Abstract
Elevated inflammatory cytokines and high mobility group box 1 (HMGB1) production are associated with chronic periodontitis (CP). Glycyrrhizin is the major constituent of Glycyrrhiza glabra. L. (Fabaceae) root with anti-inflammation activities. This study evaluated the effects of glycyrrhizin on CP. TNF-α-treated human periodontal ligament stem cell (hPDLSC) model was established, and was administrated with 1, 2 or 5 mM glycyrrhizin for 24 h. After treatment, the expression of HMGB1and inflammatory cytokines was monitored. Significantly increased HMGB1 (median: 5646.4, range: 1918.2-8233.7 vs median: 204.5, range: 98.7-283.6, pg/mL), TNF-α (median: 345.5, range: 161.0-567.9 vs median: 93.5, range: 58.1-159.3, pg/mL), IL-1β (median: 2014.6, range: 209.5-4308.1 vs median: 224.5, range: 48.8-335.8, pg/mL) and IL-6 (median: 1223.6, range: 398.2-2183.8 vs median: 240.4, range: 105.2-400.5, pg/mL) were detected in gingival crevicular fluid from CP patients. Glycyrrhizin significantly prevented TNF-α-induced expression of HMGB1 (691.5 ± 136.4 vs 142.8 ± 57.3 pg/mL), IL-6 (388.1 ± 85.2 vs 189.4 ± 61.2 pg/mL) and IL-1β (176.3 ± 47.2 vs 53.9 ± 25.7 pg/mL) in hPDLSC. In CP rats, glycyrrhizin significantly decreased HMGB1 (5795.6 ± 1121.5 vs 586.4 ± 436.8 pg/mL), TNF-α (421.8 ± 93.7 vs 87.9 ± 21.6 pg/mL), IL-6 (1423.8 ± 235.2 vs 622.6 ± 176.1 pg/mL) and IL-1β (1562.8 ± 334.3 vs 733.5 ± 265.1 pg/mL) in gingival crevicular fluid. Glycyrrhizin suppresses inflammatory activities in CP rats and represents a promising molecule for controlling CP.
Introduction
Chronic periodontitis (CP) is an infectious disease which causes inflammation in gingiva and other periodontal tissues. 1 CP is the major oral disease and affects more than 60% of the population over the world. 2
Multiple factors have been implicated in CP, including bacterial infection, environmental and host factors. Inflammation is the major characteristic of CP, which is usually initiated by Gram-negative bacteria Porphyromonas gingivalis. L. (Porphyromonadaceae). 3 The cell wall components lipopolysaccharide (LPS) could directly destruct the periodontal tissues and induced inflammatory cytokines production, which are critical in developing periodontitis. 4 TNF-α is a pro-inflammatory cytokine which is detected with increased level in CP patients’ saliva and crevicular fluid. It is well described that TNF-α contributed to inflammatory response, connective tissue detachments, and bone resorption. 5 IL-6 is induced by LPS and TNF-α stimulation and exerts significant effect in CP pathogenesis including inducing angiogenesis, activating immunocyte and inducing osteoblast differentiation. The altered production of IL-6 is associated with CP. 6 Increased IL-1β secretion is detected in periodontal tissue of patients with CP. IL-1β stimulates the release of multiple factors which contribute to extracellular matrix degradation, bone resorption. 7 Therefore, besides antibiotics treatment, the anti-inflammation approaches have been utilized for CP treatment. 8
High-mobility group box-1 (HMGB1) is a nuclear protein with DNA binding ability. HMGB1 could function to regulate transcription, DNA bending and DNA repair when it is in the nucleus. In addition, HMGB1 could be secreted out of the cells like a pro-inflammatory cytokine after stimulation by lipopolysaccharides (LPS), TNF-α or other stimuli. 9 Extracellular HMGB1 could induce inflammation and plays important roles in many inflammatory diseases. 10,11 The association of HMGB1 with CP has been described and neutralization of HMGB1 by antibodies has been shown to attenuate the CP, suggesting HMGB1 could be another potential target for CP treatment. 12
Glycyrrhizin is the major component of licorice root with multiple biological activities including antioxidative, antiinflammation, anticancer and antiviral. 13 -17 Glycyrrhizin has also described to target HMGB1 and block its functions. 18,19 These findings suggest potential effects of glycyrrhizin on inflammation in CP. In present study, the potential roles of glycyrrhizin in inflammation of CP were evaluated.
Materials and Methods
Patient Recruitment
Patients were recruited from April 2018 to August 2018 in Cangzhou Central Hospital. CP patients were selected strictly following the criteria described previously by Li et al. 20 All the patients were age ≥ 21 years and the presence of at least 20 teeth. CP patients should have multiple sites with a probing depth (PD) of ≥ 5 mm, attachment loss (AL) of ≥ 2 mm and bone loss confirmed by radiography. The individuals with the following conditions were excluded from the study: (1) current smokers or who quit smoking less than 6 months before recruitment; (2) individuals who used antibiotics, aspirin or anticoagulants in the preceding 3 months; (3) pregnant women; and (4) individuals with systemic conditions including diabetes mellitus, obesity, rheumatoid arthritis, coronary heart diseases, or malignancies. All patients were healthy and had not used antibiotics during the previous 6 months. All participants were asked to give their consent in written form after the purpose and protocol of the study were instructed. The control specimens (healthy control) were obtained from impacted third molar extraction surgery. All selected patients did not use antibiotics at least 6 month and were informed the purpose and protocol of the study. The samples from third molar extraction surgery were used as control. This study was approved by Ethics Committee of Cangzhou Central Hospital.
Clinical Recordings
Before crevicular fluid collection, the supragingival plaque was scored using plaque index (PI) as described previously. 21 After crevicular fluid collection, the gingival inflammation was scored using gingival index (GI) as described previously. 22 A conventional periodontal probe was used to measure the probing depth (PD) and attachment loss (AL). AL measurements were made from the cemento-enamel junction to the bottom of the sulcus.
Sample Collection
To collect gingival crevicular fluid, the supragingival plaque was removed. Then, a pre-weighed paper strip was inserted into the selected site. When there was resistance, the strip was kept for 30 s. If bleeding was associated with sampling, the samples were excluded for study. After GCF sampling, the strips were weighed to calculate GCF amount and then immediately stored at -80°C.
Human Periodontal Ligament Stem Cells (hPDLSCs) Isolation, Culture and Treatment
To isolate the PDLSCs, disease-free premolars were extracted and then kept in DMEM (Thermo Fisher, Waltham, MA, USA). After washing with phosphate-buffered saline (PBS) for 5 times, the periodontal ligament was scraped from the root surface and put in DMEM containing 4 mg/ml collagenase (Sigma, St. Louis, MO, USA) and 3 mg/mL dispase (Sigma, USA) for digestion. After incubation at 37°C for 1 h, cell suspension went through 70 µm cell strainer (Thermo Fisher, USA). Cells were pelleted by centrifuge and finally cultured in complete culture medium (DMEM plus 10% heat-inactivated fetal bovine serum), with 1% penicillin/streptomycin (Thermo Fisher, USA). The PDLSCs were defined as STRO-1+, CD146+, CD90+, CD31-, CD45- and CD14-, which was described previously. 23 PDLSCs were seeded in 96 or 24-well plates. Next day PDLSCs were treated with either TNF-α or glycyrrhizin, or TNF-α/glycyrrhizin together as indicated.
Cell Viability Assay
Cell counting kit-8 (Dojindo China Co. Ltd, Shanghai, China), was used to monitor the cell viability. PDLSCs were seeded in 96-well plate and treated with various concentration of glycyrrhizin (0.5, 1, 2, 5 and 10 mM) for 24 or 48 h, or treated with different concentration of TNF-α (2.5, 5, 7.5, 10, 15 and 20 ng/mL) for 24 h. After treatment, 10 µL CCK-8 solution was added to each well. After 2 h incubation, the 450 nm absorbance was measured.
Rat Model of Chronic Periodontitis
Female Sprague-Dawley rats with 10 weeks age were obtained from Charles River (Shanghai, China) and kept in environment with controlled temperature (23 ± 2°C), humidity (55 ± 5%) and 12 hlight/dark cycle. Rat had free access to food and water.
To establish the chronic periodontitis, rats were anesthetized by intraperitoneally injected with 4 mL/kg 10% chloralhydrate. Then the subgingival of the first and second molars were ligated with (3/0) silk sutures (Thermo Fisher, USA), which was pre-soaked in Porphyromonas gingivalis (Pg) solution, with a continuous “∞” approach on either side of the maxilla bone. The ligatures were kept for 4 weeks and were checked twice per week. If the ligatures were displaced or loosened, they were replaced. After 4 weeks, the ligatures were removed and rats were intravenously injected 0.5 mL glycyrrhizin with concentrations of 2 mg/kg, 4 mg/kg and 10 mg/kg in tail vein with 5 times per week for 4 weeks at indicated dosage. Animal studies were reviewed and approved by Cangzhou Central Hospital (#DWSYLL6712).
ELISA
PDLSCs were seeded in 6-well plate and treated with glycyrrhizin or TNF-α as indicated. After treatment, the cell supernatant was collected. HMGB1 level was measured using HMGB1 ELISA Kit (LSBio, Seattle, WA, USA). IL-6 and IL-1β were measured using commercial ELISA kit from R&D systems (Shanghai, China). The HMGB1, IL-6, TNF-α and IL-1β level in gingival crevicular fluid (GCF) of rats were measured by commercial ELISA kit following manufacturer’s protocols.
RT-PCR
The total RNA from periodontal tissues or PDLSCs was extracted using NucleoSpin® RNA Plus kit (Takara, Beijing, China). PrimeScript™ II 1st Strand cDNA Synthesis Kit was used for cDNA synthesis (Takara, China). The real time PCR was set up by using SYBR® Green Master Mix (Qiagen, Germantown, MD, USA). The primers used in present study were: Human HMGB1 Forward: 5’-CGCCATGAGAACTTCCTACC-3’, Reverse: 5’-CACTTGGCCTT CCCTCTGTA-3’. Human TNF-α Forward: 5’-CCCATGTTGTAGCAAACCCTC-3’, Reverse: 5’- TATCTCTCAGCTCCACGCCA-3’. Human IL-1β Forward: 5’-CCACCTCCAGGGACAG GTAT-3’, Reverse: 5’-TGGGATCTACACTCTCCAGC-3’. Human IL-6 Forward: 5’-CAATGAGGAGACTTGCCCTGG-3’, Reverse: 5’-TGGGTCAGGGGTGGTTATTG-3’. Human β-actin Forward: 5’-TGGCACCCAGCACAATGAA-3’, Reverse: 5’-CTAAGTCA TAGTCCGCCTAGAAGCA-3’. Rat HMGB1 Forward: 5’-CTGATGCAGCTT ATACGAAG -3’, Reverse: 5’-TCAGGTAAGGAGCAGAACAT-3’. Rat TNF-α Forward: 5’-TCTTCTCAT TCCTGCTCGTG-3’, Reverse: 5’-GAGGCTGACTTTCTCCTGGT. Rat IL-1β Forward: 5’-CTATGTCTTGCCCGTGGAG-3’, Reverse: 5’-CTGCTTGAGAGGTGCTGATG-3’. Rat IL-6 Forward: 5’-CCACTGCCTTCCCTACTTCA-3’, Reverse: 5’-TCTTGGTCCTTAGCCACTCC-3’. Rat β-actin Forward: 5’-CCCATCTATGAGGGTTACGC-3’, Reverse: 5’-TTTAATGTCAC GCACGATTTC-3’.
Western Blot
Total proteins from gingival tissues were extracted and 25 µg proteins were subjected to SDS-PAGE and then transferred to nitrocellulose membrane. After transfer, the membranes were blocked with 5% nonfat milk in PBST. Primary antibodies including anti-HMGB1 (R&D systems, China), anti-β actin (Sigma, USA) were used in current study. Corresponding HRP-conjugated secondary antibodies were used for detecting immune-reactive bands. ImageJ was used for quantitation analysis.
Statistical Analysis
The statistical difference was determined using Student’s t-test or one-way ANOVA followed with a Tukey’s post hoc test. When p value is less than 0.05, the difference was considered as significant.
Results
Elevated HMGB1 Expression in Gingival Tissues From Chronic Periodontitis Patients
Chronic periodontitis (CP) patients were recruited clinical characteristics were recorded (Table 1). The HMGB1 level in gingival crevicular fluid (GCF) was measured by ELISA. We detected significantly elevated HMGB1 level in GCF from CP than that from control health people (Figure 1A) (median: 5646.4, range: 1918.2 to 8233.7 vs median: 204.5, range: 98.7 to 283.6, pg/mL). Similarly, significantly increased HMGB1 protein level was detected in gingival tissues from CP when compared to the gingival tissues from control health people (Figure 1B). In addition, significantly increased level of inflammatory cytokines including TNF-α (Figure 1C) (median: 345.5, range: 161.0 to 567.9 vs median: 93.5, range: 58.1 to 159.3, pg/mL), IL-6 (Figure 1D) (median: 1223.6, range: 398.2 to 2183.8 vs median: 240.4, range: 105.2 to 400.5, pg/mL) and IL-1β (Figure 1E) (median: 2014.6, range: 209.5 to 4308.1 vs median: 224.5, range: 48.8 to 335.8, pg/mL) was detected in GCF from CP patients. Therefore, elevated HMGB1 and inflammatory cytokines expression were associated with CP.
Clinical Characteristics of Chronic Periodontitis Patients and Healthy Control Enrolled in the Trail.

Data are presented as mean ± SD or n (percentage, %).
CP: chronic periodontitis, PD: probing depth, AL: attachment loss. PI: plaque index, GI: gingival index, GCF: Gingival crevicular fluid.
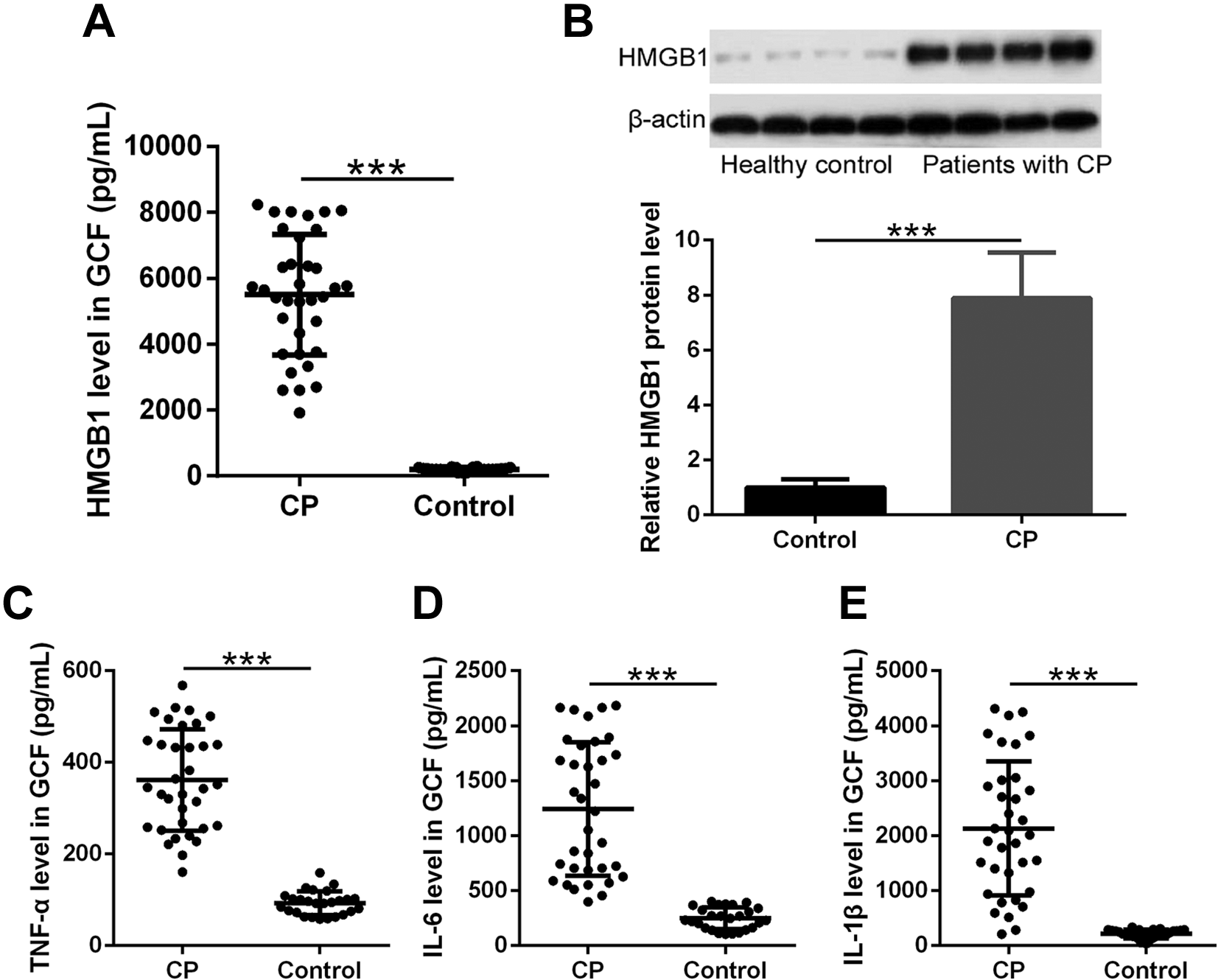
HMGB1 expressions in gingival crevicular fluid (GCF) and gingival tissues from the healthy controls and chronic periodontitis (CP) patients. A, ELISA was used to measure the HMGB1 levels in GCF (n = 35 for CP group and n = 26 for control group). B, Western blotting was used to measure the HMGB1 protein levels in gingival tissues, β-actin was used as loading control (n = 4 for each group). C-E, ELISA was used to measure the TNF-α, IL-6 and IL-1β levels in GCF (n = 35 for CP group and n = 26 for control group). Data are presented as mean ± SD with all data points presented. ***p < 0.001 between the indicated groups.
Titration of Glycyrrhizin Concentration on Cell Viability of Human Periodontal Ligament Stem Cells (PDLSCs)
To determine the best dosage of glycyrrhizin in our PDLSCs model, the PDLSCs were treated with various concentrations of glycyrrhizin for 24 or 48 h. After treatment, the cell viability was monitored. As shown in Figure 2A, 0.5, 1, 2 and 5 mM glycyrrhizin treatment for 24 h did not affect cell viability while 24 h treatment with 10 mM glycyrrhizin significantly decreased cell viability of PDLSCs. Similar results were obtained from the samples treated with glycyrrhizin for 48 h (Figure 2B). Therefore, we adopted the concentration of 1, 2 and 5 mM for our experiments. TNF-α, which was known to induce HMGB1 expression and used as the potential marker of periodontal disease, 24,25 was used as control in current study. We treated PDLSCs with different concentration of TNF-α for 24 h and found that 2.5, 5, and 7.5 ng/mL TNF-α treatment did not cause significant difference of cell viability when compared to non-treatment while 10, 15, and 20 ng/mL treatment significantly decreased cell viability (Figure 2C). We also confirmed that TNF-α treatment promoted HMGB1 expression with time increased. 6 h post TNF-α treatment (289.8 ± 63.5 vs 126.2 ± 23.1 pg/mL), significantly increased HMGB1 expression was detected (Figure 2D).

Cell viability of Human periodontal ligament stem cells (PDLSCs) after individual treatment with glycyrrhizin for 24 h (A) and 48 h (B) and different concentrations of TNF-α for 24 h (C). D. ELISA was used to measure the HMGB1 levels after PDLSCs stimulated by 10 ng/mL TNF-α for different times. n = 12 for each group. Data are presented as mean ± SD. Every experiment has been repeated for at least 3 times. **p < 0.01, ***p < 0.001 compared to control.
Glycyrrhizin Suppressed TNF-α-Induced Inflammation In Vitro
To test the effects of glycyrrhizin on TNF-α-induced production of inflammatory cytokines and HMGB1, the PDLSCs were treated with 10 ng/mL of TNF-α, together with different concentration of glycyrrhizin (0, 1, 2 and 5 mM). Protein level of secreted HMGB1, IL-6 and IL-1β in supernatant was measured by ELISA and mRNA level of HMGB1, IL-6 and IL-1β in cells was measured by RT-PCR. TNF-α treatment significantly increased both protein and mRNA level of HMGB1 (Figure 3A and B) (691.5 ± 136.4 vs 127.4 ± 29.7 pg/mL), IL-6 (Figure 3C and D) (388.1 ± 85.2 vs 39.5 ± 20.4 pg/mL) and IL-1β (Figure 3E and F) (176.3 ± 47.2 vs 24.7 ± 12.2 pg/mL). Glycyrrhizin treatment inhibited TNF-α-induced production of HMGB1, IL-6 and IL-1β. 5 mM Glycyrrhizin treatment had the best inhibitory effects as the lowest level of HMGB1, IL-6 and IL-1β were detected in these samples (HMGB1: 142.8 ± 57.3 pg/mL; IL-6: 189.4 ± 61.2 pg/mL; IL-1β: 53.9 ± 25.7 pg/mL). Taken together, our data demonstrated that glycyrrhizin suppressed TNF-α-induced production of HMGB1, IL-6 and IL-1β.

The effects of glycyrrhizin on TNF-α-induced PDLSCs. ELISA was used to measure the levels of HMGB1 (A), IL-6 (C) and IL-1β (D) after 24 h. HMGB1 (B), IL-6 (E) and IL-1β (F) mRNAs were subjected to real-time PCR analysis after 24 h. The expression levels were normalized to control groups. n = 12 for each group. Data are presented as mean ± SD. Every experiment has been repeated for at least 3 times. ***p < 0.001 compared to control. #p < 0.05, ##p < 0.01, ###p < 0.001 compared to single TNF-α stimulation.
Glycyrrhizin Decreased HMGB1, TNF-α, IL-6 and IL-1β Level in Gingival Crevicular Fluid From Rats With Chronic Periodontitis
We continued to evaluate the effects of glycyrrhizin on inflammation in rats with CP. After glycyrrhizin treatment, the protein level of HMGB1, TNF-α, IL-6 and IL-1β in gingival crevicular fluid (GCF) was measured by ELISA. We detected significantly increased HMGB1 (Figure 4A) (5795.6 ± 1121.5 vs 347.5 ± 98.4 pg/mL), TNF-α (Figure 4B) (421.8 ± 93.7 vs 87.9 ± 21.6 pg/mL), IL-6 (Figure 4C) (1423.8 ± 235.2 vs 215.6 ± 24.4 pg/mL) and IL-1β (Figure 4D) (1562.8 ± 334.3 vs 122.7 ± 42.3 pg/mL) in GCF from rats with chronic periodontitis. Glycyrrhizin treatment significantly decreased the level of HMGB1 (Figure 4A), TNF-α (Figure 4B), IL-6 (Figure 4C) and IL-1β (Figure 4D) in GCF. The lowest protein level was detected in rats treated with highest dose of glycyrrhizin (10 mg/kg) (HMGB1: 586.4 ± 436.8 pg/mL; TNF-α: 177.3 ± 67.2 pg/mL; IL-6: 622.6 ± 176.1 pg/mL; IL-1β: 733.5 ± 265.1 pg/mL). Collectively, our data demonstrated that glycyrrhizin decreased the production of inflammatory cytokines in GCF.

The effects of glycyrrhizin on inflammatory response in gingival crevicular fluid (GCF) from chronic periodontitis rats. ELISA was used to measure the levels of HMGB1 (A), TNF-α (B), IL-6 (C) and IL-1β (D) in GCF. Data are presented as mean ± SD. n = 8 for each group. ***p < 0.001 compared to control. #p < 0.05, ##p < 0.01, ###p < 0.001 compared to model group. The experiments have been repeated for 3 times to confirm the results.
Glycyrrhizin Decreased HMGB1, TNF-α, IL-6 and IL-1β Level in Gingival Tissues From Rats With Chronic Periodontitis
Finally, we monitored the expression level of HMGB1, TNF-α, IL-6 and IL-1β in gingival tissues from CP rats after glycyrrhizin treatment. We detected significantly increased mRNA level of HMGB1 in gingival tissues from CP rats. Glycyrrhizin treatment significantly decreased mRNA level in a dose-dependent manner (Figure 5A). Consistently, glycyrrhizin significantly decreased protein level of HMGB1 in gingival tissue (Figure 5B). Similarly, we detected significantly decreased mRNA level of IL-6 (Figure 5C), TNF-α (Figure 5D) and IL-1β (Figure 5E) in gingival tissue of chronic periodontitis rats treated with glycyrrhizin. Collectively, our data demonstrated that glycyrrhizin decreased HMGB1, TNF-α, IL-6 and IL-1β level in gingival tissues from CP rats.

The effects of glycyrrhizin on inflammatory response in gingival tissues from chronic periodontitis rats. HMGB1 (A), IL-6 (C), TNF-α (D) and IL-1β (E) mRNAs in gingival tissues were subjected to real-time PCR analysis. B, Western blotting was used to measure to the HMGB1 protein expressions in gingival tissues. Data are presented as mean ± SD. n = 8 for each group. **p < 0.01, ***p < 0.001 compared to control. #p < 0.05, ##p < 0.01, ###p < 0.001 compared to model group. The experiments have been repeated for 3 times to confirm the results.
Discussion
CP, which is usually caused by oral bacteria, is the major cause of tooth loss. 26 CP is an inflammatory disease in which pro-inflammatory cytokines play essential role. The released TNF-α could stimulate cell to produce downstream factors including HMGB1 for CP development. In present study, we detected significantly upregulated protein level of HMGB1, TNF-α, IL-1β and IL-6 in gingival crevicular fluid from CP patients, confirming the association of HMGB1 and inflammation with CP. Using a CP rat model, elevated protein level of HMGB1, TNF-α, IL-1β and IL-6 in gingival crevicular fluid, and elevated mRNA level of HMGB1, TNF-α, IL-1β and IL-6 in gingival tissues from CP rats. These findings demonstrated the association of HMGB1 and inflammation with CP.
TNF-α is one of the important inflammatory cytokines which contributes to CP development. The oral organisms stimulate the monocytes and macrophage for TNF-α production. It has been accepted that the released TNF-α from the polymorphnuclear neutrophils and macrophages could be used as biomarker for CP diagnosis. 5 TNF-α stimulated osteoclasts formation and then promoted the destruction of alveolar bone. 27 TNF-α also regulated matrix metalloproteinase (MMPs) expression, resulting in connective tissues degradation. Elevated TNF-α level has been associated with CP, 28 which was also confirmed in present study. In addition, using human periodontal ligament stem cells (hPDLSCs), we demonstrated that TNF-α treatment up-regulated the both mRNA and protein levels of IL-1β and IL-6, suggesting the crucial role of TNF-α in regulating inflammatory response in CP. Previous studies have demonstrated that blockage of TNF-α had a desirable effect on periodontal conditions in CP, indicating inhibiting TNF-α could be a therapeutic treatment for CP.
The association of HMGB1 and CP was described previously. Deng and colleagues reported that increased expression of HMGB1 was detected in the gingival tissue and GCF in CP and they suggested that HMGB1 could be used as a therapeutic target to treat CP. 29 Our results also confirmed these findings as we detected significantly increased mRNA and protein level of HMGB1 in GCF and periodontal tissues of patients and rats with CP, and in TNF-α-treated PDLSCs. HMGB1 is a nuclear protein but also has cytokine-like activity. HMGB1 could bind to receptor to regulate inflammatory responses. After stimulated by TNF-α and LPS, HMGB1was released and then bound to TLR-2, TLR4, TLR-9, which lead to activation of the downstream factors such as ERK, Akt, and NF-κB, and finally resulted in production of pro-inflammatory cytokine and amplification of inflammation response. 30,31 Targeting HMGB1 and suppressing its activity by neutralization antibody have been shown to ameliorate the inflammation in CP.
Glycyrrhizin is with great anti-inflammatory activities. It has been reported that glycyrrhizin blocked the CpG-induced pro-inflammatory cytokines production RAW 264.7 cells. 32 Glycyrrhizin also prevented the reactive oxygen species (ROS) production in neutrophils and following inflammatory response. 15 Glycyrrhizin could directly bind to HMGB1 through its HMG boxed, which resulted in the loss of function. 33 Gong and colleagues (2014) described that glycyrrhizin protected the ischemia-reperfusion injury by inhibiting inflammation, oxidative stress and cell apoptosis, through targeting HMGB1. In present study, we found glycyrrhizin blocked the TNF-α induced expression of HMGB1, IL-1β and IL-6. In addition, after administration of glycyrrhizin in CP rats, the level of inflammatory cytokines including TNF-α, IL-1β and IL-6 in gingival crevicular fluid and gingival tissue was significantly decreased. These findings further confirmed the anti-inflammatory activities of glycyrrhizin and suggested glycyrrhizin could be used as a potential therapeutic reagent to treat CP.
Conclusion
Our results demonstrated that glycyrrhizin suppressed the inflammatory response in chronic periodontitis, strongly suggested that HMGB1 could be used as a potential therapeutic target of CP and glycyrrhizin could be used as a therapeutic treatment for CP. However, there are still lots of work need to be done before applying glycyrrhizin to clinical trial. For example, the kinetics of glycyrrhizin needs to be characterized; the dosage with great efficacy needs to be determined.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
