Abstract
The effect of acute X-ray irradiation with 2 Gy or fractionated exposure with 0.2 Gy continuously for 10 days (0.2 Gy × 10 = 2 Gy) was evaluated in the postnatal day 21 (P21) BALB/c mouse model. Both acute and fractionated irradiation induced impairment of cell proliferation and neurogenesis in the subgranular zone of the dentate gyrus labeled by Ki67 and doublecortin, respectively. Parvalbumin immunopositive interneurons in the subgranular zone were also reduced significantly. However, the 2 patterns of irradiation did not affect animal weight gain when measured at ages of P90 and P180 or 69 and 159 days after irradiation. Behavioral tests indicated that neither acute nor fractionated irradiation with a total dose of 2 Gy induced deficits in the contextual fear or spatial memory and memory for novel object recognition. Animal motor activity was also not affected in the open-field test. The disparity of the impairment of neurogenesis and unaffected cognition suggests that the severity of impairment of neurogenesis induced by acute or fractionated irradiation with a total dose of 2 Gy at P21 may not be worse enough to induce the deficit of cognition.
Introduction
Acute high-dose irradiation of rodents induces impairment of neurogenesis and cognitive impairment. Fractionated or chronic irradiation with the same dose for acute exposure may reduce acute radiation-induced negative brain effect. However, there are still controversies regarding the effect with different patterns of radiation. For instance, γ-rays irradiation of mice with a total dose of 5 Gy in different patterns, that is, acute and fractionated exposures, induced impairment of neurogenesis which could not return to the control level when examined at 6 months after radiation exposure. Chronic γ-rays irradiation of wild populations of voles with mean lifetime doses of 4 to 6 Gy, with individual doses in long-lived animals up to about 10 Gy, did not result in any clear impacts over about 3 generations. 1 Acute irradiation of C57/Bl6 mice with 0.5 Gy of X-rays did not affect neurogenesis when examined 2 hours after irradiation, but fractionated irradiation with the same total dose of 0.5 Gy (0.05 Gy of X-rays per day for 10 days) caused impairment of neurogenesis in the dentate gyrus (DG) compared to the control mice. 2
In the present study, adolescent radiosensitive BALB/c mice (postnatal day 21 or P21 mice) were irradiated with a total dose of 2 Gy by acute (2 Gy × 1) or fractionated (0.2 Gy × 10, ie, 0.2 Gy/d, and continuous for 10 days) exposure, animal weight gain, behavioral changes, and brain neurogenesis were evaluated. We chose adolescent BALB/c mice because human adolescent brain is vulnerable to stress, and dysfunction of the neurobiological factors involved in adolescent change can increase the individual’s susceptibility to impaired judgment, drug addiction, and psychiatric disorders. 3 Furthermore, extensive studies have been done to evaluate radiation effect on the neonatal and adult rodent brain, 4 -11 and there is a dearth on the similar study in adolescent brain. A total radiation dose of 2 Gy was chosen because this is a conventional fractionated radiotherapy dose for postsurgical treatment of brain tumor reoccurrence or prophylactic prevention of metastasis of cancer cells from other organs. 12 -14 Clinical studies have shown that radiotherapy of patients with glioma induced cognitive impairment with 8.2%, 4.6%, and 5.3% in the first, second, and fifth years after treatment, respectively. 15 Animal experimental studies have also shown that whole-body 56Fe ion exposure with 2 Gy induced oxidative stress and behavioral deficits in young adult male Kunming mice 1 month after irradiation. 16,17 Most of the behavioral tests and investigation of neurogenesis were done in short periods after irradiation, which may be different from the long-term effect as radiation-induced inflammation, apoptosis, and neurodegeneration are progressive. In the present study, we did different behavioral tests from 2 months after radiation exposure.
Materials and Methods
A total of 30 BALB/C mice were used for this study. For acute irradiation, mice (n = 10, 5 females and 5 males) at P21 were irradiated with Elekta’s Precise Treatment System (Cyprus) with 2 Gy (dose rate: 1.2 Gy/min). For fractionated irradiation, mice at P21 (n = 10, 5 females and 5 males) were irradiated with 0.2 Gy (1 Gy/min). Mice (n = 10, 5 females and 5 males) at the same age without irradiation were used as the normal control. Animal weight was measured on P21, P90, and P180, and increased weight percentages were calculated on P90 and P180. Behavioural tests including the open field (locomotor testing), novel object recognition, Morris water maze, and fear conditioning were carried out from P69 after irradiation. Efforts were made to minimize animal suffering and to use the minimal number of animals throughout the study. BALB/C mice were provided by the Center for Animal Experiment of Wuhan University (Wuhan, China). All experimental procedures were carried out according to the Guidelines for the Care and Use of Laboratory Animals published by the National Institutes of Health and approved by the Institutional Animal Care and Use Committee of Yangtze University.
Behavioural Tests
Open-field (locomotor) test
Locomotor activity was recorded using the software from Shanghai Xinruan Information Technology Pvt Ltd in a square open-field (50 cm × 50 cm × 40 cm) box. All mice were allowed to explore the field freely for 5 minutes. Activity was measured in terms of total distance travelled. Data were then analyzed by One-way analysis of variance (ANOVA).
Novel object recognition test
Mice were accommodated in the platform for 15 minutes for 2 days. Animals were trained with identical “familiar” objects for 10 minutes on the third day and then assessed with a novel object 24 hours after training. All the tests were video recorded and scored using the software from Shanghai Xinruan Information Technology Pvt Ltd. Preference score was calculated as (time spent with novel object − time spent with familiar object)/total time spent with both objects.
Fear-conditioning test
Mice were placed in a standard conditioning box with a floor consisting of a grid of metal bars (Shanghai Xinruan Information Technology Pvt Ltd). Animals were allowed to explore the box for 2 minutes and followed by a tone (86 dB, 1 kHz) which was sounded for 20 seconds. The last 2 seconds of the tone overlapped with exposure to footshock. The footshock consisted of a pulsating 0.7 mA, 50 Hz current. Mice were tested at 24 hours after training for both cued and contextual fear memories. For contextual fear memory, mice were exposed to the same context (same test chamber) for 280 seconds. For cued fear memory, the animals were exposed to a different test chamber for up to 280 minutes, during which the tone was sounded for 2 minutes. The movement of the animal was monitored. The time spent immobile was recorded using FreezeScan (Cleversys, Reston, Virginia). The percentage of time spent freezing was calculated as time spent freezing (seconds)/total duration of the time period (seconds) and was compared between the control and the experimental mice.
Water Maze Test
Water maze test was carried out using SuperMaze animal behaviour record and analysis system (Shanghai Xinruan Information Technology Pvt Ltd) in a quiet room with dim light and constant temperature environment. The circular water tank with a diameter of 120 cm was filled with water to 20 cm height. The white edible pigment (nontoxic) was added to the water to make it opaque. The water temperature was kept at 23°C (1°C). Different visual clues were hung around the device to help the animals determine the azimuth. The hidden platform was placed in the middle of the fourth quadrant, about 1.5 cm below the water surface, and the position of the platform remained unchanged throughout the experiment. During the training trial, the animals were placed in the start positions in the centre of the 3 quadrants that did not contain the platform. The time needed for the animal to find and climb up the platform, that is, escape latency, was recorded. If the animal could not find the hidden platform within 60 seconds, escape was assisted by the experimenter. In the first 5 days, the animals were put in the water from the first, second, third, or fourth quadrant, and the trial was repeated 4 times a day. The escape latency of the animals in water was recorded. During the test trial on the sixth day, the platform was removed, and mice were released in the water at 1 of the 3 different starting positions, facing the wall, and were required to use visual cues to navigate to the platform. The escape latency and the number of crossing the platform were recorded.
Immunohistochemical Staining
At 159 days after irradiation, animals were anaesthetized with 1% pentobarbital sodium at 0.1 mL/10 g and perfused with 4% paraformaldehyde. The brain was removed, postfixed overnight, and then transferred to 30% sucrose in 0.1 mol/L phosphate buffer (pH: 7.4). Sagittal brain sections were then cut at 40 μm and processed by immunohistochemistry to investigate the radiation-induced changes in neurogenesis using cell or neurogenesis markers such as Ki67 or doublecortin (DCX) and interneuron marker parvalbumin (PV).
For immunohistochemistry, serial sections were transferred to 0.1 mol/L phosphate buffer (pH: 7.4) in 3 different wells of a 24-well tissue culture dish. For the immunocytochemical study, free-floating sections were treated with 3% H2O2 for 10 minutes and blocked with 4% normal goat serum (for Ki67 and PV) or 2% donkey serum (for DCX) for 2 hours at room temperature. The sections were then incubated with primary rabbit antibodies for Ki67 (1:200; Gene Tex, Hsinchu City, Taiwan) and PV (1:4000; Swant, Switzerland) or goat antibodies for DCX and NeuroD (Santa Cruz Biotechnology Inc, California) in 0.1 mol/L phosphate buffer saline (PBS) with 0.1% Triton X-100 (PBS–TX) overnight. The sections were then washed in PBS–TX and placed in biotinylated goat anti-rabbit or horse anti-goat secondary antibodies for 1 hour. After 2 washes in PBS–TX, the sections were placed in avidin–biotin complex reagent (Vector Laboratories Inc, Burlingame, California) in PBS–TX for 1 hour and then washed in PBS-TX and reacted in DAB peroxidase substrate (Vector Laboratories Inc) for 10 minutes. After immunostaining, the sections were mounted, counterstained with hematoxylin, and then covered with a coverslip.
Statistical Analysis
Animal weight change was monitored at postirradiation days 69 and 159 after acute irradiation and days 69 and 159 after first fractionated irradiation. The weight gain percentage was calculated according to weight at age of 12 or 24 weeks/weight at age 3 weeks. One-way ANOVAs followed by Student t-test were done to compare the differences among animals with different treatments.
For counting of DCX, Ki67, and PV-labeled immunopositive cells in the subgranular zone (SGZ), 8 sections of hippocampus from each animal were used. The mean value of the number of immunopositive cells in each section was calculated. All the data were then analyzed by One-way ANOVA followed by Student t-test. Statistical significance was considered at P < .05. All quantitative data were indicated as the mean+/- standard deviation (SD).
Results
Animal Weight Gain
Acute (2 Gy × 1) and fractionated (2 Gy × 10) irradiation with a total dose of 2 Gy did not induce animal death or mortality. There was also no significant difference in animal weight gain percentage among the control, acute, and fractionated radiation exposure groups at postirradiation days 69 and 159 (Figure 1).

Acute irradiation with 2 Gy at postnatal day 21 (P21) or fractionated irradiation with 0.2 Gy × 10 from P21 to P30 does not affect animal weight gain percent among the normal control without irradiation and acute and fractionated radiation exposure groups at postirradiation day 69 (12-week-old) and day 159 (24-week-old; P > .05).
Open-Field (Locomotor) Test
Both acute (2 Gy) and fractionated (0.2 Gy × 10) irradiation did not induce any difference in the distance traveled among the 3 groups of animals (Figure 2).

Acute irradiation with 2 Gy at postnatal day 21 (P21) or fractionated irradiation with 0.2 Gy × 10 from P21 to P30 does not induce any significant difference in the total distance animal traveled within 5 minutes in the open-field test at the age of 12 weeks when compared to the normal control without irradiation (P > .05).
Novel Object Recognition Test
The acute and fractionated irradiation with a total dose of 2 Gy did not induce a significant main effect on preference score (Figure 3).

Acute irradiation with 2 Gy at postnatal day 21 (P21) or fractionated irradiation at 0.2 Gy × 10 from P21 to P30 do not induce any significant difference for the preference score in the novel object recognition test (P > .05) when compared to the control.
Fear Conditioning Test
Neither acute nor fractionated irradiation with 2 Gy or 0.2 Gy × 10, respectively, affected contextual and cued fear memories (Figure 4).
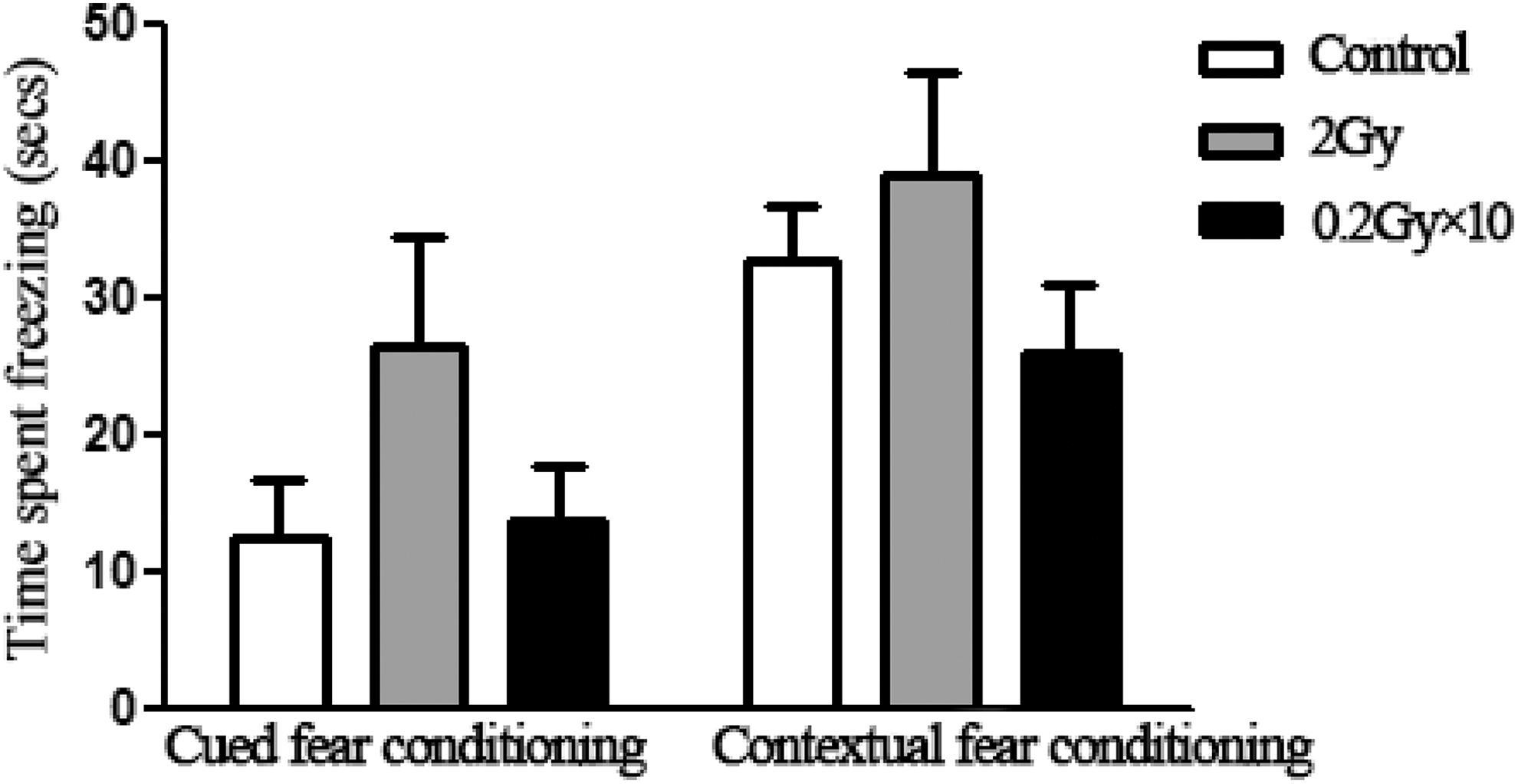
In the cued or contextual fear conditioning test, the percent of time spent freezing does not show significant difference among the control and acutely and fractionatedly irradiated mice (P > .05).
Water Maze Test
Compared to the normal control group, both acute and fractionated irradiation did not induce spatial learning (Figure 5A) and memory (Figure 5B) impairment.

Morris Water Maze test indicates that the escape latency of the control and acutely and fractionatedly irradiated mice reduces with training from day 1 to day 5. There is no significant difference for the escape latency among 3 groups of mice (P > .05, A). In the space exploration experiment, the number of times that the animals crossed the platform within 60 seconds is also similar among the 3 groups of mice (P > .05, B).
Brain Neurogenesis
Ki67 Immunohistochemistry
Immunohistochemical study of cell division markers Ki67 indicated that compared to the control (Figure 6A), both acute (Figure 6B) and fractionated (Figure 6C) irradiation, induced a significant loss of dividing cells in SGZ (Figure 6D). Acute irradiation induced much more loss of Ki67 cells compared to the fractionated irradiation (Figure 6D).

Ki67 immunohistochemistry shows that compared to the control (A), acute (B; P < .01) or fractionated (C; P < .05) irradiation induced significant loss of dividing cells in the subgranular zone (SGZ) (D). Acute irradiation induced much more loss of Ki67 cells compared to the fractionated irradiation (D; P < .05). *P < .05: 0.2 Gy × 10 group versus the control group; **P < .01: 2 Gy group versus the control group; # P < .05: 0.2 Gy × 10 group versus 2 Gy group.
DCX Immunohistochemistry
The DCX immunohistochemistry demonstrated that compared to the control (Figure 7A), both acute (Figure 7B) and fractionated (Figure 7C) irradiation induced impairment of neurogenesis in SGZ (Figure 7D). Much less newly generated neurons were found after acute irradiation compared to the fractionated exposure (Figure 7D).
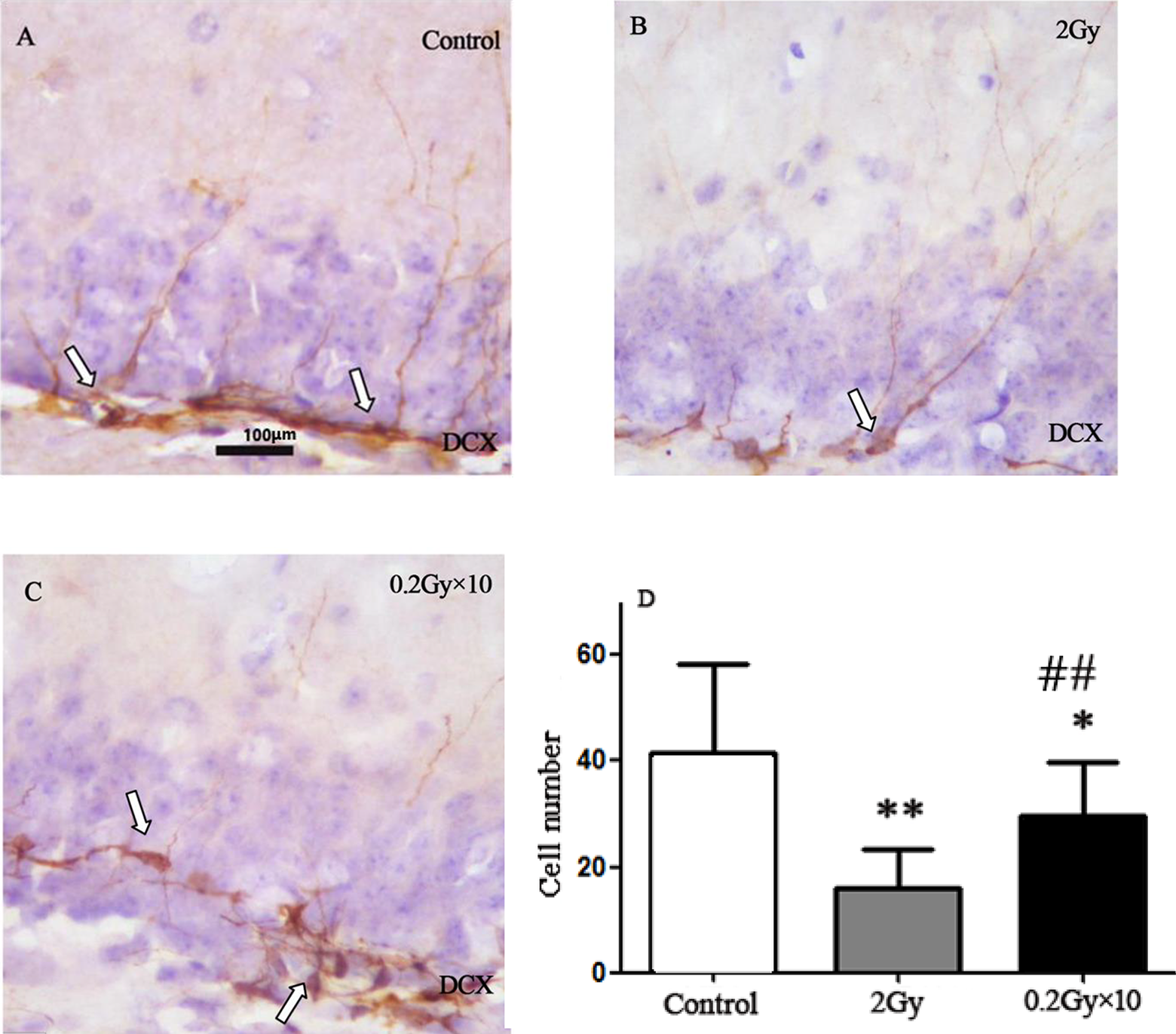
DCX immunohistochemistry demonstrates that compared to the control (A), acute (B; P < .01) or fractionated (C; P < .05) irradiation induces impairment of neurogenesis in SGZ (D). Acute irradiation causes more severe impairment of neurogenesis compared to the fractionated exposure (D; P < .01). *P < .05: 0.2 Gy × 10 group versus the control group; **P < .01: 2 Gy group versus the control group; ## P < .01: 0.2 Gy × 10 group versus 2 Gy group. DCX indicates doublecortin; SGZ, subgranular zone.
PV Immunohistochemistry
The PV immunohistochemistry showed that compared to the control (Figure 8A), both acute (Figure 8B) and fractionated (Figure 8C) irradiation induced a significant loss of PV immunopositive interneurons in SGZ (Figure 8D). Much less PV immunopositive interneurons were found after acute irradiation compared to the fractionated exposure (Figure 8D).

PV immunohistochemistry shows that compared to the control (A), acute (B; P < .01) and fractionated (C; P < .05) irradiation induces a significant loss of PV immunopositive interneurons in SGZ (D). Much fewer PV immunopositive interneurons are found after the acute irradiation compared to the fractionated exposure (D; P < .05). *P < .05: 0.2 Gy × 10 group versus the control group; **P < .01: 2 Gy group versus the control group; # P < .05: 0.2 Gy × 10 group versus 2 Gy group. PV indicates parvalbumin; SGZ, subgranular zone.
Discussions
Main Findings
Immunohistochemical study indicated that acute and fractionated irradiation with a total dose of 2 Gy at P21 BALB/c mice induced impairment of cell proliferation and neurogenesis in the SGZ labeled by Ki67 and DCX, respectively. The number of PV immunopositive interneurons in the SGZ was also significantly reduced. However, the 2 patterns of irradiation did not significantly affect animal weight gain at days 69 and 159 after irradiation. Behavioral tests did not show a significant difference in animal motor activity, novel object recognition, spatial memory, and contextual and cued fear memories.
Effect of Different Patterns of Whole-Body Irradiation on Subgranular Neurogenesis in the Adolescent Mouse Brain
In clinical practice, radiotherapy with fractionated low-dose irradiation could reduce high-dose radiation-induced side effects. However, animal experiments suggest that radiation effect may vary according to the total radiation dose, dose rate, radiation source, pattern of irradiation, age, species and strain of irradiated individuals, and the end point for examination of the radiation effect. For instance, with a total dose of 5 Gy, acute and fractionated γ-rays irradiation of adult C57BL/6 mice with different patterns of exposures (ie, 1 Gy × 5 times and 0.5 Gy × 10 times, 1.1 Gy/min/wk) reduced neurogenesis when examined at 6 months after irradiation. The number of newly generated neurons was relatively lower in the acute than the fractionated irradiation group, hence suggesting that the hippocampal neurogenesis was more susceptible to the acute than fractionated irradiation. 9 However, fractionated exposure with a total dose of 10 Gy (5 fractions of 2 Gy) was more effective in reducing neurogenesis in the DG than an acute irradiation (a single dose of 10 Gy). Moreover, a fractionated but not the acute exposure reduced the step-through latency suggesting a damaged inhibitory avoidance memory. 18 The authors attributed the worse effect of fractionated radiation exposure than acute irradiation to combined anesthesia, avoidance test, and radiation. Similarly, acute irradiation of C57/Bl6 mice with a total dose of 0.5 Gy of X-rays did not affect neurogenesis when examined 2 hours after irradiation. However, fractionated irradiation with the same total dose of 0.5 Gy (0.05 Gy of X-rays per day for 10 days) caused impairment of neurogenesis in the DG when compared to the control mice. 2 This effect may be attributed to the perturbations in extracellular signal-regulated kinases 1/2, protein kinase B (AKT), and cyclic adenosine monophosphate-responsive element binding. While acute irradiation with 0.5 Gy of X-rays did not affect neurogenesis when examined 2 hours after irradiation, 2 the same dose induced a significant increase in apoptotic cells and decrease in neurogenesis when examined 12 hours after irradiation of male ICR mice. 19 Further study indicated that within 2 weeks after acute irradiation at 0.5 and 2 Gy, neurogenesis returned to the level of the normal control mice, thereby suggesting that reduced neurogenesis in the DG might be transient and reversible after irradiation at doses of ≤2 Gy. In Nestin-CreER(T2)/R26R-YFP transgenic mice, single and fractionated exposures of 56Fe-particle irradiation with acute 1 Gy or 5 fractionated 0.2 Gy (0.2 Gy × 5) were similarly detrimental to adult-generated neurons, neither acute nor fractionated exposure decreased the number of adult neural stem cells when compared to the sham exposure. 20 In the present study, both acute (2 Gy) and fractionated (0.2 Gy × 10) irradiation significantly reduced cell proliferation and newly generated neurons as indicated by Ki67 and DCX, respectively. The number of PV immunopositive interneurons was also reduced. We showed consistently that acute irradiation induced much more cell loss than the acute exposure. It suggests that fractionated irradiation with 0.2 Gy × 10 does mitigate acute irradiation-induced impairment of neurogenesis.
Cognitive Impairment After Whole-Body Irradiation of Adolescent Mouse
Cranial radiotherapy of childhood cancer induces functional deficits in memory, attention, and executive function that severely affect the patient’s quality of whole life. 21 -24 Extensive animal experimental data support the clinical findings. For instance, acute irradiation (at radiation doses ranging from 1 to 20 Gy) of immature mice at P10 (6 Gy to the brain), 25 P11 (8 Gy to the left hemisphere), 26 P14 (8 Gy to the brain), 27 P21 (5 Gy to the brain), 28 and P30 (20 Gy to the rat brain) 29 induced hippocampus-dependent memory deficit and increased anxiety levels when animals were tested at mature ages. Acute (at dose of ≥2 Gy) 19 or fractionated (a total of 20-45 Gy) 24,30 -33 irradiation of young or aging adult rodents also induces significant memory deficits. Unfortunately, the radiation doses used in the most of previous studies are more than 2 Gy.
With radiation doses less than 2 Gy, that is, 0.5 and 1 Gy, acute radiation exposure to P3, P10, or P19 mice caused functional defects at adult age in mice exposed at the 2 earlier time points (P3 and P10), while no alteration in behavior was evident when mice were exposed at P19. This suggests that P3 to P10 may be a specifically vulnerable period to radiation-induced neurotoxicity in mice. 34,35 The dose of 350 mGy has been considered a threshold to induce cognitive impairment. However, irradiation of periadolescent Wistar rats at 6 to 7 weeks of age with a lower dose of 0.2 Gy of high-linear energy transfer particles induced persistent deficits in hippocampal-dependent spatial learning. 36 Mice exposed to 0.1 and 0.25 Gy of 16O also failed to distinguish the novel arm, spending approximately the same amount of time in all 3 arms during the retention trial during Y-maze testing. 37 At cellular and molecular levels, irradiation with doses of 0.1 and 2 Gy in the postnatal ages of day 10 or week 10 may induce specific alterations in hippocampal neurogenesis, microvascular density, and mitochondrial functions depending on ages at irradiation. 38
In the present study in P21 mice, while acute (2 Gy × 1) and fractionated (0.2 Gy × 10) irradiation stages of animal life on induced impairment of neurogenesis in the SGZ of the DG, behavioral tests did not show any abnormal changes in the novel object recognition, spatial memory, and cued or contextual fear memory. It suggests that with a total dose of 2 Gy, the severity of impairment of neurogenesis after both acute and fractionated radiation exposure may not be bad enough to cause any cognitive impairment. The organism’s compensative mechanism may also play a role to recover radiation-induced brain functional changes. The possibility that the cognitive impairment may occur at the later early stages of animal life instead of about 2 months after irradiation in the present could not be excluded. Previous studies have suggested that loss of hippocampal interneurons including PV immunopositive cells may underlie behavior deficits. 39 -43 In the present study, although a significant loss of PV immunopositive interneurons occurs after both acute and fractionated radiation exposure, no cognitive deficits were detected by different behavioral tests. It remains to be confirmed whether there is a causal relationship between the loss of interneurons, in particular, PV immunopositive cells and cognitive deficits.
Comparing radiosensitivity between the human and the mouse is critically important in the clinical–experimental neuroradioembryological research. Specifically, according to Bond et al 44 and Prasad, 45 the average half-lethal dose (LD50/30, radiation dose under which 50% of exposed animals die within 1 month) in the human is 3 or 2.7 to 3 Gy, while in the mouse, the LD50/30 is 6.4 or 8.5 to 9 Gy, respectively. There is a coefficient of 2 to 3 between the human and the mouse. Therefore, the human equivalent dose of 2 Gy for acute and 0.2 Gy for fractionated radiation exposure for mice will be 0.67 to 1 Gy or 0.067 to 0.1 Gy, respectively. The latter range falls to the low radiation dose range of ≤100 mGy. Further study with long-term monitoring of animal behavioral changes is still needed to clarify whether radiation exposure with a total dose of 2 Gy by either acute or fractionated radiation exposure will not induce any neuropsychological disorders.
Footnotes
Authors’ Note
Shuang Peng, Bo Yang, and Meng Yun Duan are the co-first authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by grants from the National Natural Science Foundation of China (no. 81772223) (RBX) and the National Research Foundation of Singapore to Singapore Nuclear Research and Safety Initiative (TFR).
