Abstract
Background:
The anterior interosseus nerve (AIN) to ulnar motor nerve transfer has been popularized as an adjunct to surgical decompression in patients with severe cubital tunnel syndrome (CuTS) and high ulnar nerve injuries. The factors influencing its implementation in Canada have yet to be described.
Methods:
An electronic survey was distributed to all members of the Canadian Society of Plastic Surgery (CSPS) using REDCap software. The survey examined 4 themes: previous training/experience, practice volume of nerve pathologies, experience with nerve transfers, and approach to the treatment of CuTS and high ulnar nerve injuries.
Results:
A total of 49 responses were collected (12% response rate). Of all, 62% of surgeons would use an AIN to ulnar motor supercharge end-to-side (SETS) transfer for a high ulnar nerve injury. For patients with CuTS and signs of intrinsic atrophy, 75% of surgeons would add an AIN-SETS transfer to a cubital tunnel decompression. Sixty-five percent would also release Guyon’s canal, and the majority (56%) use a perineurial window for their end-to-side repair. Eighteen percent of surgeons did not believe the transfer would improve outcomes, 3% cited lack of training, and 3% would preferentially use tendon transfers. Surgeons with hand fellowship training and those less than 30 years in practice were more likely to use nerve transfers in the treatment of CuTS (P < .05).
Conclusions:
Most CSPS members would use an AIN-SETS transfer in the treatment of both a high ulnar nerve injury and severe CuTS with intrinsic atrophy.
Introduction
Cubital tunnel syndrome (CuTS) is the second most common compressive neuropathy and affects the ulnar nerve at the elbow.1,2 First-line treatments aim to reverse exacerbating factors through nonsurgical measures such as positional changes and splinting. 2 However, a number of patients are refractory to these interventions and require surgical treatment in the form of decompression or nerve transposition. 3 Although this stops ongoing compression and axonal loss, it does not guarantee the recovery of normal function. With recovery from nerve injuries being correlated with the distance from the location of injury to the target muscles or sensory end organ, pathologies at the elbow can result in debilitating sequalae in hand function, despite an appropriate intervention.2,4 In addition, the duration of dysfunction plays a significant role in outcomes, a factor that is difficult to quantify in CuTS given the nontraumatic insidious onset of this pathology.
Although many strategies exist to improve outcomes in these types of injuries, 5 one option is to use an end-to-end nerve transfer of the anterior interosseus nerve (AIN). With the AIN being an expendable donor nerve in close proximity to the intrinsic musculature of the hand, it is an ideal donor. The drawback of this approach is the loss of potential recovery from proximal nerve fascicles of the native nerve. An alternative option is the reversed or “supercharged” end-to-side nerve transfer (SETS), which has been popularized by Mackinnon and Novak. 6 In the case of the ulnar nerve, the AIN is coaptated in an end-to-side fashion to allow the ingrowth of fascicles and neurotropic factors.7-9 Clinically, the AIN-SETS transfer has shown promising results;4,9-17 however, without a clear proven mechanism in vivo, the implementation of this approach is controversial.18,19
In appropriate patients, this technique may have potential benefit as an adjunct in the surgical treatment of CuTS. In the American health care system, surgical complexity and ambiguity in coding have been cited as barriers to the implementation of the AIN-SETS transfer. 20 The practice patterns of Canadian plastic surgeons has yet to be investigated, and the purpose of this work is to investigate the utilization of this technique and gain insights into factors or barriers associated with its use.
Methods
Ethics approval was obtained from the Western University institutional review board (2020-2022) before the commencement of this study. Using REDCap software (Vanderbilt University, Nashville, Tennessee), a 21-item survey was electronically distributed to all members of the Canadian Society of Plastic Surgeons (CSPS) on June 21, 2021 (Supplemental Appendix A). Recipients received follow-up reminder emails at 3 and 6 weeks. All data were anonymized apart from practice location.
The survey consisted of 4 major components: previous training and level of experience with nerve surgery, annual volume of peripheral nerve pathologies and available facilities (i.e., availability of electromyographer [EMG]), experience with nerve transfers, and approach to ulnar neuropathy (i.e., CuTS).
Statistics
Descriptive statistics were calculated using Microsoft Excel (Redmond, Washington). Direct comparisons were carried out with Student’s t tests using SPSS computational software (IBM, Chicago, Illinois) and a significance of P = .05.
Results
A total of 49 CSPS members completed the survey (12% response rate). Most respondents (n = 23) were from Ontario, followed by British Columbia (n = 11), and Alberta (n = 5). Of all, 63% (n = 31) of surgeons were practicing in academic centers, with the remainder working in a combination of community and private practices (Figure 1).
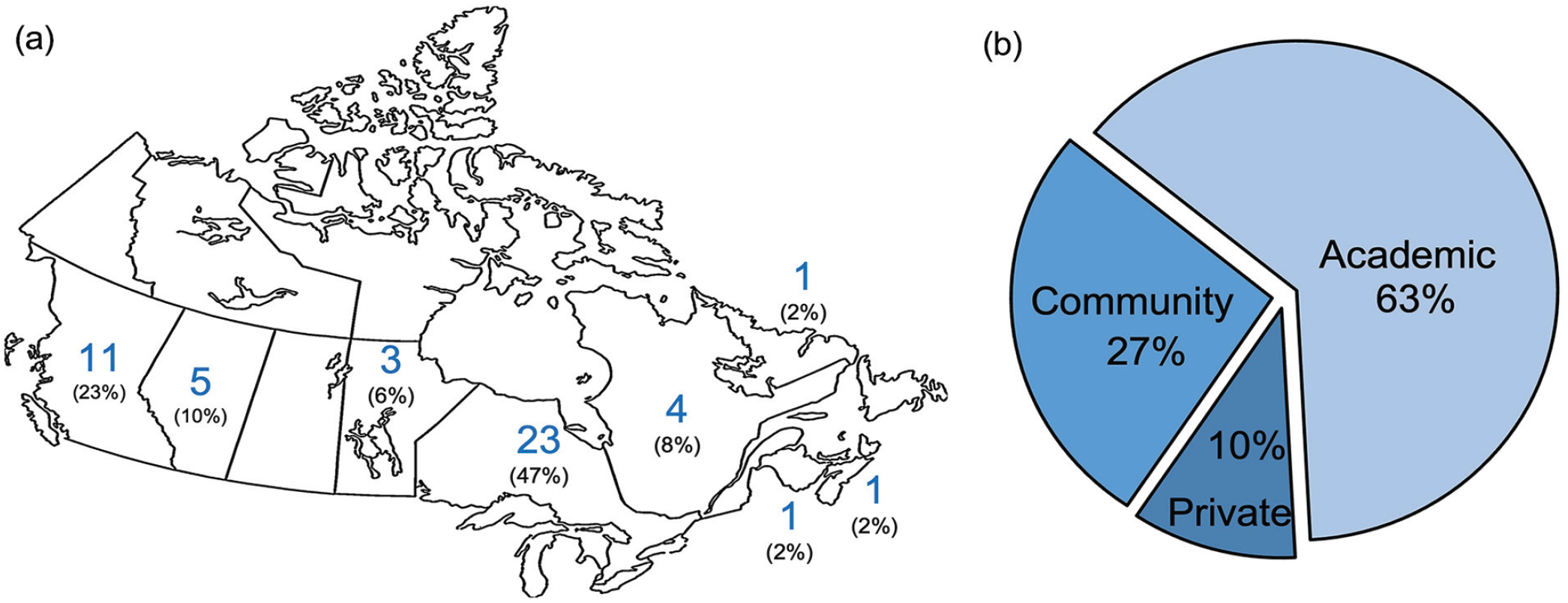
Demographics of respondents by geography (a) and practice type (b).
Microsurgery was the most common fellowship (n = 30), followed by general hand (n = 29), peripheral nerve (n = 23), wrist (n = 23), trauma (n = 21), and congenital hand surgery (n = 14), with most surgeons obtaining more than 1 fellowship. Of all, 35% (n = 17) of surgeons were within the first 10 years of practice, 29% (n = 14) between 10 and 20 years, and 36% (n = 18) had more than 20 years of experience (Figure 2). More than half (n = 32) of the respondents use nerve transfers, with 15 respondents (31%) identifying as having a peripheral nerve practice. All but one surgeon had access to EMGs. One third of surgeons used EMGs in a specialized peripheral nerve clinic (32%), one third were able to obtain an EMG within a month (17% within 2 weeks, 15% within 1 month), and the remainder took 1 month or greater.
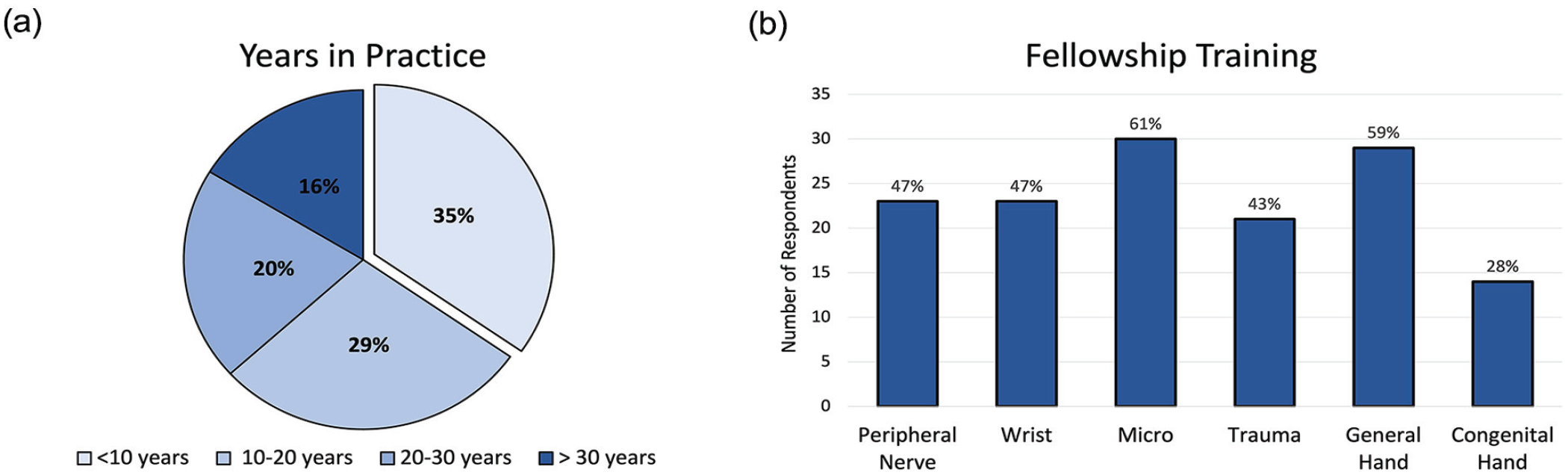
Survey respondents practice duration (a) and fellowship training (b).
Approach to Routine Ulnar Neuropathy
In the treatment of patients with CuTS with clinical signs of intrinsic muscle atrophy, 75% of 32 surgeons who use nerve transfers would perform an AIN-SETS transfer. Most surgeons (81%, n = 26) would also release Guyon’s canal during this operation (n = 20 routinely, n = 4 with EMG evidence of compression, n = 2 with clinical evidence of compression) (Figure 3). Comparing surgeons who used the AIN-SETS transfer for CuTS to those who do not, there was no difference in community versus academic practices (P = .08). However, surgeons with formal hand surgery training and those in practice for less than 30 years were more likely to use nerve transfers (P < .05) in treating CuTS.
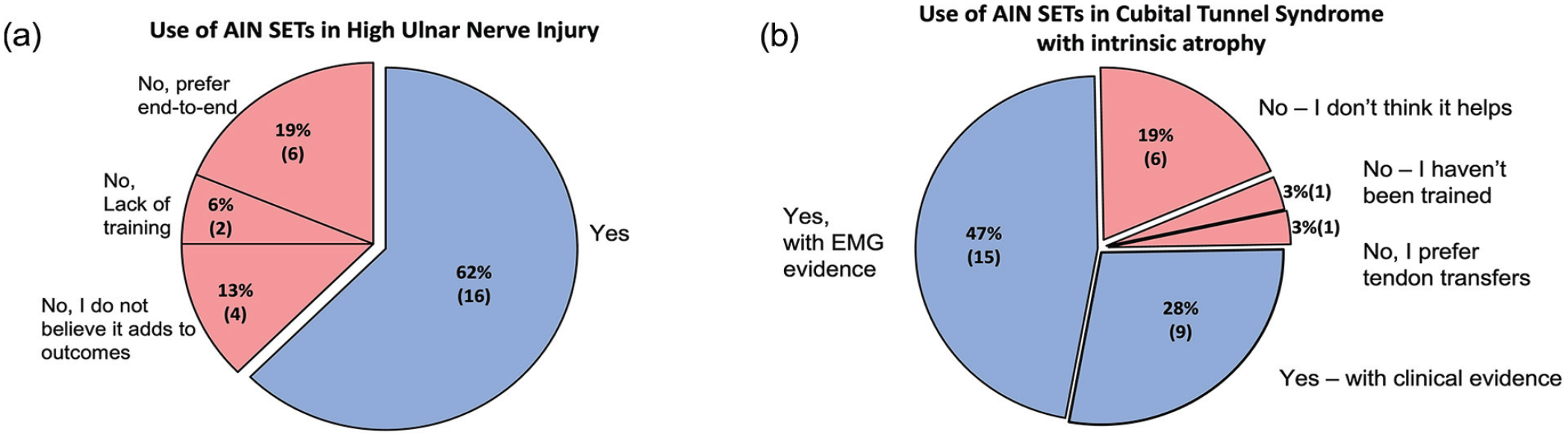
Surgeon’s preference for treatment of high ulnar nerve injuries and cubital tunnel syndrome with intrinsic atrophy. Note. AIN = anterior interosseus nerve; SETs = supercharge end-to-side; EMG = electromyographer.
AIN-SETS Technique
When performing an end-to-side nerve transfer, 56% (n = 18) of surgeons created a perineural window to the ulnar motor fascicle; 28% (n = 9) also introduced a partial fascicular injury, 3% (n = 1) introduced a full fascicular injury, and 6% (n = 2) only performed end-to-end transfers. Most surgeons (74%, n = 23) believe that specific placement of the AIN relative to the motor fascicle makes a difference with the following preferences: volar aspect n = 5, radial aspect n = 4, ulnar aspect n = 1, dorsal aspect n = 1. Twelve other respondents had specific locations of preference including using intraoperative stimulator guidance as well as specific quadrants of the nerve (i.e. volar-radial, volar-ulnar, dorsal-radial, etc) (Figure 4).
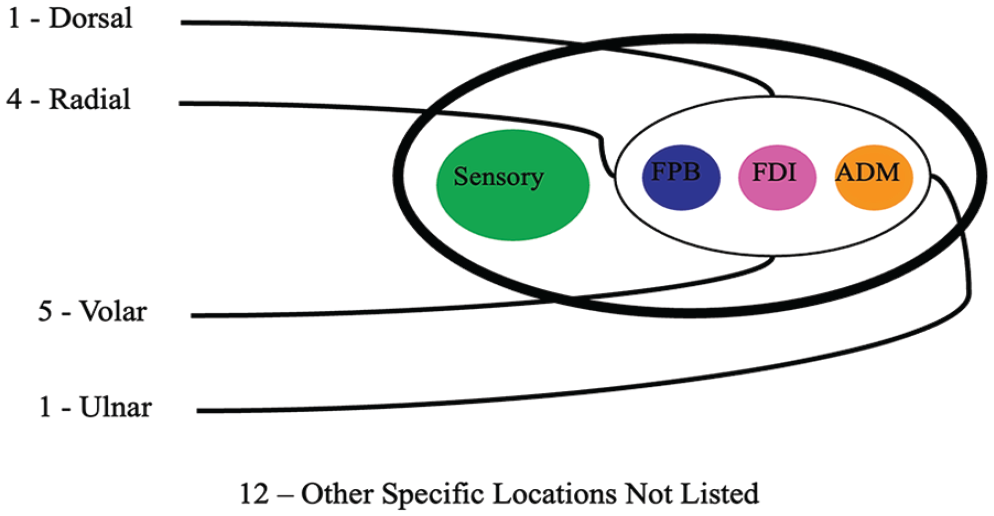
Pictorial representation demonstrating the preferred coaptation site of the anterior interosseus nerve to the ulnar nerve.
Discussion
The approach to upper limb pathology requires a breadth of surgical skills and creativity. As new techniques are introduced, their uptake is predicated on sufficient clinical evidence, knowledge translation, and training. The AIN-SETS transfer has shown early promising results.4,10-14 This survey of the CSPS found that 62% of surgeons would perform an AIN-SETS transfer for a high ulnar nerve injury, and 75% would perform it for severe CuTS with intrinsic atrophy.
Cubital tunnel syndrome represents the second most common entrapment neuropathy treated by hand and upper limb surgeons, 1 and when ongoing compression results in muscle atrophy, outcomes are less positive.9,21 Originally championed by MacKinnon et al 6 , there have been numerous articles from Canadian institutions reporting the results of the AIN-SETS transfer. Davidge et al 13 investigated 55 patients and postulated the utility of this transfer in patients with in-continuity lesions of the ulnar nerve. At 8 months of follow-up, all 39 patients with sufficient data demonstrated both clinical and subjective improvements in function with no appreciable loss of pronation power. Doherty et al 14 reported on 30 patients who underwent this transfer in conjunction with subcutaneous ulnar nerve transposition for severe CuTS. Of the 30 patients, 73% attained Medical Research Council grade 3 or greater at 18.6 months postoperatively. Similarly, Head et al 12 reported on a cohort of 17 patients and noted significantly increased strength and motor amplitude in first dorsal interossei and abductor digit minimi muscles. Recently, Dengler et al 22 examined 42 patients with chronic CuTS and demonstrated promising result while highlighting the need for appropriate patient selection as outcomes were related to total duration of neuropathy. Basic science experiments have also supported this clinical data and demonstrated that end-to-side neurorrhaphy facilitates axonal invasion from the donor nerve.7-9
In our study, 18% of surgeons did not believe that the addition of the transfer would improve clinical outcomes of treatment for CuTS. An additional 3% of surgeons would not perform the transfer because of lack of experience, and 3% would use tendon transfers alone instead for reconstruction. Opponents of the AIN-SETS transfer question whether the observed recovery of the intrinsic musculature is the result of direct reinnervation from the proximal ulnar nerve rather than from the AIN. While it is difficult to definitively elucidate where the regenerating axons originate, Doherty et al 14 reported the augmentation of motor unit numbers with active forearm pronation in 77% of patients undergoing an AIN-SETS transfer. This suggests that the transferred AIN fascicles are in part contributing to functional improvement. 14 In the setting of a high ulnar nerve injury, 38% of surgeons would not add a distal AIN transfer. However, the majority of these surgeons would refer to a colleague with subspecialty training, indicating perhaps a lack of comfort with the procedure rather than a lack of enthusiasm for its outcome. As with other surgical innovations, the uptake and application of new techniques is variable and can be related to the Gartner Hype Cycle which comprises 5 stages: (1) innovative trigger; (2) peak of inflated expectations; (3) trough of disillusionment; (4) slope of enlightenment; and (5) plateau of productivity. 23 End-to-side nerve transfers have garnered significant enthusiasm and are being applied broadly without long-term clinical outcome studies. 24 Anterior interosseus nerve supercharge end-to-side transfer appears to be in the second stage of this cycle, but only time and long-term data will determine the depth of the trough of disillusionment.
The current study also highlights the technical variability of how surgeons perform the AIN-SETS transfer, which may explain the varied results in the literature. In MacKinnon’s original description, the epineurium and perineurium of the motor fascicular group of the ulnar nerve are opened widely to create a perineurial window; however, no fascicular injury is induced. 15 Although most surgeons (56%) in our study used a perineural window, 28% also introduced a partial fascicular injury, and 3% created a full fascicular injury. There was also significant variability in the exact placement of the end-to-side neurorrhaphy. Twenty-five percent of surgeons indicated they did not place it consistently in the same place. Others indicated a variety of preferred locations (volar [16%], radial [13%], ulnar [3%], dorsal [3%]). This is not well explained by this survey, but possible reasons include variability in surgical training, preferred geometry of nerve coaptation, utility of surgical assistances, and differences in surgical opinion. Although the internal topography of the motor branch of the ulnar nerve has been described and specific placement of the transfer may influence outcomes,12,25 without definitive evidence to direct location, this remains a difference that requires further evaluation.
The primary limitations of this study are a low survey response rate of 12% and potential responder bias. However, the CSPS is composed of plastic surgeons from all specialties in all practice settings, so this percentage may represent a large proportion of peripheral nerve surgeons despite being a small fraction of the entire society nationally. There was also variability in the geographic location of respondents, but the provinces best represented have the highest population density in Canada, along with the highest number of nerve surgeons compared with other provinces. In addition, peripheral nerve surgeons not registered with the CSPS were not included in the survey, which may introduce bias by not sampling the orthopedic surgeons of Canada. The strengths include the diversity of questions asked, the anonymous nature of the survey, and the simple electronic format.
Conclusion
Among Canadian plastic surgeons, the AIN-SETS transfer is commonly used in the care of patients with high ulnar nerve injuries and for severe CuTS.
Supplemental Material
sj-pdf-1-han-10.1177_15589447231174482 – Supplemental material for Anterior Interosseus to Ulnar Motor Nerve Transfers: A Canadian Perspective
Supplemental material, sj-pdf-1-han-10.1177_15589447231174482 for Anterior Interosseus to Ulnar Motor Nerve Transfers: A Canadian Perspective by Spencer B. Chambers, Kitty Y. Wu, Douglas C. Ross and Joshua A. Gillis in HAND
Footnotes
Authors’ Note
Spencer B. Chambers is also affiliated to St. Joseph’s Health Care, London, Ontario, Canada.
Ethical Approval
This study was approved by our institutional review board.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Informed consent was obtained from all individual participants included in the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
