Abstract
Influenza A virus infections can cause acute lung injury (ALI) in humans; thus, the identification of potent antiviral agents is urgently required. Herein, the effects of salidroside on influenza A virus-induced ALI were investigated in a murine model. BALB/c mice were intranasally inoculated with H1N1 virus and treated with salidroside. The results of this study show that salidroside treatment (30 and 60 mg/kg) significantly attenuated the H1N1 virus-induced histological alterations in the lung and inhibited inflammatory cytokine production. Salidroside also decreased the wet/dry ratio, viral titers, and Toll-like receptor 4 expression in the lungs. Therefore, salidroside may represent a potential therapeutic reagent for the treatment of influenza A virus-induced ALI.
Introduction
As one of the most important human pathogens, influenza A virus infections can result in seasonal and pandemic morbidity and mortality. Influenza A viruses infect millions of people each year, leading to severe respiratory distress and death. Severe influenza virus infection, acute respiratory distress syndrome (ARDS), bilateral pulmonary infiltration, and hypoxemia can occur clinically. 1 Moreover, hypoxic respiratory failure is the main cause of death. Recently, human and avian influenza viruses that continue to infect humans, causing acute lung injury that results in a large number of deaths, have attracted worldwide attention and an urgent need to identify potent antiviral agents. 2
Rhodiola rosea is a common folk medicine, primarily used to treat symptoms of altitude sickness, altitude hypoxia, and hypoxia. 3 The main effective component of Rhodiola rosea is salidroside (SDS) (Figure 1A), which is a phenylpropanoid glycoside. A large number of studies have shown that SDS has multiple pharmacological effects, including anti-inflammatory, anti-viral, anti-hypoxia, anti-tumor, anti-fibrosis, and anti-aging properties. 4 Moreover, SDS has been shown to exhibit anti-viral activity against dengue virus by inhibiting viral protein synthesis and boosting host immunity. 5 Our previous research demonstrated that the anti-inflammatory effects of SDS against lipopolysaccharide-induced acute lung injury (ALI) in mice are attributed to its ability to inhibit the Toll-like receptor 4 (TLR4)-mediated nuclear factor-kappa B (NF-κB) signaling pathway. 6

Chemical structure of salidroside (SDS) (A) and protective efficacy of salidroside treatment in a prophylactic setting against influenza virus challenge in a mouse model. Weight loss (B) and viral titers in the lungs (C) of influenza virus-infected mice. Weight loss and survival (D) were measured daily from the date of challenge until 14 days after challenge. The dashed line represents 75% of the initial body weight, which was defined as the humane endpoint.
In this study, we examined the protective effects of SDS in a mouse model of influenza virus-induced ALI and clarified the potential anti-inflammatory mechanism. The results of this study may provide a novel method for the clinical treatment of influenza virus-induced ALI.
Materials and Methods
Animal Preparation and Treatment
Six-week-old healthy female BALB/c mice (n = 96) were maintained in a pathogen-free facility and housed in cages containing sterilized food and drinking water, as described previously. 7 A/Puerto Rico/8/1934(H1N1) virus (PR8, mouse-adapted virus) was grown and titrated in 10-day-old embryonated chicken eggs. SDS was purchased from the National Institute for the Control of Pharmaceutical and Biological Products, Beijing, China.
The mice were randomly divided into the following groups: 1) control; 2) virus infected; 3) SDS (15, 30, and 60 mg/kg) + virus infected; and 4) oseltamivir + virus infected, as described previously. 8 There are 6 groups in this study, and each group contained 16 mice. BALB/c mice were intranasally inoculated with 106 50% tissue culture infective doses (TCID50) per 0.1 ml of PR8 virus, as described previously. 7 The mice were treated with 15, 30, and 60 mg/kg of body weight/day of SDS or 20 mg/kg/day oseltamivir (as a positive control) twice daily via intraperitoneal injection (i.p.) for 5 days. Treatment was initiated at the same time.
On Days 3 and 6 post-viral challenge, the plasma and lung tissues from 5 mice per group were collected and the viral titers and cytokine levels were measured. The survival rate, degree of weight loss, and disease symptoms were observed in the remaining 6 mice for the 14 days following inoculation as described previously. 7 The lungs were collected from the mice for virus titration in embryonated chicken eggs using the Reed and Muench method. 9 All animal experiments were performed in accordance with the recommendations of the Office International des Epizooties (OIE). 10
Histopathological Analysis
Tissue sections were subjected to histopathological analysis. Lung tissues from virus-inoculated mice were fixed in 10% neutral buffered formalin for at least 24 h before processing. The tissues were embedded in paraffin using standard tissue processing procedures. Sections (4-µm thick) were cut and fixed on glass slides. Standard hematoxylin and eosin (HE) staining was performed as previously described. 7
Lung Wet Dry Ratio
The lung wet dry ratio (W/D ratio) was used to evaluate the severity of pulmonary edema. Dry ice was used as narcotic and euthanasia to alleviate the pain of mice. The lungs were removed and the wet weight was measured. The lungs were then placed in an incubator at 60°C for 24 h to obtain the dry weight as previously described. 11
Cytokine Measurement
After mice were anesthetized with dry ice as previously described, 12 a trachea cannula was inserted into the lungs, through which PBS was injected and aspirated to collect BALF. The fluid recovery rate was greater than 80%. The BALF was collected and centrifuged, and the cytokine levels in the supernatant were subsequently analyzed. The levels of the proinflammatory cytokines IL-1β, IL-6, and TNF-α in the BALF supernatant were evaluated using an ELISA in accordance with the recommended manufacturer instructions. The protein concentrations in the supernatant of the BALF were quantified using a bicinchoninic acid protein assay kit to evaluate the vascular permeability in the airways of mice as previously described. 6
Western Blot Analysis
The lungs of the mice were removed and immediately frozen in liquid nitrogen until homogenization. Protein was extracted from the lung tissue homogenates and quantified using a bicinchoninic acid protein assay kit, per the manufacturer’s instructions. The same amount of protein was loaded into each well of a sodium dodecyl sulfate polyacrylamide gel. The protein was transferred to polyvinylidene difluoride membrane. The immunoactive proteins were detected by using an enhanced chemiluminescence Western blot detection kit per the manufacturer’s instructions, as described previously. 13
Statistical Analysis
All data were expressed as the mean ± SD and statistical analysis was performed with SPSS 17.0. The differences were analyzed using a one-way ANOVA (Dunnett’s t-test), and a threshold of P < 0.05 was considered to be statistically significant.
Results
Clinical Symptoms
On day 3 post-inoculation, the infected mice exhibited similar clinical symptoms (inactivity and wrinkled fur). On day 5, the infected mice developed more severe clinical symptoms, including inactivity, wrinkled fur, hunched posture, loss of appetite, shortness of breath, and loud crackling. Treatment with SDS or oseltamivir relieved these clinical symptoms and virus titer of lung (Figure 1B and 1C). From day 5 post-inoculation, the infected mice began to die, and the body weight of the infected mice was significantly decreased on day 6 post-inoculation compared with the control group (Figure 1D).
Histopathological Analyses
To evaluate the histological changes in the influenza virus-infected mice after SDS treatment, the lung tissues were subjected to HE staining. Histopathological analyses revealed that lung tissues from the control mice exhibited a normal structure with no histopathological changes under a light microscope. At 3 days post-inoculation, the lung tissue had a multifocal mild or moderate interstitial inflammatory hyperemia and exudative pathological changes. By 6 days post-inoculation, the lesions in the lung tissue became enlarged and multiple patchy lesions had fused (Figure 2). These influenza virus-induced pathological changes were significantly attenuated by both SDS (30 and 60 mg/kg) and oseltamivir treatment.
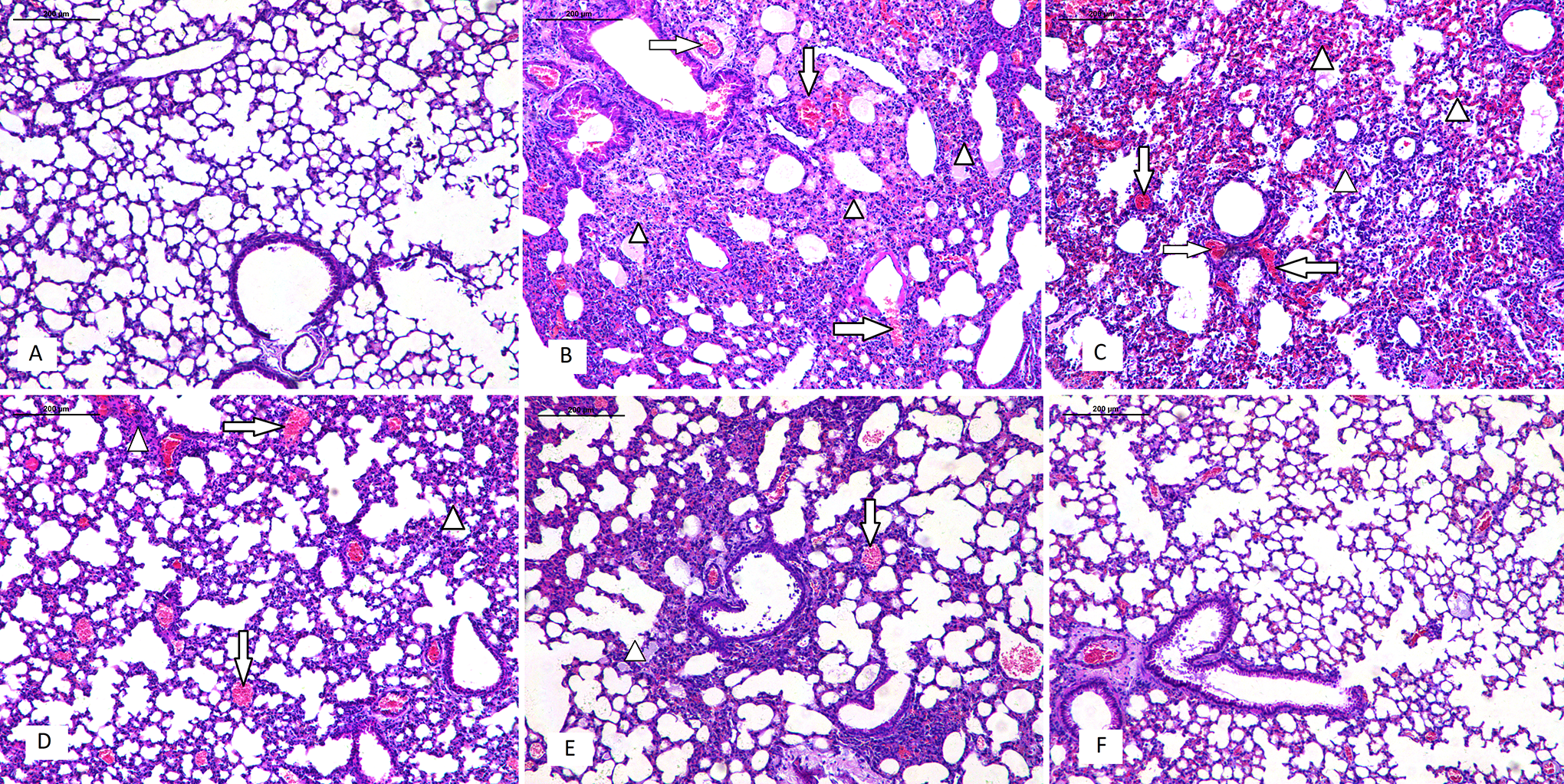
Effects of salidroside (SDS) treatment on histopathological changes in the lung tissues of influenza virus-induced acute lung injury mice at 6 days post-infection. (A) Control group; (B) virus infected group; (C-E) SDS (15, 30, and 60 mg/kg) + virus infected group; (F) oseltamivir + virus group. Representative images of lung sections from inoculated mice stained with hematoxylin and eosin are shown. The influenza virus-infected mice displayed severe interstitial pneumonia in the lung tissues, which exhibited alveolar lumen flooded with dropout of alveolar cells, erythrocytes, and inflammatory cells (triangle); and congestion in the blood vessels (arrow).
Lung W/D Ratio
The results showed that influenza virus infection could significantly increase the lung W/D ratio (Figure S1A). However, treatment with SDS (30 and 60 mg/kg) or oseltamivir significantly decreased the lung W/D ratio (P < 0.05) compared to those in the virus infected group. The total protein concentration in the BALF was significantly increased in the virus infected group (Figure S1B). In contrast, treatment with SDS (30 and 60 mg/kg) or oseltamivir significantly decreased the total protein concentration (P < 0.05) compared to the virus infected group.
Levels of Proinflammatory Cytokines
Proinflammatory cytokines appear during the early phase of an inflammatory response, which play a critical role in ALI and contribute to the severity of lung injury. Previous studies have shown that increased levels of proinflammatory cytokines, TNF-α, IL-6, and IL-1β in the BALF are observed in ARDS patients, and the persistent elevation of proinflammatory cytokines in individuals with ALI has been associated with more severe outcomes. 12 After 6 days post-inoculation, the effect of SDS on IL-1β, IL-6, and TNF-α production was analyzed by ELISA. The results showed that the concentrations of IL-1β, IL-6, and TNF-α in the BALF were significantly increased after influenza virus inoculation (Figure S1C-E). However, treatment with SDS (30 and 60 mg/kg) or oseltamivir significantly reduced the level of IL-1β, IL-6, and TNF-α production compared to those in the virus infected group (P < 0.05).
Discussion and Conclusion
TLR4 is one of the most well-characterized TLRs, and is located on the plasma membrane. 14 Previous studies have shown that influenza virus infection can lead to activation of various TLR signaling pathways. 14,15 In particular, inactivated H5N1 influenza virus can induce ALI through the TLR4-NF-κB signaling pathway. 14 As an important transcription factor, NF-κB plays a critical role in diverse cellular processes. Inhibitor of NF-κB (IκBs) and NF-κB p65 are important factors involved in the TLR4-NF-κB signaling pathway. A Western blot analysis was performed to detect the expression of TLR4, IκB-α, and NF-κB p65 (Figure 3A). Our results show that influenza virus significantly increased TLR4 expression and IκB-α and NF-κB p65 phosphorylation, whereas treatment with SDS (30 and 60 mg/kg) significantly decreased TLR4 expression, as well as IκB-α and NF-κB p65 phosphorylation (Figure 3B).

Salidroside (SDS) pretreatment inhibited influenza virus-induced activation of NF-κB by Western blot (A) and SDS treatment protects against influenza A virus-induced acute lung injury in mice (B). The dotted line indicates the phosphorylation process.
ALI is characterized by severe hypoxemia, pulmonary edema, and neutrophil accumulation in the lungs, and there are few effective measures for successful treatment. Therefore, the development of novel therapies for ALI is urgently required. 16 -19 In this study, an ALI model was established by influenza virus infection in mice, and we observed the effects of SDS on influenza virus-induced ALI. Thus, SDS may represent an effective clinical treatment for influenza virus-induced ALI.
Supplemental Material
Supplemental Material, sj-tif-1-dos-10.1177_15593258211011335 - Salidroside Protects Against Influenza A Virus-Induced Acute Lung Injury in Mice
Supplemental Material, sj-tif-1-dos-10.1177_15593258211011335 for Salidroside Protects Against Influenza A Virus-Induced Acute Lung Injury in Mice by Rufeng Lu, Yueguo Wu, Honggang Guo, Zhuoyi Zhang and Yuzhou He in Dose-Response
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Scientific Research Fund of Zhejiang Province Chinese Medicine (2019ZA045).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
