 14 The circulating arsenic metabolizes in hepatic cells by series of oxidative methylation and reduction steps to yield different methylated products: monomethyl arsenic (MMA) and dimethyl arsenic acid (DMA). The excretion of arsenic is due to its methylation yielding DMA. The arsenic undergoes oxidative methylation by consumption of S-adenosyl methionine (SAM) and glutathione (GSH) and causes many epigenetic changes in the body. 15
Arsenic compounds can also inactivate the endothelial nitric oxide synthase through interaction with thiol groups and resulting in increased level of reactive nitrogen species which perform its action in combination with ROS resulting in lipid peroxidation and apoptosis. 16 The arsenic lethal dose impairs the functions of liver 11 and also induces renal toxicity. 17 The high level of oxidative stress because of chronic exposure of arsenic leads to the activation of key signaling of kinases, p38 mitogen-activated protein kinase (p38 MAPK), and C-Jun N-terminal kinases (JNK) resulting in the apoptosis of hepatic cells. 18 The increased content of ROS induces lipid peroxidation and leads to toxicity in hepatic and renal cells. 19
The compounds having antioxidant activities slow down the process of oxidation. Many medicinal plants as substantial therapeutic agents have antioxidant potential to shield from development of many diseases. 20 Natural polyphenols have antioxidant activities to suppress the oxidative stress. The antioxidant properties of polyphenols are because of their chemical structure. 21 Resveratrol (RSV), chemically known as tran-3,4,5-thihydroxystibene, is stilbenoid polyphenol having 2 phenol rings that are linked with one another via an ethylene bridge. 22 RSV is commonly found in highly pigmented fruits and vegetables. This compound has many biological activities such as antiviral, antitumor, phytoestrogenic, and antioxidant. 23 It has also proved that RSV can shield antioxidant system of liver against reperfusion and ischemia-induced oxidative stress. 24 Ascorbic acid (AA) inhibits the microsomal mixed function oxidase and cytochrome content. 25 It has been attributed that supplementation of arsenic-exposed rats with AA provides a protective effect against oxidative stress. 26
Although the therapeutic applications of RSV have been well defined but till date the mechanism by which RSV protects the toxicity against cytotoxic effects of ATO is not known. Nevertheless, according to our knowledge, no reports have found which describe the precise and comprehensive biological actions of RSV against arsenic-induced toxicity. In this study, we have focused on RSV underlying mechanism of action for treatment of arsenic toxicity, especially with respect to oxidative stress and accumulation of arsenic. This research conducted to evaluate the antioxidant effect of RSV in liver and kidney of ATO-induced rats by subsequent estimation of lipid peroxidation and oxidative stress markers in tissue homogenates along with accumulation of arsenic in liver and kidney and their histopathological examination. This study will be helpful to provide idea to the researchers to attain their attention for further exploration of therapeutic activities of bioactive compounds in both traditional and modern medicines. The aim of this project is to explore the possible mechanisms of RSV for protection of renal and liver function against arsenic-induced rats.
Methods
Chemicals and Assay Kits
ATO (Farco Chemical Laboratories, Singapore), RSV (Chem-Impex Int’l Inc), AA (Honey Well ), sodium borohydride (NaBH4) (Merck), alkaline phosphatase (ALP) activity assay kit (Bioactiva diagnostic), alanine aminotransferase (ALT/GPT) activity assay kit (Bioactiva diagnostic), aspartate aminotransferase (AST/SGPT) activity assay kit (Bioactiva diagnostic), creatinine assay kit (Bioactiva diagnostic), malondialdehyde (MDA) (Catalog Number; E-EL-0060, Elabscience®), superoxide dismutase (SOD) (Catalog Number; E-BC-K020, Elabscience®), catalase (CAT) (Catalog Number; E-BC-K106, Elabscience®), GSH (Catalog Number; E-EL-R2491, Elabscience®), and glutathione peroxidase (GSH-Px) (Catalog Number; :E-BC-K096, E-EL-R2491, Elabscience®)
Experimental Design
To conduct this study, initially, twenty-four albino Wistar rats weighing 200–250 g were purchased and kept in animal house of Government College University, Faisalabad (GCUF), Pakistan, at an ambient temperature (25 ± 5°C) and relative humidity (70 ± 5%). To carry out research, all experimental procedures were performed in accordance with the approved laboratory animal Bio-safety guidelines, Bioethics committee and Institutional review board (GCUF/ERC/2189) from GCUF. For appropriate housing of experimental animals, all the guidelines were followed, and all rats were fed on the normal diet along with water ad libitum. Before the start of experiment, all experimental rats were acclimatized for 2 weeks. According to experimental protocols, toxicity was induced in 6 rats by ATO injection intraperitoneally (3 mg/kg/day) and designated as ATO group. The 6 rats were given RSV (8 mg/kg/day) as pre-treatment 30 minutes before inducing arsenic toxicity and designated as RSV+ATO group. The other 6 rats were given AA (standard) as pre-treatment (25 mg/kg/day) 30 minutes before inducing arsenic toxicity, designated as AA+ ATO group while remaining 6 rats were treated as control group and received normal diet. The dose of AA
27
and RSV
28
was calculated on the basis of the literature as described previously. The calculated doses of RSV and AA were administered into the experimental rats via oral gavage for consecutive 8 days. On day 8, rats were fasted overnight and sacrificed mercifully by cervical dislocation. The abdomen was dissected to remove kidney and liver for analysis of biochemical parameters and arsenic retention in liver and kidney along with histological examination. The schematic plan of this research work is shown in Figure 1. Schematic representation of study design.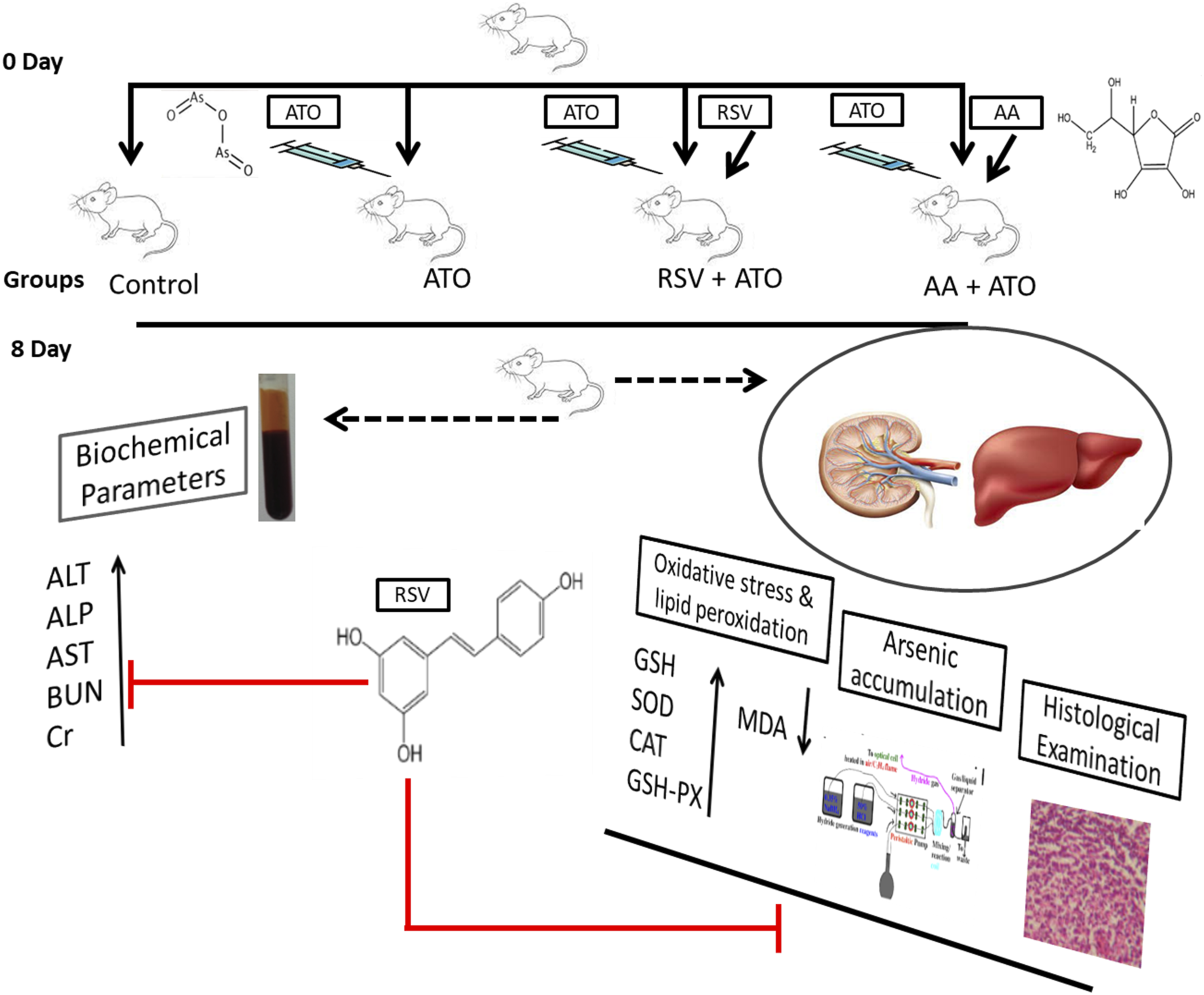
Determination of Arsenic Accumulation in Tissues
The determination of arsenic accumulation in tissues was carried out using hydride generation-atomic absorption spectroscopy technique (HG-AAS technique).
Acid Digestion of Tissue Homogenates
This method is selected to minimize the loss of arsenic metal and to avoid the contamination. All the apparatus including beakers, Pyrex tubes, boiling tubes, and conical flasks, which were required for acid digestion, was soaked in 10% nitric acid for 24 hours then rinsed 5 times with Milli-Q water before the start of experiment. The .5 g of liver and kidney samples were weighed and added into the boiling tubes. Then, acids were added in the boiling tubes in following order: (a) added 10 mL of concentrated HNO3, (b) added 10 mL of pre-formulated acid mixture of concentrated HNO3 and HCl (3:1), and (c) added 10 mL of pre-formulated acid mixture of concentrated HNO3 and H2O2 (1:1).
Then, treated samples were predigested at 25°C for 24 hours. After 24 hours, treated samples were digested by heating on hot plate at 120°C for 60 minutes. Samples were cooled and then filtered by Whatman No. 541 filter paper into the volumetric flasks. The filters and tubes were rinsed and made the volume of filtrate up to 50 mL with de-ionized water stored at 4°C for the analysis of arsenic content by following the method as described previously. 29 The samples (standards, control, reagent, and calibration blank) were introduced into the atomic absorption spectrophotometer. The calibration of instrument was also analyzed periodically at frequency of 5 readings. 30
Hydride Generation-Atomic Absorption Spectroscopy Technique
The contents of arsenic were determined in the sample by using HG-AAS technique. The generation of hydride was performed by using 3% (w/v) NaBH4 in 1% NaOH solution. The radiation source of AAS has hollow cathode lamp of arsenic which was being used at a wavelength of 193.7 nm and spectral split width of about of .7 nm.
Estimation of Lipid Peroxidation and Oxidative Stress Biomarkers From Tissue Homogenates
The liver and kidney tissues were collected and subjected to homogenization. The homogenization process was carried out by using .1 M phosphate buffer saline (pH = 7.4) and centrifuged at 10 000 r/min for 10 minutes at 4°C. After centrifugation, the supernatant of liver and kidney tissue homogenate was collected and used for determination of lipid peroxidation and oxidative status including MDA, GSH, SOD, CAT, and glutathione peroxidase (GSH-GPx) through ELISA kit by following manufacturer’s instructions.
Blood Sampling for Biochemical Analysis
Blood samples were obtained by cardiac puncture after cervical dislocation but before dissection of rats. The blood samples were allowed to stand for a period of 30 min at room temperature to allow the formation of clot before centrifugation. The serum was isolated from the blood after centrifugation at 3000 r/min for 20 minutes. Then, the obtained serum was stored at −20°C untill further analysis of abovementioned biochemical parameters.
Estimation of Liver Biomarkers
For evaluation of pre-treatment effect of RSV and AA on the liver of arsenic-exposed rats, liver function biomarkers (AST, ALT, and ALP) in the serum were estimated at the end of experimental period by using assay reagent kits via MicroLab-300 chemistry analyzer.
Estimation of Kidney Biomarkers
We investigated the pre-treatment effect of RSV and AA on the kidney of arsenic-exposed rats. For this purpose, we determined the serum creatinine and blood urea nitrogen level through MicroLab-300 chemistry analyzer by using respective assay reagents kits.
Histopathological Examination
The sections of liver and kidney tissues from experimental rats were fixed in 10% neutral buffered formalin for overnighted period at 37°C. Then, washing of fixed tissues was performed followed by dehydration in alcohol. The dehydrated tissues were placed in xylene. The infiltration of cleared tissues with melted paraffin was done for 2 hours and then embedded in the paraffin wax. The tissues were cut with microtome into thin slices of about 5 μm. Then, sections were mounted on slide and strained with hematoxylin and eosin stains and covered with cover slip. Then, slides were observed at 4×, 10× and 40× for histopathological examination and photographs were taken.
Statistical Analysis
Statistical evaluation was performed by using GraphPad Prism 5. The values of differently measured biochemical parameters were expressed as mean ± SD. The differences among all the groups were carried out by using one-way analysis of variance (ANOVA) followed by Tukey’s multiple comparison test. The level of significant difference was set as P < .05.
Results
Effect of Pre-Treatment on Accumulation of Arsenic in Tissues
In the present study, we investigated the effect of RSV and AA pre-treatment on arsenic accumulation in liver and kidney of arsenic-induced rats (Figure 2). The concentration of arsenic in ATO-treated group increased significantly (P < .001) as compared to control group. The pre-treatment significantly reduced (P < .001) the accumulation of arsenic in the liver as compared to ATO group. Effect of pre-treatment on accumulation of arsenic in (A) liver and (B) kidney of experimental rats groups exposed to ATO. The hepatic and nephrotic level of arsenic retention was measured at the end of treatment period. The level of significance was set at P < .05 using one-way ANOVA followed by Tukey’s multiple comparison test. The results are expressed as mean ± SD, n = 6. *** represents P < .001, ** represents P < .01, * represents P < .05, and ns = non-significant.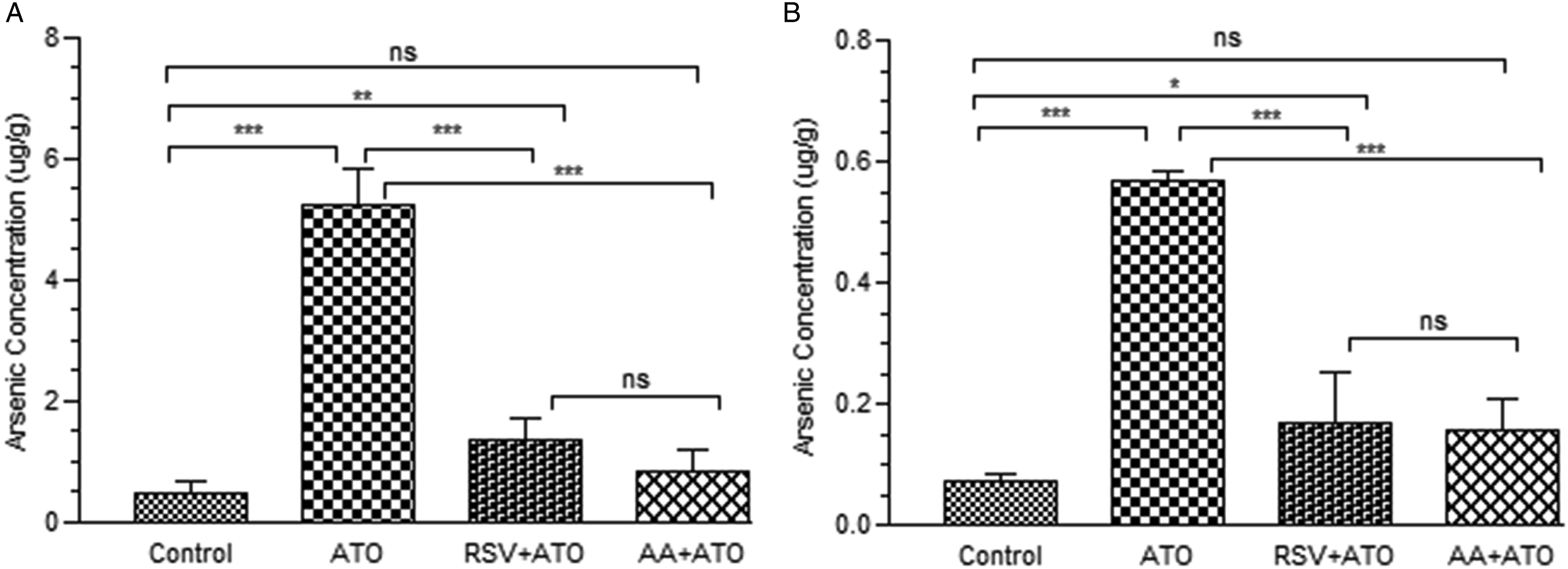
Effect of Pre-Treatment on Lipid Peroxidation and Oxidative Stress in Liver
We determined a marked elevation (P < .001) in MDA after exposure of arsenic as shown in Figure 3A. We found a significant (P < .001) decrease in the level of GSH, SOD, CAT, and GSH-Px in arsenic-exposed group in comparison to control group (Figure 3). But RSV and AA played a marked role in achieving significantly normal level of all abovementioned lipid peroxidation and oxidative stress biomarkers (Figure 3). In present study, it has been declared that RSV treatment significantly decreased (P < .001) the level of MDA while increasing (P < .001) the levels of GSH, SOD, CAT, and GSH-Px in comparison to ATO group. Effect of pre-treatment on hepatic (A) MDA, (B) GSH, (C) SOD, (D) CAT, and (E) GSH-Px of experimental rats groups exposed to ATO. The hepatic level of MDA, GSH, SOD, CAT, and GSH-Px was measured in tissue homogenate at the end of treatment period. The level of significance was set at P < .05 using one-way ANOVA followed by Tukey’s multiple comparison test. The results are expressed as mean ± SD, n = 6. *** represents P < .001, ** represents P < .01, * represents P < .05, and ns = non-significant. Abbreviations: MDA: malondialdehyde, GSH: glutathione, SOD: superoxide dismutase, CAT: catalase, GSH-Px: glutathione peroxidase, ATO: arsenic trioxide, RSV: resveratrol, AA: ascorbic acid.
Effect of Pre-Treatment on Lipid Peroxidation and Oxidative Stress in Kidney
We observed a marked elevation (P < .001) in the level of MDA after arsenic exposure (Figure 4A). We also assessed a highly significant decrease (P < .001) in the level of GSH, SOD, CAT, and GSH-Px in arsenic-exposed group as shown in Figure 4(B-E). The pre-treatment with RSV and AA played a significant role in restoring the normal level of all abovementioned lipid peroxidation and oxidative stress biomarkers in arsenic-induced rat model (Figure 4). The results showed that treatment with RSV significantly decreased (P < .001) the level of MDA and increased (P < .001) the levels of GSH, SOD, CAT, and GSH-Px relative to ATO group to achieve normal oxidative stress. Effect of pre-treatment on renal (A) MDA, (B) GSH, (C) SOD, (D) CAT, and (E) GSH-Px of exoerimental rats groups exposed to ATO. The nephrotic level of MDA, GSH, SOD, CAT, and GSH-Px was measured in tissue homogenate at the end of treatment period. The level of significance was set P < .05 by using one-way ANOVA followed by Tukey’s multiple comparison test. The results are expressed as mean ± SD, n = 6. *** represents P < .001, ** represents P < .01, * represents P < .05, and ns = non-significant. Abbreviations: MDA: malondialdehyde, GSH: glutathione, SOD: superoxide dismutase, CAT: catalase, GSH-Px: glutathione peroxidase, ATO: arsenic trioxide, RSV: resveratrol, AA: ascorbic acid.
Effect of Pre-Treatment on ATO-Induced Liver Damage
The serum level of ALT, AST, and ALP were measured to evaluate the effect of arsenic-induced toxicity and treatment of AA and RSV on ATO-induced toxicity. The effect of RSV pre-treatment on liver functions in arsenic-induced rats is shown in Figure 5. We observed that treatment with arsenic significantly increased (P < .001) the serum level of ALT, AST, and ALP relative to control group. The pre-treatment with RSV and AA resulted in restoring the serum levels of these parameters as compared to ATO group. Effect of pre-treatment on serum level of (A) ALT, (B) AST, and (C) ALP in ATO-exposed groups. The serum level of ALT, AST, and ALP was measured at the end of treatment period. The level of significance was set at P < .05 using one-way ANOVA followed by Tukey’s multiple comparison test. The results are expressed as mean ± SD, n = 6. *** represents P < .001, ** represents P < .01, * represents P < .05, and ns = non-significant. Abbreviations: ALT: alanine aminotransferase, AST: aspartate transaminase, ALP: alkaline phosphatase, ATO: arsenic trioxide, RSV: resveratrol, AA: ascorbic acid.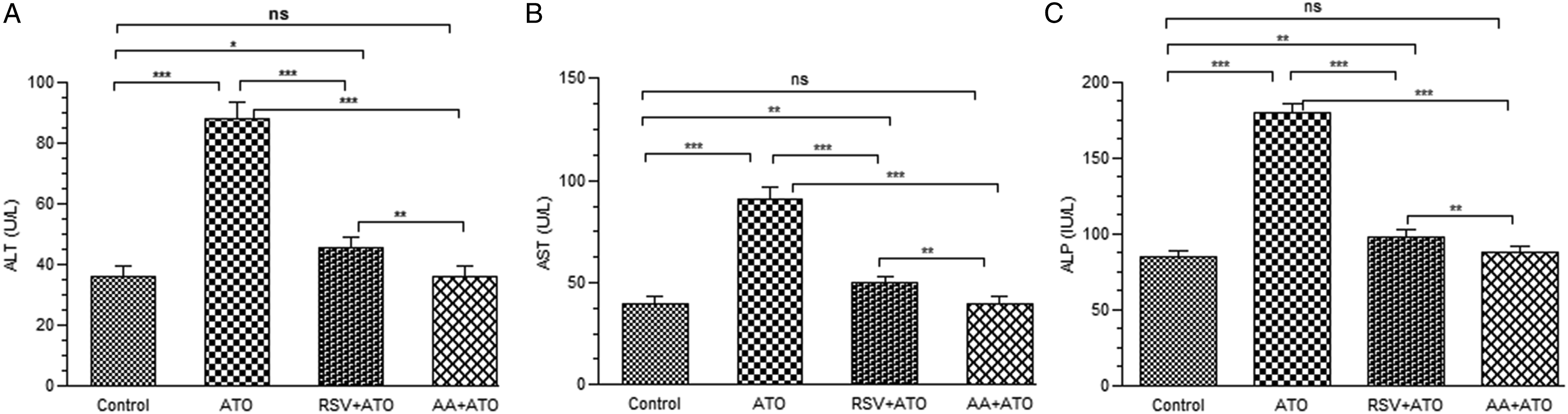
Effect of Pre-Treatment on ATO-Induced Renal Damage
The serum level of BUN and creatinine was measured to evaluate the effect of arsenic-induced toxicity in kidney and pre-treatment effects of AA and RSV on arsenic-induced toxicity (Figure 6). Arsenic significantly increased (P < .001) the serum level of BUN and creatinine relative to control group. However, RSV treatment significantly declined (P < .001) the serum levels of these parameters as compared to ATO group. The results showed the non-significant difference (P > .05) between the control group and pre-treated AA group. Effect of pre-treatment on serum level of (A) BUN and (B) Cr in experimental rats groups exposed to ATO. The serum level of BUN and creatinine was measured at the end of treatment period. The level of significance was set at P < .05 using one-way ANOVA followed by Tukey’s multiple comparison test. The results are expressed as mean ± SD, n = 6. *** represents P < .001, ** represents P < .01, * represents P < .05, and ns = non-significant. Abbreviations: BUN: blood urea nitrogen, Cr: creatinine, ATO: arsenic trioxide, RSV: resveratrol, AA: ascorbic acid.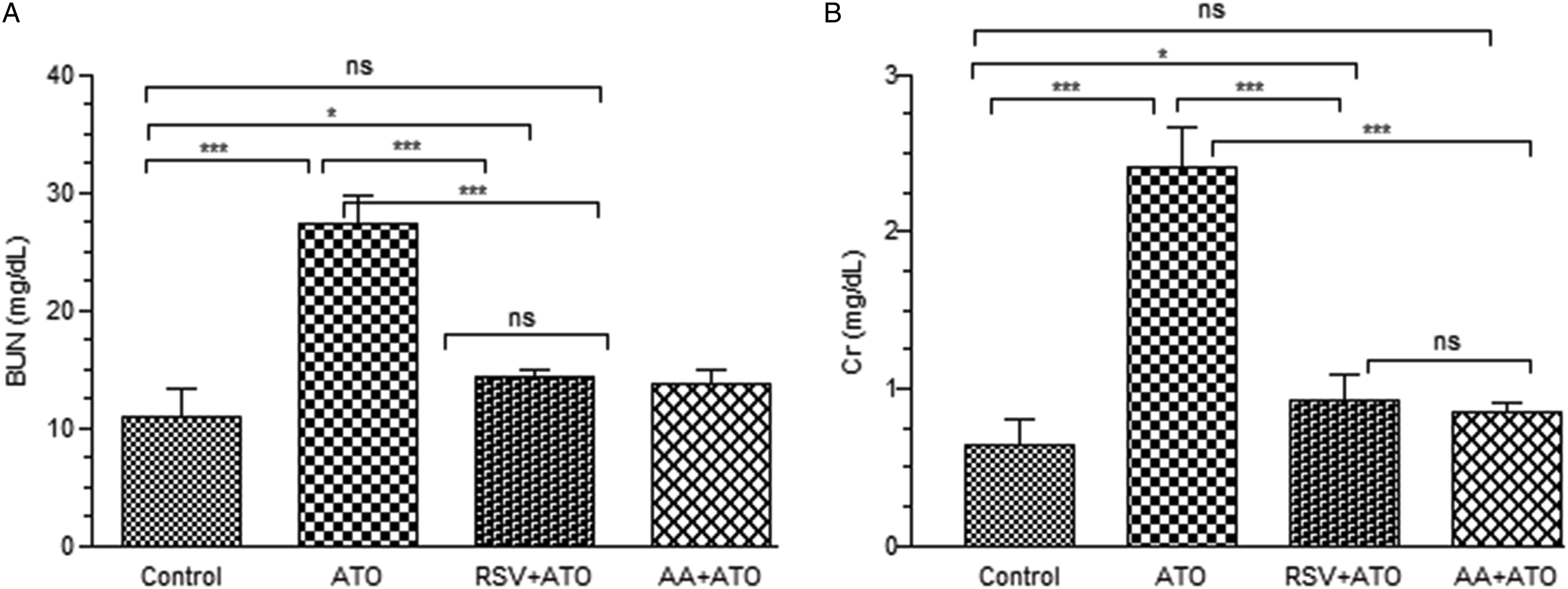
Histopathological Examination
The alterations in abovementioned liver function biomarkers due to exposure of arsenic are correlated with histological alterations in liver (Figure 7). The liver sections of control group showed normal architecture having hepatocytes regular arrangement with characteristic pattern of hexagonal lobules, visible central vein, and nuclei (Figure 7A). The arsenic-exposed group showed abnormalities in liver including steatosis, cytoplasmic vacuolization, infiltration of inflammatory cell, and focal necrosis (Figure 7B). Our findings suggested that the pre-treatment with RSV and AA showed mild focal necrosis focal and cytoplasmic vacuolization as shown in figures 7C and 7D. The figure shows the pre-treatment effect on liver structure of arsenic-exposed rat. The histology of control group (A) showing normal architecture of liver tissues. The arsenic-exposed group (B) showing steatosis (brown arrow), cytoplasmic vacuolization (red arrow), infiltration of inflammatory cell (red circle), and focal necrosis (black circle). The effect of pre-treatment with RSV (C) and AA (D) showing mild alterations as compared to arsenic-exposed group.
We have also observed that change in the serum level of kidney function biomarkers due to exposure of arsenic has impact on morphology of kidney as shown in Figure 8. But pre-treatment with RSV and AA has effect to restore the normal structure. The control group showed that normal structure of renal tissue having glomerulus embedded in large number of various tubules and no morphological changes are observed (Figure 8A). The arsenic-exposed group showed tubular dilation, hemorrhage, sever nephritis, and disruption of glomeruli (Figure 8B). The pre-treatment with RSV and AA group showed no hemorrhage and mild nephritis as shown in Figures 8C and 8D. The figure depicts the pre-treatment effect on kidney structure of arsenic-exposed rat. The histology of control group (A) showing normal architecture of renal tissues. The arsenic-exposed group (B) showing tubular dilation (red arrow), hemorrhage (black arrow), sever nephritis (red circle), disruption of glomeruli (brown arrow), and interstitial edema (yellow arrow). The effect of pre-treatment with RSV (C) and AA (D) showing mild tubular dilation and nephritis.
Discussion
The results of present study revealed that hepatic and renal oxidative damage induced by As2O3 ameliorated by pre-treatment with RSV and AA. The results also suggested that RSV decreased the arsenic accumulation in liver and kidney of As2O3-exposed rats which may be a contributing factor for reducing hepatic and renal toxicity. As2O3 has been used efficaciously for treatment of APL. But dose-dependent toxicity of As2O3 is responsible for its limited therapeutic uses. 31 The arsenic exposure for prolonged period can induce multiple toxic effects on various organs via complicated mechanisms. 32 The metals can induce toxicity in healthy individual by various mechanisms. The clear mechanism by which As2O3 can induce the nephrotoxicity and hepatotoxicity is not still clearly understood. However, oxidative stress is considered as crucial contributing and triggering factor for inducing metal toxicity and has strong association with accumulation of metals in organs. 33 Metal accumulation in body has persistent potential for damaging different organs and tissues. Arsenic can accumulate in many organs including heart, liver, lungs, kidneys, spleen, and muscles.34,35 In human, ingested arsenic can interfere with many biochemical reactions and disturb various physiological activities in different organs of body.36,37
Currently, there are a large number of bioactive compounds used as metal-chelators and synthetic antidotes to reduce metal-induced toxicity. In recent years, natural bioactive compounds have gained attention and studied to investigate their mechanism of action through which they attenuate toxicity and different pathological conditions. 38 Natural antioxidants, mainly flavonoids, have potential to prevent or reduce oxidative damage in different pathological conditions. Flavonoids chemically contain one or more aromatic hydroxyl groups which play vital role in performing antioxidant activity and produce beneficial effects on health. 39 RSV is a polyphenolic compound having bioactive effects on human health. RSV has many therapeutic applications to reduce various types of renal and hepatic injuries in animal model. 40 In this study, we investigated that RSV has ameliorating effect against arsenic-induced toxicity in rat model with variations in biochemical parameters, oxidative stress, and retention of arsenic along histopathology of kidney and liver.
Arsenic can accumulate in targeted tissues and lead to abnormalities in structure as well as functions of organs. The pre-treatment with RSV is significantly helpful in reducing the concentration of arsenic and restoring the functions of targeted tissues. RSV facilitates the metabolism of arsenic by reducing consumption of glutathione. Previous studies affirmed that arsenic-containing compounds metabolize and generate ROS in excessive amount. The increased level of ROS induces lipid peroxidation of MDA and oxidative stress leading to impairment in endogenous antioxidant defense systems and damages the macromolecules in cell including proteins, DNA, and lipids resulting in abnormalities in cellular functions and structure.41,42 This study affirmed that administration of arsenic-induced oxidative stress in liver and kidney of rats which is indicated by perturbations in renal and hepatic anti-oxidative biochemical parameters, and these can be treated with RSV, prior to administration of arsenic. MDA is considered as an index to access oxidative damage. However, this study showed that pre-treatment with RSV in arsenic-exposed rats prominently decreased MDA level in renal and hepatic tissues as compared to ATO-treated group. Arsenic has high affinity for thiol group and it binds with GSH. 43 SOD is an enzyme that prevents toxic effects of superoxide ion by enhancing its dismutation into less toxic compound (hydrogen peroxide) and prevents the generation of excessive free radicals. Hydrogen peroxide is converted into non-toxic molecules; water; and oxygen by CAT. 44 GPx is an antioxidant enzyme and reduces lipid hydroperoxides into lipid alcohols in the membranes. 45 During metabolism of arsenic, reduction in activity of SOD in liver and kidney of arsenic-exposed animals has been observed. 46 The increased level of superoxide radical inhibits the CAT activity. 47 Depletion in activity of GSH, SOD, CAT, and GSH-Px has been observed in arsenic-treated rats and these enzymes have vital role in oxygen metabolism against toxic effects of arsenic. The results of this study are consistent with previous reported papers.48,49 The pre-treatment with RSV in arsenic-exposed rats restored the level of GSH, SOD, CAT, and GSH-Px in liver and kidney.
This study aimed to explore the mechanism by which RSV improves the hepatic and renal functions. RSV showed protective effects against ATO toxicity by modulation in cellular redox system via increasing free radical scavenging activity and improving antioxidant defense system. 50 RSV performs free radical scavenging activity by competing with coenzyme Q for reduction of oxidative chain complex, which is major site for production of ROS and scavenging of superoxide produced in mitochondria. 51 The findings of our study as mentioned above support this suggestion. RSV has many beneficial effects in improving activities of antioxidant enzymes and reducing ROS level 50 as well as attenuating mitochondrial complexes I, III, and IV activities. 52 It has also been suggested that RSV activates peroxisome proliferator–activated receptor gamma coactivator 1-alpha which induces the expression of various ROS detoxifying enzymes such as GPX, CAT, and SOD. 53 It has also been proved that RSV inhibits NF-kappa B, JNK, c-Jun/AP-1, activation of caspase, and apoptosis which induce due to products of lipid peroxidation. 54 Additionally, RSV decreases genomic instability and inflammation and increased respiration and biogenesis. 55
Arsenic metabolism occurs in liver and undergoes biotransformation by binding with thiol groups of enzymes or proteins in liver. The metabolism of arsenic disrupts the hepatic plasma membrane integrity leading to leakage of ALT and AST in the serum. 56 In case of liver inflammation or injury, ALT and ALP levels in serum increased. 57 Our results demonstrated that ALP, AST, and ALT levels in serum of arsenic-exposed rats increased. However, pre-treatment with RSV in arsenic-exposed rats showed that RSV could significantly prevent elevation of these enzymes in liver as our findings comply with previously reported studies.57-60
Arsenate can replace phosphate group in the glycolytic and cellular respiration pathways because it is a chemically phosphate analog. 61 Arsenite can bind sulfhydryl groups of many proteins and ultimately disturb glucose metabolism. 62 It also interferes in many biochemical processes including uptake of glucose, gluconeogenesis, oxidation of fatty acid, and pyruvate dehydrogenase activity leading to decreased activity of citric acid cycle and reduced production of cellular ATP. 63 The methylated products of arsenic can cause damage of DNA via exchange of sister chromatid, aberrations of chromosomes, and DNA hypo- and hyper-methylation. 64 Depletion of ATP, depolarization of mitochondrial membrane, and apoptosis of endothelial cells cause damage to glomerulus membrane 65 which is indicated by changes in histopathology of liver along with increased level of BUN and creatinine. Kidney is a major organ for excretion of number of compounds and has large perfusion and that is why kidney is vulnerable to arsenic-induced damage and increased arsenic concentration in renal tubular cells. 66 Our results suggested that level of BUN and creatinine in serum of arsenic-exposed rats increased significantly leading to renal damage. The pre-treatment with RSV in arsenic-exposed rats demonstrated that RSV could restore the level of BUN and Cr in serum. Our results are in accordance with previous published finding.67,68
The histopathological examination of liver and kidney tissues demonstrated that many deformities in structure of liver and kidney have been observed due to arsenic exposure. The observed alterations in histology of liver are consistent with previous findings58,68,69 that include steatosis, cytoplasmic vacuolization, infiltration of inflammatory cell, and focal necrosis. The observed alterations in histology of kidney are corroborated by similar observations of kidney pathology in rats, 31 which include tubular dilation, hemorrhage, severe nephritis, and disruption of glomeruli. The histological aberrations in tissues of kidney could be due to over-accumulation of arsenic in renal tissues. Our findings confirmed that pre-treatment with RSV in arsenic-exposed rats prevented hepatic and renal toxicity. We have also investigated a relation between accumulation of arsenic in targeted tissues (liver and kidney) and ability of RSV to reduce arsenic concentration. We observed that RSV has significant role in reducing arsenic concentration and showed hepatoprotective and nephroprotective effect by recovering the abnormalities in architecture of liver and kidney. RSV showed comparable results with standard and that is why it can be used as remedial agent to reduce toxicity induced by arsenic. RSV has protective effect against arsenic which is dependent upon glutathione-dependent efflux from cell. 70 Arsenic metabolism depletes GSH content which enhances the arsenic retention in the mammalian cells and increases sensitivity to arsenic. 71 Our findings explained that pre-treatment with RSV in arsenic-exposed rats maintained GSH level in cells. 24
The mechanism by which RSV mediated arsenic toxicity is as follows and shown in Figure 9. GSH as a reducing agent plays a role in methylation of arsenic and also conjugates with arsenic leading to depletion of GSH and production of ROS.
72
RSV helps in methylation pathways of arsenic by reducing the depletion of GSH and induction of ROS. GSH conjugated arsenicals export to extracellular space against concentration gradient by ATP-binding cassette transporters; multidrug-resistant P-glycoproteins; and multidrug-resistant proteins.
73
RSV also facilitates the metabolic pathway of arsenic by enhancing respiration and mitochondrial biogenesis which inhibit the depletion of ATP and supply sufficient amount of energy for excretion of arsenic.
74
RSV also decreases the possibility of DNA damage by upregulation of As+3 methyltransferase that involves in arsenic methylation and catalyzes glutathione S-transferase pi 1 which was involved in detoxification of arsenic via an efflux system.
75
Graphical summarization of RSV to ameliorate arsenic-induced toxicity. Abbreviations:GSH: glutathione, SAM: S-adenosyl methionine, MMA: monomethyl arsenic, DMA: dimethyl arsenic, ROS: reactive oxygen species, SOD: superoxide dismutase, CAT: catalase, GPx: glutathione peroxidase.
Conclusively, the experimental study revealed that RSV pre-treatment in arsenic-exposed rats effectively mitigated toxic effects of arsenic by recovering antioxidant system, modulating the redox pathway, and decreasing the arsenic accumulation in targeted tissues. However, ATO, an anticancer agent, can be used clinically in combination with RSV to protect an individual from hepatotoxicity and nephrotoxicity. RSV can be used as remedial therapy for an individual exposed to arsenic in environment.
Conclusion
It has been concluded that metabolism of arsenic is responsible for elevation of oxidative stress owing to depletion of GSH and increased level of ROS. However, this study confirmed the molecular pathway by which arsenic-induced oxidative damage improved by RSV pre-treatment. RSV did not only ameliorate the liver and kidney function but also improved their architecture by decreasing arsenic retention in them. Consequently, it has been affirmed that RSV has a significant therapeutic approach as an antioxidant to attenuate the arsenic toxicity and demand additional investigation to use as remedial agent in different pathological conditions. The outcomes of RSV are appreciable and there is need to explore its maximum output to focus on its medicinal uses and properties. This study may be helpful in future for improvement in the prognostic and novel therapeutic interventions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the publication of this article. While, this research work has been financially supported by the research grant (5661/Punjab/NRPU/R&D/HEC/2016, 6429/Punjab/NRPU/R&D/HEC/2016 and 8365/Punjab/NRPU/R&D/HEC/2017) received from Higher Education Commission (HEC) of Pakistan.
Compliance With Ethical Standards
All applied procedures were approved by the Bioethics committee and Institutional review board (GCUF/ERC/2189) from Government College University, Faisalabad, Pakistan.
