Abstract
The purpose of this study is to investigate the effects of melatonin on the radiosensitivity of HeLa cells. Concentration from 10 to 1000 µM of melatonin was used on HeLa cells before X-rays irradiation (IR). The cellular inactivation effect was analyzed by clonogenic assay, and cell growth was measured by MTT assay at various concentrations. Ten micrometer melatonin promoted the cell-killing effects of IR, while 1000-µM melatonin prevented IR-induced cellular inactivation. Further analysis revealed that 1000-µM melatonin protected the cells from IR-induced reactive oxygen species damage, as the oxidative stress measured by fluorescent microscopy and fluorescence-activated cell sorting using 2,7-dichlorofluorescein diacetate staining. This is further confirmed by melatonin receptor agonist, which has no antioxidant capacity. A 10-µM melatonin, on the contrary, enhanced the cell-killing effects of IR by activating c-Jun NH2-terminal kinase (JNK) signaling. c-Jun NH2-terminal kinase signaling activation was indicated by Western blot of phosphorylated JNK. We used JNK inhibitor to further confirm the involvement of JNK signaling in the cell-killing enhancement of 10-µM melatonin administration. Our results suggest the importance of dose-dependent effects in melatonin application for radiotherapy.
Introduction
Melatonin is a natural indoleamine that regulates circadian rhythms in living organisms. Besides its role in light/darkness responses regulation, melatonin has been shown to exert antitumoral effects in many cancer cells. 1 –3 Its effect on cancer cells varies depending on its concentration. At low concentrations, melatonin inhibits cancer cell proliferation. 4,5 At high concentration, it enhances apoptosis in some certain cancer types. 6 –8
The mechanism involved in the antitumoral property of melatonin in high and low concentrations is different. Low concentration of melatonin shows oncostatic effects on tumors, which works mainly via melatonin receptors on the membrane through multiple pathways. 9 The high concentration of melatonin induces apoptosis via the early increase in reactive oxygen species (ROS), as well as a reduction in intracellular glutathione (GSH). 10 The observed redox disturbance of high concentration of melatonin on cancer cells is counterintuitive since millimolar of melatonin is effective ROS scavenger that can protect normal cells from oxidative damage.
Melatonin can either activate or inhibit signal transduction in a receptor-dependent or receptor-independent manner. It binds with high affinity to the membrane receptors in the picomolar range, in the nanomolar range it can translocate to the nuclear receptors (RZR/ROR) as well as to calmodulin. At millimolar concentrations, it can serve as an antioxidant compound. 11 Many cancer types has been shown to be susceptible to inhibition by melatonin, including, but not limited, to breast cancer, 12 ovarian cancer, 13 pancreatic cancer, 14 and hepatic cancer. 15 Considering its antitumoral potential and its protection against ROS damage for normal cells, melatonin could be a good candidate for radiotherapy to enhance antitumoral effects, as well as to limit the collateral damage of ionizing radiation (IR), if the correct dosage can be applied. The c-Jun NH2-terminal kinase (JNK) pathway is one of the major signaling cassettes of the mitogen-activated protein kinase signaling pathway. It regulates several cellular processes, including proliferation and apoptosis. Irradiation can stimulate JNK cascade that leads to apoptosis. 16,17 Similarly, melatonin can also induce cell apoptosis through activation of JNK. 18 Coadministration of melatonin has been shown to improve the sensitivity of cancers to inhibition by conventional drugs. It is, therefore, interesting to investigate whether JNK signaling participates in the effect of cell death by the combination of melatonin and IR.
Materials and Methods
Cell Culture and IR
The human cervical cancer cell line HeLa was purchased from the American Type Culture Collection and maintained in Dulbecco’s modified Eagle’s medium (DMEM; Gibco) with 10% fetal bovine serum (FBS). Cells were maintained at 37°C using humidified air supplemented with 5% CO2. Cells in exponential growth were irradiated with 250 kVp X-rays with a dose rate of 1 Gy/min. Different concentrations of melatonin were added to the medium 2 hours before IR.
Survival Assay
The sensitivity of cells to X-rays radiation was measured using a colony-formation assay. Confluent cultures were replated within 1 hour post-IR into 100-mm dishes in quadruplicate at numbers estimated to yield 50 to 100 clonogenic colonies per dish. After incubation for 2 weeks, cultures were fixed in methanol and stained with crystal violet. These stained colonies were observed with an Olympus light microscope. Colonies with 50 or more cells were scored as clonogenic colonies.
MTT Assay
HeLa cells were grown in 96-well culture plates and incubated for 1 to 3 days with DMEM/serum containing various concentrations of melatonin. MTT (5 mg/mL in water, 10 μL/well) was then added into each well. After incubation for 4 hours at 37°C, the supernatant was abandoned and 100 μL of dimethyl sulfoxide was added into each well to dissolve the produced formazan. After shaking the plates for 5 minutes, absorbance values of the produced purple formazan were recorded with a microplate reader at 490 nm.
Western Blot Assay
Cells were lysed in RIPA buffer (Beyotime, Nanjing, China). Proteins were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to a methanol-activated polyvinylidene difluoride membrane. The membrane was blocked for 1 hour in Tris-buffered saline (TBST) containing 0.5% FBS and subsequently probed with Phospho-SAPK/JNK (Thr183/Tyr185) antibody (Cell Signaling, California) at 4°C overnight with shaking. After 1-hour incubation with HRP-conjugated secondary antibody (Santa Cruz), membranes were washed 3 times for 15 minutes each with TBST. Reactive proteins were visualized using a chemiluminescence kit (Santa Cruz) according to the manufacturer’s instructions.
Reactive Oxygen Species Measurement
The fluorescent probe, 2,7-dichlorofluorescein diacetate (DCFH-DA, Sigma), was employed to quantify the level of ROS. Confluent cultures were washed with D-Hank buffer solution complemented with 1% FBS at 37°C for 2 minutes, stained with 2 M DCFH-DA for 40 minutes in the incubator and then washed with cold D-Hank twice. The staining was captured under microscopy.
For fluorescence-activated cell sorting (FACS) assay, cells were harvested by trypsin–EDTA solution to produce a single-cell suspension. The cells were pelleted by centrifugation and washed twice with phosphate-buffered serum to remove the background fluorescence. Stained cells were analyzed using a flow cytometer (Becton Dickinson, Heidelberg, Germany) in combination with Flowing (http://flowingsoftware.btk.fi/).
Statistical Analysis
Data represent mean (standard deviation) of 3 replicates. Independent samples
Results
Different Effects of Melatonin on Cell Survival After IR in a Dose-Dependent Manner
Melatonin showed no significant inhibitory effect on cell growth, except a temporal growth inhibition at 24 hours at a concentration of 1 mM (Figure 1A). Dose-dependent cytotoxicity on clonal formation was observed with IR alone (Figure 1B). Melatonin has antitumoral effects by inducing apoptosis at a millimolar concentration. 10 To test whether melatonin can enhance the cell-killing effect of IR, various concentrations of melatonin were used. To our surprise, 10-µM melatonin administration markedly reduced the clonal formation after 4 Gy IR, and 1-mM melatonin protected HeLa cells from radiation-induced clonal formation inhibition (Figure 1C).

The dosage effects of melatonin on IR-induced cellular inactivation of HeLa cells. A, Cell growth measured by MTT from 1 to 3 days with a presence of melatonin from 10 to 1000 µM. B, Dose-dependent reduction of survived clones after IR. C, The effects of melatonin on clonogenic survival of irradiated cells, cells were irradiated by 4 Gy IR. * Significant difference versus control at
Millimolar Melatonin Prevent Radiation-Induced Cellular Inactivation by Its Antioxidant Capacity
Ionizing radiation is used to kill cells by inducing DNA damage via ROS generation. It is possible that the protective effect of melatonin against IR could be due to its antioxidant ability. The DCF signal is much weaker in 4 Gy-irradiated cells treated with 1-mM melatonin compared to untreated cells (Figure 2A). The FACS result also confirmed the reduction of ROS in 1-mM melatonin-treated group (Figure 2B). Agomelatine, a melatonin receptor agonist, was unable to protect the cells from IR-induced cellular inhibition, but instead enhanced the cell-killing effect of IR (Figure 2C).

The antioxidant capacity of high concentration melatonin protected HeLa cells from IR-induced cellular inactivation. A, Representative images of cells stained by DCFH-DA 2 hours after 4 Gy IR. HeLa cells were treated with IR and/or 1000-µM melatonin. B, FACS measurement of DCF signal 2 hours after IR with/without 1000-µM melatonin after 4 Gy IR. C, The relative index of survived clones after 4 Gy IR with/without melatonin receptor agonist agomelatine. * Significant difference between 2 groups at
Micromolar Melatonin Activated JNK Signaling to Promote IR-Induced Cellular Inactivation
The synergistic effect of IR and melatonin agonist suggests that MT-mediated signaling plays an important role in the boosted cell-killing effect by melatonin. Melatonin can activate JNK signaling through melatonin receptors on the membrane. Since JNK signaling pathway can drive cells to apoptosis, we postulate that the increased cellular inactivation by 10-µM melatonin treatment after IR could be due to JNK activation. The JNK phosphorylation was detected with the presence of melatonin from the range of 10 to 1000 µM (Figure 3A). The induction of JNK phosphorylation was independent of IR (Figure 3B). SP600125, a specific p-JNK inhibitor, abrogated the enhancement of cell killing in cells treated with 10-µM melatonin and IR (Figure 3C).
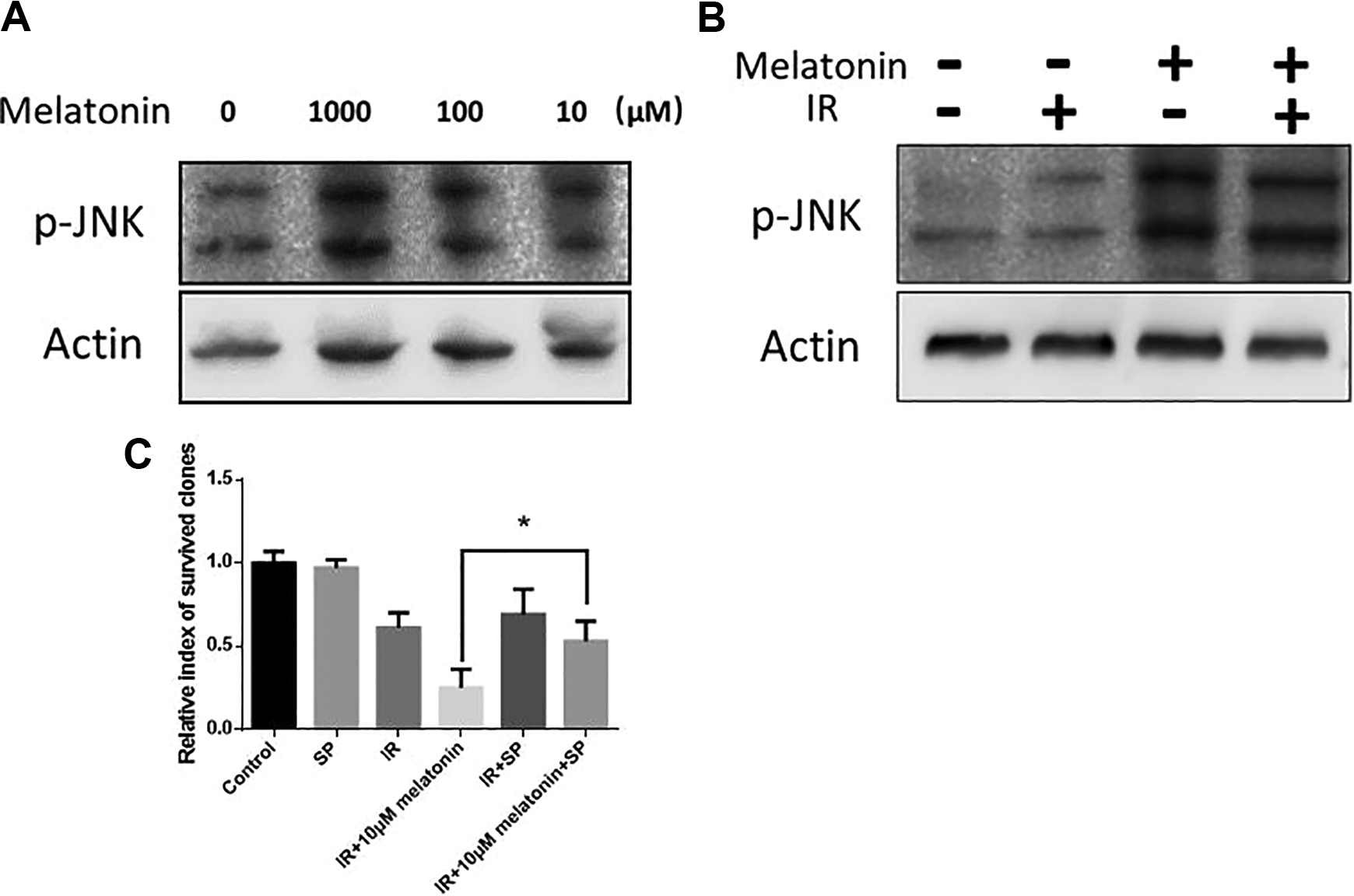
A 10-µM melatonin promoted IR-induced cellular inactivation via JNK signaling. A, Representative image of p-JNK blotting with different concentrations of melatonin. The cells were collected 4 hours with melatonin treatment. B, Representative image of p-JNK blotting after treatment of 10-µM melatonin with/without IR. The cells were collected 2 hours after 4 Gy IR. A 10-µM melatonin was added 2 hours prior to the IR. C, Relative survived clones treated with 10-µM melatonin with/without 100-µM JNK inhibitor SP600125 (SP) after 4 Gy IR. * Significant difference between 2 groups at
Discussions
The physiological concentration of melatonin, which is found in the plasma in animals and the human body, is within the nanomolar range. However, our initial experiments found no changes in HeLa cells if melatonin was administrated at nanomolar (data not shown). This could be due to its short half-life estimated (20-50 minutes in the body). Sainz et al reported that 1-mM melatonin does not promote IR-induced cell death due to increased GSH content. 19 Similarly, we found that 1-mM melatonin prevented IR-induced cell death. The protective effect is due to enhanced antioxidant capacity by the presence of a high concentration of melatonin. Also, we found a low concentration of melatonin (10 µM) efficiently enhanced the cell-killing effect of IR, with the induction of JNK signaling. This is the first report showing that melatonin at a relative lower concentration can enhance the efficiency of cellular inactivation by IR.
The researchers reported that melatonin-induced cell death is associated with an early increase in ROS at a concentration of 1 mM. 20 –25 The cell growth was not significantly affected by melatonin up to 1 mM in HeLa cells, indicating the cellular inhibition effect of melatonin could be cell-type specific. Another explanation is that HeLa cells can tolerate the early increase in ROS induced by melatonin. But, since we are looking for the long-term effect of radiotherapy, this ROS generation in the early exposure to melatonin was not investigated.
Our data showed that melatonin, as well as the receptor agonist, can activate the melatonin receptor-mediated JNK pathway, which can enhance the cell-killing effect of IR. These data indicate the potential role of melatonin receptor-mediated signaling pathway in radiotherapy. Clinical applications of melatonin for cancer, such as breast cancer, have shown promising results. However, the potential application of melatonin in radiotherapy is still under its way. Especially, the dosage effects of melatonin in combination with IR has not been studied. It will be ideal that an appropriate dose of melatonin can be administrated on cancer and its adjacent normal tissue during radiotherapy. The high concentration of melatonin presented in the normal tissue can abrogate the side effects of IR, while the presence of nanomolar melatonin can activate signaling pathway that enhances the killing effect of IR. The plasma concentration of melatonin is negatively related to age. 26 It will be interesting to investigate whether a supplement of melatonin to the aged patient could enhance the efficiency of radiotherapy.
In summary, we found that the melatonin can enhance the cell-killing effect of IR at low concentration, while the high dosage of melatonin protected cells from IR-induced death at high dosage. These different effects of melatonin are due to the versatile nature of melatonin. The MT-mediated JNK signaling accounts for the enhanced cell-killing effect, but melatonin can also prevent DNA damage induced by radiation by its ROS scavenging ability at higher concentration. These results indicate that melatonin can be used to promote radiotherapy efficiency by enhancing cancer cell killing and protecting the normal tissue from radiation damage.
Footnotes
Acknowledgments
The authors thank every researcher in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The radioactive source was supported by the Radiation Center of the Lanzhou University Second Hospital.
