Abstract
Many plant-derived compounds have been used to treat microbial infections. Staphylococcus aureus a common cause of many organ infections, has generated increasing concern due to its resistance to antibacterial drugs. This work was carried out to explore the susceptibility of 6 strains (LN872136, LN872137, LN871238, LN871239, LN872140, and LN871241) of methicillin-resistant Staphylococcus aureus to aqueous extract of Lepidium sativum seeds in vitro. Various concentrations (5-20 mg/mL) were used to evaluate the effect of the extract on bacteria growth via the assessment of the microbial biomass and the inhibition zone (IZ). The results showed that the plant extract at 15 or 20 mg/mL, significantly decreased the the biomass of S aureus strains after 24 or 48 hours exposure period. Staphylococcus aureus (LN871241) showed the largest IZ at 20 mg/mL and documented by scanning electron microscope. The current work may suggest that L sativum seed extract can be candidate as a promising antimicrobial agent to treat infection with methicillin-resistant S aureus.
Keywords
Introduction
Bacterial resistance to various antibiotics has been increased, and this become threatens the health of many population throughout the world as it decreases the efficacy of many antibiotics. 1 Staphylococcus aureus is a harmful gram-positive bacterium accountable for many illness and death. 2,3 Infection with this bacteria is one of the leading cause of a range of human diseases, including, bacteremia, food poisoning, sepsis, brain abscess, toxic shock syndrome, osteomyelitis, endocarditis, keratitis, and pneumonia. 4 Methicillin-resistant Staphylococcus aureus (MRSA) is a multiresistant strain that has been known all over the world, exhibiting resistance to many categories of antimicrobial drugs. 5 Methicillin-resistant Staphylococcus aureus has disseminated all over the world due to over- and inappropriate use of antibiotics. 6 Infection by MRSA is difficultly treated because of their resistance to many antibiotics including the most powerful effective antibiotics, vancomycin. 5 So it is urgent to look for new approaches to overcome the MRSA infectious problems.
Natural compounds of plant origin are considered sources of therapeutic value. 7 Treatment of such antibiotic resistance microbes, using plant-derived compounds with antimicrobial properties, may consider a new effective strategy for MRSA infectious eradication.
Lepidium sativum (family, Brassicaceae) is a popular plant used by Arabic countries, including Saudi Arabia, for its nutritional and medicinal values. 8,9 Lepidium sativum is known in ancient India and Saudi Arabia for treating various conditions, such as bone fracture healing, inflammation, arthritis, and many others. 8 The seed extracts have been utilized in local conventional medicine, to treat dysentery, gastrointestinal disorders, stomachache, indigestion, febrile disease, and skin disorders. 10 Also, the antimicrobial effectiveness of L sativum seed extracts versus various microbial pathogens has been documented. 11,12
Although the impacts of L sativum extracts have been confirmed against the pathogenic microbes, the effect of the aqueous extract of L sativum seeds against MRSA is still uncertain.
The current work was carried out to investigate the bioactivity of different concentrations (5-20 mg/mL) of L sativum seeds extract as a potent antipathogenic agent to fight MRSA strains in vitro.
Materials and Methods
Plant
Lepidium sativum
Lepidium sativum seeds were obtained from the local market, Jeddah, Saudi Arabia, during the period 2014 to 2015. The plant seeds were authenticated by a taxonomist in the Biological Science Department, King Abdulaziz University, Jeddah, KSA.
Preparation of L sativum seed extract
Fifteen grams of the of dried, powdered seeds of L sativum were steeped in 150 mL of sterilized bidistilled water for 3 days and then filtered through 8-layered of muslin fabric. The filtrate was centrifuged at 4000×g for 20 minutes. The extract was then gathered and freeze dried using a lyophilizer. 12
Staphylococcus aureus
Six strains of MRSA were isolated from contact lenses, kohl samples, and patients with corneal infection. Identification and characterization of S aureus were carried out using morphological microscopic study, coagulase, and catalase tests. 13 All the bacterial strains were examined for methicillin resistance. The disk-diffusion assay 14 was utilized with a 1 µg oxacillin disk (Oxoid). The sizes of IZ were recorded after 24 hours at 35°C. Strains with IZ ≤8 mm were considered methicillin resistant. 15 The various strains of S aureus were also identified by analysis of 16S ribosomal RNA (16S rRNA) genes for bacterial species. 16
Influence of L sativum extract on the biomass of S aureus strains
Seed extract (5-20 mg/mL) was added into MRS broth medium containing S aureus strains individually (106 CFU/µL). Negative control was represented by the bacterial culture without the addition of the plant extract. After 24 and 48 hours, the cultures were centrifuged at 5000×g for 15 minutes to obtain the cell pellet. The pellets of bacterial strains were dried at 80°C till constant weights were obtained. All experiments were carried out 3 times. The bacterial biomass was represented as mg dry weight/mL. 17
Antimicrobial assay of L sativum extract
The MRSA strains were grown in Tryptic Soy Broth overnight (18 hours) at 37°C. The agar-well diffusion assay was carried out for evaluation the bactericidal efficacy of L sativum seed extract. 18 Suspensions of S aureus 6 strains (106 CFU/mL, each) were spread on Mueller Hinton Agar medium. Wells with 10 mm diameter were made in the agar medium and put onto the incubated plates. Lepidium sativum seed extract (20 mg/mL) was added into the labeled medium plates. Controls of bacterial strains without the plant extract were also involved in the experiment. The plates were maintained at 4oC for 2 hours, then at 37°C for 24 hours. The susceptibility of S aureus strains to the extract was assayed by estimating the IZ (mm) postincubation time. Triplicate experiments were carried out for each S aureus strain, and the average of IZ was calculated. 19
Scanning Electron Microscopy
Scanning electron microscopy (SEM) was performed on S aureus (LN871241), the most sensitive strain to extract. The bacterial cells in nutrient broth media were treated with the plant extract (20 mg/mL) and kept for 24 hours at 37°C. Bacteria without extract were utilized as a control. Centrifugation was carried out to collect the microbial cells and then washed using a sterilized phosphate-buffered saline. The cells were fixed overnight at 4°C in glutaraldehyde (2.5%) in phosphate buffer (0.1 M, pH 7.3) and then in 1% osmium tetroxide for 1 hour at room temperature. The bacterial samples were then processed for SEM (XL20-Philips, USA).
Statistical Analyses
Results were analyzed utilizing 1-way analysis of variance (ANOVA), and Bonferroni test as a post-ANOVA test. Values were calculated as mean (standard deviation). Variations between values were significant at P ≤ .05.
Results
Effect of L sativum Extract on the Biomass of S aureus Strains
The influence of various concentrations (5-20 mg/mL) of L sativum extract on the dry weights of S aureus different strains is shown in Figures 1 to 6. The results revealed that exposure of S aureus (LN872136; Figure 1) to either 15 or 20 mg/mL of L sativum was effective in reducing the dry weight of this strain after 24 hours versus control strain (zero concentration, P ≤ 0.05), while exposure of this strain to L sativum extract (5-20 mg/mL) significantly decreased the dry weight of this bacterial strain after 48 hours versus the control strain (P ≤ 0.05 for 5 mg/mL, P ≤ 0.01 for 10-20 mg/mL).

Influence of Lepidium sativum extract (5-20 mg/mL) on the dry weight gain of Staphylococcus aureus (LN872136). Results are calculated as mean (SD; n = 3). *P ≤ .05 versus the control untreated strain (zero concentration) after 24 hours exposure to the natural extract, a P ≤ .05 and b P ≤ 0.01 versus the control after 48 hours exposure to the plant extract. SD indicates standard deviation.

Influence of Lepidium sativum natural extract (5-20 mg/mL) on the dry weight gain of Staphylococcus aureus (LN872137). Data are calculated as mean (SD; n = 3). *P ≤ .05 with respect to control untreated strain (zero concentration) after 24 hours exposure to the plant extract. a P ≤ .05 compared with the control untreated strain after 48 hours exposure to plant extract. SD indicates standard deviation.

Influence of various concentrations of Lepidium sativum on the dry weight gain of Staphylococcus aureus (LN871238). Data are calculated as mean (SD; n = 3). *P ≤ .05 compared with the control untreated strain (zero concentration) after 24 hours exposure to the plant seed extract, a P ≤ .05 and b P ≤ .01, compared with the control untreated strain after 48 hours exposure to the plant extract. SD indicates standard deviation.
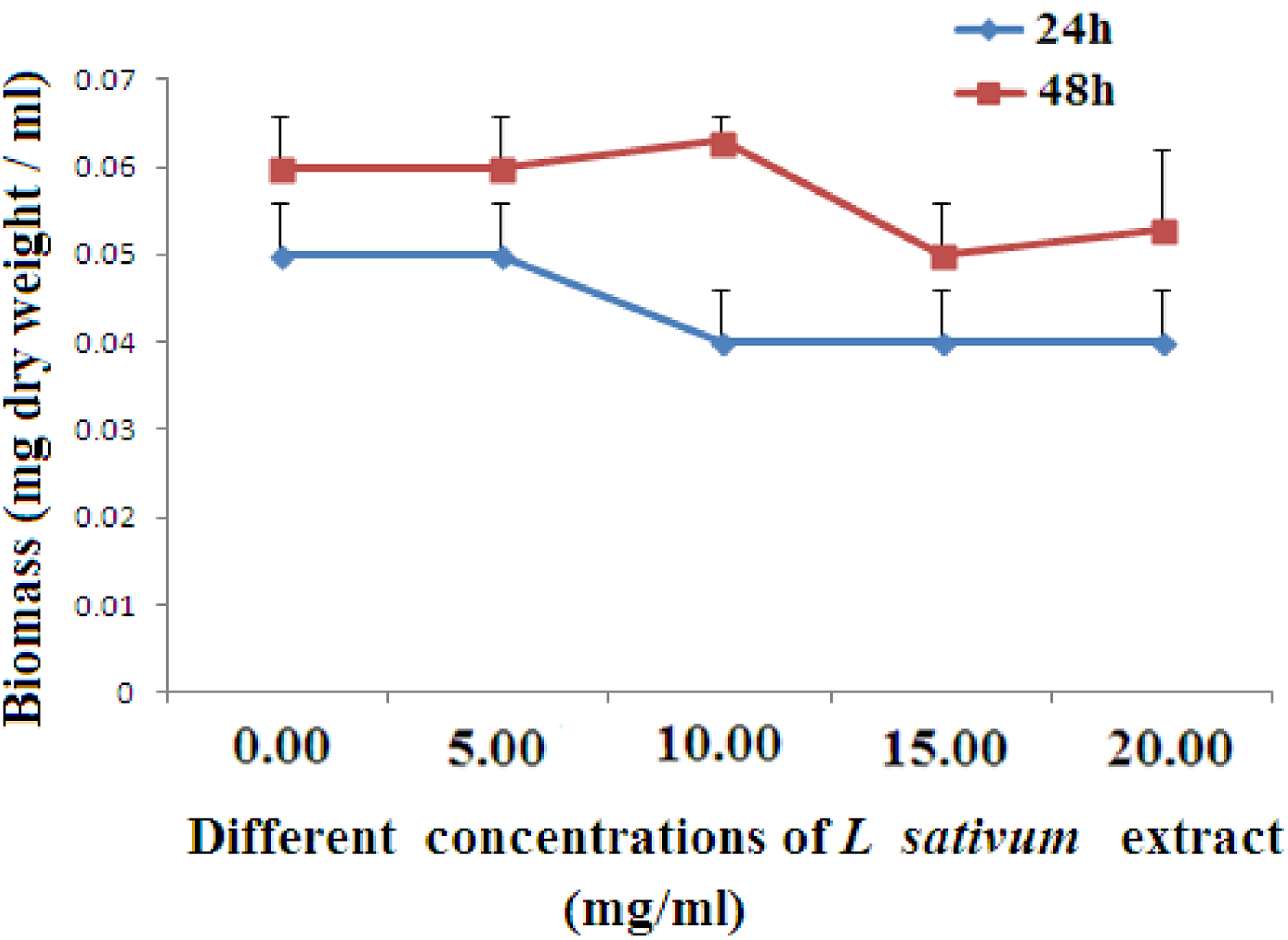
Influence of various concentrations of Lepidium sativum on the dry weight gain of Staphylococcus aureus (LN871239). Data are calculated as mean (SD; n = 3). Nonsignificant changes in the dry weight of the bacterial strain exposed to different concentrations of the plant extract after 24 or 48 hours compared with the control counterpart untreated strain (zero concentration). SD indicates standard deviation.

Influence of various concentrations of Lepidium sativum on the dry weight of Staphylococcus aureus (LN872140). Data are calculated as mean (SD; n = 3). *P ≤ .05, **P ≤ 0.01 compared with the control (zero concentration) after 24 hours exposure to the plant extract, a P ≤ .05, b P ≤ .01, and c P ≤ .001 compared with the control after 48 hours exposure to the plant extract. SD indicates standard deviation.

Influence of various concentrations of Lepidium sativum on the dry weight of Staphylococcus aureus (LN871241). Data are calculated as mean (SD; n = 3). *P ≤ .05 compared with the control (zero concentration) after 24 hours exposure to the plant extract, a P ≤ .05 and b P ≤ .01 compared with the control after 48 hours exposure to the plant extract. SD indicates standard deviation.
From Figure 2, it can be noticed that the L sativum extract was effective in reducing the dry weight of S aureus (LN872137) at 15 and 20 mg/mL after 24 and 48 hours exposure compared with the control. The results of Figure 3 demonstrated that L sativum extract at either concentration (15 or 20 mg/mL) significantly decreased the dry weight of S aureus (LN871238) after 24 hours exposure to the plant extract versus the control (P ≤ 0.05), but exposure of this strain to the different concentrations of the same extract (5, 15, and 20 mg/mL) was more effective in reducing the weight of the bacterial strain after 48 hours with respect to the control (P ≤ 0.05 for 5 mg/mL, P ≤ 0.01 for 15-20 mg/mL). Figure 4 illustrated that treatment of S aureus (LN871239) with L sativum extract (5-20 mg/mL) showed no effect on the dry weight of this strain after 24 and 48 hours exposure periods compared with control counterparts. Exposure of S aureus (LN872140) to 15 and 20 mg/mL of L sativum extract significantly reduced the dry weight of this strain after 24 hours compared with the control untreated strain, while treatment of this bacterial strain with the plant extract at 10 to 20 mg/mL, markedly reduced its dry weight after 48 hours exposure period (Figure 5). The result of Figure 6 demonstrated that the plant extract at either 15 or 20 mg/mL was effective in reducing the dry weight of S aureus (LN871241) after 24 and 48 hours exposure periods.
Antibacterial Impact of L sativum seed Extract
The growth of the different strains of S aureus at 20 mg/mL of the plant extract in term of IZ is depicted in Table 1 and Figure 7. The extract mostly suppressed the growth of S aureus
Influence of Lepidium sativum Extract on the Growth Rate of Staphylococcus aureus Strains.a

aResults are calculated as mean (SD; n = 3).
b P ≤ .001 compared with S aureus (LN872140).
c P ≤ .05 compared with S aureus (LN872137).
d P ≤ .05 compared with S aureus (LN871238).

Effect of Lepidium sativum extract on the growing of Staphylococcus aureus showing the IZ of the different strains. (A) IZ of S aureus (LN872136), (B) IZ of S aureus (LN872137), (C) IZ of S aureus (LN871238), (D) IZ of S aureus (LN871239), (E) IZ of S aureus (LN872140), and (F) IZ of S aureus (LN871241). IZS indicates inhibition zone.
Scanning Electron Microscope Observation
The most susceptible S aureus strain (LN872141) to L sativum extract was chosen to evaluate the major alterations in the morphological surface microstructure of the extract-treated bacterial cells, sing SEM (Figure 8B and C). Morphological disorganization was observed on exposure of this strain to the plant extract (20 mg/mL), which represented by formation of wrinkles in the cell wall or even separation of the cell wall. Decreases in the diameters of the bacterial cells were also observed versus control bacteria, which revealed normal morphology (Figure 8A).

Scanning electron micrographs of Staphylococcus aureus (LN872141) bacterial cells treated with Lepidium sativum aqueous extract at 37oC for 24 hours. A, Micrograph of untreated S aureus (LN872141) showing normal shape. B, C, Micrographs of the bacterial strain exposed to L sativum extract showing abnormal morphology, (B) showing rough of the cell wall due to wrinkles (arrows), and (C) showing separation of cell wall and a decrease in the size of the bacterial cell diameter (arrows).
Discussion
The search for bactericidal substances for infectious illness therapy is necessitated by the inherent ability of the pathogens to develop mechanisms of resistance against antibiotics. The potential hurtful adverse impacts related to the utilization of the newly synthesized drugs and the unsustainably high costs of drug development are slowly shifting the focus to plant-derived phytochemicals with antimicrobial significance. 20
The antibacterial impact of L sativum extract on the growth of 6 different strains of MRSA was investigated by determining the bacterial biomass as well as the IZ. The result revealed that the extract (at 15 or 20 mg/mL) was effective in suppressing the growth of most strains after 24 and/or 48 hours as indicated by the reduction in their dry weights compared with the control counterpart untreated strains.
The antimicrobial activity of L sativum seed extract on the bacterial growth in term of IZ was carried out at 20 mg/mL as it was the most effective concentration in suppressing the growth of most S aureus strains. The result revealed that the extract potentially suppressed the growth of tested S aureus strains. The extract mostly inhibited the growth of S aureus (LN871237) and (LN871241) as they recorded the highest IZ (25.33 [5.03] and 27.6 [2.51] mm, respectively) followed by S aureus (LN872140), (LN872136), and (LN871238). However, the plant extract was less effective against S aureus (LN871239) as it recorded the minimum IZ (13.33 [2.08]), compared with the IZ of other strains. Baur et al 21 have declared that the microbe is resistant if the IZ is lower than 8.00 mm and susceptible if it is more than 11.00 mm. Accordingly, the studied strains of S aureus were regarded sensitive to L sativum extract. The present antimicrobial activity of the plant extract is coped with some investigations. It has stated that aqueous L sativum extract was effective against S aureus and Proteus vulgaris. 22 In addition, Berehe and Boru 23 demonstrated that crude extract of L sativum possesses antibacterial impact versus Escherichia coli, Salmonella typhi, Bacillus subtilis, and S aureus.
Scanning electron microscopy was carried out to show the major aberrations in the microstructure of S aureus (LN871241) cells caused by the plant extract, as it is the most sensitive strain to L sativum seed extract. The result demonstrated that bacterial cells exposed to the plant extract showed morphological disorganization as observed by rough bacterial cell wall due to wrinkles, separation of cell wall, and decreases in the bacterial cell diameters compared to normal untreated bacteria, which revealed normal structure. This examination may imply that the extract could destroy the bacterial strains by damaging their cell walls. Our observation is documented by Phansri et al
24
who reported that the bacterial cell wall is the target for antimicrobial agents. Also, Riaz et al
25
found that many antimicrobial drugs can destroy many pathogens through inhibition of their proteins and nucleic acid synthesis. Previous investigation on L sativum seeds has shown that they contain many phytochemical compounds, namely, tannins, triterpenes, flavonoids alkaloids, sterols, and glucosinolates.
26
These phytoconstituents are known to have potential antimicrobial activities. Tannins can interact with proline-rich proteins, causing irreversible inhibition of protein biosynthesis.
27
Triterpenes have been found to have antimicrobial activity against clinical antibiotic-resistant bacteria.
28
Triterpenoids isolated from many plants have been found to have antibacterial efficacy versus many resistant bacterial strains, including Staphylococcus species.
28,29
Previous studies demonstrated that the antimicrobial potential of triterpenes has been related to their damaging effect on the bacterial cell membranes.
30
Also, studies have revealed that terpenes (such as citral, menthol, and thymol) could alter the bacterial cell membrane permeability via invading the lipid bilayers, disturbing lipid packing, and altering the cell membrane fluidity.
31,32
Besides, steroids and flavonoid
Conclusion
The current research illustrated that L sativum extract exhibited potential antibacterial impact against MRSA strains. This may support their potential to be used as an effective alternative medicine to treat MRSA infections.
Footnotes
Acknowledgments
The authors, acknowledge and thank DSR for technical and financial support of KAU.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah, under Grant No. 96-130-35-HiCi.
