Abstract
Lobaplatin is a diastereometric mixture of platinum (II) complexes, which contain a 1,2-bis (aminomethyl) cyclobutane stable ligand and lactic acid. Previous studies have showed that lobaplatin plays inhibiting roles in various types of tumors. However, the role of lobaplatin in prostate cancer remains unknown. Cell viability was detected by 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-H-tetrazolium bromide assay. Cell proliferation was detected by cell colony formation assay. Cell migration and invasion were determined by transwell migration and invasion assay. Cell apoptosis was detected by flow cytometry. The messenger RNA and protein expression levels were detected by quantitative real-time polymerase chain reaction and Western blot. Lobaplatin treatment inhibits cell viability, cell proliferation, cell migration, and invasion, while promotes cell apoptosis of prostate cancer cell lines DU145 and PC3. Meanwhile, lobaplatin treatment regulates apoptosis by downregulation of BCL2 expression and upregulation of BAX expression levels. Our study suggests lobaplatin inhibits prostate cancer proliferation and migration through regulation of BCL2 and BAX expression.
Introduction
Prostate cancer is one of the most common cancers in the world and leads to the second most cancer-related death for US men. 1 There are more than 3.3 million patients with prostate cancer in the United States, 2 and diagnosis of prostate cancer in United States was estimated 220 000 yearly. 3 More importantly, the increasing occurrence in developing countries including China has been noted in recent years. 4 Patients with prostate cancer are usually diagnosed by prostate-specific antigen (PSA) testing, although the efficacy of PSA screening is under debates. 5 Treatment for prostate cancer include surgery, radiation therapy, cryotherapy (cryosurgery), hormone therapy, chemotherapy, and vaccine treatment, 6 while the opinions always vary depending on disease extent and patient characteristics, such as age, comorbidity, and personal preferences. 2 Advances in understanding the epidemiology and pathogenesis of prostate cancer have been made to develop potential novel treatments. However, in clinical management, patients are likely to relapse after primary therapy, which is the most important problem. 7 Therefore, more efforts are needed to explore the molecular mechanism, which regulates prostate cancer progress for more effective therapies and improved clinical outcomes.
Lobaplatin is the third-generation platinum complex originally developed by the German firm ASTA Medica (Degussa). 8 With the DNA-alkylating activity, lobaplatin induces the DNA–drug adducts (interstrand Pt-GG and Pt-AG cross-links) formation, blocks DNA replication and transcription, and further inhibits gene expression in tumor cells, which holds the similar mechanism of action to other platinum drugs. 9 Lobaplatin has numerous advantages for clinical development, such as high antitumor activity, favorable toxicity profile, good solubility, and stability in water. 10 Previous studies demonstrated that lobaplatin plays antitumor activation in various kinds of tumors, including breast cancer, 11 esophageal squamous cell carcinoma, 12 gastric carcinoma, 13 lung cancer, 14 melanoma, 15 and ovarian cancer. 16 However, until now, no evidence has showed the role of lobaplatin in prostate cancer.
In the present study, we explored the role of lobaplatin in prostate cancer. We found that after lobaplatin treatment, the cell viability, cell proliferation, cell migration, and invasion of prostate cancer cell lines DU145 and PC3 were significantly inhibited, while cell apoptosis was significantly promoted. Mechanically, lobaplatin treatment inhibited BCL2 expression levels and increased BAX expression level. These data indicate the potential role of lobaplatin for clinical treatment of patients with prostate cancer.
Methods and Materials
Cell Lines
The human prostate cancer cell lines, DU145 and PC3, were purchased by Cell Bank of Shanghai (China) and cultured in Roswell Park Memorial Institute 1640 (Invitrogen, Carlsbad, California) in the presence of 10% fetal bovine serum (FBS), 100 units/mL penicillin and 100 g/mL streptomycin in a humidified 5% (vol/vol) atmosphere of CO2 at 37°C incubator. The cells were verified by Short Tandem Repeat method.
RNA Isolation and Quantitative Real-Time Polymerase Chain Reaction
According to the manufacturer’s instructions, total RNA was isolated from DU145 and PC3 cell lines using TRIzol reagent (Invitrogen). Complementary DNA was synthesized using the PrimeScript real-time reagent kit (TaKaRa, Dalian, China). The primers used for detecting the messenger RNA (mRNA) expression levels of BCL2 and BAX are as follows: BCL2: forward 5′-GCC CTG TGG ATG ACT GAG TA-3′, reverse 5′-TTC AGA GAC AGC CAG GAG AAA-3′; BAX: forward 5′-GCT GGA CAT TGG ACT TCC TC-3′, reverse 5′-GGC GTC CCA AAG TAG GAG AG-3′; VIM: forward 5′-GAG CTA CGT GAC TAC GTC CAC C-3′, reverse 5′-GTT CTT GAA CTC GGT GTT GAT G-3′. The mRNA expression level of GAPDH was used for normalization, and the relative expression levels of BCL2, BAX, and VIM were measured by 2−ΔΔCT method.
3-(4,5-Dimethyl-2-Thiazolyl)-2,5-Diphenyl-2-H-Tetrazolium Bromide Assay and Colony Formation Assay
3-(4,5-Dimethyl-2-thiazolyl)-2,5-diphenyl-2-h-tetrazolium bromide (MTT) assay was performed according to the manufacturer’s protocol. Briefly, 3 − 5 × 104 cells/well were cultured in 96-well plates for 24, 48, or 72 hours. At the end of culture, 50 μL of the MTT reagent (5 mg/mL) was added to each well and incubated for 4 hours, followed by adding 200 μL of dimethyl sulfoxide to each well and incubating for 15 minutes. The absorbance was read using a microplate reader (SpectraMAX Plus, Molecular Devices, Sunnyvale, California) at a wavelength of 570 nm.
For colony formation assays, 1000 cells were seeded into 6-well plates to culture for 14 days. Then, the cells were fixed using 70% ethanol and stained with 10% Giemsa (Sigma-Aldrich, St. Louis, Missouri). Only colonies containing >50 cells were counted. All experiments were carried out in triplicate.
Cell Proliferation Assay and Apoptosis Analysis
DU145 and PC3 cells were seeded at the 1 × 105/well into 6-well plates. The total cell number of each well was counted 3 times on days 1, 2, 3, 4, and 5 days using the Z1 particle counter (Beckman Coulter, Inc, Brea, California).
To perform apoptosis assay, cells were plated at a density of 2 × 106 cells per 6 cm dish and treated with 15 µM lobaplatin or the solvent control. 12 or 24 hours later, the cells were pelleted by centrifugation at 1500 rpm for 5 minutes and washed twice with phosphate buffer saline. Then, cells were stained according to the protocol of the Annexin V/PI kit, followed by flow cytometry evaluation.
Migration and Invasion Assays
The transwell system (24 wells, 8 mm pore size with polycarbonate membrane) and Matrigel were used according to the manufacturer’s protocols for invasion assays. Total of 1 × 105/well cells were seeded into the upper chamber with serum-free opti-MEM medium. In the lower compartment, opti-MEM containing 10% FBS was used as a chemoattractant. 48 hours later, cells remained in the upper chamber were scraped out. Matrigel membranes were fixed with ice-cold methanol and stained with 0.1% crystal violet solution. Five randomly fields were selected under a light microscope to count the number of cells migrated to the lower side.
For the migration assay, cells were seeded in the upper chambers without Matrigel. The rest of assay was performed as the invasion assay.
Western Blot
Cells were lysed in radioimmunoprecipitation assay buffer (Sigma) and electrophoresed in 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gel. The proteins were transferred to polyvinylidene fluoride membranes (Millipore, Billerica, MA), followed by blocking with 5% milk at room temperature. Then, the member was incubated with anti-BCL2 or BAX primary antibody at 4°C overnight, followed by incubation with horseradish peroxidase-conjugated secondary antibody at room temperature for 1 hour. Finally, signals were visualized by enhanced chemiluminescence (Pierce, Wisconsin). β-Actin (CST) was used as control.
Statistical Analysis
All data analyzed as the mean (standard deviationSD) are from at least 3 separate experiments. The statistical significance was determined using Student t test, 1- or 2-way analysis of variance analysis followed by a Bonferroni post hoc test; P <.05 was considered significant.
Results
Lobaplatin Inhibits Cell Viability of Prostate Cancer Cells
To explore the role of lobaplatin in prostate cancer cell proliferation, we treated DU145 and PC3 cells with different doses of lobaplatin and detected cell viability by MTT assay at different time points (12, 24, and 48 hours). As shown in Figure. 1A and B, the cell viability of DU145 or PC3 significantly decreased long with the increased concentrations of lobaplatin, indicating that lobaplatin could inhibit cell viability of DU145 or PC3 in a dose-dependent manner. In addition, cell viability of the 2 prostate cancer cell lines significantly decreased at 48 hours compared with that at 12 hours at the low dose of lobaplatin (5 μM). These data demonstrate that lobaplatin inhibits cell viability of prostate cancer cells, which is as similar as the effect of cisplatin and oxaliplatin (Figure 2A and B).
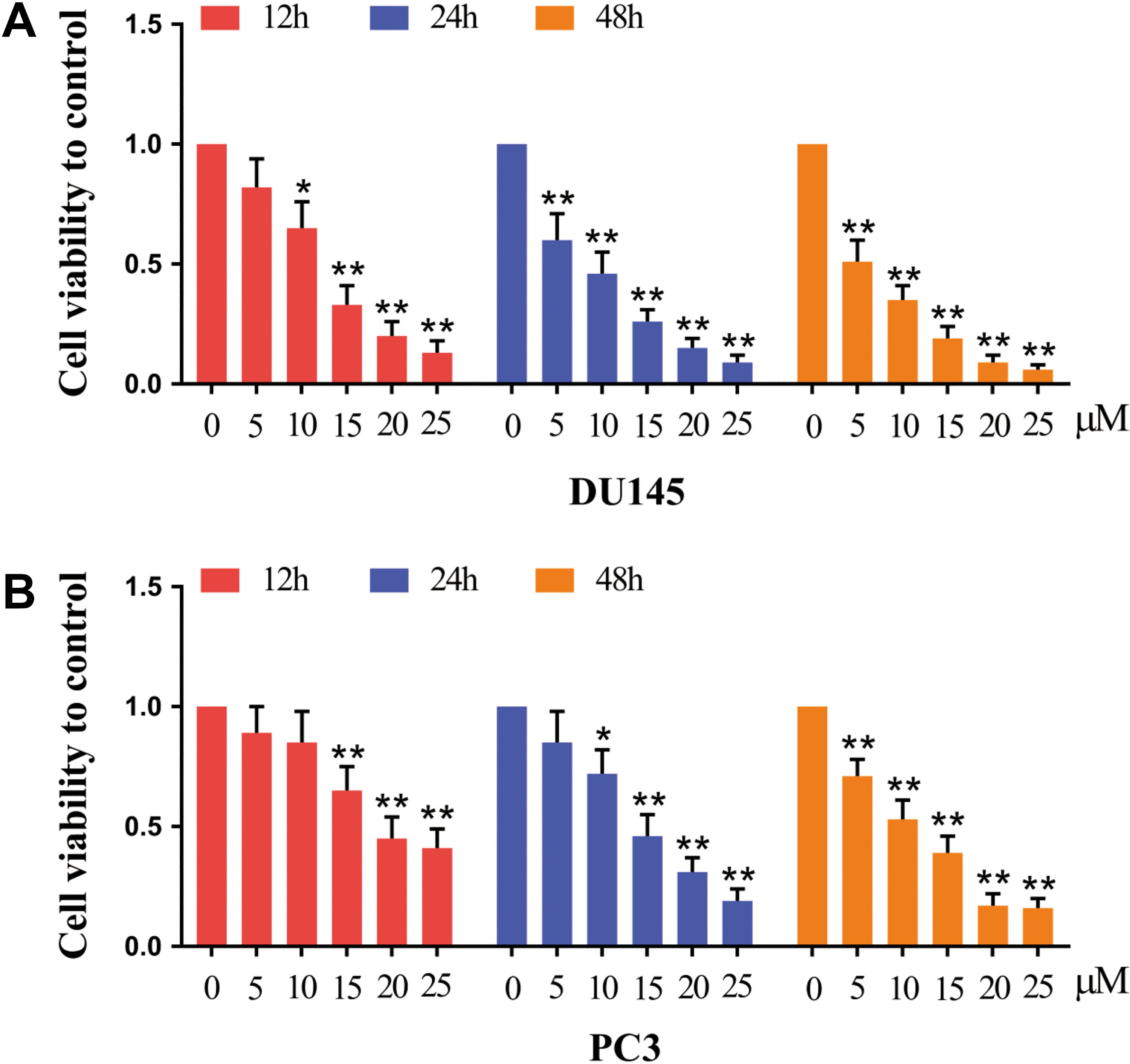
Lobaplatin inhibits prostate cancer cell proliferation. (A) DU145 and (B) PC3 cells were treated with lobaplatin and subjected to MTT assay as indicated. Data are mean (SD) of 3 independent experiments and each measurement in triplicate (*P < .05, **P < .01). MTT indicates 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-h-tetrazolium bromide; SD, standard deviation.

Cisplatin and oxaliplatin inhibit prostate cancer cell proliferation. Prostate cancer cells were treated with (A) cisplatin or (B) oxaliplatin and subjected to MTT assay as indicated. Data are mean (SD) of 3 independent experiments and each measured in triplicate (*P < .05, **P < .01). MTT indicates 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2-h-tetrazolium bromide; SD, standard deviation.
Lobaplatin Inhibits Proliferation of Prostate Cancer Cells
We next detected the influence of lobaplatin on prostate cancer cell proliferation. The cell numbers of DU145 or PC3 were much lower after lobaplatin treatment (15 µM; Figure 3A and B). The cell colony formation ability of DU145 or PC3 was also significantly inhibited by lobaplatin treatment (Figure 3C and D). These data indicate that lobaplatin inhibits proliferation of prostate cancer cells.

Lobaplatin inhibits proliferation in prostate cancer cells. (A) DU145 and (B) PC3 cells were treated with lobaplatin (15 µM) and subjected to cell number assay every 24 hours. (C) DU145 and (D) PC3 cells were treated with lobaplatin (15 µM) for 2 weeks and subjected to cell colony formation assay. Data are mean (SD) of 3 independent experiments and each measurement in triplicate (*P < .05, **P < .01). SD indicates standard deviation.
Lobaplatin Induces Cell Apoptosis of Prostate Cancer Cells
We next determined cell apoptosis after lobaplatin treatment. DU145 and PC3 prostate cancer cell lines were treated with 15 µM lobaplatin for 12 or 24 hours. The percentage of DU145 and PC3 cell apoptosis significantly increased (from approximately 4% to 7%) after lobaplatin treatment for 12 (Figure 4A and B) and 24 hours (from approximately 5% to 18%; Figure 5A and B). These data indicate that lobaplatin has the ability to induce apoptosis in prostate cancer cell lines.
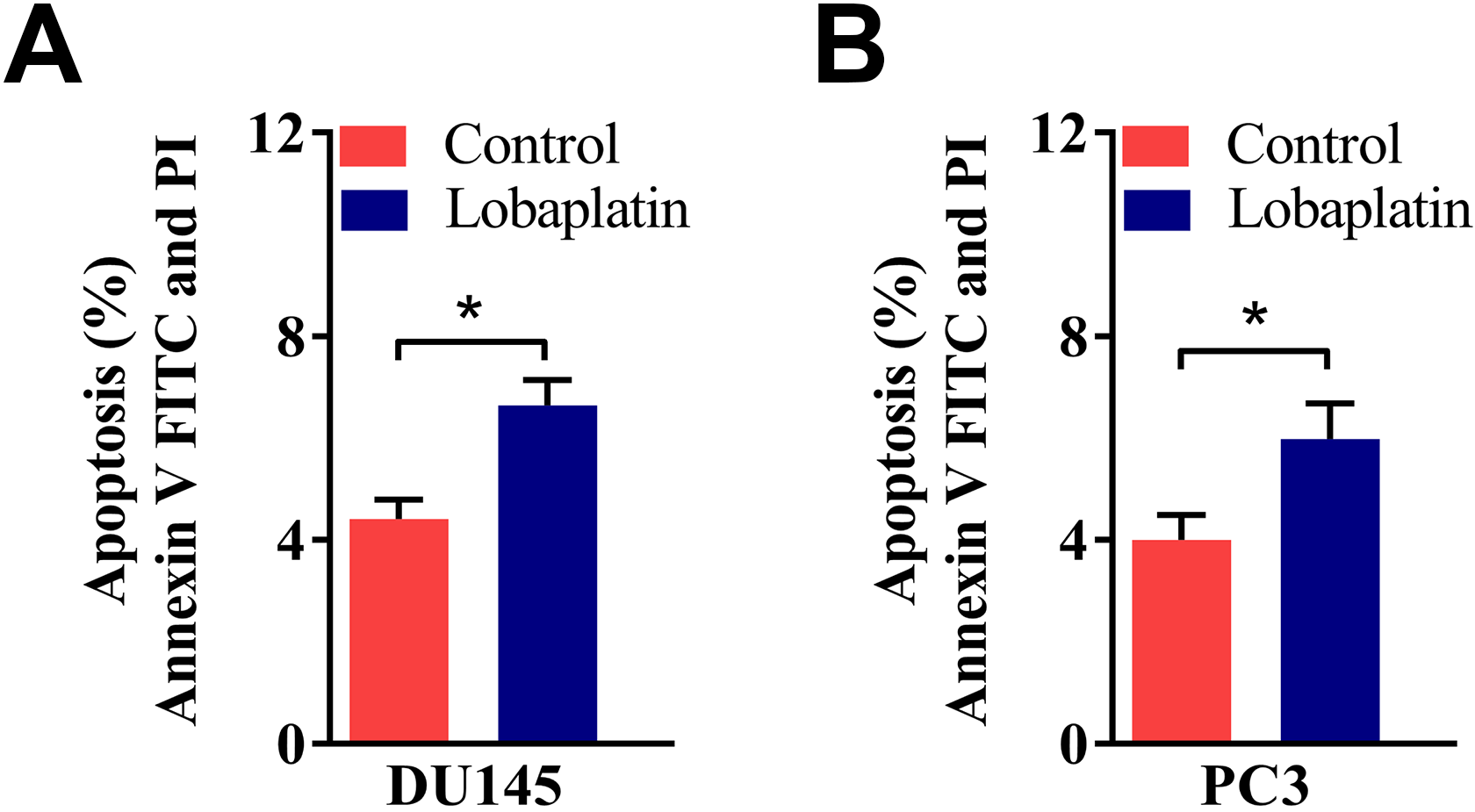
Lobaplatin induces cell apoptosis in prostate cancer cells. Representative of flow cytometry analysis of (A) DU145 and (B) PC3 cell death after cells were treated with lobaplatin (15 µm) for 12 hours. Data are mean (SD) of 3 independent experiments and each measured in triplicate (**P < .01). SD indicates standard deviation.

Lobaplatin induces cell apoptosis in prostate cancer cells. Representative of flow cytometry analysis of (A) DU145 and (B) PC3 cell death after cells were treated with Lobaplatin (15 µM) for 24 hours. Data are mean (SD) of 3 independent experiments and each measurement in triplicate (**P < .01). SD indicates standard deviation.
Lobaplatin Suppresses Cell Migration and Invasion of Prostate Cancer Cells
We next detected the influence of lobaplatin on prostate cancer cell migration and invasion, which are important for cancer progress. DU145 and PC3 cells were treated with 15 µM lobaplatin, followed by cell migration and invasion assay. As shown in Figure 6A and B, lobaplatin treatment significantly inhibited cell migration and invasion of the 2 cell lines, demonstrating that lobaplatin inhibits prostate cancer cell migration and invasion.

Lobaplatin suppresses cell migration and invasion in prostate cancer cells. (A) DU145 and (B) PC3 cells were subjected to transwell migration and invasion assay. Scale bar, 20 µm. Data are mean (SD) of 3 independent experiments and each measurement in triplicate (*P < .05, **P < .01). SD indicates standard deviation.
Lobaplatin Regulates BCL2, BAX, and VIM Expression Levels
Given that lobaplatin promotes prostate cancer cell apoptosis and suppresses migration, we next detected whether lobaplatin has the ability to regulate apoptosis and epithelial–mesenchymal transition (EMT)-related gene expression. DU145 and PC3 cell lines were treated with 15 µM lobaplatin for 48 hours. The mRNA level of BCL2 and VIM significantly decreased after lobaplatin treatment, while BAX mRNA level increased in both of the prostate cancer cell lines (Figure 7A and B). Western blot analysis showed similar decreased BCL2 and VIM protein levels and increased BAX protein levels after lobaplatin treatment (Figure 7C). These data demonstrate that lobaplatin promotes prostate cancer cell apoptosis and suppresses migration through regulating BCL2, BAX, and VIM expression levels.

Cellular expression of apoptosis-related gene was analyzed after prostate cancer cells were exposed to lobaplatin (15 µM) for 48 hours. (A) (B) BCL2, BAX, and VIM levels were measured via qRT-PCR and normalized to the level of GAPDH. (C) Western blot analysis of BCL2, BAX, and VIM expression. β-Actin serves as an internal control. Data are mean (SD) of 3 independent experiments and each measurement in triplicate (*P < .05). qRT-PCR indicates quantitative real-time polymerase chain reaction; SD, standard deviation.
Discussion
Lobaplatin is the third-generation water-soluble platinum compounds, appears to be more stable, less toxic, with a better therapeutic index, and may overcome tumor resistance as compared with the first and second generation of platinum compounds. 17,18 Because of superior pharmacokinetic parameters in Chinese populations versus Western populations, 10,19 lobaplatin has been approved in China for the treatment of chronic myelogenous leukemia, metastatic breast, and small cell lung cancer. 20,21 However, there is still no report about the application of lobaplatin in prostate cancer treatment. In the present study, we explored the role of lobaplatin in prostate cancer. We found that lobaplatin treatment inhibits cell viability, cell proliferation, cell migration, and invasion of prostate cancer cell lines DU145 and PC3, while promoted cell apoptosis. Lobaplatin treatment regulates the expression levels of apoptosis and EMT-related genes, BCL2, BAX, and VIM. To the best of our knowledge, this is the first study to explore the role of lobaplatin in prostate cancer.
Cell migration and invasion into surrounding tissue and vasculature is a crucial step for tumor development 22,23 ; so a pressing goal in tumor biology is how to inhibit the activity of migration and invasion of tumor cells. In this study, we provided evidence that lobaplatin treatment inhibits cell migration and invasion of DU145 and PC3 cell lines, indicating that lobaplatin may inhibit prostate cancer via inhibiting cell migration and invasion.
Apoptosis is an ordered and orchestrated programmed cell death that occurs in physiological and pathological conditions, which plays crucial roles in maintaining survival/death balance in cells. 24 During the process of antitumor activity, there always be apoptosis of the tumor cells. 25 Defect in apoptosis can promoted cancer development, while enhanced apoptosis may inhibit cancer progress. 26 We found that lobaplatin treatment significantly increased the percentage of apoptosis in both DU145 and PC3 cell lines, indicating that lobaplatin plays important roles in promoting cell apoptosis.
BCL-2 (B-cell lymphoma 2) is one of BCL-2 family members that regulates cell death by either inducing or inhibiting apoptosis. 27,28 Bcl-2 has clinical significance in lymphoma, specifically be considered to inhibit apoptosis during tumor development. 29 In human colon cancer cell line HCA-7, PGE2 treatment leads to increased clonogenicity, inhibits programmed cell death via inducing BCL-2 expression levels. 30 ABT-199, a potent and selective BCL-2 inhibitor, inhibits BCL-2 dependent tumor growth in vivo, and leads to tumor lysis within 24 hours in 3 patients with refractory chronic lymphocytic leukemia, indicating the promise of BCL-2 inhibition in the treatment of BCL-2–dependent hematological cancers. 31 The expression levels of BCL-2 were increased in miR-143-downregulated cervical tissues as compared with normal cervical tissues. Overexpression of miR-143 in HeLa cells resulted in downregulated Bcl-2 expression, while miR-143 knockdown upregulated Bcl-2 expression, indicating that miR-143 plays crucial roles in the pathogenesis of cervical cancer through regulating BCL-2 expression. 32 BAX (Bcl-2–associated X protein) functions as an apoptotic activator, showed lower expression in breast cancer cells. The interaction of BAX with p53 and caspase-3 is associated with active apoptosis in breast cancer cells. 33 The dissociation of BAX from Bcl-xL and activation of caspase families were essential for quercetin caused apoptotic processes in human prostate cell line (LNCaP). 34 In our study, the most important question is the mechanism of how lobaplatin promotes apoptosis. We detected the mRNA and protein levels of apoptosis-related genes, BCL2 and BAX. BCL2 expression levels decreased, while BAX expression levels increased after lobaplatin treatment, indicating that lobaplatin promotes apoptosis via regulating apoptosis-related genes expression.
It is important to note that the effects of lobaplatin in prostate cancer were performed in 2 androgen receptor negative prostate cancer cell lines (DU145 and PC3), while androgen receptor positive prostate cancer cell lines, such as 22RV1, should be used for further detection. More importantly, the data are from prostate cancer cell lines in vitro, no in vivo data. The prostate cancer mouse model and the clinical samples should be used to verify the effects of lobaplatin in prostate cancer, which will provide theoretical basis for the potential use of lobaplatin for the treatment of patient with prostate cancer.
Taken together, we found that lobaplatin treatment significantly inhibits the cell viability, cell proliferation, cell migration, and cell invasion of prostate cancer cell lines DU145 and PC3 and promotes cell apoptosis of the 2 cell lines. Mechanically, lobaplatin treatment inhibited BCL2 and VIM expression levels and increased BAX expression level. These data indicate the potential role of lobaplatin for clinical treatment of patients with prostate cancer.
Conclusion
Lobaplatin inhibits prostate cancer proliferation and migration, promotes cell apoptosis through regulation of BCL2 and BAX. These data indicate the potential role of lobaplatin for clinical treatment of patients with prostate cancer.
Footnotes
Authors’ Note
Hongwen Cao and Yigeng Feng contributed equally to this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by The Third Batch of Young Chinese Name Training Program of LongHua Hospital Shanghai University of Traditional Chinese Medicine (Chen Lei; subject number: RC-2017-01-14); Shanghai Municipal Health and Family Planning Commission Special Subject of Chinese Medicine Research (subject number: 2016JP014); and National TCM Clinical Research Base Dragon Medicine Scholars (nursery plan) of LONGHUA Hospital Shanghai University of Traditional Chinese Medicine (subject number: LYTD-56).
