Abstract
Earlier studies have shown that combination of antibodies to S100 protein and to cannabinoid receptor type 1 in released-active form (Brizantin) may possess anxiolytic properties and decrease nicotine dependence. Released-active form of antibodies is a novel approach that permits to modify natural functions of the target molecule (antigen) under investigation. The aim of the present study was to evaluate the anxiolytic-like effect of Brizantin in the light–dark test in mice, according to its ability to influence the number of entries into the lit compartment and the total time spent there. Three doses of Brizantin (2.5, 5, and 10 mL/kg) were compared with diazepam (1 mg/kg), placebo, and vehicle control. Anxiolytic-like effect of the tested drug was shown to be dose dependent, with an increasing trend from 2.5 to 10 mL/kg. Brizantin in its highest dose significantly increased studied behavioral parameters, although its effect was less pronounced than that of the reference drug diazepam (1 mg/kg).
Keywords
Introduction
Comorbidity between smoking and emotional disorders is one of the important public health concerns 1 considering continuous efforts to reduce nicotine addiction and prevalence of cigarette smoking in various studied populations and risk groups. 2,3 Several meta-analyses and systematic reviews have been published confirming a strong bidirectional association between cigarette smoking and anxiety disorders. 4 –6 These findings point out that assessment of emotional status may require special attention in order to achieve a successful smoking cessation. 1 Multiple pathways can underlie the link between nicotine dependence and anxiety symptoms and disorders; up to date, there are several, albeit alternative explanations of this association. It can be underpinned by shared vulnerability factors, which predispose toward smoking and development of anxiety disorders. 1,7 –10 Under certain conditions, nicotine exerts anxiolytic effect and smoking appears to be a compensatory behavior to relieve anxiety and distress, 1,10 –13 especially in individuals with emotional vulnerabilities. 1 The long-term nicotine exposure affects various brain structures, pathways, and neurotransmitter systems that are linked to the pathology of anxiety disorders. 10
Brizantin is a novel drug developed for smoking cessation, which possesses anxiolytic properties. 14,15 Its active ingredient is a combination of released-active form of antibodies to S100 protein (RAF of Abs to S100) and to cannabinoid receptor 1 (RAF of Abs to CB1), produced according to the previously patented technology. 16 Released-activity phenomenon was first described and studied in late 1990s, 17 –20 and since that time it was successfully used in the development of several medicinal products. In vivo and in vitro studies have successfully demonstrated that antibodies in released-active form are able not to neutralize their target, but to modify natural functions of the antigen and to change interaction of molecule (antigen) with its respective target by the mechanisms of conformation modification. 17 –23 Although the technology used in the manufacturing process of released-active drugs is consistent with routine procedures used in production of homeopathic medicines (ultra-high dilutions), there are important differences between them. 24
RAF of Abs to S100 (the ingredient of Brizantin) previously demonstrated anxiolytic properties confirmed in a number of clinical and preclinical investigations. 21,25 –29 As antibodies in released-active form are able to modulate the activity of an antigen, the effects mentioned above may be considered in light of numerous functions of S100 protein in the nervous system. 30 –33 Also in vivo and in vitro studies have shown interactions of RAF of Abs to S100 with γ-aminobutyric acid (GABA), 34 –36 serotoninergic 36,37 and dopaminergic system, 36 and glutamate and sigma receptors. 36
Cannabinoid receptor type 1 (CB1) antagonists were suggested to have a therapeutic potential in the treatment of drug dependence and, particularly, nicotine dependence. Previously published preclinical rodent studies showed that observed decrease in nicotine self-administration and nicotine-seeking behavior was attributed to administration of a CB1 antagonist rimonabant. 38 Randomized multicenter clinical trials 39,40 revealed modest effectiveness of rimonabant in achieving smoking cessation. Added benefits were noted in control of withdrawal, including cravings and mitigation of postcessation obesity in study subjects, when rimonabant was used together with a nicotine patch. 41 Late-stage clinical development of CB1 antagonists (rimonabant, Acomplia and Zimulti) was put on hold after clinically reported adverse events, including nausea, drug–drug interaction in HIV-afflicted individuals, and infectious complications of the upper respiratory tract. 42 –44 Nonetheless, research suggests that the endocannabinoid system is tightly involved in mediating nicotine effects and nicotine addiction. 45 –47
It may be a promising approach to combine RAF of Abs to S100 and RAF of Abs to CB1 to simultaneously alleviate both nicotine cravings and anxiety/agitation. The ability of antibodies in released-active form to reduce adverse or toxic effects attributed to the target molecule would also be an advantage as it may help to avoid undesirable clinical side effects of CB1 antagonists that caused their premature market exit in the past. Our novel drug Brizantin, which contains combination of RAF of Abs to S100 and RAF of Abs to CB1, is in advanced preclinical development. Up-to-date experiments showed that Brizantin alleviated nicotine dependence in rats 14 and also possessed anxiolytic and antidepressant properties in animal models of anxiety and depression. 15
The aim of the present study was to determine the dose dependence of anxiolytic-like effect of Brizantin in a simple animal test. For this purpose, light–dark test was chosen as a common protocol allowing reproducible assessment of relative anxiety status of mice. 48,49 This test is based on a conflict between the innate aversion of rodents to brightly illuminated areas and their spontaneous exploratory behavior. Mice instinctively tend to prefer dark and enclosed spaces. The time spent in lit and dark compartments during the test and respective transitions between compartments can be indicative of anxiolytic-like or anxiogenic-like activity attributed to tested compounds. 48,49
Methods
Animals
Experiments were carried out on 90 male CD-1 mice (body weight 26-34 g, 4-5 weeks old at the start of experiments), supplied by Janvier Labs (Le Genest-Saint-Isle, France). Animals were housed in groups of 8 in Eurostandard type III cage (425 mm × 266 mm × 155 mm; Tecniplast, Decines-Charpieu, France) with Abedd Espe Midi (Sniff, Soest, Germany) as a bedding material. The animals were maintained in a room with controlled temperature (21°C-22°C), the relative humidity of 30% to 70%, and a reversed light–dark cycle (12 hours/12 hours; lights on: 17:30-05:30; lights off: 05:30-17:30) with food and water available ad libitum. The experimental procedures and animal housing conditions corresponded to the regulations established by the European Convention for the Protection of Vertebrate Animals (Strasbourg, 1986) used for Experimental and other Scientific Purposes and the Guide for the Care and Use of Laboratory Animals. 50
The animals were acclimated in an isolated room of the animal facility for at least 3 days prior to dosing. During this period, the animals were exposed to daily visual and clinical examinations before their respective assignment to groups.
At the end of experimentation, animals were subjected to euthanasia by CO2 overdose. This procedure was performed by the responsible person (animal care assistant) according to the instructional guide of the euthanasia device manufacturer (CO2 euthanasia automat; Tem Sega, Pessac, France).
The study was performed in compliance with the European Directive 2010/63/EU. All procedures performed on the animals in the course of the study were reviewed and approved by the Animal Ethics Committee CEEA35 (Project 120.02, March 11, 2015).
Light–Dark Test
The apparatus consists of 2 polyvinylchloride boxes (19 cm × 19 cm × 15 cm) covered with Plexiglas (Bioseb, France). One of the boxes is darkened, and the other is transparent and illuminated. An opaque plastic tunnel (5 cm × 7 cm × 10 cm) separates the dark box from the illuminated one. Mouse was placed individually into the lit box, with the head directed toward the tunnel. The total number of entries and the time spent in the lit box over a 5-minute period were recorded by video tracking system (Viewpoint, Lissieu, France). As the total number of animals was big (n = 90), tests were performed in 4 consecutive days. Each day, 3 to 4 mice from each group were tested to ensure the uniformity of testing conditions in all groups.
Compounds
Four test samples were manufactured by OOO “NPF “Materia Medica Holding” (Moscow, Russian Federation) and supplied as ready-to-use water solutions. The samples were coded by manufacturer and tested blindly. One sample contained placebo, 3 other samples contained active pharmaceutical ingredient of Brizantin: nondiluted, diluted in purified water 1:1 (vol/vol), and diluted in purified water 1:3 (vol/vol). That was done to carry out a blind experiment with placebo (10 mL/kg) and 3 different doses of Brizantin (10, 5, and 2.5 mL/kg corresponding to the dilution level) in the same administration volume of 10 mL/kg.
Active pharmaceutical ingredient of Brizantin is composed of RAF of Abs to S100 and RAF of Abs to CB1 (1:1). Affinity-purified rabbit polyclonal antibodies to S100 protein and affinity-purified rabbit polyclonal antibodies to CB1 were manufactured in accordance with the requirements of current Good Manufacturing Practice for drug substances 51 by AB Biotechnology (Edinburgh, United Kingdom), licensed by Medicines and Healthcare Products Regulatory Agency contract GMP manufacturing facility, and were used as a starting substance.
Active pharmaceutical ingredient of Brizantin was produced from the antibody substances (2.5 mg/mL) according to the technology described in the US patent 8 535 664. 16 Briefly, the concentration of the starting substance was reduced by consecutive dilutions under specific conditions in either water–ethanol or water solutions with either 12, 30, or 200 defined steps of centesimal dilution (ie, reduction of their initial concentration by 1024 times at least). The final solutions contained mixture of 12, 30, and 200 centesimal dilutions of antibodies to S100 and CB1. In case of the placebo, purified water was used as starting substance and underwent the same technological steps. All dilutions were prepared in glass vials under sterile conditions and controlled environment, avoiding direct intense light, and were stored at room temperature.
Diazepam (Valium; Roche, batch F1107F01, Boulogne-Billancourt, France) was dissolved in saline (0.9% NaCl) to prepare a solution of 0.1 mg/mL. Saline (0.9% NaCl) was used as a vehicle control. All substances were stored in a dry, dark, controlled access area at the room temperature.
Dose–Response Experiment
Mice were randomly assigned to one of the 6 experimental groups (15 animals per group) according to their body weight. Brizantin at 3 different doses (2.5, 5, and 10 mL/kg) and placebo (10 mL/kg) were administered per os once daily for 5 consecutive days, last day—60 minutes prior to testing. The reference compound diazepam at an optimal therapeutic dose of 1 mg/kg 52 and vehicle were administered per os acutely 60 minutes prior to testing. All treatments were given from 9:00 to 12:00 via oral gavage at the same volume of 10 mL/kg. A 5-day administration regimen of Brizantin was chosen for the study based on the previous experimental data of its component RAF of Abs to S100, which was not effective at acute administration in the Vogel test and elevated plus-maze test in rats, but had a pronounced effect after a 5-day oral treatment. 29
Statistical Analyses
Statistical differences between studied groups were assessed using analysis of variance. The individual who analyzed the data was blinded to sample description. Post hoc Tukey honestly significant difference test was used for multiple comparisons. Dose–response effect was assessed with generalized linear model (negative binomial regression) on log–log scale. The P value ≤.05 was considered as statistically significant. All statistical tests were performed with the use of R version 3.4.2 software. 53
Results
No mortality or abnormal behavior was noted in any of the studied groups, and all animals were included into the final data analysis. Diazepam, administered acutely 60 minutes before commencing the light/dark test, clearly increased the time spent in the lit box and the number of entries into the lit box as compared to the respective control group (P < .05 vs vehicle; Table 1). Brizantin given per os for 5 days dose dependently changed the behavior of mice in the light/dark test as compared with their respective control—placebo (Table 2). The lowest dose of the test drug (2.5 mL/kg) was no different from placebo control. The middle dose (5 mL/kg) demonstrated a tendency to increase the number of entries into the lit box, but not the time spent there, when compared with placebo control. Only high dose of Brizantin (10 mL/kg) was able to significantly increase the number of entries into the lit box as compared with the placebo group (by 37.2%, P < .05). Brizantin in the high dose did not significantly change the time spent in the lit box, but had a tendency to increase it (by 23.4%). Diazepam significantly increased locomotion as compared with the vehicle control (Table 1), whereas Brizantin, at 10 mL/kg dose, was only moderately able to affect this parameter (Table 2).
Effect of Diazepam (Acute per os Administration) on the Light–Dark Test Parameters in Mice Compared With the Respective Control (Vehicle).a
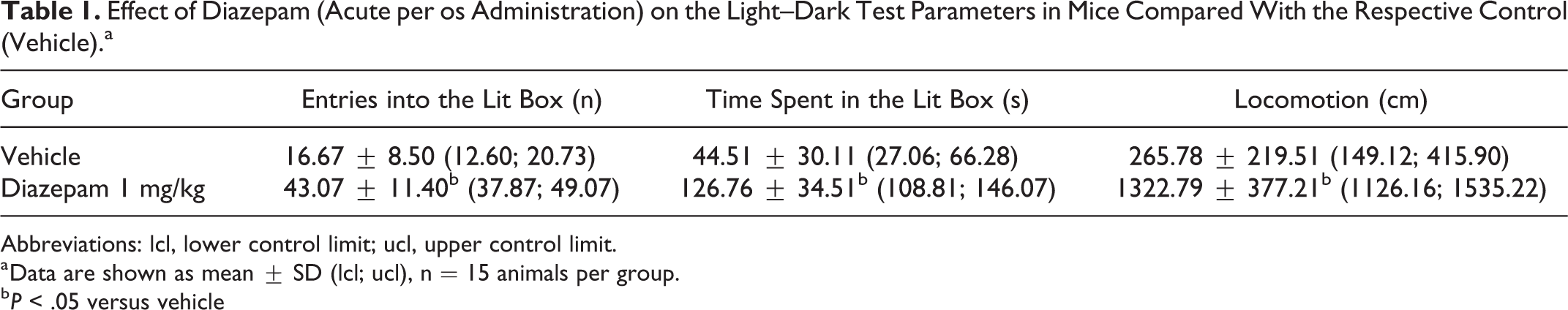
Abbreviations: lcl, lower control limit; ucl, upper control limit.
a Data are shown as mean ± SD (lcl; ucl), n = 15 animals per group.
b P < .05 versus vehicle
Effect of Brizantin (5 Days per os Administration) on the Light–Dark Test Parameters in Mice Compared With the Respective Control (Placebo).a

a Data are shown as mean ± SD (lcl; ucl), n = 15 animals per group.
b P < .05 versus placebo.
c P < .05 versus Brizantin 2.5 mL/kg.
Regression analysis was used to assess a dose–response relationship, based on the number of entries into the lit box being the most sensitive light/dark parameter attributed to the effect of Brizantin. The results of linear regression model (Table 3) confirmed the visualized data (Figure 1). The model showed statistically significant slope of the linear regression line (3.54 ± 0.08, F 1,43 = 19.31, pseudo R 2 = 0.30, P < .05) relating the natural logarithm of Brizantin dose to the effect. This finding supports the evidence of the dose–response relationship between the dose of Brizantin and its anxiolytic-like effect.
Generalized Linear Model (Negative Binomial Regression) of Brizantin Dose and the Number of Entries Into the Lit Box.a,b

Abbreviations: Constant, y-intercept; SE, standard error.
a Model showed statistically significant slope on log(Dose), which suggests a dose–response effect.
b F 1,43 = 19.31, pseudo R 2 = 0.30.

The effect of Brizantin (5 days per os administration) in 3 studied doses on the number of entries into the lit box in light–dark test in mice. Data are shown as mean ± standard deviation, n = 15 animals per group. The dose–response curve was plotted as y = e3.540 + 0.449×log(x).
Discussion
Results of the present study demonstrated anxiolytic-like properties of Brizantin in the light–dark test in mice. The dose–response relationship revealed during testing was shown to be linear, with significant effect of the higher dose (10 mL/kg) compared to placebo. It was also demonstrated that our chosen high dose was somewhat less effective than the 1 mg/kg reference dose of diazepam (both compounds were compared with their respective controls). Results obtained in the present study support potential benefit of Brizantin and its component RAF of Abs to S100 in animal models of anxiety-like behavior. 15,28,29 RAF of Abs to S100 most likely exerts influences on GABA and serotoninergic systems, involved in anxiety. Blockade of GABAA receptors attenuated anxiolytic-like activity of the drug 34 and, in contrast, blockade of GABAB receptors enhanced it. 35 RAF of Abs to S100 did not affect radioligand binding to GABAA receptors and inhibited radioligand binding to GABAB receptors acting as GABAB antagonist in the functional in vitro assay. 36 The role of GABAA system in anxiety has long been established, and now a spectrum of new studies support the role of GABAB system in stress and anxiety. 54 As for the serotonin system, blockade of 5-hydroxytryptamine (5-HT)2 receptors resulted in reduced anxiolytic-like activity of RAF of Abs to S100, 37 while binding of specific radioligands to 5-HT1 and 5-HT2 receptors was found elevated in the presence of the drug. 36 In the functional assay, RAF of Abs to S100 acted as an agonist of 5-HT1A and an antagonist of 5-HT1B receptors. Activation of 5-HT1A and inhibition of 5-HT1B receptors is considered to be a promising avenue in the treatment of clinical anxiety and depression. 55 –57 Stimulation of 5-HT2 receptors was reported to produce serotonin-selective reuptake inhibitor-like effects in behavioral and neurogenic models 58 and to manifest in anxiolytic-like activity. 59
Elucidation of the dose–response relationships of released-active drugs remains a topic of considerable interest for our current ongoing research. In previous experiments done with RAF of Abs to S100, noted dose–response characteristics were ambiguous and depended on the type of the test used. 29 In elevated plus-maze test in rats, we protocoled a linear dose–response pattern, with the highest dose used being the most effective (5 < 7.5 < 10 mL/kg). In the Vogel conflict test, RAF of Abs to S100 increased punished drinking in rats at 5 mL/kg (P > .05) and 7.5 mL/kg (P < .05), but not at 2.5 and 10 mL/kg, which allowed to assume an inverted U-shaped dose–response relationship. In vitro RAF of Abs to S100 significantly inhibited specific binding of radioligand to the native human sigma-1 receptors in a dose-dependent manner. 36
Different dose–response relationships were revealed in experiments conducted with other released-active drugs: RAF of Abs to NO synthase (NOS) and RAF of Abs to interferon-γ (IFN-γ). RAF of Abs to NOS was able to improve sexual incentive motivation in old Fisher 344 male rats when administered at 3 mL/kg, but not at 1 mL/kg, 60 while in young Wistar male rats, effective dose was 3 mL/kg, but not 9 mL/kg. 61 The complexity of the experimental procedure limited the number of doses tested in those studies. Despite differences in their respective designs, both experimental studies, when viewed side by side, suggested a nonlinear dose–response relationship. RAF of Abs to IFN-γ demonstrated a dose-dependent activity in the murine model of influenza. 62 Both survival and mean life span of infected mice rose with the increment of the RAF of Abs to IFN-γ dose from 0.13 to 0.4 mL/mouse/day, but further increment of the dose to 0.8 mL/mouse/day did not offer any significant advantage. Thus, in different studies, both linear and nonlinear dose–response relationships of released-active drugs were shown.
Application of antibodies in order to block pharmacological effects of nicotine is a new approach for treatment of nicotine addiction and for relapse prevention. Immunotherapeutic prevention of nicotine access to brain seems promising because it potentially allows not only to block reinforcing effects of nicotine but also to avoid adverse effects, which were reported for other smoking cessation medications. 63,64 To date, several nicotine vaccines are under development; however, their efficacy in clinical trials was reported to be relatively low. 65 The effect of nicotine antibodies depends on their serum concentrations and on their ability to induce strong and uniform nicotine-specific immune response; to ensure this, a wide array of strategies are being evaluated. 66,67 Brizantin, containing antibodies-derivative active ingredient, suggests one more approach to immunotherapy of nicotine addiction. 14 The dose–response relationship revealed in the present study could also be an important step for selecting an effective dose of the drug in future research. The highest tested dose of Brizantin versus placebo demonstrated efficacy during the study; however, Brizantin’s anxiolytic properties in tested doses were less pronounced, when compared to the reference drug diazepam.
Conclusions
The present study identified a dose-dependent anxiolytic-like effect of Brizantin in the light–dark test in mice. This observation is in line with prior evidence concerning anxiolytic potential of Brizantin and its component RAF of Abs to S100.
Footnotes
Authors’ Note
The study was conducted by Neurofit SAS, France.
Acknowledgments
The authors acknowledge Etienne Poiraud and Bertrand Huyard for their technical expertise in animal experiments; Evgeniy Gorbunov and Aleksandra Emelyanova for scientific discussions during preparation of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E. V. Kardash, I. A. Ertuzun, G. R. Khakimova, A. N. Kolyadin, and S. A. Tarasov are employees of OOO “NPF “Materia Medica Holding”. O. I. Epstein is founder of OOO “NPF “Materia Medica Holding”. OOO “NPF “Materia Medica Holding” sponsored the study, performed statistical analysis, made a decision to publish the work, and covered the current article processing charges. Brizantin is a commercial drug produced and marked by OOO “NPF “Materia Medica Holding”. Patents on Brizantin belong to O. I. Epstein.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by OOO “NPF “Materia Medica Holding”, Moscow, Russia.
