Abstract
Animals exposed to significant stress express multi-modal responses to buffer negative impacts. Trans-generational impacts have been mainly studied in maternal lines, with paternal lines having received less attention. Here, we assessed paternal generational effects using irradiated male crickets (Acheta domesticus), and their F1 offspring (irradiated males mated to unirradiated females). Paternal transmission of radiation impacts emerged in multiple life history traits when compared to controls. Irradiated males and their F1 offspring expressed hormetic responses in survivorship and median longevity at mid-range doses. For F0 males, 7 Gy & 10 Gy doses extended F0 longevity by 39% and 34.2% respectively. F1 offspring of 7 Gy and 10 Gy sires had median lifespans 71.3% and 110.9% longer, respectively. Survivorship for both F0 7 Gy (p < 0.0001) and 10 Gy (p = 0.0055) males and F1 7 Gy and 10 Gy (p < 0.0001) offspring significantly surpassed that of controls. Irradiated F0 males and F1 offspring had significantly reduced growth rates. For F0 males, significant reductions were evident in 4Gy-12 Gy males and F1 offspring in 4 Gy (p < 0.0001), 7 Gy (p < 0.0001), and 10 Gy (p = 0.017). Our results indicate paternal effects; that irradiation directly impacted males but also mediated diverse alterations in the life history features (particularly longevity and survivorship) of F1 offspring.
Introduction
Organisms have evolved to manage diverse internal and external disturbances. 1 Stress can be considered any disturbance (chemical, physical, or biological), that inflicts damage, disrupts homeostasis or elicits compensatory responses. 1 -3 Stress responses can resist or suppress stressors and improve or stabilize organismal and cellular functions. 1,2 However, if stress is prolonged or tolerance is exceeded, dysfunction and damage may result. 1
Ionizing radiation acts as a cellular stressor via direct damage (especially DNA), and via the creation of reactive oxygen species’ (ROS) at concentrations that exceed cellular tolerance. 4 On a cellular level, high doses cause excess ROS’s that disrupt cellular processes, compromise genomic stability and disrupt protein and lipid structures and functions. 4 At an organism level, excess ROS’s can cause a plethora of detrimental impacts including cancer, increased dominant lethal mutations, reduced survival, fertility and death. 5 -9 However, at low doses, ionizing radiation has been shown to elicit beneficial (hormetic) effects. This biphasic model of radiation exposure is characterized by low-dose benefits and high-dose harm. 1
At low doses, ionizing radiation may increase fitness via several life-history parameters. In Acheta domesticus, low-dose juvenile radiation can increase female fecundity, as well as offspring size and performance. 10 Longevity is a well-studied life-history trait relevant to ionizing radiation. Increased longevity by low-dose radiation has been demonstrated in males and females of Tribolium confusum, Drosophila subobscura, D. melanogaster, Musca domestica, Acheta domesticus, and Bracon hebetir. 11 -18 Low-dose radiation has also been shown in different species to both enhance and reduce growth. 1,19
Radiation impacts across generations has become an area of focus in radiobiology due to expanding nuclear power generation and the Chernobyl (1986) and Fukushima (2011) disasters. 20 Studies in Chernobyl and Fukushima have shed light on generational impacts of species inhabiting these radiation zones. Studies have indicated cellular impacts, including an increase in chromosomal aberration frequency, mutation rate, and morphological abnormalities. 21,22 As well, physiological, developmental, morphological, and behavioral impacts are evident. 23 Ecological impacts include a decrease in general species abundance and diversity. 23
In regard to generational impacts specific to insects much less is known when compared to vertebrates, especially regarding paternal contributions to subsequent generations, possibly because they generally lack male contributions to juvenile care. 24 -26 Most generational research in insects has mainly focused on the SIT or maternal effects. The SIT refers specifically to radiation exposure that can sterilize males without inflicting mortality. The release of large numbers of these sterilized males reduces the fertility of females and thus reduces the target population. 27 Research surrounding the SIT has indicated the impacts of paternal radiation on F1 offspring to include decreased fecundity, fertility, increased mortality, and increased development time. 27 Less is known about radiation impacts on other non-pest species, life-history features or the causal mechanism(s). 1
Although not analyzed in this experiment there are diverse modes of transmission of the paternal environment that have been identified in the literature. Utilized by the SIT, the induction of mutations can impact individuals and their offspring causing sterility. 27 Other mechanisms of paternal transmission likely include factors in male ejaculate. In crickets, ejaculate quantity and quality may influence both offspring size and embryo viability. 28,29 Other mechanisms include epigenetics (e.g., DNA methylation, histone modifications, non-coding RNA’s). 26
Here, we examine the impacts associated with ionizing radiation exposure spanning doses across 2 Gy to 12 Gy on F0 fathers as well as subsequent the F1 offspring of these irradiated males with normal females. We assessed life history traits (survivorship, longevity, and growth rate) of both F0 males and F1 offspring to detect potential paternal trans-generational impacts. As radiation impacts are highly species specific, this research aims to enhance our knowledge of paternally inherited life-history impacts in a relatively large-bodied insect, the House Cricket.
Methods
Animal Husbandry—Acheta domesticus were generated in a large breeding colony housed in an acrylic terrarium (93 x 64.2 x 46.6 cm), insulated with 1.5 cm thick Durofoam insulation. Fans provided air circulation. The colony was maintained at 29 °C ± 2 °C on a 12 h day-12 h night photoperiod. Food consisted of ad libitum 17% protein MultiFowl Grower chick feed (Quick Feeds Feed Mill, Copetown, Canada) which was replenished daily. Distilled water was maintained ad libitum via soaking cellulose sponges (replaced daily). Crickets were provided with egg-carton shelters, and paper towels sprayed daily with distilled water and replaced weekly. The colony was provided with oviposition medium, Organic Garden Soil (Swiss Farms Products Inc., Marysville, USA) in small plastic containers (7 x 7 x 7 cm). These were collected daily and incubated until hatching providing cohorts of nymphs of known age.
Experimental Groups—All experimental animals were generated from a single breeding colony oviposition container that was collected after a 24 h period. Individuals once hatched (∼ 14 d) were removed from the soil after 24 hours, to ensure the same age individuals were used. They were then randomly assigned one of 6 radiation groups between 0–12 Gy. The maximum radiation dose of 12 Gy was chosen due to previous data from our lab indicating that higher doses cause the inability for individuals to mature and successfully reproduce. Other doses were staggered until reaching 0 Gy as to better understand the dose response relationship. Experimental groups were housed in the same conditions as the breeding colony.
Irradiation—Specific radiation exposures for the 6 groups were achieved using a Cs-137 source at a dose rate of 0.25Gy/min at the Taylor Radiobiology Source at McMaster University. Crickets were 14 d of age (4th instar). Dose was applied at the dose rate of 0.25Gy/min until total dose was achieved (0, 2, 4, 7, 10, 12 Gy). Acheta domesticus typically complete 7-9 instars. The source is calibrated by Health Physics and Facility Management. Due to the source being well known the calibrations are not done very often, with the last one taking place in January 2008. Make and model are not provided due to security reasons related to the use of such a large source. All control and irradiated groups were then immediately brought to McMaster’s Life Sciences Building (LSB) where they were maintained for life. At approximately 30-40 d of age (when sex is indicated by ovipositor development), females were removed from experimental groups.
Dosimetry—The radiation fields of the Taylor Source are accurately known. Exact doses can be obtained by exposing subjects to a specific placement for a specific period. In general, dosimetry measurements during irradiations have proven counterproductive due to their interference with correct positioning of the subjects and inaccuracy related to the difficulty of having the same geometry for the dosimetry as for the subjects (with reference to the source fields). Here, position and orientations of the subjects was facilitated by placing specimens in a tube apparatus contained 7 individual tubes tied into a circle (with the center tube remaining empty). These were placed equidistant from the circular opening beneath the source (16.4 cm). Crickets were confined in each tube, allowing some freedom of movement to reduce stress while ensuring sufficient restriction to calculate the received dose with confidence.
Life-History Traits—Experimental groups were checked daily for maturation indicated by their adult molt (expression of wings and sexual maturity). Mass was measured at maturation with an Accuris analytical balance with a readability of 0.001 g ± 0.002 g. Maturation mass (g) and development time (days to mature) were employed to calculate growth rate. Once mature, males were removed into adult containers (one for each dose) to ensure males were not recorded twice. Juvenile containers were monitored several times a day to detect mature males. Sample sizes for F0 growth rate were: 0 (n = 52), 2 Gy (n = 45), 4 Gy (n = 55), 7 Gy (n = 62), 10 Gy (n = 63), 12 Gy (n = 29) and for F1 offspring: 0 Gy (n = 211), 4 Gy (n = 113), 7 Gy (n = 368), 10 Gy (n = 36). Survivorship was recorded daily until all individuals died. Longevity was calculated as the median lifespan of all individuals within groups. Sample sizes for survivorship and longevity measures were F0: 0 Gy (n = 119), 2 Gy (n = 117), 4 Gy (n = 128), 7 Gy (n = 106), 10 Gy (n = 127), 12 Gy (n = 120)) and for F1: 0 Gy (n = 501), 4 Gy (n = 500), 7 Gy (n = 500), 10 Gy (n = 57).
Mating—14-16 days post maturation, males from each group were paired with 20-30 non-irradiated virgin females of the same age that were previously separated from the same oviposition container as control F0 males. 14-16 days was chosen to allow time for males to recover from the mature molt and for females to produce mature eggs for oviposition. Females were isolated prior to maturation, thus ensuring virginity. Females were only used once. Groups were provided with oviposition medium for 24 h. Females in all groups laid eggs in this 24 h period. Following the 24 h mating period females were removed, and experimental males were placed in new containers to continue life-history monitoring. Oviposition containers were removed and monitored for hatchlings. All groups producing offspring had similar hatching times (∼14 days). Once F1 crickets for various treatment groups hatched, soil was removed after 24 h they were maintained to obtain F1 life history features; growth, longevity, and survivorship as described above. Data for both male and female life-history traits in offspring were recorded. Due to large sample sizes in the F1 generation, only 4 doses were examined for the F1 generation. Oviposition containers and virgin females were provided to all groups, but no F0 12 Gy males produced viable offspring.
Statistics—Growth rates are presented as mean maturation mass (mg)/maturation time (d) ± standard error for each experimental group. F0 and F1 growth rate data were analyzed with one-way ANOVA followed by a Dunnett’s multiple comparison test to detect differences among irradiated males compared to controls. For survivorship curves, a Gehan-Breslow-Wilcoxon survival analysis was applied to detect differences between irradiated F0 groups and F1 offspring compared to controls. Differences in median longevity of F0 and F1 groups were analyzed with one-way ANOVA followed by a Dunnett’s multiple comparison test to detect differences among irradiated males compared to controls. To determine the effect of sex on F1 results, a 2-way ANOVA followed by a Tukey’s Multiple Comparisons Test was applied between dose and sex for both longevity and growth rate. To determine differences between males and females for F1 survivorship a Gehan-Breslow-Wilcoxon test was applied between the sex’s in each dose. All statistical analyses were carried out with Prism Graph Pad 8.
Results
F0 Irradiated Males—Growth rates were collected for all irradiated and non-irradiated F0 males and are reported as mean growth rate of each group ± SEM (Figure 1A). A one-way ANOVA indicated significant differences among groups F(5, 300) = 86.53, p < 0.0001. A Dunnett’s multiple comparison test detected significant (p < 0.0001) differences between 4 Gy, 7 Gy, 10 Gy, and 12 Gy groups compared to non-irradiated males. The 2 Gy sub-group showed no significant reductions. The Gehan-Breslow-Wilcoxon test showed significant differences in survivorship for 7 Gy (p < 0.0001) and 10 Gy (p = 0.0055) groups compared to controls (Figure 2A). Longevity was reported as medium longevity with 95% confidence intervals. A one-way ANOVA indicated significant differences among groups F(5,711) = 6.242, p < 0.0001. A Dunnett’s multiple comparison test indicated significant increases in the 7 Gy (p = 0.0002) and 10 Gy (p = 0.0016) groups compared to controls (Figure 3A).
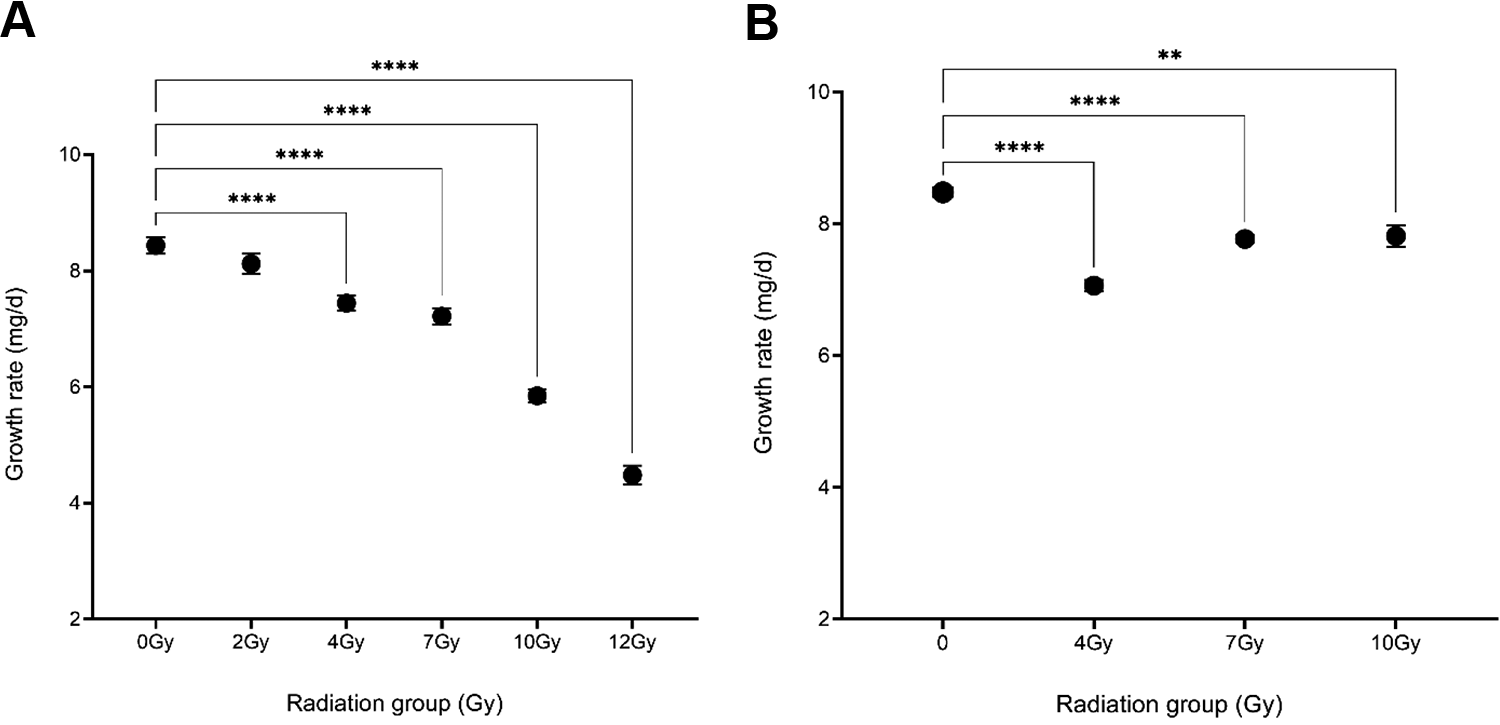
Dose-response effects of early juvenile radiation on juvenile growth rates of F0 male Acheta domesticus. Values represent the mean growth rate of each group ± SEM. Growth rates were calculated by dividing the mass at maturation (mg) by the time taken to reach maturation (days) for each individual male. A one-way ANOVA indicated significant differences between groups F(5, 300) = 86.53, p < 0.0001, with a Dunnett’s multiple comparison test indicating significant reductions in the 4 Gy, 7 Gy, 10 Gy, and 12 Gy groups (p < 0.0001) compared to non-irradiated males. (A) Dose-response effects of paternal early juvenile radiation on growth rates of F1 Acheta domesticus. A one-way ANOVA indicated significant differences between groups F(3,724) = 45.99, p < 0.0001. A Dunnett’s multiple comparison test indicated significant reductions in the 4 Gy (p < 0.0001), 7 Gy (p < 0.0001), and 10 Gy (p < 0.0017) groups compared to F1 controls (B).

Kaplan-Meier survival curves for F0 populations of male Acheta domesticus. A Gehan-Breslow-Wilcoxon test showed significant differences in survivorship in both 7 Gy (p < 0.0001) and 10 Gy (p = 0.0055) compared to the 0 Gy group (A). Kaplan-Meier survival curves for F1 offspring of F0 irradiated males and non-irradiated females. A Gehan-Breslow-Wilcoxon test, indicating the variation in survivorship curves showed significant differences in 7 Gy (p < 0.0001) and 10 Gy (p < 0.0001) groups compared to the F1 control group (B).

Dose-response effects of early juvenile radiation on longevity of F0 male Acheta domesticus. All values are the median lifespan of each group with 95% CI. A one-way ANOVA indicated significant differences between groups F(5, 711) = 6.242, p < 0.0001, with a Dunnett’s multiple comparison test indicating significant increases for the 7 Gy (p = 0.0002) and 10 Gy (p = 0.0016) compared to controls (A). Dose-response effects of paternal early juvenile radiation on longevity of F1 offspring. All individuals were offspring of corresponding F0 irradiated males mated with virgin females. A one-way ANOVA indicated significant differences between groups F(3, 1554) = 103.4, p < 0.0001, with a Dunnett’s multiple comparison test indicating significant increases in median longevity for 7 Gy (41.3%) and 10 Gy (110.9%) groups (p < 0.0001) relative to controls (B).
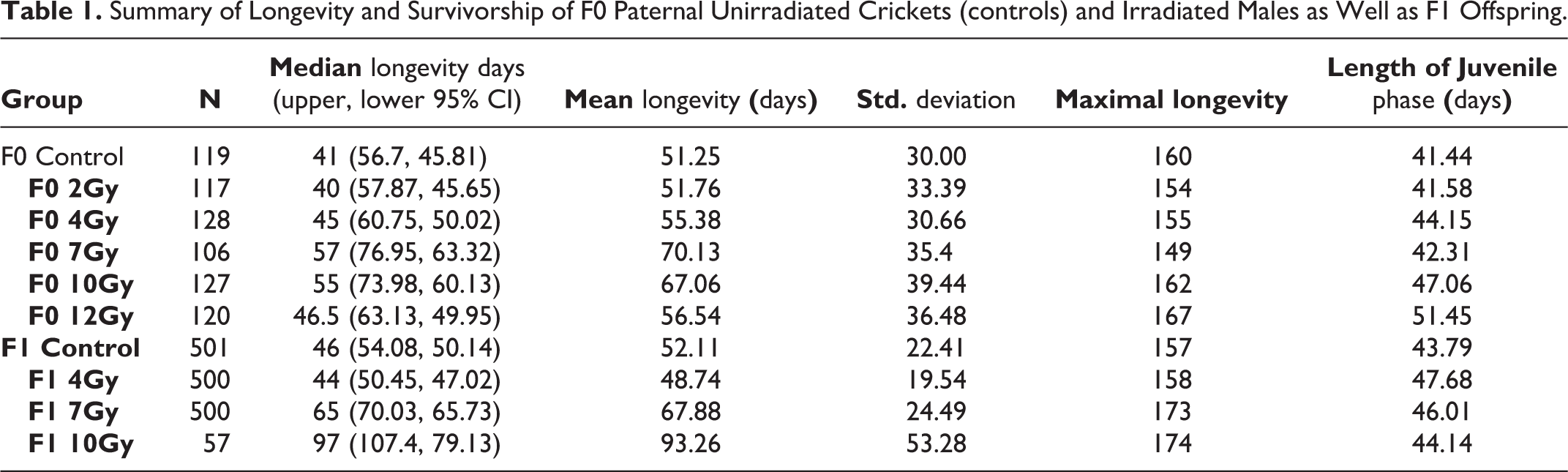
F1 Offspring—Juvenile growth rates for F1 groups (both females and males) are reported as mean growth rate of each group ± SEM Figure 1B. A one-way ANOVA detected significant differences among groups F(3,724) = 45.99, p < 0.0001 and a Dunnett’s multiple comparison test found significant differences for 4 Gy (p < 0.0001), 7 Gy (p < 0.0001) and 10 Gy (p = 0.017) groups compared to offspring from non-irradiated fathers. For survivorship the Gehan-Breslow-Wilcoxon test indicated significant differences in F1 survivorship for 7 Gy and 10 Gy groups (p < 0.0001) compared to controls (Figure 2B). Longevity was reported as medium longevity with 95% confidence intervals. A one-way ANOVA for median longevity indicated significant differences between groups F(3,1554) = 103.4, p < 0.0001. A Dunnett’s multiple comparison test indicated significant increases for 7 Gy and 10 Gy groups compared to offspring of non-irradiated males (p < 0.0001) (Figure 3B). A summary of both F0 and F1 life-history data are presented in Table 1 and Table 2.
Summary of Longevity and Survivorship of F0 Paternal Unirradiated Crickets (controls) and Irradiated Males as Well as F1 Offspring.
Summary of Growth Rates of F0 Paternal Unirradiated Crickets (controls) and Irradiated Males as Well as F1 Offspring.

Sex: To determine the effect of sex on impact’s to F1 offspring, a 2-way ANOVA was conducted on F1 growth rate and longevity between sex, dose, and the interaction between the two. For growth rate, significant sources of variation for both dose F(3, 720) = 42.22, p < 0.0001 and sex F(1, 720) = 29.41, p < 0.0001 were identified. This contributed to 14.52% (dose) and 3.015% (sex) of total variance. No significant effects were indicated for dose-sex interaction. A Tukey’s multiple comparisons test determined significant differences in growth rate between sex’s in the Control p = 0.0001, and 7 Gy groups p < 0.0001. Results of the growth rate 2-way ANOVA are summarized in Table 3. The effect of sex on F1 longevity were analyzed using a 2-way ANOVA. Results indicated significant sources of variation for dose F(3, 1285) = 143.9, p < 0.0001, sex F(1, 1285) = 3.849, p = 0.05, and interaction between these variables F (3, 1285) = 5.010, p = 0.0019. This accounted for 25.09% (dose), 0.2237% (sex), and 0.8736% (interaction) of total variance. A Tukey’s multiple comparison test detected significant differences in longevity between sex’s in the 10 Gy group (p = 0.0164). Results of the 2-way ANOVA on longevity are summarized in Table 4. Sex differences between male/female survival curves of each F1 group were analyzed using a Gehan-Breslow-Wilcoxon test. Results indicated significant differences in survival curves between male and females in the 4 Gy group (p = 0.0188). Although not insignificant, sex represented no more then 3% of total variance in growth rate and longevity within groups and are described in Table 3 and Table 4. All F1 graphs are reported as male/female combined results.
F1 Offspring 2-Way ANOVA Results for Interaction Between Sex and Dose for Growth Rate.

F1 Offspring 2-Way ANOVA Results for Interaction Between Sex and Dose for Longevity.

Discussion
Insects vary widely in responses to ionizing radiation. Here we characterized responses of Acheta domesticus associated with ionizing radiation for both males exposed as early juveniles and their offspring. As well, impacts on male and female offspring were assessed to determine potential sex differences. We found that ionizing radiation had an impact on both exposed males as well as their offspring for several life-history traits. We found a reduction in growth rate for F0 males as well as F1 offspring. In both generations, reductions were significant in doses higher than 4 Gy (Figure 1). Longevity and survivorship indicated hormetic responses. For survivorship, both F0 and F1 7 Gy and 10 Gy groups showed increased survivorship compared to control groups (Figure 2). This is similar to the results evident for longevity; both F0 and F1 7 Gy and 10 Gy groups had significantly longer median lifespans (Figure 3). Notably, F1 7 Gy and F1 10 Gy groups had median life spans (41.3% and 110.9% respectively), longer then F1 controls. Doses < 7 Gy had no significant change in survivorship or longevity compared to controls in either F0 or F1 generations.
Interestingly, the overall impacts on F1 offspring at hormetic doses (7 Gy and 10 Gy) were generally beneficial. Offspring, despite having significantly reduced growth rates, largely avoided the large growth rate declines evident in irradiated parents. F0 males had growth rate declines of 14.4% (7 Gy) and 30.7% (10 Gy), in relation to their offspring’s 8.3% (7 Gy) and 7.82% (10 Gy) reduction, when compared to non-irradiated controls.
The reduction in growth rate for both F0 males and F1 offspring seemed to be associated with both extended development time and mass at maturation (Table 2). 7 Gy and 10 Gy offspring also maintained increased longevity and survivorship seen in their fathers (Table 1). However, a significant decline in growth rate in F1 offspring suggests a negative cost associated with the increased survivorship and longevity evident in some groups. It is also important that these benefits clearly diminished above 10 Gy as F0 12 Gy males produced no viable offspring.
Similar to previous studies on irradiated insects, life-history features such as longevity and survivorship showed low dose hormesis. 5,11 - 18 As well, our work is consistent with the persistence of life-history impacts on subsequent generations from early life parental exposure. 1 However, it is clear from the negative impacts on growth rate that these beneficial doses in some F0 and F1 traits are not exclusively beneficial. Differential impacts to life-history traits has been shown in D. melanogaster in which persistent decreases in body weight in F0 and F1 flies were observed but increases in other features such as resistance to future stresses, and metabolic rate were also present. 2 Examining impacts beyond the F1 generation, as well as a more diverse array of traits would be of interest for future research. Looking past the F1 generation could also highlight potential reproductive impacts as inherited sterility has also been detected in several species. 27,30 A notable generational impact that was not analyzed in this study was the reproductive output of the F1 and future generations. As mentioned, this is an important area for future research as results would highlight the potential for the hormetic responses seen in 7 Gy and 10 Gy offspring to be offset by potential reproductive loss. Studies such as the one conducted on irradiated male Plodia interpunctella mated with normal females showed a reduction in the number of progeny produced as dose increased in F1, F2, and F3 offspring. 31 In this study male moths were irradiated at doses below what would cause sterility (sub-sterilizing doses). Other studies surrounding the use of the SIT in pest control have indicated similar impacts in irradiated males exposed to sub-sterilization doses. This is a topic of economical interest as using a lower dose reduces the amount of time and money needed to achieve the goal of reducing pest populations. In several species of lepidopterans (which as the most abundant of the “pest” species), sub sterilization doses used on males showed inherited impacts to reproduction in future generations. 30 This indicates the potential for reproductive impacts in sub-sterile doses. This is relevant as here, offspring of 7 Gy and 10 Gy males showed some beneficial responses to sub-sterilization doses, however, this may be diminished if the cost of these benefits is reduced reproductive output, especially if it persists through future generations as shown by Brower (1979). Since most work of inherited sterility pertains to species within Lepidoptera it is important to continue to gather data in other species using a variety of endpoints.
Although this study describes evidence for paternal effects in F1 offspring in Acheta domesticus these results are not generalizable to all species. The literature notes that responses to radiation are species specific, and often differ considerably between even closely related species. 32,33 In wasps, only 5 Gy is required to impact oocytes in females and decrease offspring number, but in codling moths it takes 300-400 Gy to completely sterilize a female and reduce male fertility. 34 For other life-history factors such as mortality, Lumbricus terrestris has a LD50/30 value of 680 Gy with a closely related species of Eisenai foetida having a LD50/30 value of 650, a 30 Gy difference. 34 Some invertebrate species have also been shown to be extremely resistant to radiation, while others such as Daphnia Magna have shown severe negative impacts at doses as low as 100 and 1000 mGy. 8,9 Generally, it has been noted that adult LD50 values for insect can range from 20-3000 Gy with subadult stages showing impacts at doses as low as 1-2 Gy for certain species. 35,36 The literature therefore indicates the diversity in reproductive and life history responses as it pertains to different invertebrate species and that its likely that one model or dose-response relationship will not coincide to all species.
The 2-way ANOVA’s for sex and dose in F1 life history traits indicated a significant contribution of sex in overall variation in growth rate (3%) and longevity (0.87%) Table 3/4. However, most variation can be attributed to dose for growth rate (14.5%) and longevity (25.09%). A Gehan-Breslow-Wilcoxon test applied to sex in survival curves only found significant differences between males and females in the 4 Gy group (p = 0.0188). For growth rate, sex differences emerged only in controls (p = 0.0001), and 7 Gy group (p < 0.0001) and for longevity only the 10 Gy group (p = 0.0164). Although an overall trend is not evident, results are consistent with literature suggesting that sex indeed plays a role in radiation susceptibility in insects. Several studies identified differential susceptibility of males and females to radiation in terms of reproduction and lifespan, with females generally being more sensitive then males. 18,27,37 Less is known, however, about impacts on male and female offspring from single-parent irradiation. Here we see that differential responses to radiation based on sex are relevant and likely varies with the life-history trait being assessed.
Although we did not address mechanisms, our results are consistent with trans-generational impacts of radiation stress. 38 This work aimed to further knowledge of paternal effects in a large-bodied insect, Acheta domesticus, as it pertains to ionizing radiation stress on life-history traits and trans-generational impacts. Further work to assess further generational impacts (past F1) as well as exploring possible mechanism(s), regulatory alterations mediating these impacts is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mitacs Elevate Grant [IT05851], and the NSERC Discovery Grant [RGPIN-05693-2015].
