Abstract
Purpose:
This study aimed to investigate radiation-induced lesions on the skin in an experimental animal model.
Results:
Infiltrate inflammatory process was observed in D5 and D10, for the 40 Gy and 60 Gy groups, and a progressive increase of transforming growth factor β1 is associated with this process. It could also be noted a mischaracterization of collagen fibers at the high-dose groups.
Conclusion:
It was observed that the lesions caused by ionizing radiation in rats were very similar to radiodermatitis in patients under radiotherapy treatment.
Advances in Knowledge:
This study is important to develop strategies to prevent radiation-induced skin reactions.
Introduction
Skin is one of the most commonly affected organs in radiotherapy treatment. 1 Radiation therapy can cause various histological skin changes such as an acute inflammatory response with edema, dermatitis, desquamation, ulceration, late radiation-induced fibrosis, necrosis, and secondary tumors such as sarcoma, basal, and squamous cell carcinomas. The appearance of these lesions depends, among other causes, on the radiation dose deposited in the skin, and on biological factors inherent to the patient. 2 –4
In general, normal skin is composed of approximately 75% of collagen fibers. Type 1 collagen makes up a considerable part of the extracellular fibers, responsible for the tensile strength of the skin. When there is an inadequate increase in the number of these fibers, the skin remodeling is impaired, resulting in fibrosis. 5 –7 Fibrosis is a process characterized by proliferation and differentiation of fibroblasts, besides higher synthesis of collagen, proteoglycans, and other extracellular matrix (ECM) components. 8 –10
Radiation-induced fibrosis is a multistage process, maintained through different cell interactions, especially endothelial cells, macrophages, and fibroblasts. In a few hours after irradiation, these cells begin to express several growth factors such as platelet-derived growth factor, interleukin 1, transforming growth factor β1 (TGF-β1), and tumor necrosis factor α. 7,10,11 Transforming growth factor β1 cytokine triggers the process, by inducing the proliferation of fibroblast, 12,13 collagen, and other components of the ECM synthesis. 14,15
Although the skin damage induced by radiation is well known, the effects of ionizing radiation on the skin have been poorly explored in the literature. In the present study, we have evaluated the irradiated skin remodeling process, through the quantification of collagen fibers, TGF-β1, and the analysis of the ECM structure.
Methods and Materials
Animals
Male Wistar rats aged 3 months (n = 60), weighing approximately 300 g were kept in appropriate cages with 4 animals each, on a 12-hour light/dark cycle with food and water provided ad libitum. Animals were randomly divided into 4 groups according to radiation exposure in gray (Gy): control (C), 10 Gy, 40 Gy, and 60 Gy. The study was carried out according to the Guide for the Care and Use of Laboratory Animals (Department of Health and Human Services [DHHS], public No. [National Institutes of Health] 85-23, revised in 1996, Office of Science and Health Reports, Bethesda, Maryland), and all experiments were reviewed and approved by the ethics committee for Animal Research of the Rio de Janeiro State University.
Irradiation and Euthanasia
Animals were anesthetized by intraperitoneal injection using xylazine/ketamine (1 mL/100 g), shaved on the back, and irradiated, with single–dose electron beams of 10, 40, or 60 Gy. A Clinac 2100 Varian linear accelerator (Palo Alto, California) with 4 MeV of nominal energy and dose rate of 240 cGy/min was used, the distance between the rats and the irradiation source was 50 cm. The irradiation field was 3 × 3 cm2 located in the shaved region, and a bolus of 0.5 cm was used in order to deposit the maximum percentage depth dose in the skin. We didn’t need to protect the unshaved region, as we used multileaf collimators to define the radiation field. After irradiation, the animals were assessed daily, and the wounds were recorded by a digital camera Nikon Coolpix L1 (Melville, New York). Phenotypic scores of animals were done according to Randall and Coggle 16 at the indicated time after irradiation, rats were evaluated for a skin reaction according to a phenotypic scale (1: normal, 2: hair loss, 3: erythema, 4: dry desquamation, 5: <30% moist desquamation, and 6: >30% moist desquamation). Hair loss was not evaluated because of previous rats depilation, in order to minimize beam interaction with nonskin structures. On 5, 10, and 25 days after irradiation (D5, D10, and D25), rats were euthanized, and the wound area was excised up to the subcutaneous layer.
Light Microscopy
The excised wounds (epidermis, dermis, and hypodermis) were fixed in 10% neutral buffered formalin for 8 hours and transferred to 70% ethanol before paraffin embedding and sectioning (5 µm sections through the center of the wound). Hematoxylin and eosin (H&E)-stained sections were analyzed using an Olympus BX51 (Shinjuku, Tokyo, Japan) light microscope and the images captured by an Olympus DP-72 camera, using its own software. To analyze the collagen content and the architecture of irradiated skin, deparaffinized sections were stained for 1 hour with a 0.1% solution of sirius red F3BA (Chroma Gesellschaft, Munster, Germany) in saturated aqueous picric acid, washed in 0.01 N HCl and viewed under polarized light.
Immunohistochemistry
Immunolocalization of extracellular TGF-β1, using the Santa Cruz Biotechnology (Santa Cruz, California) antibody TGF-β1–rabbit polyclonal immunoglobulin G (IgG; 200 μg/mL), was performed as described elsewhere. Briefly, deparaffinized sections were blocked using tris-buffered saline and 10% rabbit serum. Then, it was incubated overnight at 4°C with the primary antibody (14 μg/mL) in blocking buffer. Sections were washed, incubated with biotinylated rabbit antigoat IgG, washed again, incubated with alkaline phosphatase-conjugated streptavidin followed by Vector Red alkaline phosphatase visualization substrate (Vector Laboratories, Burlingame, California), and photographed under bright-field illumination. Negative controls, which included replacing primary antibody with antibody plus blocking peptide or with normal IgG, were performed in parallel assays.
Scanning Electron Microscopy
The skin sections were washed with 0.2 M phosphate-buffered saline (PBS; pH 7.2) and fixed in 0.2 M PBS containing 2.5% glutaraldehyde at 4°C for 24 hours prior to electron microscopic observations. The specimens were washed in PBS (pH 7.2) and in cold distilled water to remove the glutaraldehyde; they were then dehydrated through an ascending acetone series. The fragments were subsequently dried in a critical point dryer (CPD2; Tousimis, Cambridge, Massachusetts), coated with gold in an ion-sputtering apparatus (Sputter Coater 108; Cressington, Watford, United Kingdom), and observed using a scanning electron microscope (1450 VP, LEO Electron Microscopy Ltd, Clifton Road, United Kingdom).
Results
Macroscopic Skin Evaluation
We didn’t observe skin macroscopic alterations, for any group, at the first 24 hours after irradiation. However, in D5-10 Gy, the skin macroscopic evaluation revealed erythema and dry desquamation (Figure 1A). In D10-10 Gy, 4 of the 5 rats presented spontaneous recovery and 1 animal also showed erythema (Figure 1B). On D25-10 Gy, all remaining animals (n = 5) presented their skin completely healed (Figure 1C). In the group irradiated with 40 Gy, initial erythema was observed on D5-40 Gy, progressing to dry desquamation (Figure 1D). On D10-40 Gy, moist desquamation was observed, in up to 30% of the irradiated area (Figure 1E). On D25-40 Gy, some animals showed erythema or dry desquamation, and others were healed (Figure 1F and G). In the group irradiated with 60 Gy, it was observed that the lesions, developed in the irradiated field, showed bleeding points on D5-60 Gy (Figure 1G), sharp, and moist desquamation on D10-60 Gy (Figure 1H) and unhealed skin on D25-60G that resembled various radiological accidents (Figure 1I). Phenotypic scores of animals were evaluated and quantified as shown in the graph (Figure 1J).

Radiation-induced wound. A-C, Wound evolution in animals irradiated with 10 Gy. D-F, 40 Gy. G-I, 60 Gy. J, Phenotypic score of 0, 10, 40, and 60 Gy skin irradiation of rats (values were average of 5 rats in each group).
Light Microscopy Analysis
Hematoxylin and eosin staining
We didn’t observe skin microscopic alterations, for any group, at the day 0 of observation (the day we performed the irradiation). In the 10 Gy group, at all time points analyzed (0, 5, 10, and 25 days after irradiation), there were no changes in tissue structure (Figure 2B–D). In the 40 Gy group, alterations were observed when compared to the control group (Figure 2A). On D5-40 Gy, an intense inflammatory infiltrate composed of many polymorphonuclear cells was observed, demonstrating the acute phase of inflammatory response (Figure 2E), depletion of ECM, and thinning of collagen fibers. On D10-40 Gy, a thickening of collagen fibers in the deeper regions of the reticular dermis (Figure 2F) was observed. On D25-40 Gy, a reduction of the inflammatory infiltrate, regions with thinner fibers, and others with thicker fibers (Figure 2G) were observed. In the 60 Gy group, on D5-60 Gy (Figure 2H), it was seen an inflammatory infiltration with many polymorphonuclear more concentrated in the reticular dermis in the region nearest the papillary dermis (which was lost in the lesion) and a crust at the site of injury, it was also seen the formation of granulation tissue, in addition to regions with thinning of the fibers. On D10-60 Gy (Figure 2I), it was seen the granulation tissue in the area of the lesion, the beginning of fibrosis, and inflammatory cells across the reticular dermis. On D25-60 Gy (Figure 2J), collagen fibers are found thicker, the lesion still persisted, and morphological changes of the skin occurred, indicating the tissue necrosis. The inflammatory infiltrate was still substantial across the reticular dermis.
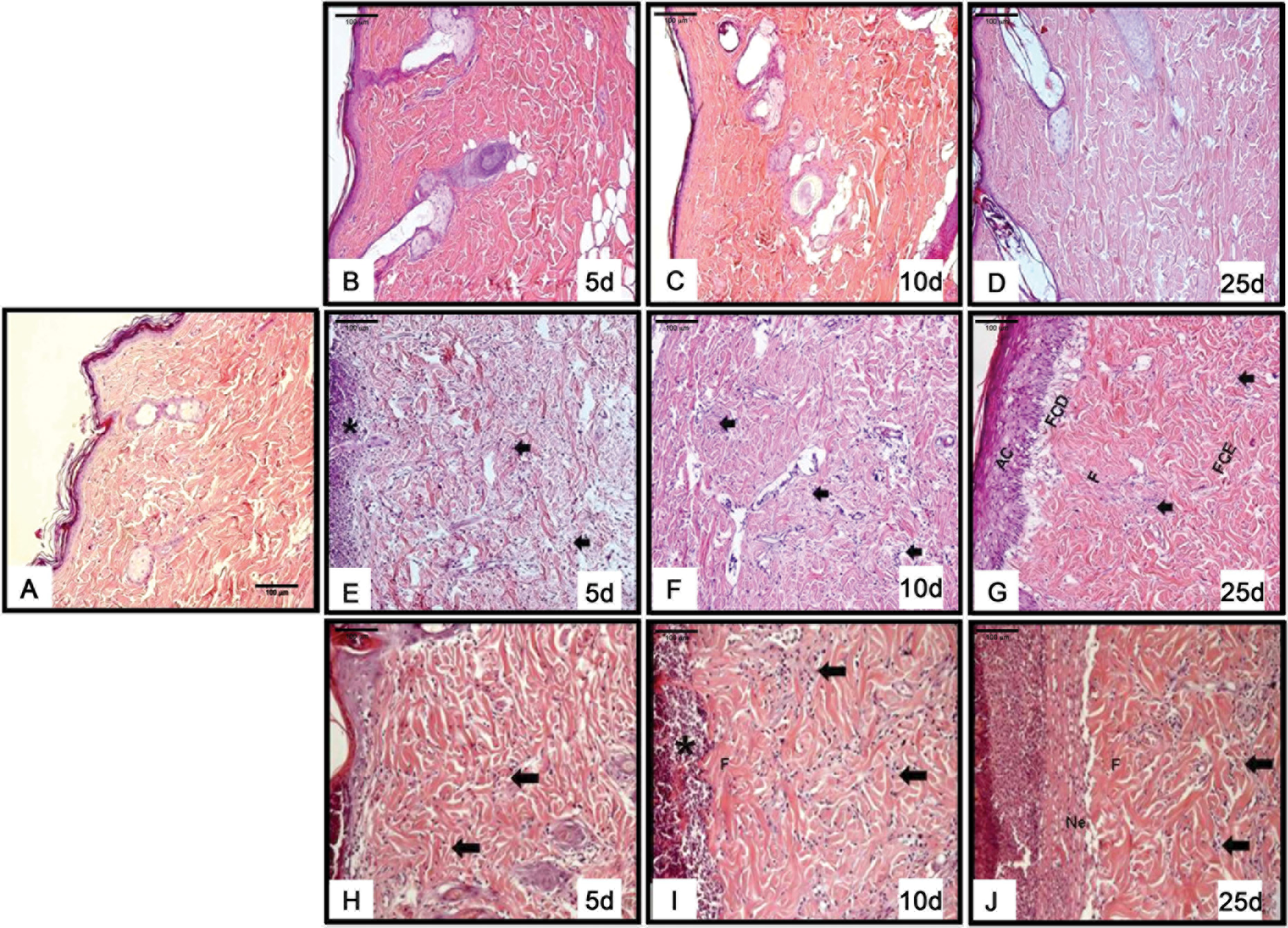
Photomicrographs of histological sections stained with HE. A, Control group. B-D, Animals irradiated with 10 Gy. E-G, Animals irradiated with 40 Gy. H-J, Animals irradiated with 60 Gy. AC indicates acanthosis; arrow, inflammatory infiltrate; asterisk, polymorphonuclear infiltration; F, fibrosis; FCD, thin collagen fibers; FCE, thick collagen fibers; H&E, hematoxylin and eosin; Ne, necrosis.
Picrosirius red staining
In the control group, the observed skin had normal architecture, characterized by fibers with prevalent red birefringence (Figure 3A). In the 10 Gy group, on D5-10 Gy, shrinkage of some collagen fibers in the papillary dermis region was observed (Figure 3B). On D10-10 Gy, the collagen fiber bundles became thicker, in this same region (Figure 3C). On D25-10 Gy, collagen fibers were oriented almost parallel to the epidermis (Figure 3D). In the 40 Gy group, on D5-40 Gy, shortened collagen fibers with a red birefringence (Figure 3E) were observed. On D10-40 Gy, the collagen fibers were more elongated, in the deeper reticular dermis, and shorter in the superficial papillary dermis, both with a yellow birefringence (Figure 3F). On D25-40 Gy, some collagen fibers were shortened and some elongated (Figure 3G). In the 60 Gy group, on D5-60 Gy (Figure 3H), it was observed perpendicular arrangement of collagen fibers in the dermis, in addition to its shortening in some regions. The group D10-60 Gy (Figure 3I) presented collagen birefringence to red which may be the indicative of collagen type 1, and it was also seen the significant shortening of these fibers, and it occurred in almost all reticular dermis. On D25-60 Gy (Figure 3J), the collagen shows a birefringence to yellow, which may indicate that the collagen is type 3, which fibers were shortened, demonstrating that the morphology of the skin is changed and with fibrosis and remodeling characteristics.

Photomicrographs of histological sections stained with picrosirius red. A, Control group. B-D, Animals irradiated with 10 Gy. E-G, Animals irradiated with 40 Gy. H-J, Animals irradiated with 60 Gy.
Qualitative TGF-β1 immunohistochemistry
There was no immunostaining for TGF-β1 in the control group, as expected (Figure 4A). In the 10 Gy group, on D5-10 Gy and D10-10 Gy, it was observed low TGF-β1 expression dispersed on the reticular dermis (Figure 4B and C). There was no TGF-β1 expression on the D25-10 Gy (Figure 4D). In the 40 Gy group, on the D5-40 Gy, the expression was more visible in some regions and dispersed on the reticular dermis (Figure 4E). On D10-40 Gy, the expression was possibly stronger in the reticular dermis, just below the injured area, where some cells expressing this cytokine (Figure 4F) were observed. In the D25-40 Gy, the epidermis cells expressed TGF-β1 in some regions (Figure 4G). In the 60 Gy group, it was observed the onset of TGF-β cytokine expression at D5-60 Gy (Figure 4H), which seemed to be enhanced, in the groups D10-60 Gy and D25-60 Gy (Figure 4I and J). This expression was diffused by the reticular dermis but envisioned to a greater expression in some areas of the papillary dermis and below the damaged area and granulation tissue. Also, this expression can be seen in the hypodermis tissue.

Photomicrographs of histological sections immunostained with anti–TGF-β1 antibody. A, Control group. B-D, Animals irradiated with 10 Gy. E-G, Animals irradiated with 40 Gy. H-J, Animals irradiated with 60 Gy. The arrows and arrow heads indicate the TGF-β1 expression; Ac, acanthosis; F, fibrosis; FCD, thin collagen fibers; TGF, transforming growth factor.
Scanning electron microscopy
In the control group, it was observed a normal distribution of collagen fibers in the skin, where the fibers were arranged in parallel or in vertical, forming a structure similar to a network in beam form (Figure 5A). In assessing the group irradiated with 10 Gy, thin collagen fibers were visualized just below the injury in D5-10 Gy (Figure 5B). The collagen fibers had their morphological features merged on D10-10 Gy and D25-10 Gy (Figure 5C and D). In the group irradiated with 40 Gy, the collagen fibers no longer have their normal morphology in D5-40 Gy (Figure 5E). While in D10-40 Gy and D25-40 Gy, it was observed that some collagen fibers were thin (Figure 5F and G). In the group irradiated with 60 Gy on D5-60 Gy, shorter collagen fibers were seen in some thinner regions (Figure 5H). On D10-60 Gy, collagen fibers no longer exhibit their characteristic morphology (in the form of beams; Figure 5I). On D25-60 Gy, aside an ECM with the morphologically altered collagen fibers without the appearance of beams, it was observed inflammatory infiltrate in the epidermis (Figure 5J).

Electronic scanning micrographs. A, Control group, preserved collagen fibers. B-D, Irradiated animals with 10 Gy. B, 5-day group, showing thin collagen fibers (FCD). C, 10-day group, general appearance of the skin tissue. D, 25-day group, skin tissue with characteristic morphology. E-G, Irradiated animals with 40 Gy. E, 5-day group showing collagen fibers without its characteristic morphology. F, 10-day group where it is observed thin collagen fibers. G, 25-day group showing thin collagen fibers. H-J, Irradiated animals with 60 Gy. H, 5-day group, overall appearance of the skin tissue. I, 10-day group, general appearance of the skin tissue, it is not possible to visualize the characteristic bundles of the collagen fibers. J, 25-day group showing an ECM with collagen fibers and altered inflammatory infiltrate. ECM indicates extracellular matrix; FCD, thin collagen fibers.
Discussion
The skin acts as a physical and biological barrier that may be severely compromised by the exposure to ionizing radiation. 17 –19 Therefore, it is of great importance to characterize the lesions, the inflammatory aspects, and the cytokines involved in the radiation–induced skin reactions. Since there are few literature data describing the evolution of dose skin reactions in animals, it becomes necessary to develop an appropriate strategy to avoid the deleterious effects that can be caused.
In the present study, early progressive macroscopic and microscopic injuries to rat skin were shown with increasing doses of radiation. Macroscopic lesions were similar to burns across irradiated area, while microscopically there were signs of acute inflammation, collagen fibers proliferation, and increased TGF-β1 expression. Spontaneous healing was observed with smaller doses of radiation. Higher doses eventually resulted in strong TGF-β1 expression and necrosis. All phases of tissue repair could be observed during the follow-up period: inflammation, which peaked between D5 and D10, granulation tissue, ECM formation, and remodeling. 14,20 –23 Healing was eventually accompanied by hypertrophy of the epidermis (acanthosis). Scanning electron microscopy corroborated findings of light microscopy.
Previous studies in rodents have shown that 5 weeks after irradiation the skin of some animals presented epidermis ulceration, severe skin inflammation, and corroborating the findings of this study, although in the present study we only followed the lesions evolution for 25 days. Our study, and others, show that the initial radiation-induced lesions are characterized by erythema, dry and wet desquamation, and ulceration resulting from infiltration of inflammatory cells, with possible acanthosis in the deposition of abnormal fibrous matrix. These fibrotic sequelae are an undesired complication of radiotherapy, generating an impact on quality of life of patients. 21,22 It was observed that the lesions caused by ionizing radiation were very similar to radiodermatitis, which often appear in patients treated with radiotherapy and in radiation accident patients. 2 To develop a mild skin radiolesion, the local dose in humans, depending on the radiation energy, should be between 3 and 5 Gy 2 and similar lesions were observed in the group of 10 Gy. In animals irradiated with 40 Gy, acute radiodermatitis was observed, which occurs in humans with absorbed dose from 6 to 12 Gy. 2 The same occurred with the 60 Gy group, where we find the similar lesions that occur in humans with doses 16 to 20 Gy. 2 Therefore, it is suggested that in animal models, using rats, for evaluation of ionizing radiation–induced injury to the skin, dose should be adjusted to be equivalent to that administered to humans. 2 The animal models with rats are widely used for the studies of radiation–induced skin reactions, but it is important to highlight that there are differences among human and rats skin. Similar to humans, the rat skin is also composed of the major layers (epidermis and dermis) of human skin. However, it does not perfectly mimic the human skin architecture because of its unique skin morphology. The skin of rodents is loosely attached to the subcutaneous muscle layer and is covered with thick fur. Anatomically, both epidermis and dermis are thinner than the corresponding structures in human skin. Despite those limitations, the animal model is still the best model to study several human pathologies, including the skin. 23,24
The expression of TGF-β1 suggests that the inflammatory process in the skin results from immune system activation. Transforming growth factor β1 is responsible for the increase and synthesis in ECM production by various cell types, mainly by differentiation fibroblasts in injured tissues, besides collagen proliferation, the radiation-induced alterations changed collagen architecture. Transforming growth factor β1 also inhibits degradation of these processes. 9,14 Exposure to ionizing radiation increases the production of TGF-β1 hours after irradiation, persisting for months. Radiation induces a cascade of inflammatory and profibrotic cytokines that lead to fibrosis. 21 Our results suggest a progressive increase of TGF-β1 associated with higher radiation doses. 25 These data are supported by studies that showed that rats deficient in TGF-β1 production are less susceptible to skin lesions caused by ionizing radiation, such as fibrosis and acanthosis. 20
The persistence of the inflammatory phase occurs mainly by the chemotactic action of TGF-β1, which causes the recruitment of more inflammatory cells. The biological effects of this cytokine are increased by its self-induced production in the ECM cells, in cases of injury. 12,20,21,26,27
The results corroborate data in the literature where the extension of the inflammation phase entails prejudice the proper skin tissue repair process, impairing healing, and lead to greater expression of TGF-β1. 12,28,29 This continued production of TGF-β1 by inflammatory cells recruited in the previous phase is essential for the next phase, the formation of granulation tissue. At this stage, there is an increased synthesis of ECM giving mechanical force to the contraction of the lesion. 30
The obtained results indicate that higher irradiation doses induced an increased expression of the profibrotic cytokine TGF-β1, and a more intense inflammatory response in the skin. The intense expression of this cytokine in irradiated skin can trigger the fibrotic process and, consequently, affect the homeostasis of this organ due to ECM accumulation.
We eventually observed regions of acanthosis, in addition to morphological alterations in irradiated tissue, indicating the occurrence of radiation-induced fibrosis, likely by type 3 collagen as suggested by their yellow birefringence. Gradually this collagen is replaced by collagen type 1, which is stronger. This work displayed collagen fibers thinning at all doses used, it was more pronounced at higher doses owing to the greater damage generated by these doses in irradiated skin. This result may indicate a weakness of the irradiated skin, due to the fact the skin is composed of 75% collagen, besides even be responsible for the firmness and skin integrity. This result displayed is corroborated by the data in the literature. 31,32 Shortening seen in intermediate groups that skin no longer has its normal morphological feature, which may result in the loss of its function and consequently a decrease in their ability to withstand physical shocks, which is displayed in some radiological accidents, where the patient is affected by recurrent lesion days after healing. 33 After complete reepithelialization, the phase of remodeling begins. This phase is characterized by decreased granulation tissue in the injured tissue, as can be observed in groups in higher doses (40 and 60 Gy), on D25. In these groups, a decrease in ECM components is observed, when compared to the control group. 11 Our study also showed a dose–response effect relationship with skin tissue repair. In the 40 and 60 Gy groups, the wounds were more severe with a more pronounced inflammatory infiltrate and most disfigured fibers, causing a delay in healing.
Our results support the need for protection of the skin of patients undergoing radiation therapy and the importance of the development of protection strategies for the skin of these patients.
In conclusion, we have seen that in irradiated rats, the presence of skin tissue repair is found. Higher doses (40-60 Gy) correspond to the most deleterious. The study showed that higher doses induced a significant inflammatory process in the skin, activating the immune system and thereby causing an increase in the expression of TGF-β1 cytokine profibrotic. The extension of the inflammatory phase generated by ionizing radiation affects the skin tissue repair. It occurred thinning of collagen fibers observed in light microscopy and confirmed in scanning electron microscopy.
Footnotes
Authors’ Note
Each author contributed equally to the article. All authors have approved the final article. This work has not been published previously, and it is not under consideration for publication elsewhere. This publication is approved by all authors and tacitly or explicitly by the responsible authorities where the work was carried, and if accepted, it will not be published elsewhere, including electronically in the same form, in English or in any other language, without the written consent of the copyright holder.
Acknowledgments
The authors thank CNEN—Comissão Nacional de Energia Nuclear and CAPES—Coordenação de Aperfeiçoamento de Pessoal de Nível Superior.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
