Abstract
In hypertension, the combination therapy is frequently used to obtain a better therapeutic effect and reduce adverse effects. One effective combination is with inhibitors and β-blockers of renin–angiotensin system. Although the mechanisms of action of each drug are already known, the antihypertensive mechanism is more complex and therefore the combined treatment mechanism is unclear. Specifically, the effect of the treatments of angiotensin-converting enzyme inhibitor or AT1 receptor antagonist with β-blocker on the angiotensin II and bradykinin reactivity has not been studied. For this reason, we evaluated the interaction between propranolol and captopril or losartan on vascular reactivity to bradykinin and angiotensin II in spontaneously hypertensive rat. We constructed concentration–response curves to angiotensin II and bradykinin after treatment of SHR with propranolol–captopril or propranolol–losartan by using rat aortic rings. While losartan or captopril with propranolol potentiated bradykinin-induced vasodilation effect, the propranolol–losartan interaction decreased the angiotensin II-induced vasoconstriction. In addition, the combinations did not reduce the heart rate significantly. These results suggest that the combined therapy decreased blood pressure to normotensive values and showed less effect for angiotensin II and greater effect for bradykinin than monotherapy which could contribute in the antihypertensive effect.
Introduction
Several societies of cardiology and hypertension have proposed the guidelines for management, diagnosis, and treatment of hypertension. 1,2 These guidelines indicate no preference for specific drugs, so any first-line can be used. Some of these treatments involve the initial use of angiotensin-converting enzyme inhibitors (ACEIs), AT1 receptor antagonists (ARAs), calcium blockers, diuretics, or β-blockers. 2 In hypertension, drug combinations mentioned above are recommended if the treatment with a first-line drug does not comply with an adequate control of blood pressure (BP). 1,2 In this sense, the use of these combinations allows to decrease the individual dose of each drug as well as the reduction of adverse effects and improves the life quality. 3 These treatments induced a hypotensive effect and diminished the probability of heart failure. 4 However, there are combinations that are not effective, for example, the use of ARA with ACEI, 5 and diltiazem or verapamil with β-blockers. 6 The recommended combinations that have shown efficacy include the use of β-blockers with an ARA or an ACEI, but its mechanism has been insufficiently studied at molecular and physiological levels. 2
Although the drug action mechanisms mentioned above have been described, the antihypertensive mechanism is more complex. Indeed, the use of ACEIs is associated with an ACE reduction and ACE2 increase in expression and the production of Ang (1-7) and bradykinin, and all these mechanisms contribute to antihypertensive effect. 7 –9 The effect of AT1 blockage is related to the reduction in ACE and the increment in ACE2 expression. 10,11 The β-adrenergic blockage is associated with a renin reduction, which diminishes the angiotensin I (Ang-I) levels. 12 All these mechanisms could conduce to an improved Ang-II or bradykinin vascular reactivity.
In this sense, when a combined treatment is used for hypertension, the interaction mechanism is even more complex. Specifically, the effect of the combined treatment with propranolol and losartan or propranolol and captopril to Ang-II and bradykinin reactivity in hypertension is unknown. For this reason, the aim of this work was to evaluate the effect of such combinations on BP and vascular reactivity to Ang-II and bradykinin in aortic ring of SHR.
Methods
Animals
Thirty-six male spontaneously hypertensive rats (SHRs) aged 6 to 8 months (157.83 ± 2.043 and 120.44 ± 1.76 mm Hg, systolic blood pressure [SBP] and diastolic blood pressure [DBP], respectively) and 6 Wistar Kyoto (WKY) rats (119 ± 1.33 and 80.46 ± 0.4 mm Hg, SBP and DBP, respectively) were obtained from Cinvestav-IPN, Sede Sur and maintained in the FES Cuautitlan, UNAM. All animal procedures were conducted according to the Federal Regulation for Animal Experimentation and Care (SAGARPA, NOM-062-ZOO-1999, Mexico). In all animals, the BP was evaluated noninvasively using a Medical Equipment Services (SPEM) apparatus, and the data were collected using SIEVART 1 software (from the Instituto Nacional de Cardiología “Ignacio Chavez,” Mexico). The animals were randomly distributed in 7 groups: WKY vehicle (n = 6), SHR vehicle (n = 6), SHR propranolol (n = 6), SHR captopril (n = 6), SHR losartan (n = 6), SHR propranolol–captopril (n = 6), and SHR propranolol–losartan (n = 6).
Reagents
Captopril, losartan, propranolol, Ang-II, and bradykinin were obtained from Sigma-Aldrich (St Louis, Missouri). Captopril, losartan, and propranolol were dissolved in physiological saline solution, and the concentration of each was adjusted to correspond with the dose administered (in milligram per kilogram) in a final volume of 0.1 mL. Ang-II and bradykinin were prepared the same day of experimentation; they were dissolved in a Krebs-Henseleit solution and kept at 4°C to avoid degradation.
Isolated Organ Bath Experiments
The thoracic aorta was quickly excised and placed in a freshly prepared Krebs-Henseleit solution containing 118 mM NaCl, 4.7 mM KCl, 1.2 mM KH2PO4, 1.2 mM MgSO4-7H2O, 2.5 mM CaCl2-2H2O, 25 mM NaHCO3, 11.7 mM dextrose, and 0.026 mM calcium disodium EDTA bubbled with 95% O2/5% CO2. The aorta was cleaned of surrounding tissues and cut into rings of 4 to 5 mm long. The aortic rings were mounted in 10 mL isolated tissue baths filled with Krebs-Henseleit solution at 37°C (pH 7.4) and were continuously aerated with 95% O2/5% CO2. To record the development of semi-isometric force, the rings were suspended on 2 wire hooks (Nicrom wire, Quiminet, Mexico City), and one was fixed at the bottom of the chamber and the another to a force transducer (BIOPAC TSD105; BIOPAC systems Inc, Santa Barbara, California) connected to a BIOPAC MP100WSW system (from BIOPAC Systems Inc). The aortic rings were adjusted to a tension of 3 g. The response to 10−6 M phenylephrine was recorded every 20 minutes until the maximal reactivity was observed, which usually occurred after 1 hour. The integrity of the endothelium was evaluated by measuring the vasodilator response after the administration of 10−5 M acetylcholine to the aortic rings that had been pretreated with 10−6 M phenylephrine.
Evaluation of Antihypertensive Effect Induced by Treatments
The SHRs (n = 30) were treated with propranolol (2.5 mg/kg/d/im), losartan (1 mg/kg/d/im), captopril (1 mg/kg/d/im), propranolol–captopril (1.15 and 0.7 mg/kg/d/im, respectively), or propranolol–losartan (1.69 and 0.53 mg/kg/d/im, respectively), and the BP was measured noninvasively at the end of the treatment. The treatment was administered each day at the same time for 5 days. The individual treatments selected reduced the BP to normotensive values and were calculated through dose–effect curves using SHR, and the combination doses were calculated through isobologram method. 13 The results were compared with those obtained in the normotensive and hypertensive vehicle groups.
Evaluation of Vasoconstrictor Effect on Ang-II
Concentration–response curves of Ang-II (10−10 to 10−6 M) were obtained using the rat aortic rings with intact endothelium obtained from SHR previously treated with the vehicle, propranolol, losartan, captopril, propranolol–captopril, or propranolol–losartan.
Evaluation of Vasodilator Effect on Bradykinin
Concentration–response curves of bradykinin (10−10 to 10−7 M) were obtained using the rat aortic rings with endothelium previously precontracted with phenylephrine (10−8 M) from SHR treated with the vehicle, propranolol, losartan, captopril, propranolol–captopril, or propranolol–losartan.
Statistical Analysis
Data were expressed as the mean ± standard error (SEM) of N = 12 rings from 6 rats; the differences were analyzed using 2-way analysis of variance followed by Tukey test. In all cases,
Results
Systolic Blood Pressure, DBP, and Heart Rate Values in Normotensive and SHR With and Without Treatment
Table 1 shows the SBP, DBP, and heart rate values from normotensive and hypertensive rats with and without captopril, losartan, propranolol, propranolol–captopril, propranolol–losartan treatments. By comparing with the SHR vehicle group, it is found that all treatments reduced significantly the SBP and DBP to normotensive values (
Systolic and Diastolic Blood Pressure and Heart Rate Values Before and After Pharmacological Treatment.a

Abbreviations: DBP, diastolic blood pressure; HR, heart rate; SBP, systolic blood pressure; SHR, spontaneously hypertensive rat.
aResults are shown as mean values SEM from n = 12 and 6 rats.
b
Effects of Captopril, Propranolol, Losartan, Propranolol–Captopril, and Propranolol–Losartan on Contraction Induced by Ang-II
Figures 1 and 2 show the effect of several treatments on the vascular contraction induced by Ang-II, which produced a concentration-dependent contraction in aortic rings in both WKY rats and SHR, and the response was greater in SHR than in WKY. Table 2 shows the Emax (Maximum effect) values increased but not the EC50 (Effective Concentration 50) values; these results are associated with the curves of Figures 1 and 2. The propranolol and its combination with captopril increased the Ang II contractibility
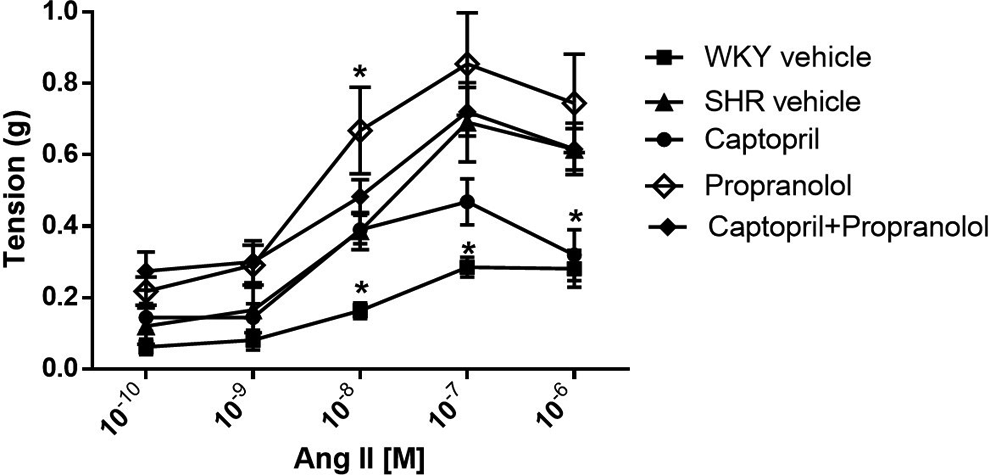
Cumulative concentration–response curve to Ang-II in aorta from rats treated with captopril (•), propranolol (◊), the captopril–propranolol interaction (♦), Wistar Kyoto (WKY) vehicle (▪), and SHR vehicle (▴). The results are shown as mean values SEM from n = 12 and 6 rats. *

Cumulative concentration–response curve to Ang-II in rat aorta from rats treated with losartan (•), propranolol (◊), the losartan–propranolol interaction (♦), Wistar Kyoto (WKY) vehicle (▪), and SHR vehicle (▴). The results are shown as mean values ± EE from n = 12 and 6 rats. *
The Emax and EC50 Values by Ang-II and Bradykinin With Different Pharmacological Treatments.a

Abbreviations: Ang-II, angiotensin II; BK, bradykinin; SHR, spontaneously hypertensive rat.
aThe results are shown as mean values ± EE from n = 12 and 6 rats.
Effects of Captopril, Propranolol, Losartan, Propranolol–Captopril, and Propranolol–Losartan on the Vasodilation Induced by Bradykinin
Figures 3 and 4 show the effect of several treatments on the vasodilation induced by bradykinin. From figures it can be seen that bradykinin induced a concentration-dependent vasodilator response in combined treatment, while the individual treatments did not modify such relaxation. The values of Emax and EC50 for all groups are shown in the Table 2.

Cumulative concentration–response curve to bradykinin in aortic rings precontracted with phenylephrine (10−8 M) from rats treated with captopril (•), propranolol (◊), the captopril–propranolol interaction (♦), and SHR vehicle (▴). The results are shown as mean values ± EE from n = 12 and 6 rats. *

Cumulative concentration–response curve to bradykinin in aortic rings precontracted with phenylephrine (10−8 M) from rats treated with losartan (•), propranolol (◊), the losartan–propranolol interaction (♦), and SHR vehicle (▴). The results are shown as mean values ± EE from n = 12 and 6 rats. *
Discussion
Results indicate that combination therapies were effective because of reduction in hypertension. The mechanism of the antihypertensive effect involved not only a reduction in reactivity to Ang-II but also an increased reactivity to bradykinin. Interestingly, the combination therapies avoid the bradycardic effect induced by propranolol.
Hypertension is a chronic disease with high prevalence and mortality in the world; this is considered as the main risk factor for other illness such as heart disease and stroke. 15 Hypertension in human and experimental models is characterized by structural and functional changes. 16 For example, the dysfunction of renin–angiotensin system promotes an increase in the vascular reactivity to Ang-II, AT1 receptor expression, and ACE activity and a decrease in bradykinin activity 17 ; in smooth muscle cells, the CaMKII has a role in Ang-II-dependent small artery remodeling. 18 The antihypertensive therapy could reverse these changes. 19 In this sense, it has been seen that ACEI as captopril or a new thiomorpholine compound decreases ACE messenger RNA and reactivity of Ang-II in SHR to levels obtained in normotensive rat; 20 propranolol treatment reduces the renin 21 and ACE levels in infantile hemangioma; 22 losartan, an ARA, attenuates cardiac oxidative stress induced by Ang-II. 23 According to these evidence, our data obtained corroborated the results mentioned above, due to the fact that captopril, losartan, and propranolol reduced the BP values (Table 1) and Ang-II induced reactivity only with the 2 first while propranolol increased it (Figures 1 and 2). This effect could be related to the results obtained by Vittorio et al, where the treatment with metoprolol and carvedilol produced an increase in BP in response to Ang-II infusion. 24 In relation to the combination therapy, the antihypertensive mechanisms are not sufficiently studied. In this way, through synergist mechanism, the use of interactions decreased the BP by using lower doses of captopril and propranolol, 25 these results were corroborated by us with this combination. However, there is little evidence about the rational mechanism that supports its use. For example, nebivolol, a third-generation β1 adrenergic blocker, offers an additional BP reduction when it was administered in a combination therapy or as an add-on drug to the ongoing therapy with ACEI or ARA. 26 There are few studies that describe the use of propranolol in combination with cilazapril or captopril, and results show that both combinations reduced SBP, DBP, and plasmatic renin activity. 27,28
One of the mechanisms that could explain the antihypertensive effect is through changes in the vascular reactivity. In this sense, it has been found that in mesenteric bed, the renin–angiotensin system increases the norepinephrine release in SHR, and β2 adrenergic receptors are involved in this effect, promoting an increase in vascular reactivity through perfusion pressure augment. According to these results, the use of propranolol and captopril inhibited the norepinephrine release and reduced the perfusion pressure, the BP, and the vascular reactivity in SHR by diminishing the Ang-II level. 29 However, it has been found that the addition of propranolol does not increase the antihypertensive effect of captopril in the combination, as in the interaction of captopril with a diuretic, but it prolongs the action of ACEI, suggesting a pharmacokinetic interaction. 30 However, and in agreement with our results, it has described that treatment with captopril and a β-blocker is not very effective and might be associated with little change in the vascular reactivity to Ang-II in SHR. 31 In addition, in patients with primary hypertension, the propranolol treatment might be associated with reduction in heart rate, 32 and captopril has shown a positive chronotropic effect by using isolated dog heart. 33 In our study and in a similar way, propranolol showed the bradycardic effect which was avoided by the combination with captopril (Table 1).
The antihypertensive effect of losartan is by blocking the AT1 receptor, reducing the Ang-II efficacy, 34 suggesting that AT1 receptor downregulation could contribute to antihypertensive effect. In agreement with these results and by using human tubular cell line (HK-2), Ang-II was able to upregulate ACE and downregulate ACE2, which were blocked by losartan but not by an AT2 receptor antagonist. 35 With respect to interaction with β-blocker and ARA, there is evidence in which an additive effect on BP reduction was described; such effect was only observed with the third-generation β-blocker but not with the first or second one. 36 In this sense, it has been described in hypertensive patients that valsartan in combination with nebivolol increased the antihypertensive efficacy. 37 These results are in agree with our results based in that losartan–propranolol induced to lower doses an antihypertensive effect than that used as single treatment (Figure 2). However, this combination did not modify the response to Ang-II induced by losartan.
In addition, it has been described that hypertension is associated with reduction in vasodilator mechanisms. For example, it has been described that acetylcholine-stimulated cyclic guanosine monophosphate levels, and vasodilator activity are diminished in the aorta of SHR which was attributed to endothelial Nitric oxide (NO) reduction. 38 In relation to bradykinin, Ognibene et al found that the kinin-induced vasodilation in mesenteric bed was lesser in old SHRs than in young SHRs. 39 This difference is attributed to endothelial dysfunction. Similar results were obtained in our study by using aorta from old SHRs and only the treatment with captopril–propranolol and losartan–propranolol increased the relaxation, indicating that endothelial function improves with combination but not with monotherapy (Figures 3 and 4). However, there is evidence that propranolol increases the kinin synthesis, 40 and the captopril treatment produces an increase in the bradykinin levels avoiding its biotransformation through ACE. 41 Additionally, ACE inhibitors could increase the B2 receptor signal pathway 42 and the release or action of NO 43 by facilitating the prostacyclin synthesis. 44 Finally, in the case of losartan, AT1 antagonism promoted an increase in AT2 signaling, which in turn increased the bradykinin activity. 45 It is important to mention that the bradykinin effect amplifies the knowledge about the possible antihypertensive mechanism of the combined treatments which had not been reported.
In conclusion, the results suggest that the combinations between propranolol and captopril or losartan reduce BP through the participation of ACE inhibition, AT1 receptor blockade, bradykinin, and B2 receptor stimulation, which in turn explain the minor vasoconstriction induced by Ang-II in hypertension.
Footnotes
Acknowledgments
The authors would like to thank C. Ramon Martinez-Gomez and Juan Martinez-Parente from CINVESTAV Instituto Politecnico Nacional for the experimental animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by grants from DGAPA PAPIIT IN224310-3, IN212213-3; UNAM PIAPI-1645 FES Cuautitlán, Universidad Nacional Autónoma de México, and the Secretaría de Investigación y Posgrado (SIP: 20140913), Comisión de Operación y Fomento de Actividades Académicas, and the Programa Institucional de Estímulo al Desempeño de los Investigadores del Instituto Politécnico Nacional.
