Abstract
This work concerns study of self-absorption factor (SAF) and dose rate constants of zirconium-89 (89Zr) for the purpose of radiation protection in positron emission tomography (PET) and to compare them with those of 18F-deoxyglucose (18F-FDG). We analyzed the emitted energy spectra by 18F and 89Zr through anthropomorphic phantom and calculated the absorbed energy using Monte Carlo method. The dose rate constants for both radionuclides were estimated with 2 different fluence-to-effective dose conversion coefficients. Our estimated SAF value of 0.65 for 18F agreed with the recommendation of the American Association of Physicists in Medicine (AAPM). The SAF for 89Zr was in the range of 0.61-0.66 depending on the biodistribution. Using the fluence-to-effective dose conversion coefficients recommended jointly by the American National Standards Institute and the American Nuclear Society (ANSI/ANS), the dose rate at 1 m from the patient for 18F was 0.143 μSv·MBq−1·hr−1, which is consistent with the AAPM recommendation, while that for 89Zr was 0.154 μSv·MBq−1·hr−1. With the conversion coefficients currently recommended by the International Committee on Radiological Protection (ICRP), the dose rate estimates were lowered by 2.8% and 2.6% for 89Zr and 18F, respectively. Also, we observed that the AAPM derived dose is an overestimation near the patient, compared to our simulations, which can be explained by the biodistribution nature and the assumption of the point source. Thus, we proposed new radiation protection factors for 89Zr radionuclide.
Introduction
Immuno-PET imaging uses monoclonal antibodies (mAbs) labeled with radionuclides for positron emission tomography. Unlike 18F-FDG which is a relatively non-specific tracer, mAbs are designed to specific antigenic sites such as growth factor receptors. 18F (t 1/2 = 109 min), 64Cu (12.7 hr), 68 Ga (68 min), 76Br (16 hr), 86Y (14.7 hr), 89Zr (78.4 hr) and 124I (100.2 hr) have been used in immuno-PET. 1 Typical injected activity of 18F-FDG ranges from 100 MBq to 500 MBq.2 89Zr gives good diagnostic quality at administered activities as low as 37 MBq 2,3 and is sufficiently long-lived (t 1/2 = 78.4 hr) to allow serial imaging up to approximately 1 week post-administration. 4 It can be produced in medical cyclotrons by proton irradiation of an 89Y foil. 5 89Zr decays to 89Y through β+ emission (22.7%) and electron capture (77.3%) followed by a prompt γ-ray at 909 keV (99.04%). 6
In a Monte Carlo study, for radiation protection purposes, when using 89Zr in an imaging facility that is optimized for the annihilation photons only, the results showed that the low 89Zr injected activity (75 MBq) would deliver a lower effective dose near the patient than 18F (500 MBq) would; however, 89Zr would give a higher effective dose than the 18F outside the patient room due to the greater penetration power of the prompt γ-ray. 6
In PET procedures utilizing 18F-FDG, the 511 keV annihilation radiation is the main radiation protection concern, delivering a dose to the staff of 2.7-4.0 mSv during injection and 3.5-5.0 mSv during patient set-up. 7 The dose to the general public in the waiting area is about 0.8 mSv per procedure. 2 The dose to the staff and the general public depends on the positron emitter, the facility layout, the injected activity and the work load. The patient’s physiology affects the accumulation of the tracer while his size affects the attenuation of the radiation. Thus, the dose rate D(x) in μSv·hr−1 at a distance x, in m, from the patient can be estimated by the following equation:
where A 0 in MBq is the activity adjusted for biological and physical decay in the patient, DRC the dose rate constant in μSv·m2·MBq−1·hr−1 and SAF the dimensionless self-absorption factor. The DRC is the constant of proportionality assuming that the dose rate is inversely proportional to the square of the distance from a point source. The SAF is defined as the ratio of the amount of energy absorbed by the patient’s body to the amount of energy emitted by the radionuclide. 6 The American Association of Physicists in Medicine (AAPM) has recommended values of DRC and SAF for some commonly used radionuclides such as 11 C, 18F, 124I, for example, DRC = 0.143 μSv·m2·MBq−1·hr−1 and SAF = 0.64 for 18F. 8 The AAPM values for 18F are further supported by studies in clinical setting 2,9 and with Monte Carlo methods 10 at distances beyond 1 m from the patient. However, studies near the patient (less than 1 m) concluded that the DRC provided by AAPM was a conservative value that over-estimated the dose. 9 The DRC for 18F is not applicable to 89Zr while the 18F SAF may not be valid for 89Zr. From previous work, we found that the estimated effective dose from 89Zr is 0.57 ± 0.2 mSv/Mbq and the highest organ dose is 1.87 ± 0.2 mSv/MBq in the liver. 11
Furthermore, the AAPM recommendations are based on the ANSI/ANS fluence-to-dose conversion coefficients 8 that have been replaced by those in International Committee on Radiological Protection (ICRP) Publication 60 12 and subsequently in Publication 116. 13 The ANSI/ANS values are generally higher than the current ICRP recommendation. In particular, the difference between the 2 data sets is most prominent at energies below 0.1 MeV, which is relevant to the multiple scattered photons emerging from the patient.
The goal of this study was to investigate the SAF and the DRC for 18F and 89Zr using the Monte Carlo method and an anthropomorphic phantom.
Methods
We carried out the simulations to estimate the self-absorption factors and the dose rates in air using the Zubal voxel phantom 14 and MCNPX version 2.5.0. 15 For the benefit of computing efficiency, the simulations were done in photon-only mode. We did not model the positron source explicitly but implicitly with a combination of monoenergetic photon sources, each of which is either the annihilation photon (511 keV) or the prompt γ-ray (909 keV) emissions. Although secondary electrons were omitted, MCNPX accounted for their bremsstrahlung radiations with the thick-target bremsstrahlung model. 6 In each simulation, a monoenergetic photon source was distributed uniformly in 1 of 7 organs or to the entire body—the “total body” distribution. The results for 18F were derived from the 511 keV simulations assuming 1.93 annihilation photons per decay; those for 89Zr were derived from the 511 keV and the 909 keV simulations assuming 0.46 annihilation photons and 0.99 prompt γ-ray per decay, respectively. The photon intensities per decay came from the Nuclide Chart by National Nuclear Data Center of Brookhaven National Laboratory (www.nndc.bnl.gov/chart). Thus, the results for a radiopharmaceutical were weighted sums of the tallies according to the emission intensities and the biological distribution of the radiopharmaceutical. Statistical error doesn’t exceed 1% for all simulations carried out, with a total number of events of 10 9 for each run.
Biological Distributions
In this study, we assumed the following typical biodistribution for 18F-FDG (where the percentages refer to the percent of the administered activity): 7.5% in the brain, 4.8% in the liver, 6.8% in the bladder content and 80.9% in the rest of the body. 10 For 89Zr, we studied the distributions of 2 radiopharmaceuticals from literature 89Zr labeled chimeric monoclonal antibody U36 (cmAb) for head and neck cancer patients 4 and trastuzumab for breast cancer patients. 3 The 2 studies gave uptakes in individual organs (Table 1). The activities in the rest of the body was derived by subtracting the sum of the organ activities at each time point from the total body activity decayed from the previous time point. 11 The uptake ratio for each organ was reassigned to account for of the total body distribution. For 89Zr-cmAb, we can see that the radioactivity clearly concentrated in liver and spleen sites over time. However, kidneys, brain and liver organs presented the highest uptake after 4-5 days for 89Zr-trastuzumab. Each organ has its own specific labeling response capability.
Typical Biodistributions of 89Zr Used in This Study.

Self-Absorption Factors and Emission Spectra
For the self-absorption simulations, the phantoms were positioned at the center of a vacuum sphere of 100 cm radius. The total amount of photon energy crossing the spherical surface was calculated with the *F1 tally, which is a track length estimator modified by the particle energy so that the energy is tallied instead of the number of photons. To obtain the total amount of energy emitted by a radionuclide in multiple organs, the *F1 tallies were weighted by the biological distribution and summed together. Thus the self-absorption factor of a radionuclide was the weighted sum divided by the total amount of energy that it emitted.
Dose Rate Constants
Dose rates at 1, 4 and 8 m from the phantom center were estimated in concentric spherical shells at those positions. Outside of the phantom was modeled with air medium. The F4 track length estimators modified with fluence-to-effective dose conversion coefficients were applied in the spherical shells. The unit of the modified tallies was Sv/hr per source photon. Similar to the self-absorption and spectrum calculations, the estimation of the dose rate D at distance x took into account the photon emission intensity and biological distribution of the radiopharmaceutical.
The phantom scattered dose rate constant K that accounts for phantom scattering and self-absorption was obtained by least square fit of the estimated dose rates D(x) per MBq of activity in the phantom at x by the equation

Ideally, K should be close to the product of DRC and SAF.
Results and Discussion
Dose Rate Constants From a Point Source, ANSI/ANS vs ICRP
The dose rate at 1 m from point source of 18F was estimated to be 0.143 μSv·MBq−1·hr−1 using the ANSI/ANS conversion coefficients. Our estimated dose rate for 18F was the same as the AAPM recommended value. Since the AAPM model used a point source, the value of the dose rate at 1 m is the value for the DRC, that is, the DRC for 18F is 0.143 μSv·m2·MBq−1·hr−1. When the current ICRP conversion coefficients were used instead of the ANSI/ANS coefficients, the DRC was reduced to 0.139 μSv·m2·MBq−1·hr−1, which was 2.8% below the AAPM value. For 89Zr, the DRCs were 0.154 μSv·m2·MBq−1·hr−1 and 0.150 μSv·m2·MBq−1·hr−1 using ANSI/ANS and the ICRP conversion coefficients, respectively. The ICRP conversion coefficients reduces the dose by 2.6%.
Self-Absorption Factor
The estimated SAFs in this study are listed in Table 2. The SAF calculated with a typical biodistribution of 18F-FDG in the Zubal phantom was 0.65. Our value is between the AAPM value of 0.64 8 and the value of 0.66 when using the phantom developed by the Committee on Medical Internal Radiation Dose (MIRD). 10 Since the fraction of administered activity of 18F-FDG in total body is 0.849, the SAF is similar to the one from a uniform distribution in total body 0.66.
Self-Absorption Factors of 18F and 89Zr in Various Biodistributions for Zubal Phantom.

At the time of injection, the fraction of 89Zr-cmAb in total body was 0.845; the SAF was estimated to be 0.66 which was similar to the one from total body, 0.67. In fact, the SAF was also close to the one of 18F-FDG. After 72 hours, the SAF reduced to 0.62 because the fraction of the tracer in total body was reduced to 0.453. In the case of 89Zr-trastuzumab, its ratio in total body was further reduced to 0.128 after 4-5 days. The SAF was estimated to be 0.61 which is similar to the SAF of 89Zr-cmAb after 3 days. Hence our study suggested that the SAF for 89Zr was about 0.66 immediately after injection and 0.61 a few days later.
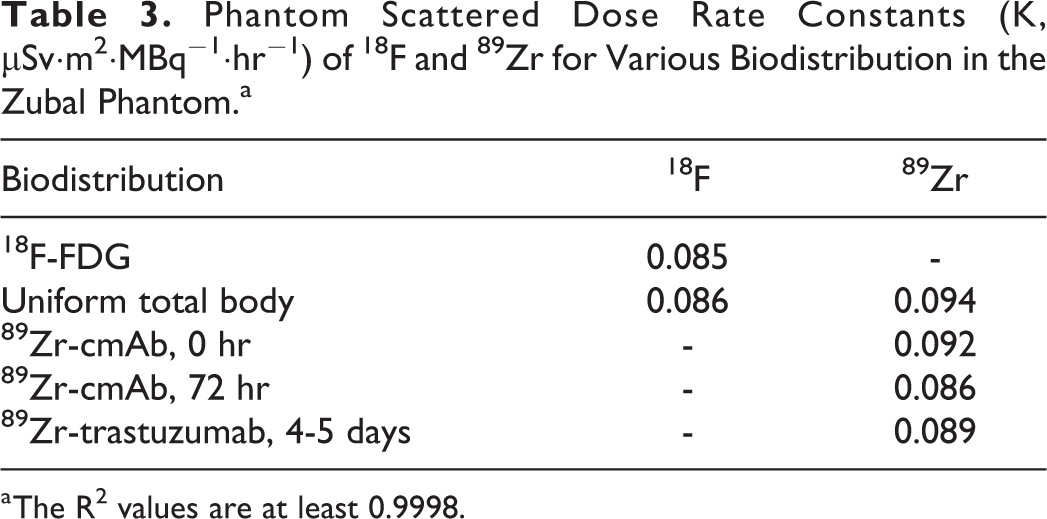
Phantom Scattered Dose Rate Constants of the Emissions From the Zubal Phantom
The phantom scattered dose rates were calculated at 1, 2, 4 and 8 m from the phantom center and normalized by the activity in the phantom. They had not been adjusted for the physical and biological decay. These data were fitted to equation (2) to obtain K. Furthermore, the ANSI/ANS conversion coefficients were applied instead of the current ICRP recommendation to maintain comparability with AAPM recommendations. Table 3 summarizes the values of K from various biodistribution. For comparison, the DRC and SAF product under the AAPM formalism gives 0.094 μSv·m2·MBq−1·hr−1 for 18F and it varies between 0.094 μSv·m2·MBq−1·hr−1 and 0.103 μSv·m2·MBq−1·hr−1 for 89Zr. Thus, the AAPM formalism yields conservative estimates near the patient.
a The R2 values are at least 0.9998.
As pointed out by Zaidi and Xu, 16 the voxel phantoms allow the modeling of any geometrical shape of the body and any complex distribution of radioactive sources inside the body. Since the phantoms are developed from scans of real patients or volunteers, the modeled distribution and the scattering of the radiation will vary according to the phantom employed in the simulation. In general, larger phantom will attenuate the emitted radiation more than a smaller one. The Zubal phantom was developed to reflect an average adult male 14 ; its size is similar to the MIRD phantom used by AAPM. Although our estimated SAF for 18F was the same as the AAPM recommended value, the distributed source in our simulation yielded a smaller value in the phantom scattered dose rate constant than the AAPM suggested DRC-SAF product.
Comparing 18F Dose With AAPM Values
A point source is a crude approximation of the radioactivity distributed in a patient. Using the Zubal phantom and a typical biological distribution of 18F, the dose rate at 1 m was 0.087 μSv·MBq−1·hr−1 anterior to the phantom, which is 40% below the estimation with the AAPM model: 0.143 μSv·MBq−1·hr−1 × 0.64 / 1 m2 = 0.092 μSv·MBq−1·hr−1. The discrepancy between the 2 models decreases with increasing distance; it reduces to 4% at 2 m, below 1% at 4 m and beyond. Thus, the dose to the nurse or technician who administer the radiopharmaceutical injection may be overestimated based on the AAPM dose rate constant.
Dose survey suggested that the dose rates at 30 cm, 1 m and 2 m from the patient were 290 μSv·hr−1, 60 μSv·hr−1 and 15 μSv·hr−1, respectively, during injection of 505 MBq of 18F-FDG. 2 During the scanning after 1 hr of uptake, the dose rates were 190 μSv·hr−1, 47 μSv·hr−1 and 20 μSv·hr−1 at 30 cm, 1 m and 1.3 m, respectively. Estimation using our results gave 332 μSv·hr−1 (30 cm), 44 μSv·hr−1 (1 m) and 12 μSv·hr−1 (2 m) during injection and227 μSv·hr−1 (30 cm), 30 μSv·hr−1 (1 m) and 19 μSv·hr−1 (1.3 m) during scanning. On the other hand, the doses derived from AAPM would be 514 μSv·hr−1 (30 cm), 46 μSv·hr−1 (1 m) and 12 μSv·hr−1 (2 m) during injection and 352 μSv·hr−1 (30 cm), 32 μSv·hr−1 (1 m) and 19 μSv·hr−1 (1.3 m) during scanning. Thus, the simulation results were broadly comparable to the measurements. Furthermore, the results were very close to the AAPM derived dose at 1 m and beyond.
The observation that the AAPM derived dose is an overestimation near the patient is also supported by Quinn et al. 9 The authors measured 0.37 μSv·MBq−1·hr−1 at 30 cm from the patient during scanning. Our simulations gave a value of 0.42 μSv·MBq−1·hr−1 while the AAPM values gave a value of 1.02 μSv·MBq−1·hr−1. Quinn et al suggested that the biodistribution might explain the discrepancy between their measured value and the one calculated using the AAPM parameters.
Our simulations showed that the highest dose rates occurred if the radionuclide concentrated in the brain (0.90 μSv·MBq−1·hr−1 at 0.3 m), the skin (0.78 μSv·MBq−1·hr−1 at 0.3 m) and in the muscle (0.64 μSv·MBq−1·hr−1 at 0.3 m). If the radionuclide was distributed uniformly over the entire body, the dose rate at 0.3 m would be 0.66 μSv·MBq−1·hr−1. When the radionuclide were distributed in deeper tissues, the dose rate predictable would be less than the foregoing values. All values were well below the AAPM dose rate, 1.02 μSv·MBq−1·hr−1. Our estimated dose rate of 0.42 μSv·MBq−1·hr−1 from a typical biodistribution was a weighted sum of the dose rates from individual organs. It is compatible with measurements interference. 8 While the biodistribution is an important factor in the dose rate close to the patient, the AAPM assumption of a point source is the main cause of the discrepancy.
Comparing 89Zr With 18F Doses
The dose rate estimated at 1 m from a point source of 89Zr in our simulation was 0.154 μSv·MBq−1·hr−1, which was 8% above the 18F dose rate. With the radionuclide distributed in the Zubal phantom, we estimated that the patient body absorbed 33%-38% of the emitted energy depending on the distribution. Therefore, the self-absorption factor was estimated to be 0.67 for a uniform total body distribution, 0.66 for a typical U36 distribution and 0.61 for the trastuzumab distribution. In contrast, we estimated the self-absorption factor for 18F was 0.65 while AAPM estimated it to be 0.66. The differences between 18F and 89Zr values were generally small because the curve of mass energy absorption coefficients for soft tissue is relatively flat in the range of 100 keV to 1 MeV. Using log-log interpolation of the NIST mass energy absorption coefficient data (https://www.nist.gov/pml/x-ray-mass-attenuation-coefficients), the values are 3.267 × 10−2 cm2/g and 3.117 × 10−2 cm2/g for 511 keV and 909 keV photons, respectively. The absorption of the prompt γ-ray in the body is only about 5% less than that of the annihilation photon.
We calculated the 89Zr dose rates for 3 biodistributions: (1) a hypothetical situation same as the typical 18F-FDG distribution by Elschot et al, 10 (2) in breast cancer patients by Dijkers et al, 4 and (3) in head and neck cancer patients by Alfuraih et al. 11 We repeated the 18F calculations assuming that it followed the 89Zr biodistributions (2) and (3) for comparison. The values are summarized in Table 4.
a Notice that the 18F would not follow the U36 or the trastuzumab distributions in reality and neither would 89Zr follow the FDG one. They are included for comparisons only.
Due to the presence of the 909 keV prompt γ-ray, the dose rates from 89Zr labeled antibodies are higher than the 18F-FDG in organ-by-organ comparisons. It is interesting to note that the dose rate from a typical 18F-FDG distribution in the body was very similar to a uniformly distributed 18F source in the entire body (0.087 μSv·MBq−1·hr−1). Although the 89Zr radiopharmaceutical accumulated more in the tumor deeper in the body, the estimated dose rates were about 3% (cmAb, 0.093 μSv·MBq−1·hr−1) to 5% (trastuzumab, 0.091 μSv·MBq−1·hr−1) below a that of a uniform total body distribution (0.096 μSv·MBq−1·hr−1).
Despite the higher dose rate of 89Zr due to the greater penetration power of the prompt γ-ray, a typical immuno-PET scan using 89Zr may not give a higher dose to the clinical staff than 18F-FDG because immuno-PET generally utilizes a lower administered activity (75 MBq) than that typically used for 18F-FDG (∼500 MBq). Table 5 compares the dose rates of the 2 radionuclides during injection and during scanning. We assumed that the radiopharmaceuticals followed a uniform total body distribution at the time of injection and subsequently accumulated in a typical distribution after 1 hour of uptake time. Because of the long half-life of 89Zr, the dose rates changed very little from 7.2 μSv·hr−1 during injection to 6.8-6.9 μSv·hr−1 during scanning. Furthermore, the low injected activity made the 89Zr dose rates per scan significantly lower than those of a typical 18F-FDG study that gave 44 μSv·hr−1 during injection and 28 μSv·hr−1 during scanning.
Comparing 89Zr and 18F Dose Rates Per Scan at 1 m Anterior to the Phantom [μSv·hr− 1 ].a
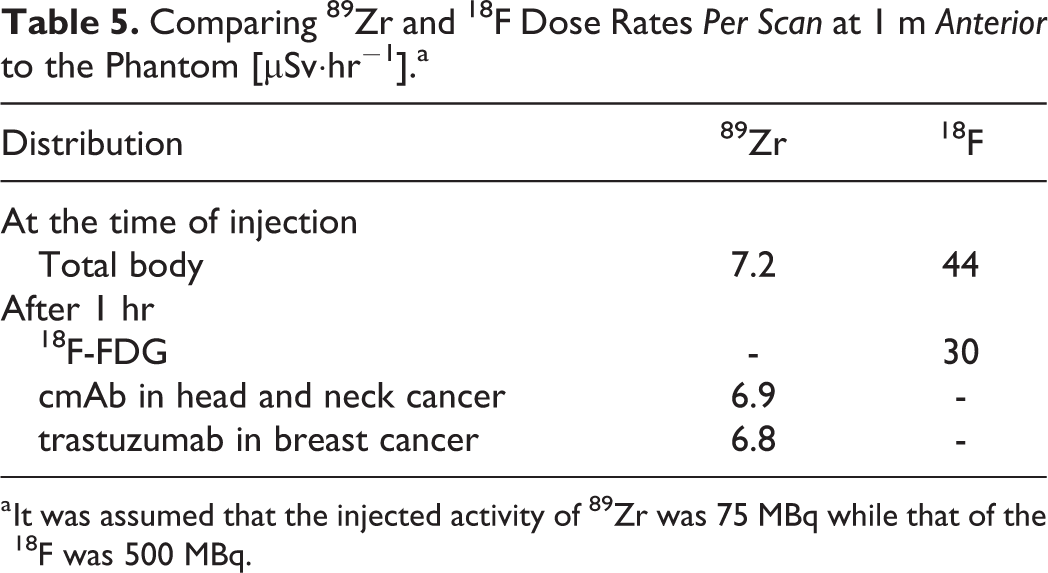
a It was assumed that the injected activity of 89Zr was 75 MBq while that of the 18F was 500 MBq.
ICRP Conversion Coefficients Applied to the Emission From the Zubal Phantom
The dose rate at 1 m from a point source of 18F and 89Zr decreased to 0.139 μSv/hr and 0.150 μSv/hr, respectively, when the current ICRP conversion coefficients were employed. They were 2.8% and 2.6% below the values obtained with the ANSI/ANS coefficients. When the estimation was estimated again with the Zubal phantom, the estimations using the ICRP coefficients were also smaller than those using the ANSI/ANS ones. However, it is interesting to note that the reduction was larger in the case of 18F (4.5%) but smaller than in the case of 89Zr (1.4%) because of the greater penetration power of the 89Zr emission. The 89Zr emissions from the distal parts of the body were less attenuated than those of 18F when they arrived at the point of calculation.
Conclusion
We have presented our study on the 18F and 89Zr dose rates and the dose rate constants from point source in air and also from an anthropomorphic phantom. The dose rate at 1 m for 18F was found to be 0.143 μSv·MBq−1·hr−1, which was consistent with the AAPM recommendation, while that for 89Zr was found to be 0.154 μSv·MBq−1·hr−1. We estimated SAF for 18F to be 0.65, which also agreed with the AAPM recommendation. The SAF for 89Zr was in the range of 0.61−0.66 depending on the biodistribution. We further examined the effect of fluence-to-effective dose conversion coefficients on the effective dose rate estimations. Also, we found that the AAPM derived dose is an overestimation near the patient, compared to our simulations, which can be explained by the biodistribution nature and the assumption of the point source. Compared to using the current ICRP coefficients, the AAPM method using the ANSI/ANS coefficients overestimated the dose rate by about 3% for both radionuclides.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research project was financially supported by the Strategic Technologies Program for National Plan for Science, Technology and Innovation (NPSTI) in Kingdom of Saudi Arabia under Contract no. 11-MED 1586-02.