Abstract
Background:
Liver injury due to ionizing radiation exposure either accidental or after radiotherapy treatment, may lead to many alterations in proteins expression related to inflammation or apoptosis. Our study investigated the curative effect of Mangosteen (MGS) extract (fruit rind) against ionizing radiation (IR) induced liver damage.
Methods:
Hepatotoxicity was induced in Wister rats by exposure to an acute single dose (6 Gy) of IR while MGS was given orally to rats (500 mg/kg bwt) and administered daily for 30 days after irradiation.
Results:
MGS treatment has significantly attenuated redox imbalance state and toxicity induced by protracted exposure to gamma-rays in liver tissues, which was substantiated by the significant amelioration of liver function tests, MDA contents, antioxidant enzymes (SOD and CAT) activities and NO level. MGS inhibited also the inflammatory markers (TNF-alpha, IL-6 and CRP) and downregulated transcriptional factor NF-Kappa-B/TGF-β1. These alterations were concomitant with an improvement of the Proliferating cell nuclear antigen (PCNA) which is a protein expressed in the nuclei of cells during cell cycle and is important for both DNA synthesis and DNA repair. These results were confirmed by amelioration in histological and ultrastructural examinations.
Conclusion:
We concluded that MGS could ameliorate via minimizing significantly the amount of oxidative damage, inflammations disturbances and pro-apoptotic alternations induced by IR. MGS may be a promising supplement with protective effects from irradiation-induced injury such as TNF-α/NF-κB/TGF-β1 management.
Introduction
Exposure to ionizing radiation accidentally, occupationally or even as a therapy makes a complex cascade of systemic and tissue-specific responses that lead direct and indirect changes. 1 Direct effects produce damage signal transduction, as well as disruption in the cells, while indirect effects result from its action on water-soluble molecules and production of free radicals on cellular level. 2 Free radicals’ production (reactive oxygen/nitrate species) induced activation of cytosolic dimer (NF-κB p65) with translocation into the nucleus via alternative IκBα phosphorylation, which may result in the degradation of IκBα through proteasome. 3 Moreover, these over production of free radicals will lead to an increase in several pro-inflammatory cytokines such as IL-1b, TNF-α and IL-6, with activation of growth factors (TGF-β), which are implicated in critical injuries. 4 Liver is one of the most radiosensitive organs; it can cause cell degeneration, apoptosis, disordered enzyme activity, biofilm destruction, metabolic disorders, and severe functional liver failure. 5 Treatment with phytochemicals possessing curative effects with minimal side effects would be beneficial in the management of radiation exposure side effects. 6 Mangosteen is a tropical tree native to Southeast Asia and Africa and includes more than 300 diverse species from which many families of bioactive compounds such as a class of polyphenols known as xanthones, flavonoids, triterpenoids, and benzophenones have been characterized and isolated. 7 Many studies have suggested that extract of MGS has more than 68 xanthones. The 2 major isolated xanthones are α-mangostin and γ-mangostin that have biological functions in terms of anti-inflammation, antioxidation, anticancer, antimicrobial, and neuroprotective activities. 8 Moreover, the chemotherapeutic and chemopreventive activities of xanthones have been widely studied due to their repressing impact on carcinogenesis process. 9
The aim of this study is to evaluate the possible curative role of MGS on the oxidative damage leading to hepatotoxic effects due to radiation exposure of rats. This goal is achieved by evaluating oxidative damage markers (MDA, SOD, CAT, and NO), the inflammatory markers (TNF-alpha, IL-6, CRP, and NF-Kappa-B), growth factors (TGF-β1), the marker of cell proliferation (PCNA) and apoptotic markers (Caspase 3). Furthermore, histopathological and immunohistochemical examinations were investigated to confirm the above results.
Materials and Methods
Materials
MGS Extract (fruit rind) are obtained from Solaray®(Park City, UT 84098 USA) and other chemicals and reagents were purchased from Sigma–Aldrich® (St. Louis, Missouri, USA).
Irradiation
The irradiation of whole-body gamma irradiation was performed at the NCRRT (Cairo, Egypt) using Canadian gamma cell-40 (137Cesium) at a dose rate of 0.67 Gymin−1.
Experimental animals
24 male Wistar albino rats, of an average weight of (150 ± 20 gm) were used, maintained in an air-conditioned (25 ± 2°C) animal house cages under standard conditions of humidity (50% ± 5%) with specific pathogen-free conditions and subjected to a 12:12-h daylight/darkness and allowed unlimited access to chow and water. All the ethical protocols for animal treatment followed by the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978) were following the guidelines for the proper treatment and use of laboratory animals authorized by the Research Ethics Committee (REC) for experimental studies (human and animal subjects) at National Center for Radiation Research and Technology (NCRRT), Egyptian Atomic Energy Agency, Cairo (Serial No.22A/20).
Rats of the present study were divided into 4 equal groups (6 rats/group) as follows:
Group (1): Untreated normal control group, Group (2): Animals received orally MGS (500 mg/kg body weight/day), 10 for 30 consecutive days, Group (3): Animals were exposed to an acute single dose of 6 Gy of γ-irradiation, Group (4): Animals were exposed to an acute single dose of 6 Gy of γ-irradiation then received MGS for 30 consecutive days.
The animals were anesthetized with ketamine/xylazine (40-90) mg/kg + (5-10) mg/kg 11 and then sacrificed after a fast night at the end of the experiment. Intracardiac blood samples were collected and serum was separated by centrifugation at 3000 rpm and stored at −80°C until analysis. Liver has been removed immediately after sacrifice; a known liver tissue weight has been homogenized, then the homogenate was centrifuged and the supernatant was collected for the further biochemical and molecular analysis.
Biochemical Assay
The liver enzymes assessment
Serum levels of alanine aminotransferases (ALT, EC 2.6.1.2), aspartate aminotransferase (AST, EC 2.6.1.1), and alkaline phosphatase (ALP EC 3.1.3.1). Enzyme activities following the manufacturer’s recommended procedures were estimated using commercial kinetic assay test kits Spectrum Diagnostic Company Cairo, Egypt.
Determination of oxidative stress markers in liver tissue
Liver tissue lipid peroxidation, malondialdehyde (MDA) product was measured by thiobarbituric acid assay by colorimetric technique according to the details given in Bio-diagnostic kits guidelines, Cairo, Egypt (Cat. No. MD 25 29). The total level of nitrate/nitrite (NOx) content was carried out according to Miranda et al. 12 The activities of antioxidant enzymes Superoxide dismutase (SOD) and catalase (CAT) (Cat. No. SD2521, No. CA 2517) were assayed via the method of colorimetric kits Bio-diagnostic, Cairo, Egypt, respectively according to the manufacturer’s instructions.
Estimation of liver TNF-α, IL-6 and serum CRP levels
The concentration of cytokines was measured by rat ELISA kits (
Molecular analysis
The level of (NF-κB p65) was measured by (Rat Cat. No. MBS2505513) from (MyBioSource, Inc. San Diego, USA) and caspase-3 activity in liver tissue was directly proportional to the color reaction by using the kit (Rat Cat. No. KHZ002, ApoTarget, Invitrogen, Carlsbad, CA, USA,) was according to the manufacturer’s methods.
DNA fragmentation assay
DNA was extracted from liver tissue lysate using the kit supplied by Qiagen following the recommended steps, then DNA fragmentation was detected in the extracted DNA through gel electrophoresis and was visualized and photographed under UV light as described previously. 13 For quantification of fragmented DNA, diphenylamine analysis as previously defined by Burton 14 was used and DNA fragmentation was expressed as percentage of total DNA.
Histopathological investigations: For light microscopic examination small pieces of the liver were immediately fixed in 10% buffered formalin. The specimens were then dehydrated, cleared and embedded in paraffin wax. Sections of 6µm thickness were stained with haematoxylin and eosin. 15
Transmission Electron Microscopy (TEM)
For TEM examination, liver was cut into small pieces of about 1 mm 3 in size and immediately fixed in 2.5% glutaraldehyde for 24-48 h. The specimens were then washed in phosphate buffer (pH 7.2-7.4) 3-4 times for 20 min each and post-fixed in a buffered solution of 1% osmium tetraoxide for 2 h. Specimens were dehydrated in ascending grades of ethyl alcohol, and embedded in epoxy resin (Epikote 812). 16 Ultra-thin sections were stained with alcoholic uranyl acetate followed by lead citrate and then examined using a TEM. 17
Immunohistochemical investigations
Immunohistochemical staining was performed in liver tissues with 4μm-thick sections that were deparaffinized and incubated with fresh 0.3% hydrogen peroxide in methanol for 30 min at room temperature. Briefly, deparaffinized tissue slides were incubated with the antibody against Proliferating cell nuclear antigen (PCNA) (diluted 1:50), NF-kB (1:300). The antibody binding sites was visualized with 3, 3′diaminobenzidine. The sections were then counterstained with hematoxylin, dehydrated using graded alcohols and xylene, and mounted. The immunostaining intensity and cellular localization of PCNA and NF-kB were analyzed by light microscopy. 18 For immunohistochemistry quantification, the positive area percentage (%) of positive brown stained cells was estimated.
Statistical Analysis
Data were subjected to statistical analysis and tests of significance performed using the statistical package SPSS (Statistical Program for Social Science) version 15.0 by applying a one-way ANOVA test followed by a post hoc test for multiple comparisons. All data expressed as a mean of 6 values; SE and difference between means were considered significant at P < 0.05.
Results
Effect of MGS on Hepatic Oxidative Stress Markers in Irradiated Rats
MGS treatment showed an improvement in the hepatotoxicity profile induced by γ-irradiation. To determine the severity of liver injury provoked by IR exposure, liver function tests (AST, ALT and ALP) were performed. Table 1 discerns a significant increase (P < .0001) in serum AST, ALT and ALP activities in IR groups when compared to the corresponding control. On the other hand, concomitant treatment of IR rats with MGS counteracted hepatic damage induced by IR exposure and significantly diminished (P < .0001) serum AST, ALT and ALP activities as revealed in MGS + IR groups when compared to IR groups.
Effect of MGS on Liver Enzymes of Rats Exposed to IR.*

* The values are means ± SE (n = 6). Symbol (a, b and c) denote significant change at P < 0.05 versus control, MGS, and IR groups, respectively.
Exposure to IR is usually accompanied by imbalance between pro-oxidant and antioxidant defense mechanisms in liver tissue. Therefore, the hepatic MDA, NO, SOD and CAT levels were investigated. As presented in Figure 1, a remarkable rise (P < .001) and (P < .0001) in hepatic MDA level by 442.5% and 893.7% associated with a pronounced decline (P < .0001) in hepatic SOD and CAT levels by 4.3% and 1.6% was observed in IR groups, respectively as compared to the control.
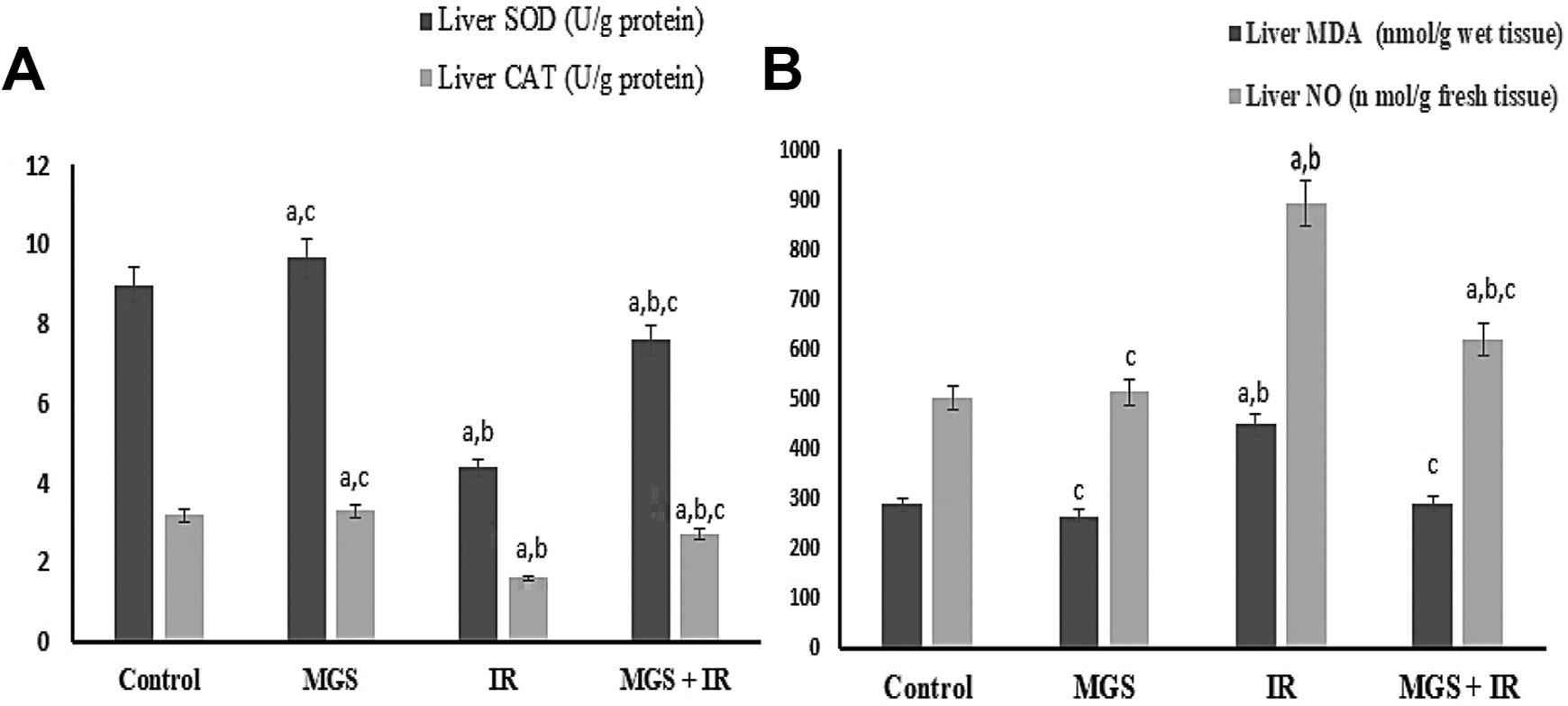
MGS attenuates IR induced oxidative stress and liver injury. The values are means ± SE (n = 6). Symbol (A, B and C) denote significant change at P < 0.05 versus control, MGS, and IR groups, respectively.
Noticeably, MGS treatment significantly abolished (P < .0001) hepatic MDA and NO level by 289.2 and 619 respectively significantly (P < .0001) associated with elevated in hepatic SOD and CAT levels as it achieved 7.6 and 2.7 as perceived in MGS+IR.
Effect of MGS on Systemic Inflammatory Mediators in Irradiated Rats
The IL-6 and TNF-α levels in liver tissue were surveyed in this study. As depicted in Figure 2D, animals of IR group showed a pronounced increment (P < .0001) in serum IL-6 and TNF-α levels by 54.3% and 63%, respectively when compared to the control set. Interestingly, concurrent treatment of IR group with MGS significantly decreased (P < 0.05) serum IL-6 and TNF-α levels by 48.5 for IL6 in MGS + IR groups 39.1 and 37 for TNF-α in MGS +IR groups, respectively (Figure 2A), suggesting that MGS treatment suppresses inflammatory mediators which are crucial for metabolic disorders associated with radiotherapy side effects. In IR groups, a significant elevation (P < 0.001) in C-reactive protein (4.7) and caspase-3 activity with a recorded (2.8) was noticed when compared to the respective control (Figure 2). Worthwhile, concurrent treatment of IR rats with MGS markedly reduced CRP level (P < 0.001) by (4.47) and caspase-3 activity (P < .0001) by 4.9 when compared to IR groups (Figure 2B). Additionally, as shown in Figure 3, IR rats group displayed a significant increase of NF-kB associated with a significantly enhanced level of the hepaticTGF-β1protein (P < 0.05) compared with the control group (Figure 2C and D). The increase of the NF-kB /TGF-β1signaling pathway was inhibited by the MGS administration clarifying the anti-fibrotic potential of MGS.
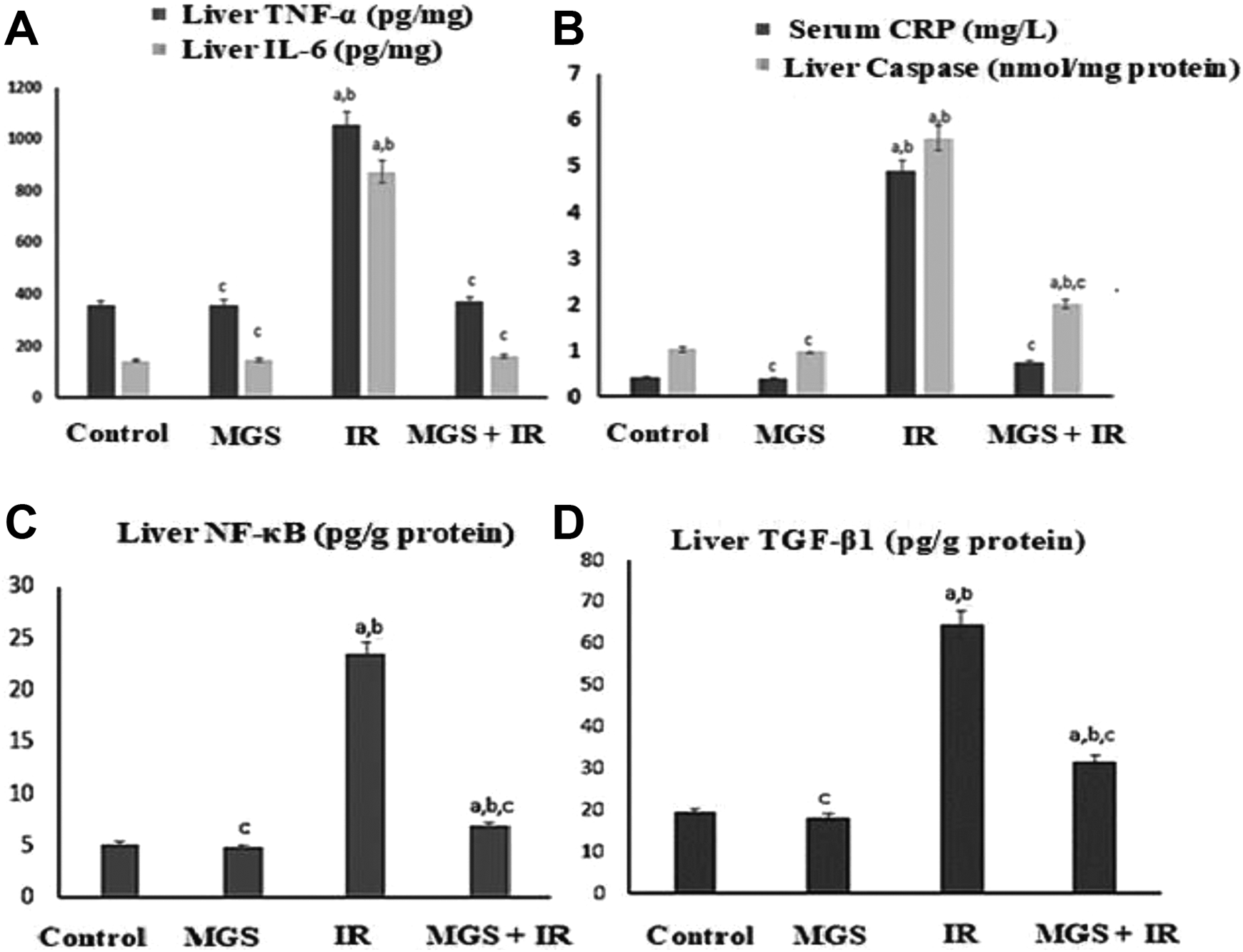
MGS down-regulating TNF-α/NF-κB/TGF-β1 signaling factors and inhibits apoptosis. The values are means ± SE (n = 6). Symbol (A, B and C) denote significant change at P < 0.05 versus control, MGS and IR groups, respectively.

The DNA fragmentation pattern was assessed in treated and untreated liver tissue on agarose gel electrophoresis. A, Representative agarose gel electrophoresis showing DNA fragmentation pattern in different studied groups, Lane M: 100 bp DNA ladder marker, Necrotic strand breaks/streaking DNA was observed in Lane (2) group of IR, but not in Lane (1) control group and treatment groups in Lane (3) MGS, and Lane (4) MGS+IR. B, The values are means ± SE (n = 6) Symbol (A, B, C) denote significant change at P < 0.05 versus control, MGS, and IR groups, respectively.
Effect of MGS on Hepatic DNA Damage
Oxidative stress mediated hepatic DNA damage was assessed in terms of DNA fragmentation (Figure 3); in which lanes 2 showed a widespread DNA fragmentation as indicated by sequestered DNA oligonuleosomal fragments corresponding to IR group. Whereas the untreated control and MGS treated groups (lane 1&3) didn’t show any DNA fragments. Interestingly, oxidative DNA damage was improved to a reduced level as evidently shown in lane 4 of MGS+IR group.
Histopathological Investigations
Histological rat liver sections of normal control group and MGS treated group manifested normal histological structure characterized by well detailed polygonal hepatocytes arranged in regular cords radiated from the central vein which is the center of liver lobule. Küpffer cells were finely arranged in between the cords Figure 4A and B.
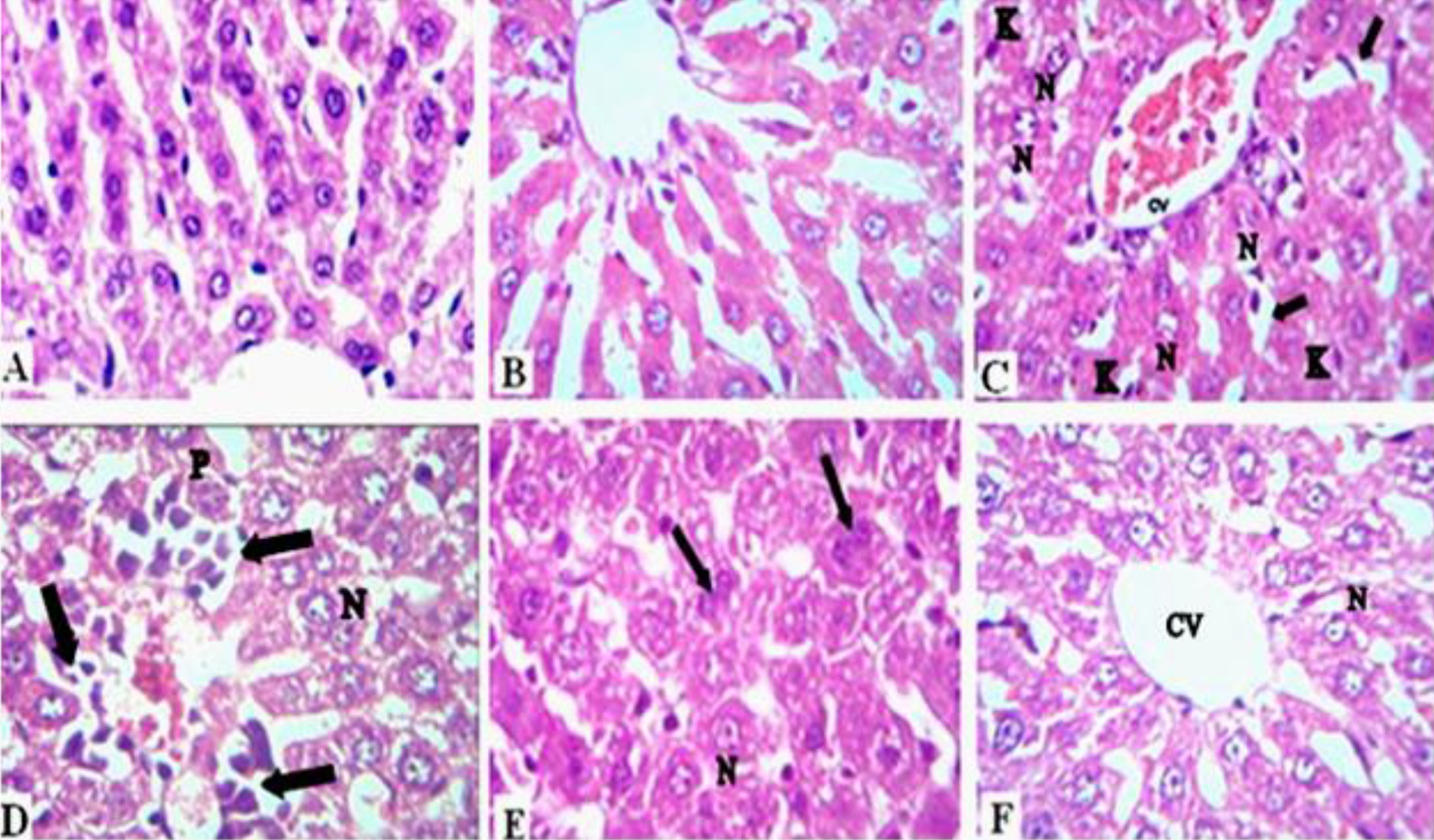
Photomicrograph of liver section. A, Normal liver section. B, Liver section of MGS treated rats. C, Liver section of γ-irradiated rats showing necrotic nuclei (N) in hepatocytes, dilated blood sinusoids (arrow), Küpffer cells (K) and severe hemorrhagic central vein (CV). D, Liver section to γ-irradiation rats showing high infiltrative inflammatory cells (arrow) with necrotic (N) and pyknotic nuclei (P). E, Liver section of γ-irradiation rats treated with MGS showing active mitosis in hepatocytes (arrow) and some necrotic cells (N). F, Liver section of γ-irradiated rats treated with MGS showing recovery in central vein (CV) with some necrotic cells (N) (Hx-E ×400, scale bar; 20 μm).
Nevertheless, rats exposed to γ-irradiation illustrated abnormalities in liver sections characterized by necrotic nuclei, dilated blood sinusoids, abundant Küpffer cells and central vein hemorrhage Figure 4C and D. Moreover, mononuclear inflammatory cells, fibroblasts, Küpffer cells and pyknotic nuclei were noticed. However, treatment with MGS was shown to protect cells against oxidative stress induced by γ-irradiation. This improvement is manifested by the presence of areas of regeneration and active mitosis in most of hepatic tissue, yet mild necrotic cells were persisted in Figure 4E. Recovery in central vein and blood sinusoids, some cells revealing normal pattern were also seen. Limited infiltrative inflammatory cells around central vein was rarely manifested in Figure 4F.
Ultrastructural Investigations
The ultrastructure of the liver of the control group is illustrated in Figure 5A and B, showing 2 neighbor hepatocytes with a narrow bile canaliculated in between cytoplasmic organelles as well as the nuclei of the hepatocytes revealed normal ultrastructural appearance. The cytoplasm contained abundant mitochondria with spherical or ovoid shape with well-developed cristae. The rough endoplasmic reticulum consisted of closely packed parallel and flattened cisternae where their outer surfaces studded with ribosomes (Figure 5A). Hepatic sinusoids localized between the hepatocytes and lined with a Küpffer cell is observed in Figure 5B. MGS group exhibited also normal hepatic architecture with normal features of nucleus with heterochromatin and euchromatin, mitochondria and rough endoplasmic reticulum faces of plasma membrane (Figure 5C). γ-irradiated rats revealed marked cytopathological alterations (Figure 5D and E), degenerated cytoplasm occupied by deteriorated mitochondria, pyknotic nuclei with loss of nuclear spherical configuration, and nuclear envelope and condensed chromatin (Figure 5D). In the same group, rats revealed dilatation of hepatic sinusoid with stagnant blood cells and activated Küpffer cell which is proliferated between hepatocytes with markedly increase in size filling the sinusoidal lumen. Devastated mitochondria and rough endoplasmic reticulum were fragmented into smaller stack and vesicles, and proliferation in smooth endoplasmic reticulum was noticed in Figure 5E. γ-irradiated rats revealed marked improvements of the cytoplasmic organelles following MGS treatment. The hepatic cells contained abundant mitochondria exhibiting close to the normal appearance. The hepatic cells revealed improvement of rough endoplasmic reticulum in the form of parallel and flattened cisternae studded with ribosomes, nucleus exhibit recovery illustrated by distinct regular nuclear envelope and chromatin material (Figure 5F).

TEM micrographs of hepatocyte. A, Control group showing the cytoplasm of a normal hepatocyte occupied by normal mitochondria (M), rough endoplasmic reticulum (RER) and part of the nucleus (N) and bile canalicule (bc). Scale bar: 2 μm, original magnification ×4000. B, Control group showing Küpffer cell (K) and blood sinusoid (BS) between hepatocytes (H). Scale bar: 10 μm, original magnification ×2000. C, MGS treated group showing normal rough endoplasmic reticulum (RER), mitochondria (M) and part of the nucleus (N) with euchromatin (EC), heterochromatin (HC) and plasma membrane (PM) Scale bar: 2 μm, original magnification ×4000. D, γ-irradiated rats showing degenerated cytoplasm, pyknotic nucleus (N), mitochondria (M) and red blood cell (RBC). Scale bar: 10 μm, original magnification ×2000. E, γ- irradiated rats showing Küpffer cell (K) proliferation between hepatocytes, small size mitochondria (M), fragmented rough endoplasmic reticulum (RER), proliferation in smooth endoplasmic reticulum (SER) and red blood cell (RBC). Scale bar: 10 μm, original magnification ×2000. F, γ-irradiated rats treated with MGS showing cytoplasm containing mitochondria (M), rough endoplasmic reticulum (RER) and part of the nucleus (N) Scale bar: 2 μm, original magnification ×4000.
Immunohistochemical Investigations
PCNA expression
Normal accumulation of PCNA is expressed by brown color in the liver cell nuclei of the control group and the group treated with MGS (Figure 6A and B). In contrast, high suppression in brown color immunostained PCNA expression was indicated in rats exposed to γ-irradiation compared to the control group (Figure 6C). However, treatment with MGS in rats exposed to γ-irradiation showed moderate accumulation of PCNA expression in the nuclei of the liver cells (Figure 6D). These results are also demonstrated by immunohistochemical quantitative percentage in Figure 7 and Table 2.

Photomicrograph of immunohistochemistry of PCNA expression in liver tissue. A, Normal control rats group. B, MGS treated rats. C, γ-irradiated rats showing a detectable decrease of PCNA expression. D, Liver section of γ-irradiated rats treated with MGS showing a detectable elevation of PCNA expression (immun;400, scale bar; 20 μm).
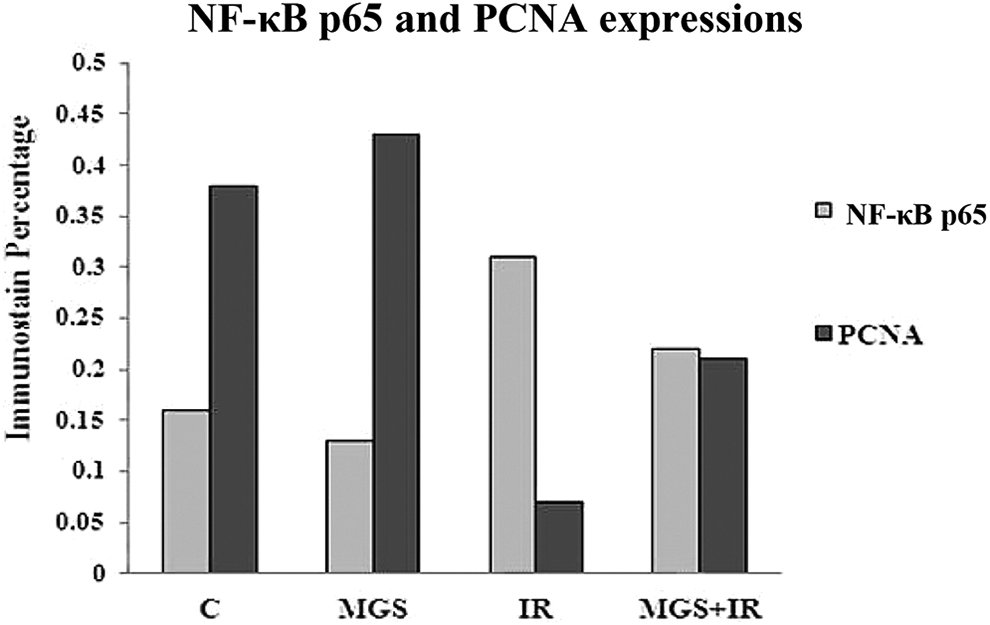
Quantitative percentage of NF-κB p65 and PCNA expressions in liver tissue.
Quantitative Percentage of PCNA and (NF-κB p65) Expressions in Liver Tissue.*

* Each value represents the mean ± SE (n6). Values with different superscript letters within the same raw are considered significantly different at P < 0.05.
NF-Kb (P65) expression
Sections from control rats and MGS treated group revealed normal accumulation of (NF-κB p65) expression (Figure 8A and B). On the contrary, high accumulation of brown color immune-stained (NF-κB p65) expression was indicated in rats exposed to γ-irradiation (Figure 8C). On the other hand, γ-irradiated rats treated with MGS revealed a decrease of (NF-κB p65) immunoreactivity in the liver nuclei (Figure 8D). These results are also indicated by immune-histochemical quantitative percentage in Figure 7 and Table (2).
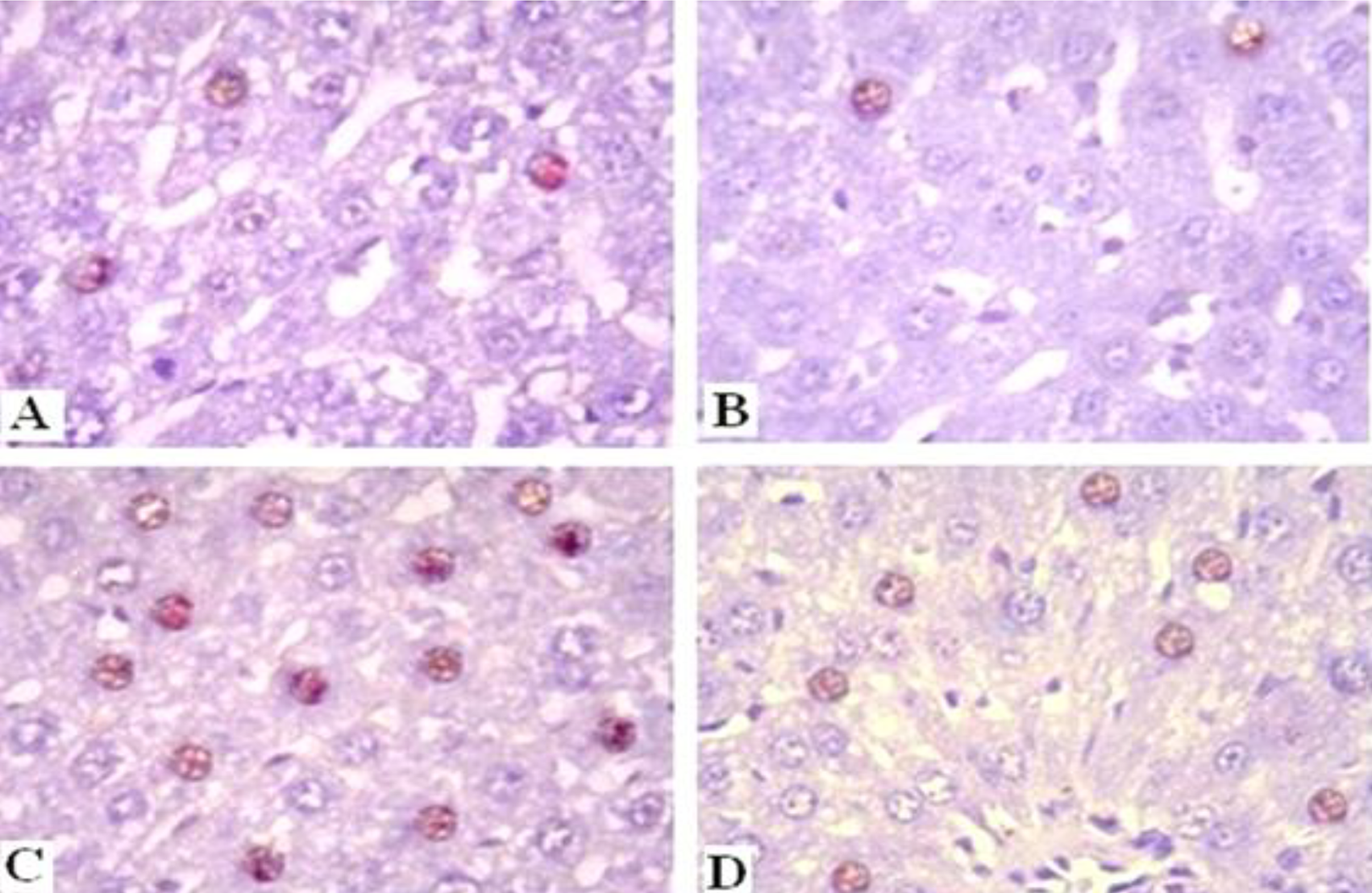
Photomicrograph of immunohistochemistry of (NF-κB p65) expression in liver tissue. A, Normal control rats’ group. B, MGS treated rats. C, γ-irradiated rats showing a detectable increase f (NF-κB p65) expression. D, Liver section of γ-irradiated rats treated with MGS showing a high reduction of (NF-κB p65) expression (immune;400, scale bar; 20 μm).
Discussion
In that era, “classic” radiation-induced liver disease is a devastating consequence of radiotherapy. Strategies have been implements to reduce the risk of liver toxicity due to radiation, opening new prospects to help patients who otherwise have limited options for liver-directed therapies. 19
In the current study, whole body gamma irradiation of rats with 6 Gy provokes oxidative stress in the liver tissue. This was confirmed by increasing liver enzymes (ALT, AST, and ALP) when compared with their corresponding values in the control group of rats. In the same line, Al-Khattab et al 20 indicated that the stimulation of liver enzymes by gamma irradiation, which is responsible for the biosynthesis of fatty acids and mobilization of fat from adipose tissues to the blood stream, leads to hyperlipidemic state. Treatment of rats with MGS for 30 consecutive days has attenuated hepatic tissue damage associated with irradiation, as revealed by the preserved structural integrity of the hepatocellular membrane and liver cells architecture in histopathological sections along with the suppressed plasma activities of liver transaminases. Recent studies indicates that α-MGS could attenuate lipopolysaccharide/d-galactosamine-induced liver pathological injury, and decrease the hepatic malondialdehyde level, serum ALT, AST, bilirubin and albumin. 21,22 The damage effect of γ-irradiation provokes oxidative stress in the liver tissue which is evidenced by the significant increase in the level of liver lipid peroxidation (MDA) concomitant with a significant decrease in the activity of the antioxidant enzymes SOD, CAT. The interaction of MDA and SOD demonstrated that SOD activities can indirectly reflect the scavenging capacity of oxygen free radicals. 23 Radiation exposure mechanism induces radiolysis of water in the aqueous media of the cells. This hydrolysis leads to the production of hydroxyl radicals (•OH) which interact with the polyunsaturated fatty acids in the lipid portion of biological membranes initiating the lipid peroxidation and finally damaged the cell membranes. 24 MGS antioxidants could significantly decrease the cellular ROS levels by increasing SOD-1 and HO-1 that resulted in boosted cell survival. 25 Also, MGS possesses potent anti-glycemic and hepatoprotective effects by reducing metabolic markers, oxidative stress markers with an improvement in hepatic glycogen level and anti-oxidant enzyme activities of type II diabetic mice. 26
Abnormalities induced by γ-irradiation in liver tissues pathology, were characterized by necrotic nuclei, dilated, blood sinusoids, mononuclear infiltration of inflammatory cells and increase in Küpffer cells. Moreover, dilatation of central veins was detects with central vein hemorrhage. These findings were in agreement with Alkhalf and Khalifa. 27 Previously explained by García et al, 28 free radicals interact with the polyunsaturated fatty acids in the lipid portion of biological membranes initiating the lipid peroxidation and finally cause cellular injury. In contrast, MGS alleviates histological alterations affected by γ-irradiation, this result concurs with Adyab et al 29 who showed the improvement of MGS in hepatocytic irregularities induced by high-fat diet which can be attributed to its potent antioxidants and anti-inflammatory effects. The decreased inflammatory cellular infiltration in MGS treatment group is demonstrated by Palakawong et al 30 who stated that xanthones found within the MGS peel prevent the development of the COX-2 enzyme the major cause of inflammatory response, prostaglandin- 2 and nitric oxide. Decreased generation of reactive oxygen species (ROS) could also be associated with a decrease in the inflammation process to repair tissue damage. 31
The fine structure of hepatic tissue of γ-irradiated rats in this study realized conspicuous hazardous alterations, including degeneration in cytoplasm that is occupied by deteriorated mitochondria, pyknotic nuclei with loss of nuclear spherical configuration, nuclear envelope and condensed chromatin, fragmentation of rough endoplasmic reticulum (ER), dilatation of hepatic sinusoid with activation of Küpffer cell. These findings agree with those obtained by Samy et al 32 Kim et al 33 showed that the main risk of the radiation exposure of cells arises from the formation of (ROS) and the damage of cellular components such as DNA, cell membranes or cell organelles leading to loss function or even cell death. Some cytoplasmic degeneration, as well fragmentation of the rough endoplasmic reticulum after the exposure of accumulated dose of γ-irradiation was also detected. 32 Miller and Zachary 34 described that cytoplasmic degeneration as a result of irradiation is due to a progressive ischemia or hypoxia. Also they attributed the fragmentation of endoplasmic reticulum to the ingression of water and solutes into the cell which was referred to dysfunction of the cell membrane permeability. ER, responsible for the protein synthesis, is extremely sensitive to different kinds of endogenous and exogenous harmful stimuli, which can further result in the disturbance of ER lumen called ER stress 35 Moreover, NF-κB (p65) is known to be mediated by the signaling pathway from endoplasmic reticulum. 36 Presently, the ultrastracture of hepatocytes of γ-irradiated rats treated with MGS revealed high amelioration in cellular organelles such as mitochondria, nucleus and rough endoplasmic reticulum which are extremely sensitive to alterations in surrounding environment that have a direct effect on their structure, integrity, and function. MGS improved mitochondrial membrane potential and suppressed levels of cytochrome c, and caspase-3 activity, thus reducing hepatocellular apoptosis. 37
Shan et al 38 also reported the effect of MGS in the regulation the antioxidant sensor the nuclear factor erythroid 2–related factor- 2 (Nrf2) regulatory in the expression of antioxidant proteins. Antioxidants could significantly decrease the cellular ROS levels by increasing manganese superoxide dismutase expression that resulted in boosted cell survival. 39 On the other hand, anti-inflammatory drugs have also the capability to renovate cellular organs such as relieving endoplasmic reticulum stress via the inhibition of NF-κB activation and inflammatory response. 40
Previous research has recognized key molecular intermediaries tangled in radiation injury. Radiation activated nitric oxide (NO), that produced by macrophages and other immune cells trigger serious damage. 41 Additionally, rises of iNOS lead to stimulate transcription factors such as nuclear factor-κB (NF-kB) resulting in increased NO production that leads to caspase-mediated apoptosis, 41 which is in agreement with our results. Macrophage activity might be a cause of NO elevated level as a source of iNOS. 42 In γ-irradiated rats, radiation can alter the numbers and functions of immune system cells (e.g. increased numbers of lymphocytes T and macrophages) which consequently stimulate numerous of inflammatory mediators. 41 TNF has major immune functions in inflammation, cell proliferation control and division. 43 In our results the up-regulation of liver TNF-α, IL-6 and serum C-reactive protein (CRP) levels after exposure to 6 Gy of γ-irradiation might stimulate a feed forward loop in the NF-kB activation in Küpffer cells leading to secretion of IL-1α, TNF and also activation of quiescent hepatic stellate cells (HSCs) via stimulated TGF-β . Moreover, oxidative stress through its own increased TGF-β has driven apoptosis and enhanced hepatic damage cellular death, 44 which might explain radiation induced DNA fragmentation associated with boost of Caspase-3; as an indicator of apoptosis.
In this study administration of MGS (500 mg/kg B.W.) substantially lowers NO levels in irradiated rats. Reduction NO levels can be caused by iNOS expression inhibitors. Our results are consistent with Adyab et al 29 who found improved lipid profile, increased total antioxidant capacity and glutathione peroxidase level and also reduction of nitric oxide concomitant with lowered plasma pro-inflammatory markers IL-6 and TNF-α in obese rats supplemented with G. mangostana extract. On the same line, Mangostana extract showed greatly reduction in TGF-β. 45 The beneficial role of MGS in blocking inflammatory cytokines and improvement of DNA has been noted in current findings which may be attributed to the impact of DNA repair and the increase of protein synthesis.
The current immunohistopathological study showed that MGS reduces NF-κB-p65 expression. This result is in concurrence with Zou et al 46 who revealed that NF-κB-p65 expression was down regulated by MGS in lipopolysaccharide induced inflammatory model of rat intestinal epithelial cells. Buhrmann et al 47 showed that ΚB-p65 expression was down regulated via inhibition of p65 phosphorylation, IKK activity, and IKK phosphorylation in NF-kB signaling, by anti-inflammatory agent. The current elevation in oxidative stress enhanced NF-κB-p65 and decreased PCNA expression production following irradiation confirmed by the immunohistopathological study. This result is in parallel with the work of Karabulut-Bulan et al 48 screening that irradiation reduced PCNA in rat’s intestinal tissues. This down regulation of PCNA was mediated through the p53 pathway, which is a protein that regulates the cell cycle and cycle arrest. 49 DNA damage and stress signals may induce the activation of p53 protein, which has 3 major roles in DNA repair, apoptosis and growth arrest by suppressing PCNA. 49 On the other hand, Sheng et al 50 stated that the disrupted PCNA binds with p21 mutant caused arrest in G1 after-irradiation, suggesting that p21-PCNA interaction prevented rather than promoted cell-cycle arrest. MGS normalized the PCNA expression; this result is in the same current of previous study reported that the regulation of PCNA leads to the promotion of cell cycle progression and the reduction of cell arrest by the antioxidant agents. 51 The treatment with antioxidant significantly inhibited the production of ROS and consequently, suppressed p53 signaling to induce cell proliferation-associated proteins PCNA which promotes hepatocyte cell cycle and facilitates liver regeneration against liver injury. 52
Conclusion: The radioprotective role of MGS is illustrated in this study by alleviating oxidative stress, regulating signaling factors and by preventing liver cell death. Trials are essential to authenticate the independent or therapies combined with the use of MGS against hepatotoxicity induced by γ-irradiation as an adjuvant therapy. Our study suggests that the mangosteen fruit requires further work to understand its complex chemical composition.
Footnotes
Authors’ Note
All the ethical protocols for animal treatment were followed by the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978) and supervised by the animal facilities, National Center for Radiation Research and Technology, Atomic Energy Authority (Serial No.22A/20).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
