Abstract
Elbow defects have a number of etiologies and present a difficult task for the reconstructive surgeon. A number of reconstructive options have been previously reported. We describe a case of a 54-year-old woman with a recurrent elbow defect secondary to prior trauma, surgical fixation, and infection. This was successfully managed with a novel chimeric flexor capri ulnaris and ulnar artery flap for coverage of the defect, which has not been previously described. This may serve as a useful guide for surgeons moving forward.
Keywords
Introduction
Elbow defects may result from traumatic injuries, oncologic resection, or prior surgical intervention.1,2 Durable soft tissue reconstruction is required for exposure of bones, joints, neurovascular bundles, or prostheses. 1 Thin, pliable tissue is preferred to allow for repetitive elbow flexion and extension. 2 A multitude of reconstructive options have been previously described, including primary closure, skin grafting, local muscle flaps, pedicled fasciocutaneous flaps, distant pedicled muscle, or myocutaneous flaps, as well as microsurgical free tissue transfer.1 -3 The more commonly described reconstructive options, including pedicled radial forearm or latissimus dorsi flaps, as well as anconeus or brachioradialis muscle flaps, are associated with limited coverage or donor site morbidity. 3 In addition, these options may be unavailable due to the zone of injury, anatomy of the defect, and the patient’s clinical course.
Alternative flap options, including the flexor capri ulnaris (FCU) flap and the ulnar artery flap, have been described to mitigate some of these limitations.3 -5 However, a pedicled chimeric flap involving the FCU muscle and fasciocutaneous tissue supplied by the distal ulnar artery has not been previously described. We describe the first reported use of this chimeric flap for a recurrent medial elbow defect in which previous flap options were inadequate.
Methods
In this article, the authors retrospectively reviewed the clinical course of one patient with a recurrent medial elbow defect, which was ultimately managed using our novel chimeric FCU and ulnar artery flap. Clinical photographs are used to demonstrate initial flap coverage, recurrent hardware exposure, and successful coverage with the chimeric flap. All health information has been de-identified.
Results
Clinical Course
A 54-year-old right-hand-dominant woman was seen in the orthopedic surgery clinic in February 2020. She had sustained a right distal humerus fracture in June 2018 for which she underwent open reduction internal fixation (ORIF). On assessment in clinic, she was found to have a failure of her hardware with fracture of the distal screws, as well as a malunion. Her medical history was significant for Crohn’s colitis, panic disorder, fibromyalgia, depression, and liver fibrosis. She had been previously taking Stelara, coversyl, citalopram, pantoloc, diltiazem, lyrica, and zopiclone.
In October 2021, she underwent removal of her previous hardware, anterior transposition of her ulnar nerve, and total elbow arthroplasty. The radial head was noted be malformed and was resected. She was doing well until 3 months postoperatively when she presented back to hospital with an open wound and evidence of hardware exposure and infection. She was started on intravenous cefazolin and taken back to the operating room for irrigation and debridement (I + D) as well as replacement of the ulnar link component. She went back to the operating room 2 weeks later for repeat I + D and placement of a vacuum-assisted closure (VAC) due to nonhealing of the wound. A 12 cm × 8 cm defect was noted at this stage. Two days later, a pedicled latissimus dorsi myocutaneous flap was inset over the defect for coverage and tenoplasty of the triceps tendon was performed using the thoracolumbar fascia for triceps reconstruction (Figure 1). Progressive ecchymosis of the flap was noted over the next 48 hours, prompting return to the operating room for debridement of the distal nonviable portion of the flap, further irrigation, and sequestrectomy of the medial epicondyle. The flap inset was loosened on POD#2 with VAC application. She underwent split thickness skin grafting with VAC application to the distal aspect of the flap 2 weeks later. During this time, she continued to improve clinically while on rifampin and cefazolin.
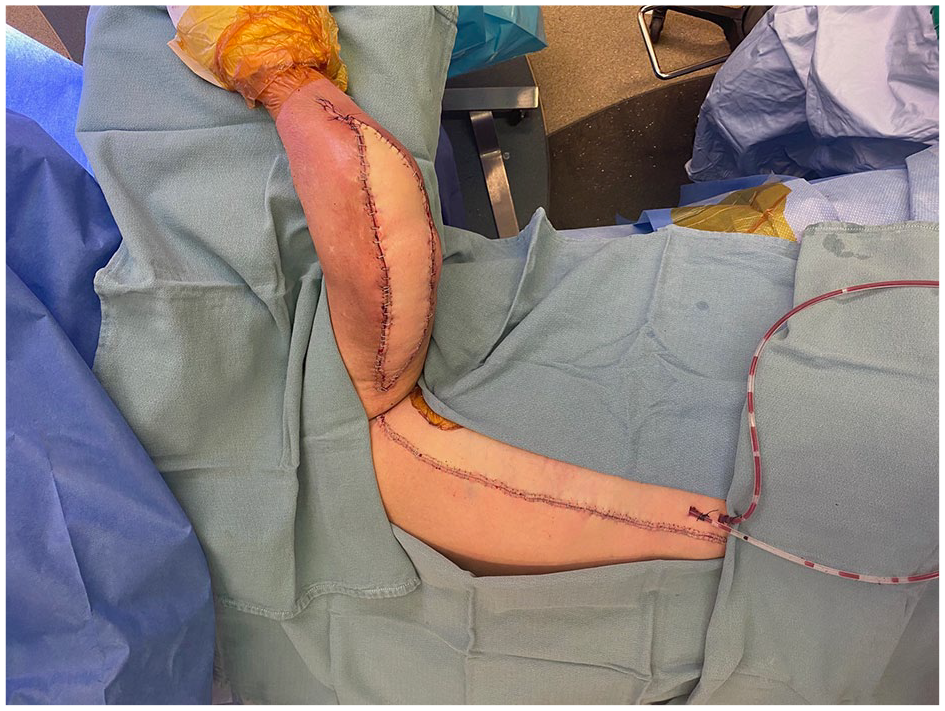
Pedicled latissimus dorsi myocutaneous flap following triceps tenoplasty and completion of inset.
She was seen in clinic 2 weeks after split thickness skin grafting, and hardware exposure was once again noted over the medial elbow (Figure 2). The arthroplasty hardware was quite prominent and significant dead space was noted posteromedially between the hardware and the previous latissimus flap. Repeat I + D and VAC application were performed. Cultures grew Corynebacterium, and vancomycin was added to her regimen. She was brought back to the operating room 2 days later with a plan for definitive soft tissue coverage and application of an external fixator. The defect measured 10 cm × 6 cm with a significant amount of dead space intraoperatively. We planned to use a chimeric ulnar artery flap for coverage with a FCU flap for dead space obliteration, as well as to bolster the thickness of the soft tissue cover.
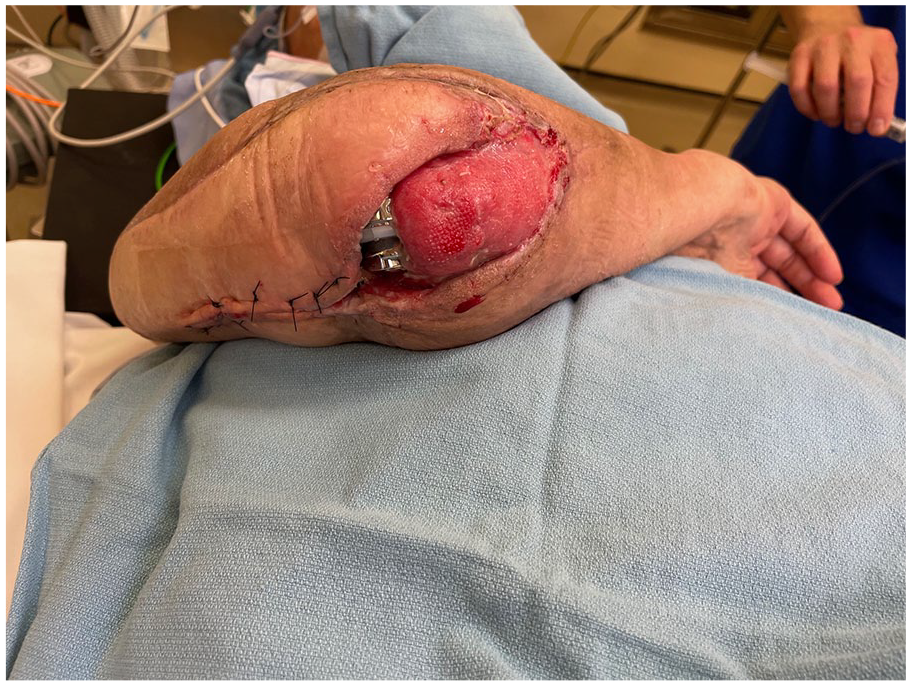
Recurrent exposure of medial elbow hardware noted in clinic.
Operative Technique
The ulnar border of the planned ulnar artery flap was incised down to the level of the fascia before fascial division. Once the septum between the FCU and flexor digitorum profundus muscle was reached, the FCU muscle was circumferentially dissected prior to transection. The FCU muscle was then elevated to the level of the mid-forearm. The ulnar artery was circumferentially dissected and clamped, confirming excellent flow to the hand through the radial artery. The radial border of the flap was incised down to the level of the fascia. Flap elevation continued in a radial to ulnar direction prior to distal ligation of the ulnar artery. Dissection then proceeded proximally, taking care to maintain all perforators supplying the flap. As we got into the proximal third of the forearm, we identified the major perforators going into the FCU muscle belly. These were dissected out circumferentially and preserved. The FCU was detached from its proximal insertion and mobilized around the ulnar nerve. At this point, the chimeric flap elevation was complete with both the FCU muscle and ulnar artery flap still attached to its common pedicle proximally (Figure 3). The FCU flap was then mobilized further and inset over the hardware for extra coverage using 2-0 Vicryl suture. The presence of muscle over the hardware helped to make coverage more robust, reduce the chance of hardware erosion, and obliterate the dead space underneath the latissimus flap. The ulnar artery flap was inset over the remaining soft tissue defect using 3-0 Vicryl and 3-0 nylon suture (Figure 4). The donor site was closed primarily. An elbow spanning external fixator was then placed from the anterolateral aspect of proximal humerus to the dorsal radial aspect of the radial shaft. This was set to maintain the elbow in an extended position given the lack of triceps function.
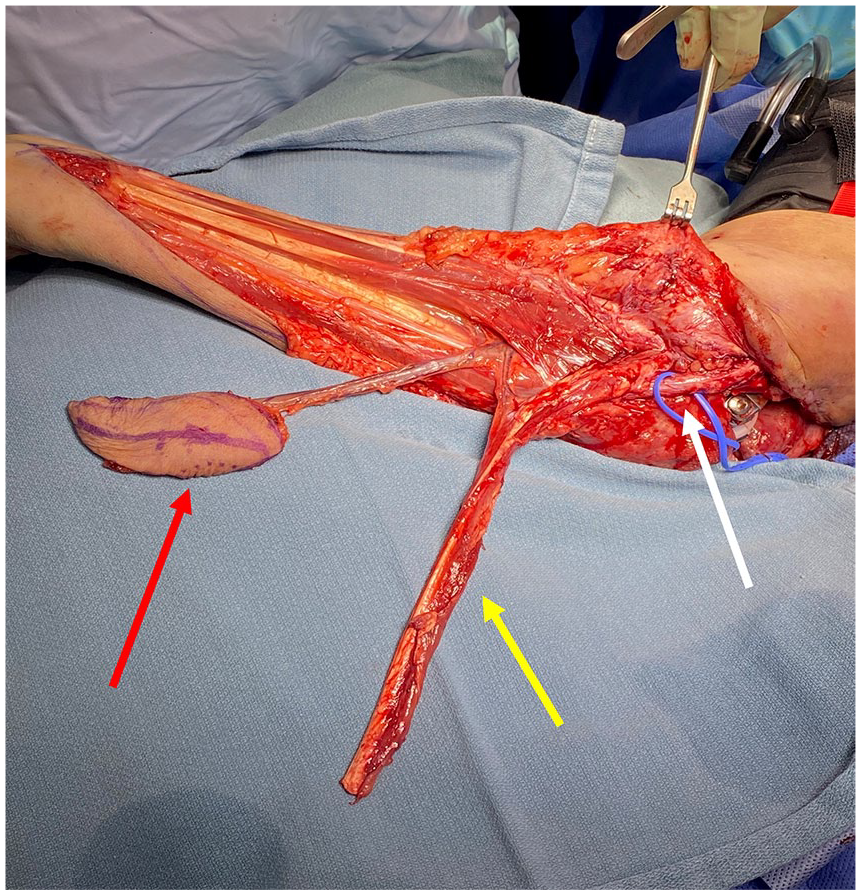
Chimeric flexor carpi ulnaris and ulnar artery flap at completion of dissection.
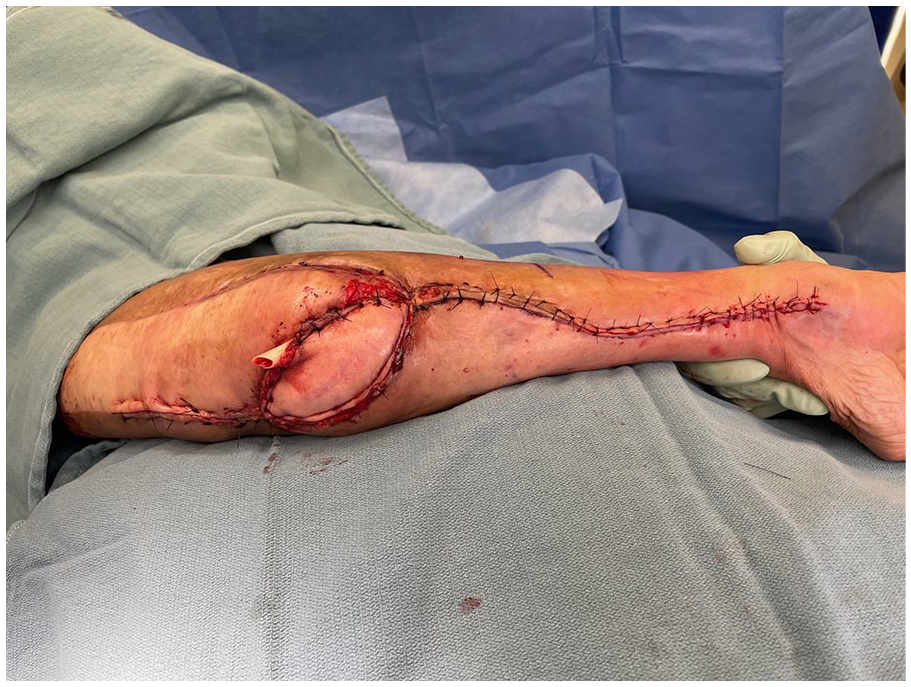
Chimeric flap at completion of inset. The donor site has also been closed primarily.
Six weeks postoperatively, she was noted to have a peri-prosthetic fracture around the distal radius pin site. The external fixator was removed and ORIF was performed using a dorsal Thompson approach. She was last seen in clinic 2 weeks following ORIF and was noted to be recovering well (Figure 5). No negative sequelae were noted despite harvest of the dominant artery to the hand.
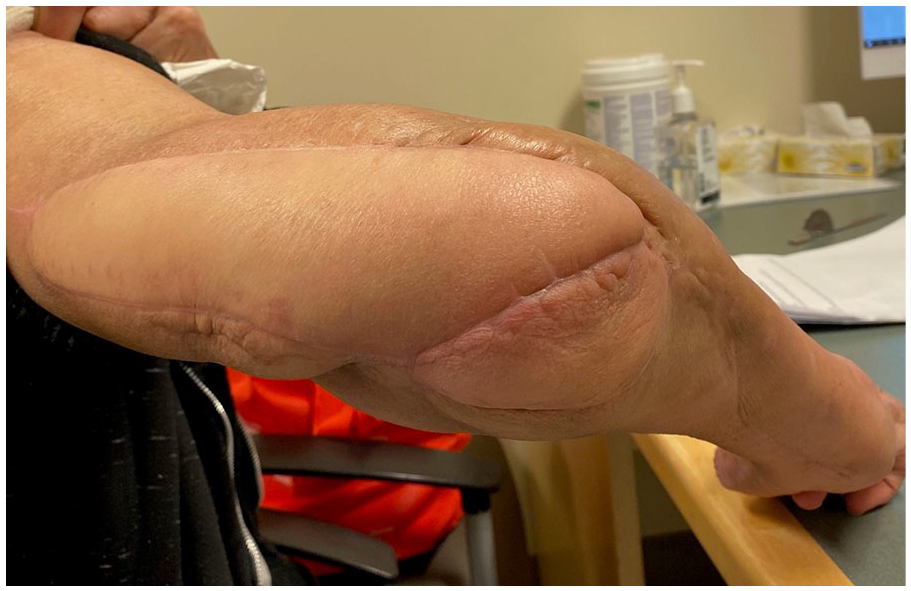
Patient at 2 weeks following external fixator removal and distal radius open reduction internal fixation. Excellent healing of the flap is evident with no signs of wound infection or breakdown.
Discussion
Elbow defects may arise from a variety of causes, including prior surgical intervention, trauma, or after oncologic resection.1,2 Numerous methods of reconstruction have been described, including local muscle flaps, pedicled fasciocutaneous flaps, as well as free tissue transfer. However, the reconstructive choice is ultimately decided based on size and anatomy of the defect, exposure of vital structures, patient comorbidities, and potential donor site morbidity. 2
The first-line reconstructive option for our patient was a pedicled latissimus dorsi myocutaneous flap. This was necessary given the large size of the defect (12 cm × 8 cm), presence of exposed hardware, need for skin resurfacing, as well as the need for restoration of triceps function. As noted by Stevanovic and Sharpe, 1 a skin paddle is necessary in the case of functional transfers to prevent muscle adhesion and improve tendon gliding. Progressive flap compromise was noted 48 hours postoperatively, requiring debridement of the distal nonviable portion of the flap. Skin necrosis of the distal latissimus dorsi flap is not uncommon for extensive elbow defects. Stevanovic et al 6 reviewed their series of 16-pedicle latissimus dorsi flaps for elbow coverage, and 3 patients experience distal flap necrosis requiring additional procedures. In a review by Choudry et al, 7 14% of pedicled latissimus dorsi flaps experienced significant distal necrosis. It is important to note that the wounds extended past the olecranon in all of these cases, 7 much akin to our patient.
The anconeus and brachioradialis have also been described as pedicled muscle flaps for elbow coverage.1 -3 While excellent results have been reported by Elhassan et al, 8 the anconeus is limited by both its small size and short pedicle, thereby not permitting coverage of medial defects. In addition, the pedicle is often compromised with a posterolateral approach to the elbow, such as in our case, and is unsuitable in the setting of multiple previous operations.2,3 The brachioradialis muscle flap is best suited for coverage of lateral defects, and may produce functional deficits in a limb with pre-existing compromise,2,5 such as the case of our patient.
The FCU flap was first described by Meals 9 in 1989 as a proximally based turnover flap for elbow coverage. Since then, it has become part of the reconstructive algorithm for elbow coverage, with numerous studies noting the consistent vascular supply, large surface area, and ease of elevation as distinct advantages.1,3,5 In a study by Bayne et al, 17 patients underwent FCU flap reconstruction with no complications or reoperations. Importantly, the same study showed no significant differences in wrist motion or strength when compared with the contralateral side. 3 Similar findings were corroborated by Raskin and Wilgis. 10 Taken together, these studies may help to alleviate concerns related to potential decrease in wrist flexion with harvest of the FCU muscle. While the musculocutaneous variant of the FCU flap has been described,5,11 the inability to close the donor site may result in exposure of the ulnar neurovascular bundle and the distribution of cutaneous perforators in relation to muscle harvest has not been described. 3 It also does not permit independent arcs of rotation of the cutaneous and muscle components. The chimeric flap design in our case was necessary given the complexity of the defect, as differential vectors were required for each of the individual flap components.
This chimeric FCU muscle and ulnar fasciocutaneous flap have not been previously described, and thereby add a new flap option to the reconstructive armamentarium. The ulnar artery flap and its associated perforators have been detailed previously, with perforators located along the axis from the pisiform to the medial epicondyle. Dominant perforators are located 7, 11, and 15.5 cm above the pisiform. 4 This flap offers numerous advantages over the radial forearm flap, including easier management of the donor site, potential to avoid skin grafting, less conspicuous scar, and hairless skin. 4 Therefore, our chimeric flap was able to combine robust dead space obliteration and additional hardware coverage with the FCU muscle component and inclusion of a reliably vascularized skin paddle, while avoiding skin grafting to the donor site. The skin paddle also obviates need for skin grafting at the recipient site. 3
Conclusion
Elbow defects have numerous etiologies and present a difficult problem for the reconstructive surgeon. Various local fasciocutaneous and muscle flaps have been previously reported for management of these defects. We describe the first use of a chimeric ulnar artery fasciocutaneous and FCU muscle flap for coverage of a recalcitrant medial epicondyle wound. This may serve as a suitable first-line option for medial wound coverage or as a reliable back-up option in the event of wound breakdown. We hope that this serves as a useful reference guide for reconstructive surgeons moving forward.
Footnotes
Authors’ Note
No writing assistance was used in the creation of this manuscript.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Informed consent was obtained from the individual included in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
