Abstract
To achieve the efficient encapsulation of microencapsulated phase change materials (mPCMs) and the one-step preparation of a phase change fabric, we prepared self-adhesive phase change microcapsules. By adding sticky polymers during the preparation of the phase change microcapsules, the microcapsule preparation and thermoregulation finishing steps were integrated into a single process to realize the fast and efficient preparation of phase change thermoregulating fabrics with high energy storage density. A sticky polymer polyvinyl alcohol solution used instead of conventional water as the continuous phase medium for emulsion polymerization to synthesize sticky mPCMs in the emulsions, enabling their application in fabrics via rapid spraying. The micromorphology, chemical structure, thermal properties, dynamic heat storage and temperature regulation performance of the phase change fabrics with mPCMs were subsequently studied, revealing the successful preparation and effective adhesion of the mPCMs to the interior of the fabric. After about 10 min under simulated sunlight, the fabric treated with mPCMs exhibited a 14°C temperature buffering impact within the phase change temperature (~26°C) compared with the untreated fabric. The treated fabric also had a 4.5°C lower surface temperature upon reaching thermal equilibrium after 30 min, lead to the slower rise in the fabric temperature. Our approach simplified the preparation method into a one-step process to provide phase change thermoregulating fabrics with high heat storage density. These findings further demonstrated the potential of microencapsulated n-octadecane for thermal management applications, including automotive interiors, heat protection suits, sports clothing, thermoregulating covers for buildings and other commercial applications.
Keywords
Introduction
The increased utilization of sustainable energy while improving the power efficiency of current processes is highly desired to address current energy shortages, especially for fossil fuels. Phase change materials (PCMs) have gained considerable interest in energy storage systems due to their capacity to absorb and release thermal energy.1,2 PCMs can be used as thermal energy storage materials to regulate temperatures in various applications3,4 such as temperature-regulated textiles, 5 heat transfer mediums, 6 temperature-controlled building materials 7 and food protection. 8 As the most common application of PCMs, temperature-regulated textiles require fabrics that provide thermal comfort at a pleasant temperature, and intelligent temperature control features, in application such as automotive interiors,9,10 biomedical product storage and transportation, 11 aerospace materials and security. As PCMs cannot satisfy the demand in certain complex application scenarios, due to their temperature independence, other materials such as phase change composites (PCCs) can also be used.12,13
Among PCMs, solid-liquid phase change materials 14 are best suited for energy storage and improving latent heat capacity. 15 As the temperature approaches the melting point, heat is absorbed through the solid-liquid phase thransition. 14 Conversely, as the temperature decreases and the PCM begins to crystallize, previously accumulated heat is discharged back into the environment through the corresponding phase shift. PCMs are typically encased in shells, 16 which maintain the liquid phase within a vessel, 17 preventing liquid depletion during the phase transition. Encapsulation techniques can reduce environmental exposure to reduce degradation, boost thermal conductivity, better integrate the PCMs with materials such as fabric while preserving their functionality and properties, and prevent issues with supercooling.18–20 The mechanical properties of the PCMs are also important, as the shell must possess sufficient flexibility to bypass the solid-liquid transition and prevent cracking. The shell of the PCM generally includes materials such as methyl triethoxysilane and polyethyleneglycol diacrylate (PEGDA), water glass, urea-formaldehyde resins and silicon dioxide. 21 The composition of the core mainly depends on the desired temperature range and expected application; n-octadecane, n-hexadecane and n-eicosane, have respective melting temperatures of about 26°C, 36°C and 18°C, which approach the human comfort temperature.16,22 Most mPCMs incorporate melamine-formaldehyde and urea-formaldehyde,23,24 toluene-2,4-diisocyanate and diethylenetriamine, 25 poly (methyl methacrylate),26–28 poly(butyl acrylate), 29 high-density polyethylene30,31 or silica.32–34 Currently, mPCMs for heat-storage fabrics are produced primarily through interfacial polymerization,35,36 emulsion polymerization37,38 and in situ polymerization.39–41 These techniques employ the polymerization or condensation of monomers, oligomers or prepolymers as raw materials to create continuous dense microcapsule shells at the oil-water interface. 21
To provide thermal comfort, mPCMs were integrated with the fabrics to achieve thermoregulation. These phase change fabrics can regulate the heat balance in the local temperature of the body to provide a comfortable environment. Moreover, the fabrics can maintain temperature balance and reduce the temperature gradient by absorbing and releasing heat when the temperature changes to provide a comfortable living environment. Traditional preparation methods include dip-rolling, coating and impregnation. Xu et al. 41 used paraffin as the core material of microcapsules and urea formaldehyde resin as the shell material to prepare mPCMs via in situ polymerization, and these were then coated onto the fabrics to obtain infrared camouflage materials. De Castro et al. 42 used polyurethanes as the core material and n-docosane as the shell material to prepare mPCMs via in situ polymerization, and there were impregnated into fabrics. However, dip-rolling incorporates low amounts of mPCMs contributing to its poor performance, while the coating and impregnation methods create surface films with poor air permeability, which negatively affects the hand feel. The surface of the microcapsules is non-adhesive, necessitating the use of adhesives to mechanically attach them to the fabric. Furthermore, these methods require the encapsulating of the phase change material into the microcapsules, followed by its extraction, dispersing and finishing onto the fabric. These processes require complex experimental procedures and have high materials and equipment costs for used in practical applications. Therefore, it is of great significance to further study the facile preparation of phase change fabrics with high heat storage density and excellent temperature-regulating properties.
Herein, PEGDA phase change microcapsules with n-octadecane cores were in situ synthesized in emulsion using an emulsion component, with a phase change temperature of 30.7°C and a thermal storage enthalpy of 178.61 J g−1. The mPCMs were immediately sprayed onto fabrics to obtain phase change thermal storage fabrics, which realized the efficient encapsulation of PCMs and one-step preparation of phase change thermoregulation fabrics. The microscopic morphology, chemical structure, thermal storage performance and thermal stability of the mPCMs and phase change thermoregulating fabrics with different microcapsule contents were characterized by scanning electron microscopy (SEM), Fourier transform infrared (FTIR) spectroscopy, differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA), and the thermal regulation ability of these fabrics was measured by simulated light experiments. The prepared phase change microcapsule emulsion had small particle size, low adhesiveness and good uniformity, making it suitable for spraying. The produced thermo-regulating fabrics can slow the temperature rise, reduce the surface temperature and improve thermal comfort, with potential application prospects in flexible thermo-regulating textiles (Scheme 1).

Schematic diagram of the one-step preparation of mPCMs and their incorporation in phase change energy storage fabrics.
Experiment
Preparation of microcapsules
The PEGDA shell was selected for its exceptional thermal stability and established elasticity to effectively counteract the temperature and volume fluctuations in the encapsulated PCM during heat absorption and dispersal. n-Octadecane was chosen as the core material due to its high thermal capacity, non-corrosive nature and transition temperature (26°C–28°C) close to the human comfort temperature, making it suitable for use in functional textiles with thermoregulation applications.
In the synthesis of mPCMs by emulsion polymerization, the emulsion design was optimized to realize the rapid and efficient preparation of phase change fabrics. The traditional solution medium was replaced with 5% polyvinyl alcohol (PVA) solution, which not only enhanced the stability of the emulsion but also granted the emulsion a desired viscosity. The emulsion could then be directly sprayed onto the fabric due to its fluidity and viscosity to achieve the efficient and firm adhesion of mPCMs to the fabrics.
The emulsion consisted of n-octadecane, PEGDA monomer and 5% PVA solution at a 1/0.3/6 ratio. Liquid n-octadecane solution was added to the 5% PVA aqueous mixture at 60°C, stirred at 12,000 rpm for 3 min, mixed with a mass fraction of PEGDA monomer and further stirred for 3 min. The emulsion was then left undisturbed for 5 min, exposed to UV light and stirred at 350 rpm for 20 min. Finally, deionized water was incorporated into the resulting emulsion for dilution.
The microcapsules were about 2–20 μm in size with respective melting and crystallization temperatures of 23.8°C and 30.7°C and a melting and crystallization enthalpy of 178.61 J g−1. The encapsulation rate was 99% n-octadecane per capsule, which was close to the theoretical capsulation rate. Significant elasticity was exhibited by the integrity and non-sticky properties of the capsules, as revealed by SEM measurements.
Preparation of phase change fabrics
A polyester/cotton fabric with a unit weight of 100 g m−2 was provided by Jiangsu Beltford New Materials Co., Ltd. (Jiangsu, China) and selected as a vector to show the thermoregulating effect of microencapsulated n-Octadecane in a PEGDA shell. To prepare the sprayed fabrics, the coating was sprayed onto the fabric for 7 s from a 5 cm distance using 0.3 mm calibre airbrushes at 9 psi (Figures S1–S4, Tables S1, S2 in the Supporting Information). The concentration of mPCMs within the textiles significantly influenced their thermoregulatory capabilities. To explore this effect, fabrics sprayed with 40 and 80 wt% mPCMs were examined. These percentages represented the weight gain relative to the pure fabric, translating to actual mPCMs concentrations of 28 and 44 wt%, respectively, in the final phase change thermoregulating fabrics following the incorporation of mPCMs. The content of mPCMs in the fabrics was further substantiated through experimental validation, utilizing the enthalpy of melting data obtained from DSC and the TGA weight loss profiles, which yielded consistent results.
Characterization
A HITACHI SU8010 scanning electron microscope was employed to determine the morphology of the fabrics and mPCMs. After the samples had completely dried, the fabrics were stuck on conductive adhesive attached to the electron microscope stage and coated with gold, while the resulting emulsion was sprayed onto aluminon foil to prepare mPCMs samples. Visual stability tests were carried out by heating the samples to 60°C, above the melting temperature of the PCM n-octadecane for 30 min and cooling them to room temperature (RT). Photos of samples were taken to observe the release of the PCM through the fabric structure. Chemical characterization of the samples was performed on a NICOLET 6700 FTIR spectrometer (Thermal Fisher Scientific, USA). The mPCMs were tested using the KBr tableting technique, while both untreated fabrics and phase change energy storage fabrics were tested via all-reflective diamond ATR method. TGA was performed using a TGA 8000 instrument (Perkin Elmer Company, USA) to evaluate the thermal stability and decomposition behaviour of the fabrics. The measurements were collected in triplicate from RT to 800°C at a heating rate of 10°C min−1 in N2 atmosphere. DSC was conducted with a DSC 8500 instrument (Perkin Elmer, USA) to determine the latent heat storage properties. DSC measurements were collected from 0°C to 50°C at a speed of 5°C min−1, maintained for 5 min, and subsequently reduced to 0°C at the same velocity. To determine dynamic heat storage performance, a xenon lamp was used to irradiate the sample at 1000 W m−2 to simulate the temperature change of the fabric surface under sunlight. The irradiation was maintained for 30 min until the surface temperature of the fabric was stable and thermal equilibrium was reached. The environmental temperature was 20°C and the dimensions of the sample specimens were 5 × 5 cm. The surface temperature was measured and recorded with an infrared thermal imager (FLIR Systems, UK). The different spraying parameters, including the spraying distances and spraying time were adjusted to optimize the DSC data, thermal properties and melting enthalpy. The breathability, moisture permeability and washing resistance of the spraying, impregnation and padding methods were compared (see Supporting Information). The microcapsule cycle stability after 100 cycles and the abrasion resistance of phase change fabrics were also tested, and the DSC images are provided in the Supporting Information.
Results and discussion
Figure 1 displays the SEM images of sprayed microcapsules (Figure 1(a1) and (a2)), untreated fabric (Figure 1(b)) and fabrics sprayed with 40 and 80 wt% mPCMs (Figure 1(c) and (d)) at different magnifications. The microcapsules were first sprayed from the emulsion to study their microscopic morphology. These microcapsules were stacked and partially adhered with relatively uniform sizes (2–20 μm), which were spherical with slight dents due to the volume change of the phase change microcapsules by the solid–liquid phase transition (Figure 1(a)). Figure 1(b1) and (b2) show the surface morphology of fabrics of 1000 and 50 µm sizes, and then the concentration of mPCMs was further increased from 40 to 80 wt% to investigate the microstructure of fabrics sprayed with mPCMs. As shown in Figure 1(c) and (d), the mPCMs were extensively adhered to the fabric surface and the inter-fibres. Figure 1(c) demonstrates SEM images of the fabrics loaded with 40 wt% of mPCMs. The cotton fibres were almost covered by microcapsules, aggregated capsules were observed along the surface of the cotton filaments, and these microcapsules filled the fibre crevices (Figure 1(c1) and (c2)). As shown in Figure 1(d), the textile fibres containing 80 wt% mPCMs presented a thick coating of microcapsules, which were densely distributed in the fabric structure, and a higher number and amount of densely distributed microcapsules was observed in comparison with Figure 1(c2). Figure S5 in Supporting Information shows visible images of fabrics with different spraying rates.

SEM images of the microscopic morphology of (a) sprayed microcapsules, (b) cotton fabric, (c) 40 wt% mPCMs and (d) 80 wt% of mPCMs sprayed into fabrics at different magnifications.
The stability of the fabrics treated with mPCMs was assessed through visual melting tests. Selected fabrics sprayed with 40 and 80 wt% mPCMs and a reference prepared by spraying 40 wt% pure n-octadecane were heated to 60°C for 30 min and then cooled to RT. Compared with the directly immobilized n-octadecane (unencapsulated n-octadecane), the sprayed structure with mPCMs showed remarkable stability. As shown in Figure 2(a2), the fabrics prepared with unencapsulated n-octadecane showed the release of n-octadecane from the fabric, while the initial appearance of the fabric sprayed with 40 and 80 wt% mPCMs remained stable (Figure 2(b1) and (c1)). And from Figure 2(b2) and (c2), there was almost no visual difference between fabrics prepared with 40 and 80 wt% mPCMs after heating to 60°C and then cooling. Compared with unencapsulated n-octadecane, the encapsulation of the microcapsule shell improved the stability of these phase change materials. As shown in Figure S6, the mPCMs can also be recycled for long periods without decreasing their thermoregulation performance (Figure 3).

Visual representations of fabrics containing 40 wt% pure n-octadecane, 40 wt% mPCMs, and 80 wt% mPCMs (a1–c1) before and (a2–c2) after heating to 60°C.

FTIR spectral analysis of untreated fabric, mPCMs and fabric with 40 and 80 wt% mPCMs.
The FTIR spectra revealed peaks corresponding to mPCMs and fabrics, which were also evident on fabrics loaded with 40 and 80 wt% mPCMs. The mPCMs peaks included –CH out-of-plane bending at 717 cm−1; –CO stretching in ethers at 1118 and 1259 cm−1; deformation or –CH2 bending and alkane scissoring at 1378 and 1471 cm−1, respectively; the maximum shell elongation peak (–C=C) at 1637 cm−1, ester –CO stretching at 1735 cm−1; symmetrical and asymmetrical –CH2 stretching vibrations in methyl and methylene at 2850 and 2919 cm−1, respectively; and –OH stretching vibrations, perhaps for residual water or alcohol, at 3432 cm−1. Peaks corresponding to the pure fabric included –CH out-of-plane bending in the benzene ring at 723 cm−1; C–C bending in the benzene ring at 873 cm−1; –CO stretching in cellulose and C–O–C skeletal vibration of the pyranose ring at 1017 and 1093 cm−1, respectively; symmetrical and asymmetrical C–O–C stretching vibrations at 1241 and 1339 cm−1, respectively; –CH2 bending or scissoring at 1409 cm−1; and C=O stretching at 1713 cm−1. The two categories of fabrics containing mPCMs were nearly identical, with the distinction lying in the higher peak values of fabrics with 80 wt% mPCMs compared to those with 40 wt% mPCMs. The observed peak intensities in the sprayed fabrics closely matched those of the original fabric and mPCMs, suggesting that the phase change microcapsules had been effectively incorporated into the fabric without triggering additional chemical reactions.
The thermal stabilities of fabrics and mPCMs were determined by TGA. The weight of the pure fabric remained stable with no weight loss as the temperature increased to 320.5°C (Figure 4). The first main decomposition step occurred at about 323.1°C–498.8°C with a weight loss of 85.2 wt%, while the second step occurred in the temperature range of 498.8°C–784.4°C, with a weight loss of 11.2 wt%. The mPCMs had only one main decomposition step from 249.6°C to 350.6°C, which involved a weight loss of 98.6 wt% from the decomposition of the capsule shell and encapsulated n-octadecane. The first main decomposition step of the fabrics sprayed with 40 wt% mPCMs occurred at about 243.4°C–349.6°C with a total weight loss of 22.2 wt% from the oxidation of the encapsulated octadecane (similar to the TGA data of pure mPCMs). The subsequent weight-loss step took place from about 349.6°C to 470.6°C, resulting in a total weight loss of 62.8 wt%, attributable to the degradation of the fabric matrix. Fabrics sprayed with 80 wt% of mPCMs exhibited a similar decomposition pattern characterized by the first significant step in the range of 249.6°C–350.3°C, with the weight loss at 33.4 wt%, which was 17 wt% higher than that of the fabric sprayed with 40 wt % mPCMs due to the higher mPCM content. The second step in the range of 350.3°C–479.4°C involved a loss of 54.1 wt%, which was associated with the decomposition of the fabric. These results show that the sprayed fabrics contain the corresponding mass fraction of phase change microcapsules, further confirming the successful loading of the fabrics with phase change microcapsules.
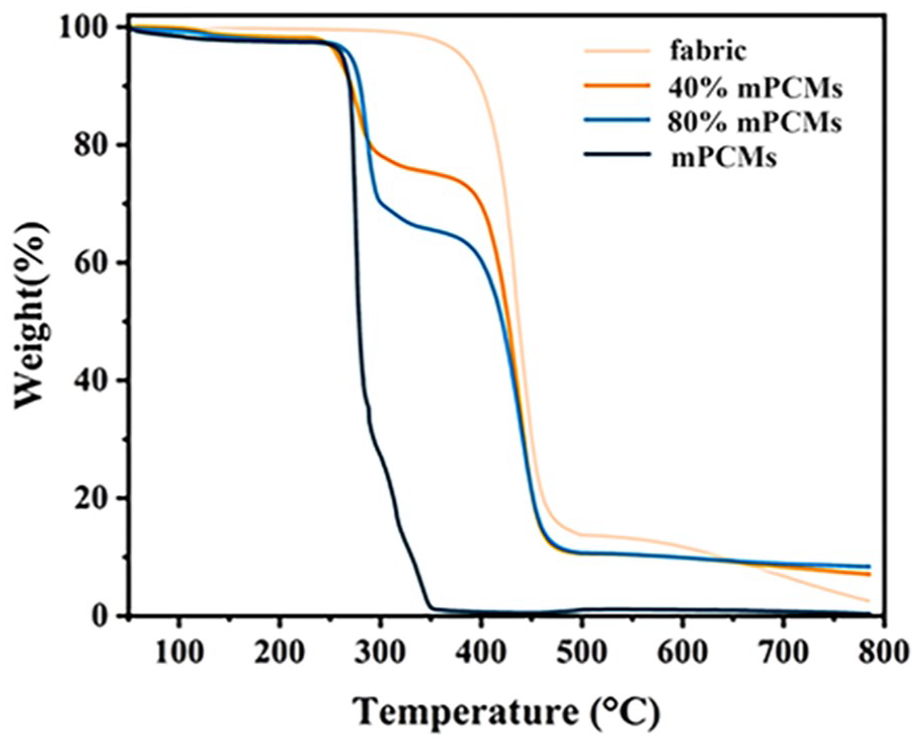
Thermogravimetric analysis of sprayed fabrics, pure fabrics and mPCMs.
DSC measurements were collected to assess the thermal capacities of the sprayed structures. The DSC thermograms for mPCMs, pure fabrics and fabrics sprayed with 40 and 80 wt% mPCMs were measured in Figure 5. The mPCMs sprayed onto the fabrics had a melting temperature of 30.7°C and a melting enthalpy of 178.6 J g−1, while the crystallization temperature was 23.8°C with a shoulder at 19°C, associated with a crystallization enthalpy of 178.6 J g−1 upon cooling. The absorption of heat and emission of energy were similar, demonstrating stable heat storage and release cycling. There was no heat absorption or release behaviour of the pure fabric during heating and cooling. The fabric sprayed with 40 wt% mPCMs exhibited a melting temperature at 29.6°C and a melting enthalpy of 48.3 J g−1 upon heating, while the crystallization temperature and enthalpy were respectively 23.6°C and 45.8 J g−1. There was an exothermic contribution during crystallization that took place below the crystallization temperature of 19.1°C, which showed as a small contributing peak corresponding to the different crystalline phases of n-octadecane. The fabric sprayed with 80 wt% mPCMs exhibited a melting temperature of 29.8°C and a melting enthalpy of 80.2 J g−1, with a crystallization temperature and enthalpy of 23.47°C and 77.9 J g−1, respectively. The DSC thermograms of the fabrics sprayed with 40 and 80 wt% mPCMs showed curves similar to those of pure mPCMs, which indicated that the mPCMs were successfully loaded onto the fabrics by spraying and attained the corresponding latent heat capacity. Compared with the pure fabric, the average latent heat values for the fabrics sprayed with 40 and 80 wt% mPCMs were 47.1 and 79.1 J g−1, respectively. Based on the mPCMs enthalpy data, thermal melting enthalpies of the fabrics prepared with 40 and 80 wt% (compared to the pure fabrics) mPCMs were 46.7 and 80.2 J g−1, respectively, which were similar to the experimental test enthalpy data of the sprayed fabrics. Therefore, the mPCMs were almost completely loaded onto the fabric and provided the fabrics with excellent heat storage performance.

Differential scanning calorimetry thermograms of pure mPCMs, untreated fabrics and fabrics sprayed with 40 and 80 wt% mPCMs.
The varying temperature patterns of untreated and 80 wt% sprayed fabrics under simulated sunlight over a 30-min heating period are shown in Figure 6. The sprayed fabric showed a temperature gap compared with the untreated fabric. At the beginning of the heating period, the temperature of the fabric with mPCMs was lower than that of the pure fabric (Figure 6(a)). In the first 5 min, the temperature of the pure fabric increased rapidly to about 40°C and then remained stable. After about 1 min, the n-octadecane entered the initial melting phase at 26°C, there was a significant temperature decrease on the fabric surface in heating speed, and a noticeable temperature variance was observed between the untreated fabric and fabric with 80 wt% mPCMs. The solid PCMs had absorbed the heat and melted upon reaching the melting point of the PCM, resulting in a significantly slower temperature increase. After about 10 min, the temperature of the sprayed fabric was nearly 27.5°C, which was 12.2°C lower than that of the pure fabric (Figure 6). The temperature variance persisted even after 30 min of melting, with the 80 wt% mPCMs fabric exhibiting a 4.5°C temperature lower than that of the pure fabric. Due to the fact that the solar reflectance of phase change fabric was higher than the pure fabric (Figure S7). The schematic diagram of this simulated sunlight test device is shown in Figure S8 of the Supporting Information. The prepared phase change energy storage fabrics possessed excellent thermal management properties, with the phase change microcapsules storing heat to slow the temperature rise caused by light exposure and effectively reducing the surface temperature of the fabrics.

(a) Dynamic temperature of pure fabric and fabric with 80 wt% mPCMs after 0, 10, 20 and 30 min and (b) dynamic heat storage measurements for pure fabric and phase change fabric sprayed with 80 wt% mPCMs.
Conclusions
In summary, we used 5% PVA solution instead of conventional water as the solution medium to produce n-octadecane mPCMs. The viscous polymers in the emulsion medium adhered to the surface of the microcapsules during spraying, enabling their effective adhesion onto the polyester/cotton fabric. The macro- and micro-morphology, chemical properties, latent heat capacity, thermal stability and thermal regulation properties of the phase change microcapsules and thermoregulating fabrics were characterized by SEM, FTIR spectroscopy, TGA, DSC and thermal imaging.
The prepared phase change microcapsules had a particle size of 2–20 μm, a phase change temperature of 30.7°C, a mPCMs thermal storage enthalpy of 178.6 J g−1 and a phase change temperature of 26°C in the phase change energy storage fabric. The mPCMs were extensively adhered to the fabric surface and the inter-fibres, which improved the mechanical properties of the fabrics. The heat capacity of the fabric treated with 40 wt% mPCMs was 47.1 J g−1, while the fabric sprayed with 80 wt% mPCMs achieved a higher heat capacity of 79.1 J g−1. Moreover, the phase change microcapsules and phase change fabrics exhibited excellent thermal stability at temperatures as high as 245°C. Compared with the untreated fabric, the fabric treated with mPCMs provided a temperature buffering effect of 14°C in the phase change temperature range. Furthermore, the fabric was about 4.5°C cooler than pure fabrics at thermal equilibrium after 30 min of simulated sunlight exposure, demonstrating its excellent heat storage and temperature regulation ability. The phase change fabrics showed low weight loss and retained their thermal properties after washing and abrasion tests, which confirmed the stability of the mPCMs coating. However, the fabrics with mPCMs had a worse hand feel than the pure fabrics and need further improvement in this regard. In summary, our study provided a new method for the facile preparation of phase change thermoregulating fabrics for practical applications, which shows great potential in energy storage and thermal management. Future research aims to focus on the study of composite inorganic materials to improve the thermal conductivity of phase change storage fabrics and the design of mPCMs with additional functionalities such as photochromic, photocatalytic and bactericidal properties for a wider range of applications.
Supplemental Material
sj-pdf-1-jef-10.1177_15589250241266976 – Supplemental material for Preparation and performance of phase change thermoregulating fabric with high heat storage density via spray coating
Supplemental material, sj-pdf-1-jef-10.1177_15589250241266976 for Preparation and performance of phase change thermoregulating fabric with high heat storage density via spray coating by Xiyan Lang, Xuexue Xiang and Jing Gao in Journal of Engineered Fibers and Fabrics
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
