Abstract
Background
Cancer of unknown primary (CUP) is metastatic at diagnosis with an unknown primary site, indicating a high degree of malignancy with a poor prognosis. The development and application of targeted therapy and immunotherapy are current research hotspots, which provide additional treatment options for CUP.
Case Presentation
A 36-year-old male presented with pain on the right hip in April 2018. After various examinations, he was diagnosed with CUP. This patient received chemotherapy, immunotherapy, and local radiotherapy in our department. However, the use of radiotherapy after immunotherapy resulted in severe pneumonia.
Conclusion
Compared with traditional treatments, immunotherapy is an effective treatment with fewer side effects and better patient tolerance. However, treating physicians should be still pay special attention to the occurrence of side effects when radiotherapy is combined with immunotherapy.
Background
Cancer of unknown primary (CUP) is a mysterious type of cancer. It is metastatic at diagnosis with an unknown primary site, indicating a high degree of malignancy with a poor prognosis. 1 Alcohol consumption and cigarette smoking have a correlation with increasing CUP risk. 2 CUP accounts for approximately 2–5% of all new tumors. 3 In a previous study, CUP was diagnosed as a malignant tumor and metastasis was confirmed through histology. The current standard treatment approaches for CUPs often need the recognition of the primary sites. 4 However, despite a comprehensive diagnostic work-up, the primary site was not determined. 5 The systemic treatment for putative primary based on RNA sequencing is not superior to standard empirical chemotherapy. 6 The development and application of targeted therapy and immunotherapy are current research hotspots, which provide more treatment options for CUP.
Recently, a patient with CUP and multiple systemic metastases was treated using immunotherapy combined with chemotherapy and radiotherapy in our department. The pathology of this patient indicated that the tumor derived from the digestive tract. However, the primary site was not found during gastroscopic and colonoscopic examinations. Moreover, chemotherapy for gastrointestinal carcinomas was not effective for this patient. Therefore, pathology was not the gold standard for CUP. Immunotherapy was an effective treatment for this patient. However, care must be exercised when radiotherapy is co-administered with immunotherapy.
Case Presentation
A 36-year-old free working male presented with pain on the right hip in April 2018. Positron emission computed tomography (CT) was performed, demonstrating probable bone metastasis and surrounding soft tissue masses in the pelvis (Figure 1(a)) and the upper lobe of the right lung (Figure 1(b)). Emission CT of the bone was performed, which revealed bone metastasis. Pelvic puncture of the soft tissue masses in the posterior superior acetabulum and the iliac crest indicated poorly differentiated mucinous adenocarcinoma. Immunohistochemistry confirmed the presence of mucinous adenocarcinoma, with some cells being signet-ringoid, cytokeratin 7 (CK7) (+), CK19 (+), thyroid transcription factor-1 (−), anaplastic lymphoma kinase-CST (−), CK20 (−), caudal-related homeodomain transcription 2 (CDX2) (−), and Napsin A (−). Pathological consultation at the Fudan University Shanghai Cancer Center indicated that the tumors were derived from the digestive tract. However, following gastroscopic and colonoscopic examination, the primary site remained unknown. The patient was subjected to immune-related gene testing. The results showed programmed cell death 1 ligand 1 (PD-L1) positivity (25%), the tumor mutational burden (10.48%), and mutation abundance of exon 5 in TP53 (49.49%). Therefore, the patient was diagnosed with CUP. Timeline of a case of CUP with multiple systemic metastases treated with a combination of immunotherapy and chemotherapy. (A) PET–CT imaging of the patient prior to any treatment, showing bone metastasis and a surrounding soft tissue mass in the pelvis. (B) The mass in the lung was a soft tissue showing a maximum cross-section of 42 × 56 mm. (C) The mass on the chest wall had a size of 15 × 15 mm. (D) Lung CT imaging of the patient after treatment with XELOX chemotherapy. The mass in the lung showed a maximum cross-section of 68 × 44 mm. (E) CT imaging of the pelvis for last review. (F) Lung CT imaging after chemotherapy and immunotherapy. The mass in the lung showed a maximum cross-section of 35 × 24 mm. CUP, cancer of unknown primary; PET–CT, positron emission tomography computed tomography.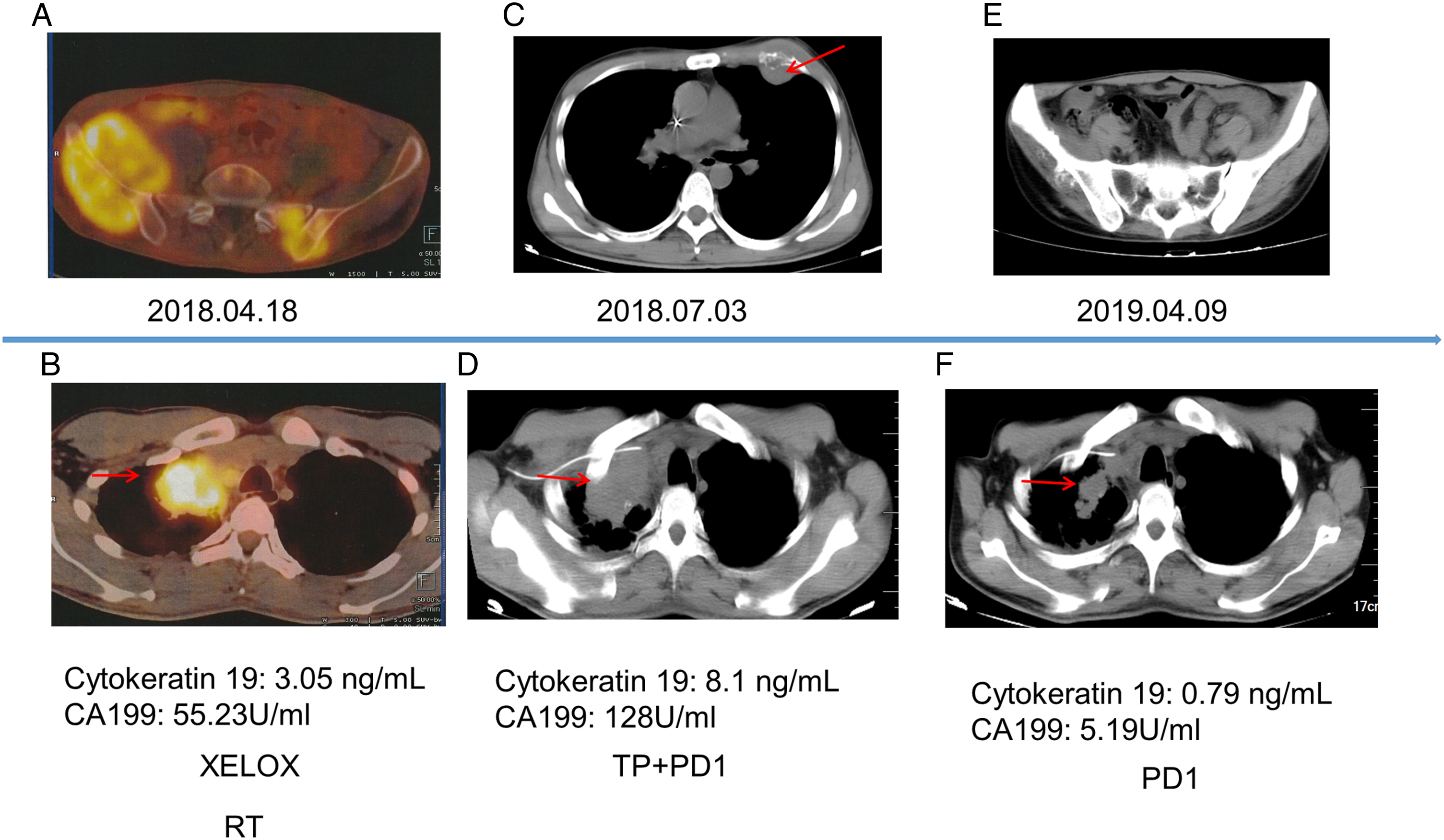
In May 2018, the patient received palliative care to alleviate the pain and special conformal radiotherapy with DT40GY/20F for the bone metastasis and surrounding soft tissue mass (Figure 1(a)). Subsequently, in June 2018, he received chemotherapy with the XELOX regimen (i.e., capecitabine 1500 mg in the morning and 2000 mg in the evening
During the treatment period, the pain in the left ilium intensified. Conformal radiotherapy with DT40GY/20F to the site was initiated on June 15, 2018. The size of the mass on the chest wall was 15 × 15 mm (Figure 1(c)), and the mass in the lung increased in size (Figure 1(d)). These findings indicated a progressive disease. A biopsy of the mass of the chest wall was performed to identify the primary lesion. Based on the morphology with H&E staining, ×40 (Figure 2(a)) and H&E staining, ×100 (Figure 2(b)), the pathological analysis indicated the presence of poorly differentiated mucinous adenocarcinoma, solid type (signet-ring cell). Immunohistochemistry results excluded mesothelium and mammary gland sources. Based on clinical endoscopy and other tests, the digestive tract was excluded as the source of the cancer. It was considered that the tumor originated in the lung. Subsequently, the patient received second-line chemotherapy (paclitaxel 200 mg d1–8 plus cisplatin 30 mg d1–3) for two cycles on July 3, 2018 and July 31, 2018. The levels of tumor marker carcinoembryonic antigen (CEA) and carbohydrate antigen 199 (CA199) progressively declined (Figure 3). The mass on the chest wall was composed of poorly differentiated mucinous adenocarcinoma of the solid type. (A) H&E staining, ×40. (B) H&E staining, ×100. H&E, hematoxylin, and eosin The changes of tumor markers during the treatment. CA199 and CEA increased after treated by “XELOX” chemotherapy, but decreased after treated by “paclitaxel + cisplatin” chemotherapy combined with immunotherapy. CEA carcinoembryonic antigen and CA199 carbohydrate antigen 199.

From September 8, 2018, the patient was treated with immunotherapy (nivolumab [Bristol-Myers Squibb, Opdivo, Brooklyn, NY, USA] 100 mg/m2) every 2 weeks. Nivolumab was approved by the China Food and Drug Administration on June 15, 2018. Meanwhile, chemotherapy was used continually, and suspended after six cycles. Subsequently, the patient continued to receive treatment with immunotherapy. The levels of CEA, cytokeratin, and CA199 progressively decreased. In addition, the size of the masses in the lung and pelvis was also reduced (Figures 1(e) and 1(f)). The observed changes in tumor markers were consistent with the therapeutic efficacy and tumor load (Figure 3).
According to the follow-ups performed between October 2018 and April 2019, the patient had been able to conduct normal daily life activities, with a markedly improved quality of life and absence of additional abnormalities.
However, the patient presented with recurrent cough and hemoptysis on April 18, 2019. Lung CT imaging revealed the presence of new tissues (Figure 4(a)). These new tissues detected in the right main bronchus were removed using an endoscope (Figure 4(b)). The size of the largest new tissue was 3 × 1 cm, and the pathological diagnosis suggested hemorrhage and fibrous exudation (Figure 4(c)). The new tissues were detected through a second respiratory endoscopy suggested the presence of malignant epithelial tumors of the salivary gland type, based on the morphology of H&E staining, ×40 (Figures 5(a)) and H&E staining, ×100 (Figure 5(b)). The immunohistochemistry results showed CD117(+/−), DOG-1(−), CK7(+), CK5/6(−), P63(−), CEA minority (+), thyroid transcription factor-1(−), Napsin A (−), CR (−), P40(−), P53 mutant, smooth muscle actin (+), S-100(−), and Ki-67(+) (approximately 50%). The masses did not show significant change in the lung CT image (Figure 4(d)). These findings indicated progressive disease. After consultation with the Fudan University Shanghai Cancer Center, the use of local radiotherapy was suggested after discontinuation of immunotherapy. In June 2019, the patient received radiotherapy with DT45GY/15F for the lesions in the right lung, and mediastinal and hilar lymph nodes. The treatment alleviated the patient’s cough and hemoptysis did not recur. Two weeks later, it was observed that the mass in the lung had decreased in size (Figure 4(e)). However, interstitial inflammation occurred after DT36Gy/12F radiotherapy (Figure 4(f)). Radiotherapy was discontinued and anti-infective and anti-inflammatory treatments were administered. The inflammation in the lung was resolved (Figure 4(g)). In the end, this patient died from recurrent pneumonia in December 2019. Timeline of a case of CUP with multiple systemic metastases treated with radiotherapy after immunotherapy. (A) New tissues in the lung CT images of the patient. (B) The new tissues in the right main bronchus under the endoscope. (C) The largest new tissue in the bronchus. (D) The mass did not show significant change in the lung CT image. (E) The mass in the lung decreased in size. (F) Interstitial inflammation appeared on the CT image. (G) Interstitial inflammation in the lung was reduced. CUP, cancer of unknown primary; CT, computed tomography. The new tissues detected through second respiratory endoscopy mainly comprised malignant epithelial tumors of the salivary gland type. (A) H&E staining, ×40. (B) H&E staining, ×100.

Discussion and Conclusion
The treatment strategy for CUP is challenging. Poorly differentiated neuroendocrine tumors are invariably characterized by aggressive clinical behaviors, and are usually associated with a poor prognosis. Typically, CUP responds to chemotherapy (cisplatin and etoposide) with 70−80% overall response rates. 7 Ohta et al 7 found that patients with CUP who received chemotherapy showed a relatively good prognosis. However, chemotherapy has no impact on overall survival. Notably, gene detection can identify the source of CUP tissues, while another promising strategy involves liquid biopsies that detect circulating tumor DNA in patients with CUP. The detection of gene mutations (e.g., mismatch repair and tumor mutational burden) may help with immunotherapy, and plays an important role in the development of treatment strategies against CUP. 8
Surgery is usually the first choice for patients with cancer. However, when this patient with CUP was diagnosed, the bone metastases was already present. Therefore, surgical resection was not possible for this patient. The chemotherapy with the XELOX regimen was performed since pathological results showed that the tumor was derived from the digestive tract. However, the levels of CEA and CA199 progressively increased, which indicated that the chemotherapy was not effective. After changing the chemotherapy regimen to paclitaxel and cisplatin, the levels of CEA and CA199 declined. The changes in tumor marker levels were related to the progression of the disease. 9
Immunosuppressive agents carry considerable potential for the treatment of patients with cancer, such as advanced non-small cell lung cancer (NSCLC) and small cell lung cancer. 10 Programmed cell death protein 1 (PD1) is a member of the immunoglobulin family B7-CD28. It is expressed in monocytes, B cells, activated T cells, and dendritic cells as an immunosuppressive receptor. 11 In healthy organisms, PD1 is mainly expressed in activated immune cells, promoting the maturation of T cells and regulating excessive immune responses through a negative regulatory mechanism. PD-L1 and PD-L2 are ligands for PD1. Over-activation of the PD1/PD-L1 pathway leads to inhibition of the immune response, 12 thereby preventing excessive inflammatory responses. PD-L1 is the major ligand, and its expression in tumor tissues (e.g., NSCLC, breast cancer, melanoma, gastric cancer, and renal cancer) is significantly higher than that recorded in normal tissues.13-17 Blockage of PD1/PD-L1 inhibits the proliferation and viability of T and B cells. It plays an important role in tumor immune escape and promotes the progression of tumors. Overexpression of PD1 ligands in tumor cells cause weakening or even death of T cells. 18 Therefore, blockage of PD1/PD-L1 enhances the effect of immune cells on tumor cells, and has shown benefits based on clinical trials.
Nivolumab is one of the drugs which block PD1/PD-L1. In 2015, it was reviewed by the U.S. Food and Drug Administration for the treatment of previously treated advanced NSCLC. The U.S. Food and Drug Administration approved nivolumab as the immune checkpoint inhibitor for the treatment of this disease. In a phase 3 clinical trial (CheckMate-017), 51,272 patients with lung squamous cell carcinoma were randomized to receive either nivolumab or platinum and docetaxel. The results showed that nivolumab had a significantly higher response rate than the docetaxel regimen (20% vs 9%; P=.008), and significantly prolonged progression-free survival and overall survival. Clinically, approximately 60% of patients with NSCLC have abnormal PD-L1 expression. 19 However, the correlation between the expression of PD-L1 and prognosis remains unclear. It is recommended to determine the expression of PD-L1 in NSCLC patients without a driver gene mutation. Immunosuppressive therapy or combined chemotherapy with PD-1 is recommended for patients with high PD-1 expression, demonstrating the new direction toward individualized therapy for NSCLC based on gene detection. Therefore, it is emphasized that gene testing should be performed to determine the presence of gene mutations and PD-1 expression in patients prior to the administration of lung cancer-targeted drugs and immune drugs. Another phase II study found that PD-L1 expression in patients with lung squamous cell carcinoma was not related to treatment efficacy. After treatment with an anti-PD-L1 agent, partial remission was achieved in 24% of PD-L1-positive patients and 14% of PD-L1-negative patients. Currently, there are many challenges in clinical practice that require urgent attention. For example, the PD-1/PD-L1 monoclonal antibody is not effective in all patients. Therefore, it is especially important to select the population that would benefit the most from this therapy. The CheckMate-227 study showed that the tumor mutational burden may be helpful in predicting the efficacy of immunotherapy. 20 PD-1 immunotherapy may be appropriate for patients carrying TP53 and KRAS mutations. 21 Currently, there is no gold standard for the detection of PD-L1, and it remains unclear whether PD-L1 could become an ideal molecular marker. Nivolumab was listed in China as second-line treatment for NSCLC in 2018. Therefore, there is less data regarding its use in clinical practice in China. The use of immunotherapy for this patient in our case report was new and challenging.
Local radiotherapy was also used in this case, resulting in a certain therapeutic effect against CUP. The patient received three-time radiotherapy. The first two radiotherapy sessions were performed to control the pain from bone metastasis using a standard dose. The last radiotherapy session was performed to treat the right lung, and mediastinal and hilar lymph nodes. Due to the immunotherapy, the total dose and duration of treatment of radiotherapy were reduced (DT45GY/15F) to decrease the side-effects. In a case of primary retroperitoneal serous adenocarcinoma, the combination of immunotherapy with radiotherapy decreased the tumor size, and subsequently led to durable disease control. 22 Another research study showed that radiotherapy may improve systemic responses to anti-PD1/PD-L1-directed immune therapy in Merkel cell carcinoma, and immunotherapy clearly showed abscopal regression. 23 Coincidentally, a case report also suggested that immunotherapy increases the chance of an abscopal effect occurring after radiation therapy in a patient with lung adenocarcinoma treated with atezolizumab and subsequently combined with brain irradiation for the treatment of metastasis. 24 However, unexpectedly, serious interstitial pneumonia in the contralateral lung field was noted in the present case, which may have been related to long-term use of immunotherapy. Traditional radiation pneumonitis is usually associated with the range of exposure and dose distribution. However, the range of interstitial pneumonia was mainly observed in the contralateral lung field in this case. Moreover, the contralateral lung only received a low-dose radiation. This suggests that low-dose radiation may be the trigger for the activation of immune pneumonia. Alternatively, the paracrine signaling of the tumor may trigger this immune pneumonia. Radiation induces bystander response and abscopal effects through a series of biological effects in irradiated cells, or cellular communication with non-irradiated local neighbors or distant cells. 25 This has been adopted by an increasing number of scholars. However, distinguishing between radiation pneumonitis and immune checkpoint blockade-related pneumonitis remains important and challenging. 26
Therefore, the combination of immunotherapy with radiotherapy requires caution due to the risk of immune inflammation. Further research is warranted to identify the optimal approaches for incorporating radiotherapy in combination treatments with respect to the dosage, as well as the fractionation and sequencing of therapies. 27
We reported a unique case of CUP with multiple systemic metastases in a 36-year-old male. In patients in whom the primary tumor site cannot be identified, apart from conventional chemotherapy and radiotherapy, immunotherapy can also be an effective treatment with fewer side effects and better patient tolerance. Moreover, the use of radiotherapy after immunotherapy requires caution due to the risk of immune inflammation.
Footnotes
Acknowledgments
Availability of Data and Materials
All datasets generated for this study are included in the manuscript and/or the supplementary files.
Author Contributions
PW and SD provided this case. QN, KL, and CP wrote, reviewed, and/or modified the manuscript. All authors have read and approved the manuscript
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Human Ethics Review Committee of Jiangsu Taizhou People’s Hospital.
Informed Consent
Written informed consent to publish this report and the associated medical images was provided by the patient’s wife.
