Abstract
Research on bisphenol A (BPA) as an environmental contaminant has now major regulatory implications toward the ecosystem health, and hence it is incumbent on scientists to do their research to the highest standards possible, in order that the most appropriate decisions are made to mitigate the impacts to aquatic wildlife. However, the contribution given so far appears rather fragmented. The present overview aims to collect available information on the effects of BPA on aquatic vertebrates and invertebrates to provide a general scenario and to suggest future developments toward more comprehensive approaches useful for aquatic species protection.
Effects of BPA on Vertebrate Species
As for many other chemicals, rivers and lakes are the major sinks for bisphenol A (BPA); therefore, the majority of data on the impact of BPA on wildlife come from studies on aquatic vertebrates, fish in particular and, to a minor extent, amphibians, with some available data also in reptiles and birds. Independent of the species, most studies were carried out in controlled settings, while few of them really focused on wild populations. However, since in the aquatic environment, (i) BPA concentrations vary dramatically, (ii) BPA is part of a complex mixture of chemical stressors, and (iii) the different vertebrate classes and species have different sensitivities to xenobiotics, undoubtedly laboratory experiments provide better insights toward the understanding of the mode of action of BPA, its dose response, and its effects on animal physiology. Some particularly sensitive species, tissues, and life stages have been addressed and useful for further investigations. Tests for screening and assessment of BPA toxicity based on fish are also under development in relation to regulatory aspects, aiming to the possible replacement of BPA with more ecocompatible compounds.
Vertebrates as Test Organisms for the Effects of BPA
Overall, BPA is reported to act as a teratogen and endocrine disruptor in vertebrate animals (Flint et al. 2012). Teratogenic effects were found at high (1-10 mg/L range; Iwamuro et al. 2003; Sone et al. 2004), while endocrine and pleiotropic effects have been observed at lower doses (within the µg/L range; Flint et al. 2012), possibly reflecting the effective concentrations present in the environment.
To establish what concentrations of BPA are to be considered as environmentally relevant has been a difficult task. The highest amounts of BPA are reported in landfill leachate and pulp mill effluents (up to 17 mg/L; Flint et al. 2012), while concentrations in the low µg/L range are found in surface waters (Crain et al. 2007; Flint et al. 2012). BPA has been detected in river and marine sediments (eg, 43 and up to 191 g/kg dw; Flint et al. 2012 and Koh et al. 2006, respectively) and at a concentration of about 100 µg/kg dw in soils; however, not enough data are available to discuss the relationships between these concentrations and the real exposure/uptake in different animals. According to a vast collection of data, some authors defined about 12 µg/L or lower as the environmentally relevant concentration in surface waters (Flint et al. 2012). This value may be adopted to discriminate among the effects in aquatic vertebrates described subsequently, in order to acknowledge particular importance to the responses observed at environmental concentrations.
Possible Mechanisms of Action of BPA as an Endocrine Disrupting Chemical in Vertebrates
In comparison to mammals, information on the mechanisms of action of BPA in nonmammalian vertebrates is far behind. Only a few studies were addressed to the interaction of BPA with cellular receptors and signaling. Furthermore, model organisms and experimental designs were different among studies, and dose–response effects were rarely measured. Available evidence suggests that BPA interactions similar to those observed in mammals can be hypothesized; however, since the exposure is more prolonged and routes of exposure to BPA are different, especially in the aquatic environment, more focused investigations are needed.
As a matter of fact, the main route of exposure in fish is not the diet, but inhalation through the gills, and metabolism of BPA via this route is not as efficient as in the liver. Thus, it is not surprising that waterborne BPA consistently produces more relevant estrogenic effects in fish, including the induction of egg yolk protein precursor vitellogenin (vtg) in males (Kang et al. 2007).
The general consensus is that the estrogenic activity of BPA is mediated through its binding to estrogen receptors (ERs) in fish (Gibert et al. 2011) as well as in frogs (Lutz and Kloas, 1999; Suzuki et al. 2004b).
A relative estrogenic activity of BPA, calculated to be about 0.008% of estradiol (E2) activity, was observed in Xenopus hepatocytes (Mitsui et al. 2007). These estimates are within the range of those reported in the hepatocyte vtg assays on fish species (for a review, Navas and Segner, 2006). It was also found that BPA has a significant antagonistic activity on ER (approximately 0.8% of tamoxifene activity). This evidence would also explain why the BPA-ER complex did not cause the postranscriptional enhancement of vtg synthesis in the Xenopus system (Mitsui et al. 2007).
More recent advances were obtained in zebrafish, where it was demonstrated that the orphan nuclear estrogen-related receptor y (ERRy) is the mediator of BPA-induced malformation of the otoliths (Tohmé et al. 2014). By blocking the ERRy function, the effects of BPA on otoliths were abolished, further supporting this hypothesis. According to this evidence, the effects induced by BPA in fish may be wider than anticipated. In particular, the metabolic effects of BPA through ERRy, that in mammals regulates the expression of gluconeogenesis genes, is also implicated in heart and skeletal muscle metabolism and in the control of insulin secretion. If this were the case also for nonmammalian vertebrates needs to be more widely addressed.
Further data suggest that BPA could also act by using mechanisms independent of ER. Waterborne BPA showed antagonism on androgen receptors (ARs) in fish (Ekman et al. 2012). At 10 and 100 µg/L, it reduced the masculinization effect of trembolone, a potent toxicant that binds fish AR (Ankley et al. 2003) and also reduced the effect of trembolone on hepatic metabolome in female fathead minnows, providing evidence for AR antagonism (Ekman et al. 2012).
Based on its structural similarity to thyroid hormones (THs) due to its 2 benzoic rings, BPA has been proposed to act as a TH antagonist or agonist and to cause disruption of the thyroid system (Zoeller et al. 2005; Hiroi et al. 2006; Jung et al. 2007). These data were mainly obtained in models of amphibian metamorphosis. Using larval stages of Xenopus, Iwamuro et al. (2006) found that BPA blocked both spontaneous and TH-induced metamorphosis
However, BPA was shown to be a weak ligand to liver TR in rodents, while being a potent inhibitor of T3 binding to human TH-binding proteins. So, as in mammals, also in other vertebrates, the TH antagonism could be played not at the receptor level, but through the ability of the chemical to suppress TR-mediated transcription and consequently to reduce TR availability (Mathieu-Denoncourt et al. 2014).
On these basis, we can conclude that BPA, besides its estrogenic effects and ability to bind ERRy, possesses anti-TH and antiandrogen hormone activities on aquatic vertebrates.
Mode of Action and Pleiotropic Effects of BPA in Vertebrates: Some Examples in Aquatic Animals
Sex and reproduction
Abnormal sex ratios (up to 11 females: 1 male) were found in zebrafish fries fed with food containing BPA, at 2000 mg /kg, which is an ecologically unrealistic dose. No effects were observed below 500 mg BPA/kg (Drastichova et al. 2005).
Treatment with 22.8 µg/L BPA induced approximately 62% to 70% of females in
Vitellogenin is a large (250-600 kDa mm) calcium-binding phospholipoglycoprotein precursor of egg yolk proteins that is common to all oviparous vertebrates and required for normal oocyte maturation. It is produced by the liver upon estrogen stimulation and released into the blood. The Vtg production is normally restricted to mature females, and little, if any, vtg is normally detected in males or sexually immature females. However, exposure to estrogenic compounds can trigger its expression in males, since they carry the vtg gene (Sumpter and Jobling, 1995). For monitoring purposes, vtg can be measured in the liver, blood, and mucus of male and female fish as well as in primary hepatocyte cultures (Navas and Segner, 2006).
BPA induced synthesis of vtg and expression of vtg messenger RNA (mRNA) in multiple species of fish and amphibians at concentrations ranging from 10 to 2000 μg/L (Hatef et al. 2012; Mihaich et al. 2012; Arukwe et al. 2000; Sohoni et al. 2001; Kloas et al. 1999; Gye and Kim 2005). No increases were observed in goldfish at concentrations below 5 μg/L (Hatef et al. 2012). However, increased vtg levels were measured in the plasma of female and male carp (
The dose–response curves of BPA on vtg expression are available for adult fathead minnows and zebrafish (Villeneuve et al. 2012). Fish were exposed to 0, 0.01, 0.1, 1.0, 10, or 100 μg/L BPA for 96 hours, and the compound significantly increased plasma vtg concentrations in both males and females of fathead minnows, at concentrations higher than 10 μg/L. Moreover, E2 concentrations were significantly decreased in plasma of females, while circulating testosterone levels were significantly reduced in males, consistently with a negative feedback response to an increasing estrogenic signal. Zebrafish appeared less sensitive to BPA, and increases in vtg were observed only in males (at 10 and 100 μg/L BPA), although to a much lower extent than in fathead minnows (Villeneuve et al. 2012).
In the same work, the ovarian transcription profiles for fathead minnows and zebrafish were evaluated (Villeneuve et al. 2012). At the lowest BPA concentrations, enrichment of differentially expressed genes associated with cell cycle control and mitosis was observed only in zebrafish. In fathead minnows, the expression of genes with functions related to intracellular signaling processes was upregulated. Conversely, reduced expression of oocyte growth-related genes, together with genes associated with cholesterol uptake, and genes coding for various matrix metalloproteinases, which play a role in ovulation, was reported in BPA-treated fish versus controls.
Expression of genes related to steroid biosynthesis was induced in fish exposed to the lowest concentrations of BPA compared to controls (Villeneuve et al. 2012). Overall, from this work it emerged that after 4 days of exposure to 0.01 μg/L BPA, approximately 4% and 2% of microarray features for fathead minnow and zebrafish females, respectively, had expression levels significantly different from controls. The level of impact on the ovarian transcriptome declined in the 0.1 and 1.0 μg BPA/L treatments and then increased dramatically at the 10 μg/L, before falling to values similar to controls at 100 μg/L BPA. It is worth noting that the effects observed at 10 μg/L are somehow consistent with the effects observed in vtg or hormone production; however, the changes in microarray features at 100 μg/L BPA are in clear contrast. Such apparent inconsistency reduces the importance of the information, at present. However, toxicogenomics data clearly exemplify an inverted U dose–response pattern to BPA, thus advising for additional studies more specifically focused on the low doses.
At upper levels of the hierarchy of the reproductive axis, a complex network of neuroendocrine systems act coordinately to regulate the reproductive functions. Gonadotropin releasing hormone (GnRH) decapeptides are produced in the hypothalamus and bind to GnRH receptors on the cell surface of gonadotrope cells. Here they stimulate the synthesis and release of the gonadotropins, which in turn act on the gonads to stimulate maturation, gametogenesis, and steroidogenesis. Adult rare minnows
Data from the field highlighted that BPA contributes to intersex conditions. Immature barbels (
Finally, examples of sex disruption by BPA are also provided in aquatic reptiles. In some species, gender is determined by the temperature of egg incubation during a critical window of development. While control embryos of
Gonad functionality
Several reports are available supporting the ability of BPA to affect gonad functionality at environmentally relevant concentrations (<12 µg/L). At about 1 µg/L BPA showed a reduced sperm density and mobility in brown trout,
In the carp
After a 14-day exposure to 10, 100, and 1000 µg/L BPA, the gonads of zebrafish had a normal external appearance; however, multiple alterations in the ovaries with degeneration of the cell components and a significant increase in atresic follicles were induced at 100 and 1000 µg/L BPA (Molina et al. 2013). These data highlight that an exposure to BPA for 2 weeks may be sufficient to affect gonadal function in the zebrafish, although the active concentrations are above the average environmental levels.
Bisphenol A has been shown to affect maturation and quality of eggs and semen in adult brown trout (Lahnsteiner et al. 2005); however, the same species seemed to be relatively insensitive to the exposure concentration in the period from fertilization until larvae swim-up (Bachmann-Bjerregaard et al. 2008). Brown trout embryos and larvae were exposed to BPA at environmentally realistic concentrations (from 1.75 µg/L) from 0 to 63 days postfertilization (dpf) corresponding to the swim stage, and then gonad development was followed up to 400 dpf. No alterations were observed after BPA exposure in these experimental conditions (Bachmann-Bjerregaard et al. 2008).
Fathead minnows exposed to nominal concentrations of 1, 16, 64, 160, and 640 µg/L BPA for 164 days (Mihaich et al. 2013) showed male gonad cell type frequencies significantly different from controls and a slight decrease in spermatocytes at 160 µg/L or higher concentrations. No further changes in growth, gonadosomatic index, or reproduction variables (eg, number of eggs, hatchability) were detected after 164 days (Mihaich et al. 2013).
Development
Acute exposure of rainbow trout eggs to 30 and 100 µg/L BPA for 3 hours prior to fertilization, in order to mimick accumulation of the compound by maternal transfer, did not modify fertilization rate but delayed hatching, yolk reabsorption, larval growth, and first feeding of larvae of about 7 days compared to controls (Aluru et al. 2010). Amounts of 32 and 420 ng BPA/oocyte were detected after a 3-day exposure and they dropped by at least 90% after 13 days postfertilization. Although BPA was cleared from the embryos within 31 days postfertilization, growth suppression persisted in juvenile fish. Moreover, adult developed from BPA-exposed oocytes displayed a disturbed plasma cortisol and glucose profile in response to stressors, suggesting the disruption of the hypothalamus–pituitary–interrenal axis and metabolic disfunctions during early embryogenesis (Aluru et al. 2010).
BPA showed dramatic effects on amphibian metamorphosis. The time of development was delayed, and body length and weight were reduced in tadpoles of
Thyroid system
Transcriptome analyses of TH-regulated genes in zebrafish during the eleutheroembryonic stage (days 2-5 postfertilization) were performed to detect potential markers of thyroid disruption. Bisphenol A (0.1, 0.4, 1, 2, and 4 mg/L) acted as a weak T3 agonist when tested alone, but it strongly enhanced the effect of subsaturating concentrations of T3, with effects at 2 and 4 mg/L (Pelayo et al. 2012). The chemical did not prevent the ability of thyroid follicles to synthesize thyroxine, a landmark for substances able to suppress thyroid gland functions.
Exposure to 200 µg/L BPA accelerated early embryonic development within 24 hours of exposure, attenuated body growth, and advanced the times of hatching and reproductive maturation in the medaka,
Conversely, BPA showed lethality in zebrafish larvae (96 hour-LC50 was 8.04 mg/L) and hatching success for zebrafish embryos was reduced by about 50% after 96 hours of exposure at BPA 5.25 mg/L (Chan and Chan 2012). However, at these high concentrations, the chemical did not change the expression of any of the mRNA related to the thyroid system, for example, thyroglobulin, TH receptors, and transthyretin (Chan and Chan 2012). By using the microarray technology, it was shown that 4 days of premetamorphic
We may note that the above-mentioned effects of BPA were caused at relatively high concentrations, and the different effects and active concentrations found in the different approaches seem to depend on the type of tissue and animal species under investigation. Therefore, whether BPA may affect the animal thyroid system in the natural environment needs to be ascertained.
Metabolism
Human obesity and related disorders have increased dramatically over the past decades, reaching epidemic proportions. The worldwide prevalence of obesity more than doubled between 1980 and 2014 (WHO 2015). Among the suggested causes is the increasing exposure to chemicals (obesogens) that more appropriately are addressed to as metabolic disrupters. This class of compounds affect mainly adipocyte physiology and alter adipocyte mass and energy homeostasis (Regnier and Sargis 2014). A common obesogen target is the peroxisome proliferator activated receptor γ (PPARγ), a nuclear receptor family member which has widely been recognized as a master regulator of adipogenesis. Some evidence for obesogenic effects of BPA has been obtained also in lower vertebrates. Zebrafish embryo exposure to BPA activated PPARγ and induced lipid accumulation as well as late-onset weight gain in juvenile zebrafish (Riu et al. 2014). Recently, a laboratory test for examining the effects potential obesogens taken through the diet and exposure to environmental contaminants has been developed in zebrafish larvae (Tingaud-Sequeira et al. 2011). The assay proved able to evaluate the obesogenic effect of PPARγ agonists and tributyltin (a well-known environmental obesogen), being useful also for BPA. However, the knowledge on the modulation of satiety, energy balance, and their main regulators (eg, leptin, a nonglycosylated peptide target of estrogens with a key role in fish appetite; Salmeron et al. 2015) in nonmammalian fauna is scarce and its relationship with BPA exposure has not been investigated yet.
Immune function
Besides being an endocrine disruptor, BPA increased lymphocyte proliferation at 5 µg/L to 50 mg/L concentrations in aquacultured goldfish,
In primary macrophages from head kidney of the common carp (
Oxidative stress
Concentrations of BPA that can be encountered in nature are capable to induce oxidative stress, leading to impaired sperm quality and DNA fragmentation in fish. Higher levels of protein and lipid oxidation and superoxide dismutase activity were observed in sterlet (
Overall, available data provide convincing evidence that BPA affects physiological homeostasis in aquatic vertebrates, also at environmental (≤12 µg/L) concentrations. A graphical summary is provided in Figure 1. The existence of main windows of susceptibility (development) and targets (gonad functionality) can be deduced, together with a dose- and time-dependent BPA effectiveness. In particular, nonmonotonic dose responses are highlighted, where low doses, encompassing the environmental range, are the most effective.

Scale bar of effects of environmentally relevant concentrations of bisphenol A (BPA; µg/L) in aquatic vertebrates. Upper panel: Values represent examples of BPA concentrations detected in different types of environmental samples. Lower panel: Values represent examples from laboratory experiments of BPA exposure with different vertebrate models. (1) Flint et al., 2012 and reference therein; (2) Xu W, Yan W, Huang W, Miao L, Zhong L.2014. Endocrine-disrupting chemicals in the Pearl River Delta and coastal environment: sources, transfer, and implications. Environ Geochem Health. 36:1095-1104. (3) Xu EG, Morton B, Lee JH, Leung KM., 2015. Environmental fate and ecological risks of nonylphenols and bisphenol A in the Cape D’Aguilar Marine Reserve, Hong Kong. Mar Pollut Bull. 91:128-138. (4) Basheer C, Lee HK, Tan KS, 2004 Endocrine disrupting alkylphenols and bisphenol-A in coastal waters and supermarket seafood from Singapore. Mar Pollut Bull. 48:1161-1167. (5) Villeneuve et al. 2012. (6) Mandich et al. 2007. (7) Lahsteiner et al. 2005. (8) Kashiwagi et al. 2008. (9) Kwak et al. 2001. (10) Roger and Mirza, 2013. (11) Haterf
Bisphenol A Bioaccumulation/Elimination in Vertebrates
It is reported that relatively little BPA occurs in animal tissues, with maximum bioconcentration factors (BCFs) well below 1000, which is the limit of concern according to United States Environmental Protection Agency (USEPA) (Staples et al. 1998). Renz et al. (2013) assessed the presence BPA in the brain of 44 of the 58 fish collected from the Greater Pittsburgh area, with a maximum value of 120 pg/g w/w, and a calculated BCF ranging from 0.7 to 143 in different species. In general, bioaccumulation is estimated to occur only at high doses of BPA (Staples et al. 1998), while low doses are biodegraded or metabolized. BPA was detectable in plasma, liver, and muscle of rainbow trout after 2 hours of exposure to 100 µg/L BPA, reaching a steady state within 6 to 12 hours and BCF values of about 3.5-5.5 (Lindholst et al. 2001). Rainbow trout injected with 154 µmol BPA/kg w/w excreted the compound mainly as BPA–glucuronid acid, and a half-life of 3.75 hours was calculated for BPA (Lindholst et al. 2001). The accumulation of BPA–glucuronid acid in the gall fluid before excretion was reported in rainbow trout (Larsson et al. 1999) and in carp (Mandich et al. 2007). Although able of readily excrete BPA, aquatic organisms may be chronically exposed to the compound, and in these cases excretion may be overwhelmed. For example, the field BCF calculated for the bile of the common carp
Furthermore, metabolism of BPA is known to produce xenoestrogenic compounds as potent as the parent molecule or even more potent, as reported in medaka,
Bisphenol A uptake was demonstrated in
Effects of BPA on Invertebrate Species
Invertebrates as Test Organisms for the Effects of BPA
In the Final Report of the State of the Art Assessment of Endocrine Disrupters (Kortenkamp et al. 2011), it was stated that assays utilizing invertebrates offer some advantages over vertebrate models, since their use involves fewer ethical considerations, doses are easier to deliver in the aquatic medium, and their small size and inexpensive cultural requirements allow larger data sets to be collected. Unfortunately, although invertebrates represent 95% of animal species, and important components of aquatic ecosystems, and are, therefore, at considerable risk of exposure, scattered information is available on the biological effects of endocrine disruptive chemicals (EDCs) in these organisms. This is due to the large number of species and diversity of endocrine systems in different phyla and scarcity of data on the synthesis, effects, and mechanisms of action of hormones, steroids in particular, in comparison with those available in vertebrates (Porte et al. 2006; Crain et al. 2007; Scott 2013).
From the first observations indicating that prosobranch molluscs are prone to the endocrine disrupting effects of BPA (Ohelmann et al. 2000), the possible adverse effects of this compound have been investigated in different invertebrate species. However, these studies indicated the absence of reproductive effects of BPA at environmental concentrations in most invertebrate groups, including cnidarians, nematodes, and crustaceans (reviewed in Crain et al. 2007). In contrast, chronic exposure to low levels of BPA induced a superfeminization syndrome (ie, increased egg mass/number) in the freshwater rumshorn snail
The controversy surrounding the validity of these studies, regarding the failure to compare data obtained in different laboratories, was partly ascribed to differences in the experimental design. In fact, different attempts to reproduce these data underlined the importance of taking into account the influence of both endogenous and environmental variables (ie, seasonal differences in reproductive stage and effects of temperature; Sieratowiczs et al. 2011).
A large number of laboratory studies have been carried out to evaluate the effects of BPA on invertebrate development and reproduction at different concentrations and times of exposure. The results have been recently reviewed by Flint et al. (2012), leading to the conclusion that the effect of BPA appears to vary considerably among related taxa. Although the results obtained from laboratory experiments are obtained at different times of exposure and involve evaluation of different biological endpoints in different species, the available data underline how some invertebrates appear to be quite sensitive to BPA (freshwater and marine molluscs, insect larvae, marine copepods in particular), with effects documented at environmentally relevant concentrations (Figure 2). Differences in sensitivity to BPA can be explained by the differences in endocrine regulation in different groups, in physiological adaptations to different environments, as well as to the diverse modes of action of this compound at the cellular level.
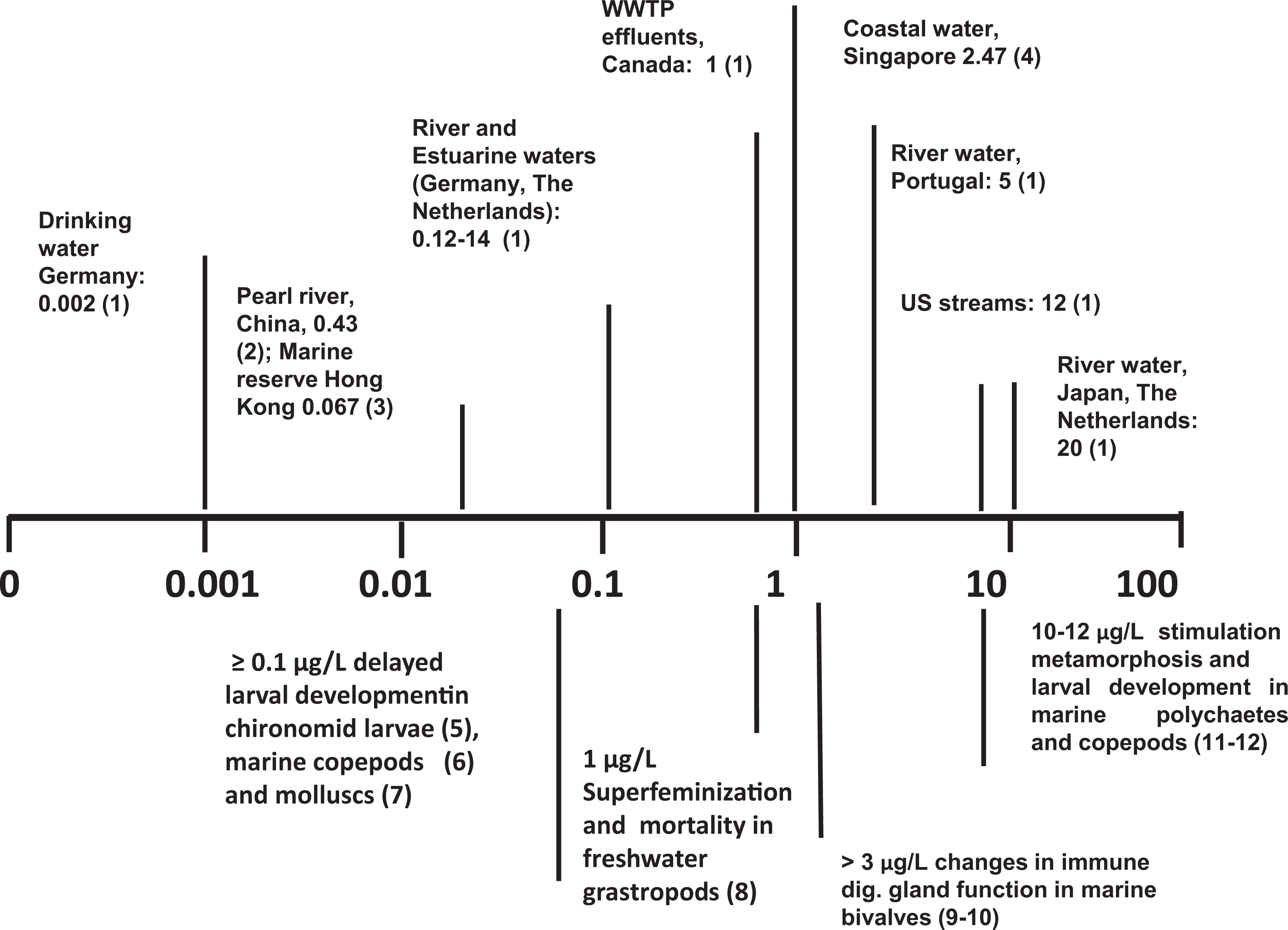
Scale bar of effects of environmentally relevant concentrations of bisphenol A (BPA; µg/L) in aquatic invertebrates. Upper panel, Values represent examples of maximal BPA concentrations detected in different types of environmental water samples. WWTP indicates waste water treatment plants. Lower panel, Values represent examples from laboratory experiments of BPA exposure with different invertebrate models. For reference 1-4 see Figure 1. (5) Watts
Possible Mechanisms of Action of BPA as an ED in Invertebrate Cells
A great deal of efforts has been made to identify the mechanisms of action of BPA at the cellular level in invertebrate models. The most comprehensively described endocrine system within invertebrates is that of insects, where different hormones act through nuclear receptors (NRs) that control multiple functions at different life stages. The midge
However, the general assumption that BPA acts as a xenoestrogen in vertebrate cells through binding to nuclear ERs, leading to changes in gene transcription (through “classical,” genomic, or receptor-dependent mechanisms of action) is in contrast with the failure in identifying functional ERs in invertebrates (Thornton et al. 2003). The ER-like sequences have been identified in different invertebrate groups, molluscs in particular; however, molluscan ERs generally show constitutive transcriptional activity and are not activated by either natural estrogens or estrogenic compounds (Bingham et al. 2014 and references quoted therein). An accurate analysis on the literature on the effects of steroids and EDCs recently concluded that there is no indisputable bioassay evidence that vertebrate sex steroids (or their agonist/antagonists) have endocrinological or reproductive roles in molluscs (Scott 2013b). A recent genetic and structural analysis of oyster ERs clearly demonstrated that the estrogen-binding site is occluded by hydrophobic residues and it is too small to accommodate the physiological agonist 17β-E2 or other similar compounds. Evolution of constitutive activity was due to 2 substitutions and subsequent mutations filled in the hormone-binding cavity, in a process of vestigialization (Bingham et al. 2014). This implies that molluscan ER is an authentically ligand-independent transcriptional activator, which exists in the active conformation in the absence of ligand or other apparent modifications. Therefore, if estrogen-like compounds, endogenous or environmental, affect mollusc reproduction and physiology, these impacts must be mediated by mechanisms other than ER activation.
Mode of Action and Pleiotropic Effects of BPA in Invertebrates: The Example of the Marine Bivalve Mytilus
The marine bivalve Mytilus is tolerant to fluctuations in abiotic and biotic factors and shows a wide range of responses to environmental stressors and therefore it is worldwide utilized as a model organism for investigating the effects of environmental contaminants, as well as a sentinel species for biomonitoring of coastal areas (Viarengo et al. 2007).
The effects of the natural estrogen 17β-E2 and environmental estrogens, including BPA, have been thoroughly investigated in mussel immune cells, the hemocytes, both
In the same
In mussel digestive gland, BPA was recently shown to affect 17 β-hydroxysteroid dehydrogenases (17β-HSDs), multifunctional enzymes involved in the metabolism of steroids, fatty acids, retinoids, and bile acid (Zhang et al. 2014). Expression of 2 novel types of 17β-HSDs (MgHsd17b10 and MgHsd17b12) identified in
Effects of BPA on Invertebrate Development
Developmental effects of BPA have been observed in invertebrates, in different exposure models, but only in few species at environmental concentrations (see Flint, 2012 and reference quoted therein). In the gastropod
Bisphenol A Bioaccumulation/Elimination in Invertebrates
Bisphenol A discharge can occur from the migration of BPA-based products into rivers and marine waters, and in particular from effluents from wastewater treatment plants and landfill sites. However, in the aquatic environment, BPA can undergo photodegradation or biodegradation through microbial or plant activities and metabolism by aquatic animals. As a result of this, relatively little environmental BPA occurs in biota compared to nonbiotic environmental compartments (reviewed by Flint, 2012). Sparse reports on BPA concentrations in aquatic invertebrates are available (Nurulnadia et al. 2014 and references quoted therein), with levels in the order of ng/g w/w, generally lower than those found for other alkhylphenols such as Nonyl-phenol. Information on accumulation and metabolism of BPA is available in molluscs. In freshwater, bivalves (
Conclusions
Laboratory studies indicate that in non-mammalian vertebrates, in particular aquatic species, BPA causes developmental and reproductive effects, including reduction of male hormones, death of testicular cells, decreased sperm density and motility, inhibition of spermatogenesis and egg production, along with delayed or absent ovulation, and impairment of sex ratio. Furthermore, the chemical proved able to affect other systems, for example, disturbed immune function and metabolism. Therefore, the compound appears to be a multifunctional endocrine disrupter. The whole animal may respond to BPA in a different way according to its developmental and physiological status, which would justify the difficulties encountered when drawing possible mechanisms of action.
With regard to invertebrate species, according to the USEPA final Report (2014), BPA showed high chronic aquatic toxicity; however, experimental studies located for daphnid used as an invertebrate test species were of insufficient exposure duration to be utilized to assign the hazard concern. Taken together, the available results indicate that neither ecotoxicity tests carried out in model species nor studies focused on a mechanistic approach based on interactions of BPA with ERs are sufficient to explain and predict the effects of BPA, or other EDCs, in different invertebrate systems. The greater susceptibility of certain invertebrate species may be explained with multiple mechanisms of action, as now recognized also in vertebrates (Rubin et al. 2011). Moreover, knowledge on the physiological status and the responses to abiotic variables of the test organism is crucial in evaluating the impact BPA in invertebrate groups. However, taken together available data support the hypothesis that environmental concentrations of BPA (low μg/L) can elicit significant responses on certain groups of aquatic invertebrates, mollusks, and copepods in particular.
Overall, from available data in both vertebrates and invertebrates, there is agreement on the fact that BPA is a chemical of potential concern for the ecosystem.
In many cases, the concentrations of BPA necessary to cause such effects exceeded the average concentrations in the environment or were in the upper range. The laboratory studies cannot account for the life-long animal exposure to BPA, since it is continuously released in large amounts. Thus, an underestimation of the effects is conceivable, also considering that wildlife species may be exposed to higher BPA concentrations in specific matrices (leachates, plants effluents, river, and marine sediments).
The present review highlights that a few reports address dose–response effects of BPA exposure. In the absence of clear dose response curves, and due to the shortcomings in experimental designs, a conclusion cannot be reached regarding the BPA impact on ecosystem. These studies would have major regulatory implications, and hence it is incumbent on scientists to do their research to the highest standards possible, in order that the most appropriate decisions are taken.
Recommendations
Develop a full toxicological assessment on BPA to determine an acceptable freshwater and marine exposure level.
Identify which animal species are most at risk to environmental BPA levels.
Perform more studies in the natural environment to evaluate real concentrations and long-term exposures.
Investigate the relative importance of different exposure pathway to BPA (digestive tract, tegument, and respiratory surfaces) for wildlife.
Evaluate the bioaccumulation potential of BPA, especially in edible species, also in the light of the latest EFSA reevaluation of BPA exposure and toxicity and concomitant reduction of the safe highest dietary exposure to BPA from 50 to 4 µg/kg of bw/d (EFSA, 2015)
Assess effects at low BPA doses and avoid narrow dose ranges; testing 1 or 2 doses may lead to erroneous conclusions since nonmonotonic dose–response curves are encountered frequently in basic endocrinology.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
