Abstract
To improve the water solubility of thymoquinone (TQ), a major constituent of Nigella sativa seed oil, a TQ-loaded self-nanoemulsifying drug delivery system (SNEDDS) was prepared. The SNEDDS formulation was optimized using almond oil (AO) (Oil; X1), tween 80 (surfactant; X2) and polyethylene glycol 200 (PEG 200) (cosurfactant; X3) compounds as independent variables. The results showed that the globule size ranged from 65 to 320 nm. In addition, a strong agreement was reached between the system estimation and the experimental values of globule size. To evaluate the gastroprotective effect of optimized TQ-loaded SNEDDS against indomethacin (Indo.)-induced gastric ulcers in comparison with non-emulsified TQ, the ulcer index and histopathological changes were estimated. Optimized TQ-loaded SNEDDS showed improved gastroprotective activity against Indo.-induced ulcers relative to the non-emulsified TQ. In addition, the gastroprotective index was improved by 2-fold in TQ-loaded SNEDDS as compared to non-emulsified TQ. This is attributed to the strong antioxidant and the cytoprotective activities of the TQ. These results demonstrate enhancement of the efficacy of TQ through the optimized SNEDDS.
Keywords
Introduction
Peptic ulcer is a common health problem that occurs in both young and old patients all over the world. The incidence of peptic ulcer disease (PUD) increases as a patient gets older with an estimated prevalence of about 10% in the general population. 1,2 PUD are described as defects or injuries in the gastroduodenal mucosa caused by peptic acid. 3 This ulceration is associated with stomach pain and often leads to gastrointestinal bleeding. PUD usually occurs at the lower part of the esophagus, lower portion of the stomach and upper duodenum. Two factors are considered as the main causes of PUD: Helicobacter pylori (H. pylori) infection and long-term use of non-steroidal anti-inflammatory drugs (NSAIDs). 4,5
Drug repurposing make potential applications for licensed or investigational products that fall beyond the original medical indication. This strategy provides numerous advantages over the production of a brand-new medication for a given 1 indication. First, and perhaps most significantly, the probability of failure is lower; since the recurrent drug has already been shown to be sufficiently safe in preclinical models and humans if early-stage trials have been completed, it is less likely to fail, at least from a safety point of view, in subsequent efficacy trials. Second, the timeline for the production of drugs may be shortened. 6 The main bioactive component of Nigella sativa L seed is thymoquinone (TQ). which present in many tropical countries. The seeds are known as black cumin, which is used as condiment and spices. Traditionally, active seeds are used in the treatment of influenza, asthma, bronchitis, cough, dizziness, hypertension, fever, inflammation, headache, and eczema. Preclinical screening for this black seed has been thoroughly studied for numerous physical disorders, including diabetes, hypertension, immunomodulatory and anti-inflammatory drugs, neuroprotective, and even cancer. 7 Since For hundreds of years, thymoquinone (TQ) has been commonly used for conditions ranging from a simple migraine to illnesses such as obesity, asthma, gastrointestinal problems, menstruation and lactation. 8,9 TQ is a member of the Ranunculaceae family, rising on Mediterranean coastal plants. Almond oil (AO) is a colloidal liquid containing proteins, minerals and vitamins. It was reported that AO affects the concentration of hydrochloric acid significantly which alters the stomach acidity environment. 10
NSAIDs are the most common prescribed medications for patients suffering from pain and inflammation 5,11,12 NSAIDs work as anti-inflammatories through the inhibition of Cyclooxygenases (COXs) enzymes. However, NSAIDs inhibit both COX-1 and COX-2 enzymes in a non-selective way. 13,14 COX-1 plays an important role in the protection of gastric mucosa by producing prostaglandins and thromboxane A2 that control the mucosal barrier in the gastrointestinal tract (GIT). On the other hand, COX-2 is responsible for prostaglandin-mediated pain and inflammation. Indo. is one of the most commonly prescribed non-selective NSAIDs, mainly used for its analgesic and anti-inflammatory effects. 15 However, ulcerations and gastric mucosal damage are the most common side effects associated with using Indo. 16 TQ is a bioactive agent that is poorly water soluble and exhibits low oral bioavailability upon oral administration. Due to the low aqueous solubility and bioavailability of TQ, TQ has been developed and tested with various self-nanoemulsifying drug delivery systems (SNEDDS) to improve its hepatoprotective effects and oral bioavailability. In rat models, hepatoprotective and pharmacokinetic trials of TQ suspension and TQ-SNEDDS have been performed. 17,18 Previous reports showed that oral administration of an aqueous suspension of TQ prevents gastric mucosal injuries caused by ethanol and strong alkalies, the most commonly employed tests in the evaluation of anti-ulcer and cytoprotective activities. It is suggested that oxygen radicals may contribute to the formation of ethanol-induced gastric mucosal lesions, and antioxidants are protective against the damage caused by these oxidants. 7,19
SNEDDS are effective nanocarriers in the drug delivery system. 20 The trial-and-error assessment of the optimum ratios of SNEDDS components is carried out based on the conventional 1-factor-by-time method. Nevertheless, this approach is time-consuming, labor-intensive and unreliable. In addition, this approach often provides insufficient data to evaluate the effect of each factor and its effect on the responses. Experimental statistical modeling designs have been developed to forecast the results of mix-related factors and the interaction of multi-component independent variables. In addition, a numerical optimization technique based on surface reaction methodology and innovative designs such as central composite, Box-Behnken, factorial and mixture designs have been developed. 21,22 Extreme vertex design is used for the cases where there are constraints on 1 or more components. Mixture design, as one of the experimental statistical designs, deals with components that are variables as proportions of the total system (mixture). Mixture designs can be utilized in the optimization of a SNEDDS formula as the system components’ percent adds up to 100%. The aim of this work was to improve the solubility of TQ by a SNEDDS-loaded formula and optimize the prepared formulations for globule size using experimental designs. A mixture design with the composition of AO (oil; X1), tween 80 (surfactant; X2), and Polyethylene glycol 200 (PEG 200) (cosurfactant; X3) as independent variables was utilized in this study. The optimized formula was investigated for gastroprotective effect in a rat model.
Materials
TQ, AO, Polyethylene glycol (PEG, MW 200) and tween 80 were purchased from Sigma-Aldrich, USA. Indo was kindly gifted by Jamjoom Pharmaceuticals, Jeddah, Saudi Arabia.
Methods
Formulation of TQ-Loaded SNEDDS
Based on the preliminary studies, multiple vehicles were screened, and the compositions of AO, tween 80, and PEG 200 were selected. For any oil, surfactant and co-surfactant, the total combination was always 100%. TQ was loaded in a SNEDDS formula (10 mg/1 g) as previously described. 23 Briefly, the SNEDDS formula was prepared by mixing AO, Tween 20 (surfactant), and PEG 200 (co-surfactant). The prepared SNEDDS were stored at ambient temperature and used for the experiments within 4 weeks. 24
Optimization of TQ-Loaded SNEDDS
Optimization of the TQ-loaded SNEDDS were performed by a mixture design using the Statgraphics plus, version 4 (Statgraphics Centurion XV version 15.2.05 software) (Manugistic Inc., PA, USA). The test was planned to use the 3 elements, including the concentrations of AO (oil; X1), tween 80 (surfactant; X2) and PEG 200 (cosurfactant; X3), that were set at 0.1%-0.3%, 0.2%-0.4% and 0.4%-0.6%, respectively. For any study, the concentrations of X1, X2 and X3 were added up to 100%. Globule size (nm) was selected as the design response.
Globule Size Determination
Globule size was analyzed by s particle size analyzer (Nano-ZS, Marlvern Instrument, Worcestershire, UK) using a dynamic light-scattering technique. For this purpose, 100 µL of each TQ-loaded SNEDDS were diluted with 10 mL of 0.1 N HCl (to simulate a gastric environment), vortexed for 2 minutes and then the size was measured. 25
Prediction and Preparation of Optimized TQ-Loaded SNEDDS Formulation
According to the optimization design, the results obtained from the total of 22 formulations were statistically analyzed obtained from the total of using Statgraphics software with ANOVA and multiple-response optimization analysis. The optimum formulation was prepared, evaluated and compared with the optimal formulation that was actually proposed by the software.
In Vivo Study
25 Sprague-Dawley male rats were (weighing 220-250 g) divided into 5 groups (gps; n = 5/gp) randomly. The analysis was performed using a slight modification of the specified procedure. 26 Before the experiment, the animals were fasted for 24 hours, but were given free access to water. All 5 groups of rat were orally gavaged, including untreated group (control, gp1), Indo (gp2), plain formula (AO, tween 80 and PEG200) SNEDDS (gp3), TQ suspended in water with dose 1.5 mg/kg (gp4), and the last group received a dose equal to 1.5 mg/kg TQ of TQ-SNEDDS (gp5). After 1 h, all rats in groups 2,3,4 and 5 received Indo (20 mg / kg, ip) to cause gastric ulcers. 27,28 The rats were sacrificed after 4 h by an overdose of diethyl ether. The adopted guidelines were in accordance with “Principles of Laboratory Animals Care” (NIH publication no. 85-23, revised 1985). The protocol for the animal studies were conducted in accordance with the ethical rules of the ethics committee at the Faculty of Pharmacy, King Abdulaziz University, Jeddah, Saudi Arabia, that approved our experiments with application no. PH-45-1440.
Peptic Ulcer Lesions Evaluation
The stomachs were rinsed with deionized water. Stomachs were cleaned, pinned on board and pictures were taken for the stomach samples then were scored for the degree of mucosal damage using Image J software. Areas of mucosal damage were expressed as a percentage of the total surface area of the examined stomach. The ulcer index (UI) is the mean ulcer score of each animal. The ulcer score was determined by measuring the length of each lesion along its greatest diameter. The ulcer index was used to calculate the preventive index of the drug which is the percentage inhibition of gastric mucosal damage produced by such drug.
Determination of Gastric Secretion Parameters
Equal volumes of gastric juices were completed to 2 ml by deionized water to allow measurement of pH using a pH meter. The initial pH of gastric juice was measured to indicate the effect of various treatments on gastric acid secretion. Then the gastric juices were titrated to pH = 7 using 0.0025 N NaOH and Toepfer’s reagent as an indicator. The total acidity was calculated using the equation:
Histopathological Analysis
Stomach samples were fixed overnight in a formalin solution buffered by 10 percent alcohol and xylene. The tissue was then wrapped under the Embedding Station (Microm EC 350, Thermo Fisher Scientific, Germany) in paraffin wax (Paraplast, Leica, Singapore). The Rotary Microtome was appended to fabric frames (3-5 m) (Accu-Cut SRM 200, Netherlands). Finally, haematoxylin and eosin (H&E) staining was performed. Briefly, a 1 cm segment of each histological section was evaluated for loss of epithelial cell (score: 0-3), mucosal edema (score: 0-3), hemorrhagic damage (score: 0-3), the presence of inflammatory cells (score: 0-3), and the total lesion (score 0-3). 29
Statistical Analysis
The findings were represented as mean ± standard deviation (SD) and evaluated using 1-way variance analysis (ANOVA) followed by multiple comparison Dunnett tests to compare therapies with the ulcer control group. Significant levels were evaluated at P < 0.05.
Results
Statistical Analysis Using Extreme Vertices Mixture Design
To achieve TQ-loaded SNEDDS with minimum globule size, AO, tween 80 and PEG 200 were the design independent variables (X1, X2, and X3, respectively; Table 1 previous manuscript referred to TQ solubility in various oils, surfactant and co-surfactants. 7 SNEDDS droplet size (nm) was the selected response. Response and contour plots showing the effects of components on SNEDDS’ properties were assigned to deduce the mixture region as shown in Figure 1. The results revealed that globule size ranged from 65 to 320 nm, as shown in Table 2.

Estimated response surface 3D (A) and contour (B) plots for the effects of independent variables on SNEDDS globule size (nm).
The Independent and Dependent (Response) Design Variables.
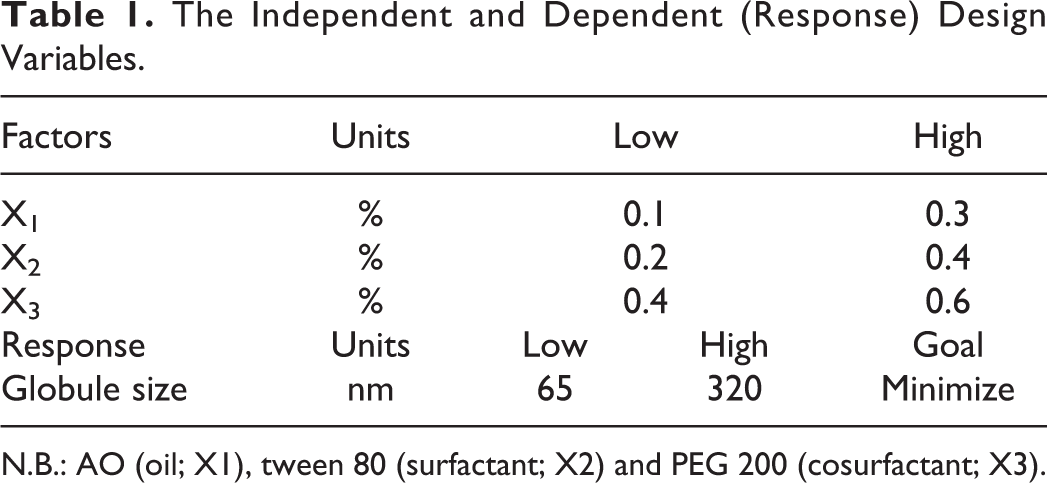
N.B.: AO (oil; X1), tween 80 (surfactant; X2) and PEG 200 (cosurfactant; X3).
Experimental Runs and the Observed Response (Actual, Predicted, and Residual Values).

N.B.: AO (oil; X1), tween 80 (surfactant; X2) and PEG 200 (cosurfactant; X3).
The results of globule size showed an positive relation with X1. As the concentration of X1 increased, globule size increased. This is indicated in F2 (30% X1), F15 (20% X1) and F4 (10% X1) where globule sizes were 320 nm, 172 nm and 65 nm, respectively (Table 2). A cubic model equation for the effects of the investigated factors (X1-X3) on the SNEDDS globule size was calculated (equation 2).

Validation of the Optimized TQ-Loaded SNEDDS Formulation
Extreme vertices mixture experimental design deduced the optimum TQ-loaded SNEDDS formulation that was prepared and evaluated for particle size in the lab (Table 2). The obtained results indicated that a combination of independent factors for the optimized TQ-loaded SNEDDS formulation exhibited the actual particle size of 67.7 nm that was comparable to the predicted size by the design of 64.8 nm with residual 2.9 (Table 3).
Optimum Levels for TQ-Loaded SNEDDS Factors and the Predicated and Actual Response Values.

Abbreviations: TQ, thymoquinone; SNEDDS, self-nanoemulsifying drug delivery system.
N.B.: AO (oil; X1), tween 80 (surfactant; X2) and PEG 200 (cosurfactant; X3).
Gross and Histopathological Evaluation of Gastric Lesions
Gross examination of the stomachs obtained from control animals showed normal gastric tissues with no apparent epithelial loss, erosions or inflammation (Figure 2A). Whole stomachs collected from Indo and plain SNEDDS-treated animals showed severe congestion, inflammations and erosions (Figure 2B and C). TQ-R showed partial protection with mild to moderate edema inflammations with scattered erosions (Figure 2D). TQ-SNEDDS afforded obvious protection as evidenced by the almost normal appearance of the stomachs with scattered edema and mild-to-almost-nil inflammation (Figure 2E). Histological examination of H&E-stained stomach sections obtained from the control group revealed normal histological architecture of the glandular gastric mucosa and submucosa (Figure 3A). Meanwhile, a severe gastric alteration was detected in the Indo-alonetreated group. The mucosa showed severe destruction of the epithelial covering and the underneath gastric acini accompanied by abundant inflammatory edema (Figure 3B). Almost similar gastric damage was observed in the plain SNEDDS group with multifocal hemorrhagic areas and golden yellow-to-brown hemosiderin pigment as well as necrosis of the surrounding tissue. Additionally, some examined sections showed aggregations of mononuclear inflammatory cells in the affected mucosa (Figure 3C). Sections obtained from animals treated with TQ-R prior to the Indo challenge exhibited moderate gastric improvement that exhibited apparently normal mucosa in some examined sections; however, mild cystic dilation of glandular acini was noticed. A few sections showed moderate expansion in the submucosa with edema, inflammatory cell infiltration and congested blood vessels (Figure 3D). Examination of the glandular stomach sections of animals in the TQ-SNEDDS group indicated the highest protection against Indo-induced injury. This was evidenced by the absence of abnormal histopathological alterations in most examined sections except for a few scattered foci of inflammatory cell aggregations (Figure 3E). Semi-quantitative evaluations of histopathological alterations are presented in Table 4. Control animals showed almost no epithelial loss, edema or inflammatory infiltrations. However, animals in both the Indo and Plain SNEDDS groups showed severe epithelial loss, edema, hemorrhage and inflammation. TQ-R treated animals showed partial protection with less edema and inflammation. TQ-SNEDDS exhibited the highest protection with mild edema and almost no hemorrhage.

Gross evaluation of gastric lesions. Untreated gp (control, gp1, A), dose 20 mg/kg Indo (gp2, B), plain formula (AO, tween 80 and PEG200) SNEDDS (gp3, C), TQ suspended in water with dose 1.5 mg/kg (gp4, D), and dose equal to 1.5 mg/kg TQ of TQ-SNEDDS (gp5, E). Groups are treated with the oral gavage. After 1 h, all rats in groups 3, 4 and 5 received Indo (20 mg/kg) orally for causing gastric ulcers.

Histological examination of untreated gp (control, gp1, A), dose 20 mg/kg Indo (gp2, B), plain formula (AO, tween 80 and PEG200) SNEDDS (gp3, C), TQ suspended in water with dose 1.5 mg/kg (gp4, D), and dose equal to 1.5 mg/kg TQ of TQ-SNEDDS (gp5, E). Groups are treated with the oral gavage. After 1 h, all rats in groups 3, 4 and 5 received Indo (20 mg/kg) orally for causing gastric ulcers.
Semi-Quantitative Evaluation of Histopathological Alterations.

Abbreviations: Indo, indomethacin; SNEDDS, self-nanoemulsifying drug delivery system; TQ, thymoquinone; R, raw.
(-) = no, (+) = mild, (++) = moderate, (+++) = severe.
Effect of Raw TQ and Optimized TQ-SNEDDS on Gastric Secretion Volume and pH
Gastric secretion volume and pH are represented in Figure 4. The TQ-SNEDDS formulation provided a 78.95% reduction in Indo-induced ulcers with only some red color in some sections (Figure 4A). and the results revealed a significant difference (P < 0.05) between the Indo group and the control group for both gastric secretion volume and pH. Additionally, the data revealed that there is a significant difference between the TQ-SNEDDS and the Indo groups (P < 0.05) for both volume and pH. The plain formula and raw TQ showed no significant difference in gastric secretion pH in comparison with the Indo group. On the other hand, the results revealed that both the plain formula and raw TQ showed a significant difference (P < 0.05) in gastric secretion volume.
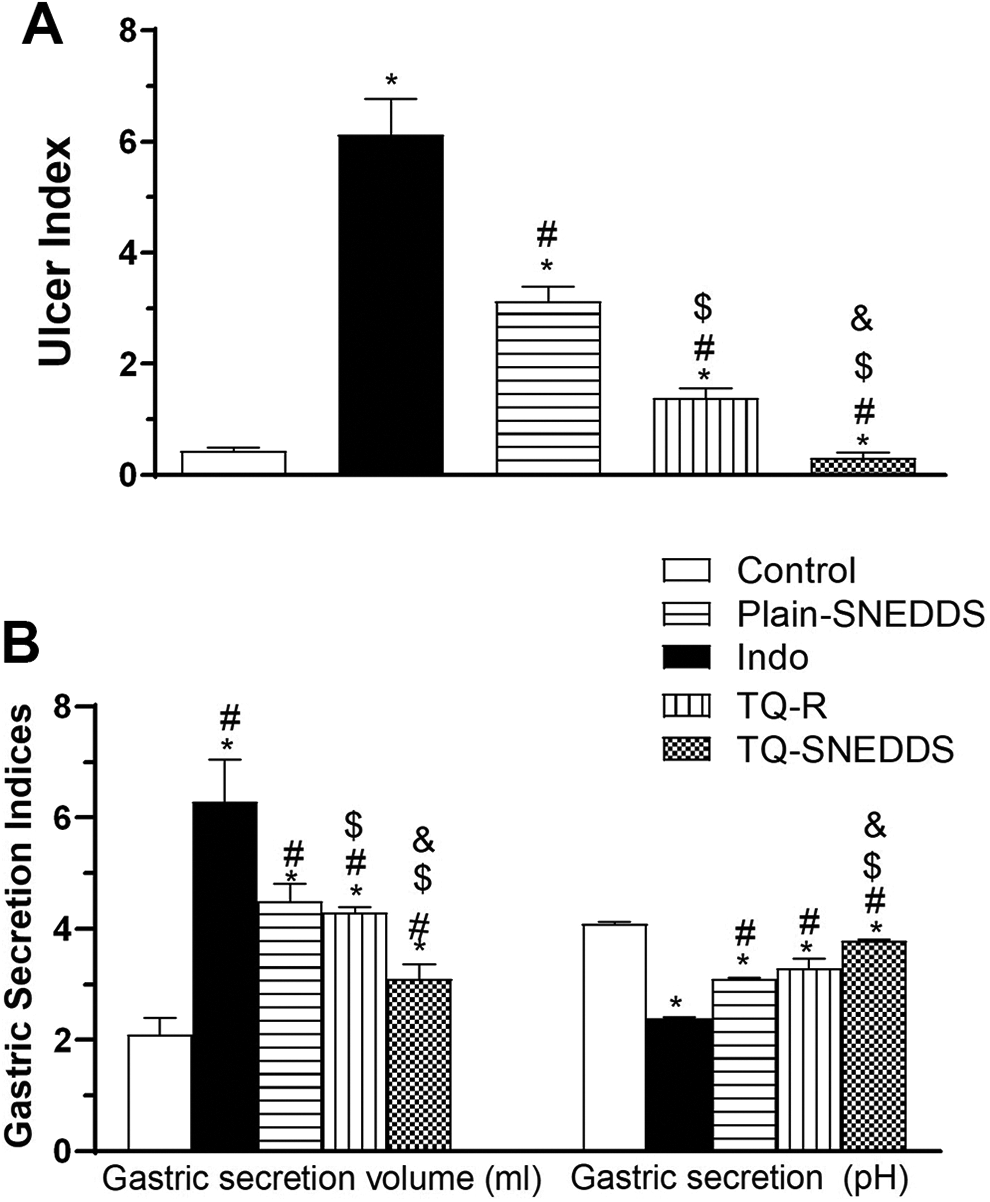
The effect of indo, raw TQ, plain formula and optimized TQ-SNEDDS formula on U.I. (A), and (B) the effect of indo, raw TQ, plain formula and optimized TQ-SNEDDS formula on gastric secretion volume (left section) and gastric secretion pH (right section). Data are presented as mean ± SE. *Significantly different from corresponding control at P < 0.05; # significantly different from indo gp. at P < 0.05; $ significantly different from plain-SNEDDS at P < 0.05., & significantly different from TQ-R at P < 0.05.
Discussion
The present study was intended to optimize the factors effecting SNEDDS formulation to improve the protective effects of TQ-loaded SNEDDS on the development of ulceration mediated by Indo in rats. The use of SNEDDS is one of the most interesting approaches to improve solubility and oral absorption of poorly water-soluble drugs. SNEDDS have gained attention due to their properties and advantages of drug-loading. 12,30 -35 To achieve TQ-loaded SNEDDS with minimum globular size, an experimental mixture design was created using AO, tween 80 and PEG 200 as the design independent variables, and SNEDDS globule size (nm) as the response (Table 2). The effectiveness of TQ in treating peptic ulcers is maximized by nano-sized oil globules. The most important factor in the drug’s bioavailability is droplet size. A small droplet size provides a large surface area that facilitates the epithelial layer’s solubilization and penetration. The size of emulsion droplets affects the distribution of the drug, thus improving the penetration of TQ into the gastric membrane. 36 The software showed that the P value was significant (P < 0.05) for the linear and cubic models when compared with the quadratic and special cubic models. The cubic model was selected on the basis of the most complicated model with a P value < 0.05. The cubic model adds other third-order terms. In addition, the statistics results showed that the cubic model maximizes the adjusted R-square value (adj. R2) when compared with other models. R2 reflects the variance of the response factor attributed to all the independent variables in the specified template (Table 5). The design deduced the optimized formulation that was prepared and compared with predicted values. After that, the formula was investigated for in vivo activity.
Cubic Model ANOVA for SNEDDS Globule Size.

Abbreviations: Df, degree of freedom; MAE, mean absolute error.
Indo is known to cause ulcers due to local or indirect action. 37,38 Local impact relates to the immediate local contact between the mucosa and the medication, contributing to inflammation, tissue cytotoxicity and mucosal injury. The indirect effect of Indo which causes ulcers is the inhibition of prostaglandin synthesis by a cyclooxygenase (COX) pathway. 39 Gross analysis of the ulcer control group revealed a few large, dark hemorrhagic streaks (Figure 2B). Among the pre-treatment groups, the TQ group showed a significant (P < .05) reduction in the ulcer region, accompanied by a plain SNEDDS gp, which showed negligible defense against Indo-mediated ulcers. The macroscopic analysis of TQ and TQ-SNEDDS revealed some red color spots erosion. As one of the benefits of SNEDDS is that it does not harm healthy human or animal cells, SNEDDS are ideal for human or animal use. In contrast, oil droplets are quickly expelled from the abdomen and facilitate a wide distribution of the medication throughout the intestinal tract, thereby reducing inflammation. SNEDDS has a positive effect on TQ distribution. 40,41 SNEDDS improves medication-remaining time in the target, enhances bioavailability, and decreases TQ degradation. The excessive hepatic first-pass effect and the release of formulations from the aqueous media of the intestinal contents are significant barriers to the absorption of poorly water-soluble lipid products because of poor aqueous solubility. SNEDDS is considered a possible vehicle for improving the oral bioavailability of medications that are poorly soluble in water. Different mechanisms for the absorption of SNEDDS from the intestine have been reported. SNEDDS provides a range of advantages, including high producibility, no organic solvents, thermodynamic stability and increased bioavailability of the oral drug. The motility of the gastrointestinal tract allows the stirring of the SNEDDs. TQ controls a wide variety of functions of the body and is a precursor to various molecules’ inflammatory process. The goal of the study was to formulate TQ as a SNEDDS, to increase the gastric ulcer defense efficacy of TQ. Direct absorption through the GI tract, which is due to small particle size and lipid content, is one of these mechanisms. The particle size of the SNEDDS formulations was almost less than 100 nm, and this decrease in particle size affects the SNEDDS surface area. Particularly in the lymphatic region of the tissue, this small size allows better absorption in the intestine, thus avoiding hepatic first pass metabolism. In the small intestine, the lipids could induce bile secretion, and SNEDDS were associated with bile salt to form mixed micelles that helped the intact SNEDDS get into the lymphatic vessels and first-pass metabolism of the liver. Significant factors in the encouraged absorption are the uptake and lymphatic transport of intact SNEDDS. Rising permeability by surfactants is another mechanism that promotes the absorption of SNEDDS. By disrupting the cell membrane and reversibly opening the tight junction of intestinal epithelial cells, surfactants can increase intestinal epithelial permeability and thus facilitate paracellular absorption. 17,42 -45
This may clarify the protective effect of TQ SNEDDS on the abdomen. 46 Nano globules exclusively adhere to inflamed tissue in gastric ulcers and duodenal ulcers. Inflammation leads to increased development of mucus in the infected area. Differences in ulcerated tissues in a normal population have therefore become less noticeable than in colitis, where variations in the volume of mucosa and bacterial clearance have a greater impact on particle compliance. The size-dependent deposition of nano globules therefore represents an important step in the development of a new drug supply strategy. 39,47 There was increased adhesion of nano globules in thicker, inflamed tissue layers, while reliance on size was found in ulcerated regions. This theory could be related to the results of studies in which TQ could stay in the stomach for hours and cause delayed absorption.
Conclusion
The formulations of TQ-loaded SNEDDS were successfully optimized using a mixture model. The prepared TQ-loaded formulation of SNEDDS, consisting of almond oil (oil; X1), tween 80 (surfactant; X2) and PEG 200 (cosurfactant; X3), demonstrated a decrease in globular size. Clear agreement was reached between the template estimation and the experimental values of the globule size used as a response variable. TQ-loaded SNEDDS improved antiulcer protection in the Indo-induced ulcer system, which is due to the existence of various phytochemicals in TQ. TQ-loaded SNEDDS have been shown to increase efficacy as an anti-ulcer agent. Overall results have shown that this combination of TQ-loaded SNEDDS could be a viable method for enhancing the protective properties of TQ, which has shown great potential for use in nutraceutical fields.
Footnotes
Authors’ Note
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, under grant no. (G-431-166-1440). The authors, therefore, acknowledge with thanks DSR for technical and financial support.