Abstract
The study explores the use of pectin from lemon peel for pigment printing cotton fabric. The optimal extraction conditions were 95°C, 90 min, 1:40 MLR, and pH 2. The extracted pectin was light yellow, with a moisture content of 13.4%, volatile compound content of 82.3%, total ash content of 3.1%, fixed carbon content of 14.6%, and a pH of 3.5. The thermal stability of the pectin binder was determined to be 230°C. The study also found that pectin cross-linking was achieved through continuous extrusion using 1% citric acid and 0.3% AgNO3. The formulated pectin binder was found to improve textile pigment printing quality and performance. The rheological properties of the pectin-based and commercial binder pastes were evaluated, showing that the pectin-based binder can produce fine designs with shear thinning but retain consistency when applied force is released, resulting in defined edges in prints. The study suggests that pectin-based formulations can be used as an optional binder to commercial binders in textile pigment printing.
Keywords
Introduction
Citrus fruits are abundant worldwide, producing 1.17 × 108 tonnes of fruit every year. However, this process produces a significant amount of waste, accounting for roughly half of the fruit’s initial quantity. This waste, which includes peels, seeds, and non-edible sections, can be processed into essential oils, pectin, animal feed, and natural fertilizers. As a result, large quantities of citrus garbage are available as by-products. Peels, when treated as waste, can cause environmental problems, particularly water pollution. This challenge may have been turned into an advantage if useful by-products like pectin could have been extracted from these wastes. 1
Pectin, a naturally occurring polysaccharide present in the cell walls of fruits and vegetables, is recognized for its sticky characteristics. Lemon peel pectin, a byproduct of the citrus fruit industry, is gaining popularity as a renewable and environmentally acceptable alternative binder in a variety of industries, including paper and textile manufacturing. Citrus fruit peel pectin-based binder can be utilized as an excellent adhesive material in place of synthetic binders due to its renewability and biodegradability. 2 Pectin derived from lemon peel was utilized in fabric finishing operations and proved to be an effective binder. A pectin-based binder could be a viable alternative to synthetic binders used in textile finishing since it provides a sustainable and environmentally friendly solution for improving textile performance.3,4
Citrus peel contains 20%–30% pectin on a dry matter basis. Pectin Yield from lemon peel can range from 5.20% to 23.59% depending on the type of extraction method used and the extraction conditions. Acidic extraction using hydrochloric or citric acid, microwave, and conventional heating methods are all options. 5 The acid extraction method use hot water (60°C–100°C) acidified with a mineral acid (such as sulfuric, phosphoric, nitric, hydrochloric, or citric acid) at a pH of 1.5–3 for 0.5–6 h, involves the hydrolysis of protopectin to pectin using acids, followed by precipitation with ethanol. The typical water heating approach is time-consuming and causes pectin breakdown, therefore it has both quantitative and qualitative drawbacks for pectin extraction. To generate pectin with improved yield and quality, it is critical to investigate fresh ways or modify existing methods that avoid employing harsh extraction conditions. However, as extraction using heating has several downsides, as an applicable and creative technology, the application of ultrasound and microwave assisted extraction. 6
Lemon peel pectin-based binders have potential applications in paper and textile improving color fastness, adhesion, and resistance to washing.7,8 Pigment printing is an ancient and easy method for printing fabrics, offering adaptability, ease of application, and compatibility with almost every fabric type. It is the oldest and easiest method for printing, and most printed fabrics depend on pigment printing. Pigment is fixed to the fabric using a binding agent called a binder, which forms a continuous film enclosing the pigment molecules. The insoluble pigments are fixed onto fibers with binding agents or binders. The success of a printing process is established by the sharpness of the mark, levelness, correct color, good hand, and high fastness. Three factors are responsible for the flow properties of print pastes: viscosity, fluidity, and viscoelasticity. The viscosity and rheology of the paste are the main factors influencing the quality of print. 9
Binders are the mechanism used to keep the color on the fabric when using pigments for printing textiles. Choosing a binder for pigment coloration is a complex but critical step in developing a recipe that will meet specific requirements.9,10 Binders used in pigment printing are typically based on styrene-butadiene, styrene-acrylate, or vinyl acetate-acrylate copolymers. The most common is poly-acrylate dispersion, which contains formaldehyde and has high hardness, poor heat and cold resistance, and poor application properties.11,12 As a by-product of printing, the release of formaldehyde into the environment creates a human carcinogen compound that is hazardous to both human health and the environment. Formaldehyde is classified as human carcinogenic by the International Agency for Research on Cancer (IARC). As a result, there is a general trend in textile processing to limit or eliminate formaldehyde-based chemical goods.13,14 The search for emulsion binders’ replacement is also an ongoing research project to overcome kerosene-related issues such as the oil crisis, both in terms of cost and availability. During drying printed fabrics Curing ovens must have enough air volume to avoid exploding, which could result in a fatal accident. Also, emulsion binders are regarded to emission of a high percentage of hydrocarbons through curing exhausts which is a major issue. 15
Bio-binders, also known as biopolymers, are compounds derived from natural resources such as plants that contain monomers linked by covalent bonds to form polymeric chains. Bio-binders have low densities and are widely used in a variety of industries for a variety of reasons, including energy efficiency, ease of handling, and low costs. Bio-binders serve as the glue for various materials. Melt flow indices, impact properties, hardness, vapor transmission characteristics, coefficient of friction, and decomposition are all attributes of bio-binders. The ability of bio-binders to absorb water is also an important parameter, particularly during binder synthesis because water is a greener and less expensive solvent. 16
A wide range of natural carbohydrates such as Pectin derived from plants, specifically citrus fruits and grapefruit have been used as common binder substitutes. These organic and renewable binders were discovered to be relatively low cost, require minimal reaction time, and do not form viscous solutions that would clog sprayers, making them an appealing binder. Carbohydrate binders, such as pectin binders, have a low binding effect. 17 With the addition of an acidic component such as citric acid, a pectin-based binder with strong cross-linking activity and equal binding effect to commercial binder is most likely to be developed. 18
While preparing the citrus fruits for extraction, drying ensures long-term storage. The highest drying temperature of 95°C and above reduces drying time but may cause bioactive compound degradation. The drying process for the material is usually recommended at temperatures around 40°C–60°C and drying times of up to 2 days. It is also recommended to keep the raw material at a low humidity level of 10% to extend the storage time and achieve a small particle size of 60 μm. Small particle size results in a higher contact surface and better extraction performance. Pectic substances are extracted from citrus fruits by breaking down the polymer and dissolving it into a proper liquid. The dissolved polymer is then washed with alcohols or organic solvents to agglomerate and evaporate to obtain the product of interest.19,20
The different methods of pectin extraction from plant cell walls are Acid extraction, Direct boiling (hot water) extraction, Thermo Mechanical extraction, and Microwave extraction. Acid extraction involves extracting the pectin using acidified water (pH up to 2) and heating the solution mixture by using a water bath shaker at temperatures of 80°C. The most commonly used acidifying materials are mineral acids, hydrochloric, sulfuric, and phosphoric acids, and organic acids such as oxalic acid, ammonium oxalate, tartaric acid, and polyphosphates. Double extraction at 85°C–88°C for 1 h using a cationic resin has been reported to give higher yields and better gel strength. Commercially, pectin is extracted by treating the raw material with hot dilute water. The extraction time of mineral acid at pH of 2, 3 varies depending on raw material, type of pectin desired, and manufacturer. Thermal and microwave extraction methods are used to separate the hot pectin extract from the solid residue as efficiently as possible, with a compromise between efficient extraction and solids separation and operating cost. Powdered pectin is produced by mixing concentrated liquid from either apple or citrus with an alcohol. Microwave extraction modifies the physical properties of material tissues, breaking down the cell structure and improving the capillary porous structure.21–23
Research on the extraction of pectin from lemon peels is limited, with most studies focusing on other fruit peels. The unique composition and efficiency of lemon peel pectin require further exploration. Pectin conversion into cross-linked silver nano binder is another underexplored area. Few studies have explored incorporating pectin into metal nanoparticles for various applications. Future applications of cross-linked pectin-silver nano binders derived specifically from lemon peels include biomedical applications, food packaging, and environmental applications. However, no studies specifically focus on these potential applications, highlighting the need for further research to evaluate their performance and feasibility.
This study helps to promote sustainable practices in the textile sector. In this study, pectin from lemon peel was extracted using an acid extraction technique, specifically hydrochloric acid. Pectin was precipitated with ethanol, methanol, and acetone to see which solvent produced the most pectin. The extracted pectin was evaluated for its physical and chemical properties as a binder, and it was used for pigment printing of cotton textiles using the flat hand screen printing technique, with the printed quality of the textile samples being compared to commercial-grade binders.
Materials and methods
Materials
The materials used in this study were 100% full-bleached plain woven cotton fabric supplied by Kombolcha Textile Share Company (KTSC) and post-consumer lemon peel. The fabric specifications used in this study are listed in Table 1 below.
Fabric specification.

Chemicals and colorants
The chemicals and solvents used in this study are all laboratory-grade reagents. To extract pectin and create a bio-binder, hydrochloric acid, ethanol, methanol, acetone, silver nitrate, and magnesium chloride were used. Cotton fabric printing uses pigment, thickener, binder, sodium carbonate, sodium sulfate, urea, and a cross-linking agent. Except for the synthetic binder, which was replaced by the pectin-based bio-binder for the comparison study, all auxiliaries, and chemicals used in pigment printing with extracted pectin binder will be used. C.I. Pigment Red 146 was used as a pigment throughout the study.
Equipment/apparatus and machines
Whatman universal filter papers, laboratory scale IKA MF 10 Basic Microfine Grinder, which is capable of achieving particle sizes of 60 μm, Advantech Sonic Sifter sieve analyzers small laboratory scale, which are capable of analyzing small particle sizes down to 1 μm, the Thermo Fisher Scientific oven dryer FLASH 2000 model for determining the moisture content of pectin according to the ASTM D6304 procedure, Coperion ZSK series twin-screw extruder was used for reactive extrusion of pectin, Silk screen printing, Brookfield model DV-111 Rheometer for testing Rheological property of pectin binder, Datacolor 850 spectrophotometer for testing color strength of printed fabrics, Testex Launder-O-Meter machine for wash fastness testing of the printed fabrics, the SDL Atlas Crockmeter 2010 for testing the rubbing fastness of the printed fabrics, TESTEX TF415 Perspirometer for evaluating the colorfastness of fabrics to perspiration, and light fastness tester were used in this study. For controlling RH% dehumidifiers and air conditioners were used. The physical properties of the printed fabric were tested using the Universal Tensile Strength Tester 3101, Digital Elmendorf tear strength tester, FX-3300 Air Permeability Tester, and S.D.L Stiffness Tester. Thermal analysis was performed using a Perkin Elmer TGA 4000.
Experimental set-up
Collection and preparation of materials
Post-consumer lemon peels were collected from Kombolcha city’s small and medium juice-making enterprises and thoroughly washed with tap water to remove impurities and dirt on the surface. The lemon peels were then exposed to sunlight to dry before being ground into a fine powder. Using dehumidifiers and air conditioners, the ground lemon peels were sieved and stored at a relative humidity of 10% (Figure 1).

Sample material preparations.
Determination of particle size
The particle size of ground lemon peel powder was determined using a sieve analyzer. A mechanical sieve shaker was used in this study to determine the particle size of a material by shaking or vibrating it. The particle size of the fine powder was 60 μm.
Extraction of pectin
Pectin was hydrolyzed from 10 g of post-consumer lemon peel powder using hydrochloric acid, that is, acid hydrolysis, with MLR (lemon peel powder weight to water volume (W/V)) ratios of 1:20, 1:30, and 1:40, extraction temperatures of 30°C, 60°C, and 95°C, extraction times of 40, 60, and 90 min, and pH levels of 1, 2.5, and 5, pH adjusted with trace hydrochloric acid addition. At the end of the extraction time; to remove the solid residues of the lemon peel from the solution, the mixture of water, acid, and lemon peel powder was filtered using universal filter paper. The filtered solution was washed with 20ml of ethanol, methanol, and acetone to precipitate pectin from the solution after it had cooled to 50°C. The mixture of the filtered solution and the solvents was kept at room temperature for around 5–10 min to allow the pectin to fully precipitate from the solution. The precipitated pectin agglomerates were carefully separated and dried overnight in an oven dryer at 50°C. The dried pectin was then crushed and weighed with an electronic weighing balance. Finally, the Pectin Yield percentage was computed using equation (1) below. The extraction factors were optimized using an OFAT (changing one factor at a time) design of the experiment based on their impact on the dependent Pectin yield percentage.

Pectin cross-linking and
Pectin cross-linking was performed using citric acid as a cross-linking agent to form strong bonds between pectin molecules, resulting in the formation of a gel-like structure. The procedure was carried out in a twin-screw extruder at a rate of 5 kg/h and a temperature of 150°C. The extracted pectin was solubilized using a continuous reactive extrusion method, which turned the very high solid paste into a thermoplastic melt phase before cross-linking and sizing the solubilized pectin molecules into nanoparticles. The Pectin Solution was made by dissolving Pectin powder in water and continuously swirling it to ensure optimum solubility and homogeneity. The solution’s pectin content was optimized to 1%, which is the ideal range for pectin solutions in pigment printing binders. To make 1 L of pectin solution, 10 g of pectin was used, which is 1% of 1000 mL. Because 1 L of 1% citric acid equals 0.01 × 1000 mL, 10 mL citric acid was used to initiate the cross-linking reaction between pectin molecules. For the pectin silver nano binder, a 1:0.3 ratio of pectin to silver nitrate was utilized. As a result, the amount of silver nitrate (AgNO3) is 0.3% of 10 or 3 g. 10 g pectin, 3 g silver nitrate, and 10 mL citric acid dissolved in 990 mL water. The pectin solution containing citric acid and AgNO3 was fed into the twin-screw extruder hopper. The rotating screws in the hopper provided the mechanical energy required to mix and homogenize the paste, and the heat generated by frictional forces and heating elements within the barrel raised the paste’s temperature to the melting point of pectin. In the twin-screw extruder hopper, citric acid reacted with pectin molecules, resulting in cross-linking and the creation of a gel-like structure. Extruding the cross-linked gel-like pectin paste with AgNO3 provided vigorous mixing and shearing between the two components. The extruder’s strong mixing and shearing broke down solid particles, resulting in the breakdown of pectin nanosilver in the liquid phase. A silver nano pectin binder was created as a result. AgNO3 was used to enhance textile pigment printing by catalyzing crosslinking, improving pigment adhesion, and enhancing wash fastness and color strength, thus enhancing the quality and performance of pigment-printed fabrics.
Chemical composition of the extracted pectin
Determination of moisture contents
The moisture content of the pectin samples was determined using the ASTM D6304 procedure. 5 g of the sample was weighed and dried in an electric oven at 105°C for 12 h to a constant weight. The weight loss was calculated as a percentage of moisture (%). The percentage of moisture content was calculated using equation (2).

Determination of ash contents
By igniting the sample in a muffle furnace, the ash content was determined. 2 g of the sample was weighed and burned in a 600°C furnace for 5 h, after which the remainder was weighed and the percentage of ash content (%) calculated. The ash content was determined using equation (3).

Where W1 is the weight of the moisture-free sample before ignition in grams (g), W2 is the weight of the sample after ignition in grams, and Wn is the oven-dry weight of the sample in grams.
Volatile matter
The volatile matter was determined using a muffle furnace. To determine the volatile content, a known amount of dried sample (10 g) was placed in a closed crucible and placed inside the muffle furnace at 660°C for 5 min. The sample’s weight loss was determined, and the percentage of volatile matter was calculated using equation (4).
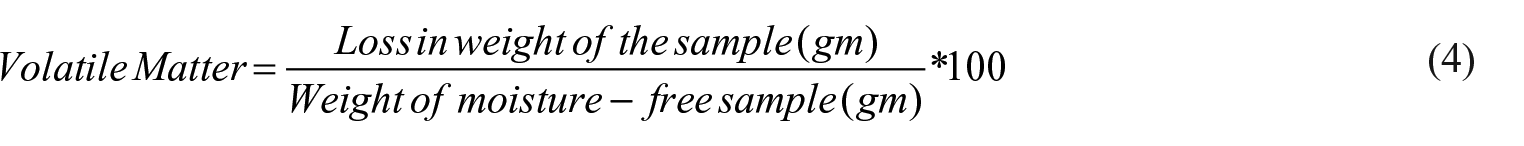
Fixed carbon
Samples’ fixed carbon was calculated by subtracting the sum of their ash content (%) and volatile matter (%) from 100. The residue left after removing the volatile matter and ash from the substance which is the fixed carbon was determined using equation (5) below.
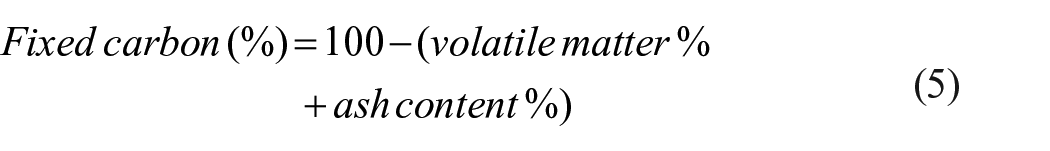
Water absorbency of binder films
By pouring the binders into Petri dishes and allowing them to dry in the air, films of pectin-based binder and commercial binder were created. The formed films were cured for 4 min at 140°C before being allowed to cool for 24 h at room temperature. The weight (W) of each binder film was determined. The films were then immersed in distilled water at room temperature for 6, 12, 18, and 24 h. After removing the surface water with filter paper, the swollen film was reweighed (W1). The water absorbency (%) was calculated using equation (6).
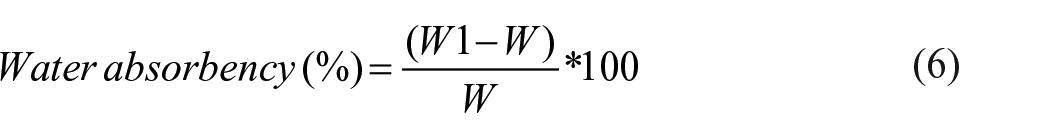
Where w is the original sample weight and w1 is the swollen sample weight.
Rheological properties
The rheological properties and apparent viscosity of prepared binders were measured at 25°C using a procedure previously described in previous research 24 by dissolving the purified samples in distilled water at a concentration of 3% (v/v) on a Rheometer, Brookfield model DV-111, at different shear rates. The apparent viscosity was calculated using the following equation (7).

Where η = apparent viscosity in poise, τ = shearing stress (dyne/cm2), D = rate of shear (s−1)
Thermal analysis
The thermal behavior of the extracted pectin was investigated using thermo gravimetric analysis (TGA 4000 Perkin Elmer). A sample weighing 10 mg was analyzed in a nitrogen atmosphere with a gas flow rate of 20 mL/min, a heating rate of 10°C/min, and a temperature range of 25°C to 600°C. TGA analysis was used to determine the curing temperature of the prepared pectin-based binder.
Formulation of the pigment printing paste
Printing pastes preparation
The printing pastes were made according to the recipe in Table 2. Urea, monobasic ammonium phosphate, and the binder were combined. The thickener was then added, and the paste was stirred for 10 min with a high magnetic stirrer to allow full viscosity to develop. The pigment was then added to the mixture and stirred for 15 min with a high magnetic mixer. The pectin-based binder was added to the pigment paste in various amounts (2.5%, 5%, 7.5%, 10%, 12.5%, 15%, 17.5%, 20%, 22.5%, 25%, 27.5%, and 30%).
Formulation of the printing paste.

Printing technique
The flat-hand screen printing technique was used to apply all printing pastes to the cotton fabrics. The printed fabrics were then air-dried before being thermally treated in a thermostatic oven.
Print fixation
The printed fabrics were air dried before being fixed at different temperatures (120°C, 130°C, 140°C, and 150°C) for different periods (2, 3, 4, and 5 min) in a thermostatic oven to fix the color through the polymerization process.
Washing
The pigment-printed fabrics were washed before the color strength test. Washing pigment printed materials before evaluating color strength offers trustworthy findings by eliminating excess pigments, maintaining consistent colorant dispersion, and simulating real-world usage conditions. The pigment-printed fabrics in this investigation were washed and agitated in cold water with a moderate detergent. Before draining, the agitated materials were immersed in a detergent and cold water solution for 10–15 min. To remove any soap residue, the wet goods were thoroughly rinsed with clean water. Finally, the surplus water is squeezed and the fabric is air dry completely before measuring color strength Avoid sunlight or heat.
Measurement of color strength
Color measurement values of dyed samples were measured on a Color data 850 spectrophotometer (D65/10°), and color yield expressed as K/S of colored samples was calculated using the Kubelka-Munk equation; see equation (8) below.

Where R is the decimal fraction of the light reflectance of the dyed fabric at 700 nm. K is the absorption coefficient and S is the scattering coefficient. 25
Determination of fastness properties
ISO 105 C06 2002 method on launder-O-meter was used to assess the wash fastness. The change in color and degree of staining was evaluated using geometric gray scales. The light fastness was evaluated with a light fastness tester according to ISO 105 B02 standard method and the rubbing fastness on the crock-meter was evaluated according to ISO 105 F09 2009 method. Perspiration fastness was tested according to ISO 105F10 standards.
Physical properties of the printed fabrics
The printed fabric’s stiffness was tested using the ASTM D1388-2007 standard method. The fabric sample’s tensile and tear strength were determined using the ASTM D5035-95 and ASTM D1423-83 standard test methods, respectively. The Universal Strength Tester was used to determine the tensile strength of the fabric in both the warp and weft directions. The air permeability was tested by ASTM D737-2004 standards. To achieve a specified air pressure difference between the two fabric surfaces, the rate of air flow passing perpendicularly through a specific area of fabric in a unit of time was adjusted. This air flow rate aids in determining the air permeability of the fabric samples.
Result and discussion
Optimization of extraction conditions
The pectin extraction conditions were optimized by holding all other variables constant and varying only one variable at a time. The goal of numerical optimization was to maximize pectin yield by taking each response value into account. The goal was to maximize the yield of the investigated independent variables within the given interval.
According to the findings of this investigation, the optimal extraction conditions were 95°C extraction temperature, 90 min extraction time, 1:40 MLR, and pH 2. A higher percentage yield of pectin was obtained under these optimized extraction conditions. The percentage of pectin yield was calculated using the above-mentioned equation (1). It was also discovered that ethanol precipitation produced a higher pectin yield than other solvents (methanol and acetone).
Pectin color
To determine the color of the pectin, a qualitative test was performed. The dried pectin samples were visually examined, and the color of the pectin is shown in Figure 2. The extracted pectin color is light yellow except Some of the acetone precipitated pectin appeared dark yellow (see Figure 2).

The Extracted wet pectin.
Chemical analysis
At 30°C, that is, room temperature; the pectin had a moisture content of 13.4%, a volatile compound content of 82.3%, a total ash content of 3.1%, a fixed carbon content of 14.6%, and a pH of 3.5. 30°C is chosen as a standard temperature for determining pectin’s chemical properties due to its biological relevance, controlled experimental conditions, and practicality. It is within the range of biological systems, allowing optimal activity of enzymes and other biological processes. 30°C is also easy to achieve and maintain in a laboratory setting, ensuring consistency and reproducibility in results. The results showed that the amount of ash content in pectin was small, indicating that the ash content will not have a significant impact on the pigment printing paste or process, as confirmed by smooth printing without any issues. Because of the high amount of hydrocarbons in the pectin, the material has a highly volatile compound and moderately fixed carbon content. Table 3 shows the chemical compositions of the pectin extract that were determined.
Chemical composition of pectin.

Water absorbency of binder films
The film formed by the pectin-based binder was clear, of even thickness, smooth, and neither too hard nor soft. It was elastic and had good adhesion to the substrate without being tacky.
Figure 3 depicts the water absorption of the extracted soluble pectin film and commercial film. The water absorption of the pectin-based binder film is greater than that of the commercial binder, as shown in this figure. This could be explained by the presence of polar groups along pectin macromolecules, specifically -OH and -COOH groups, which can form hydrogen bonds with water molecules. 26 It can be seen that the reaction is catalyzed under acidic conditions, with the binder having a pH of 5.9, and that it is reversible. Because water is a by-product of the forward reaction that leads to the fixation of the binder onto the fiber, the removal of water promotes this reaction. Curing with fresh air at high temperatures effectively removes this product and ensures the formation of the fiber-binder bond. The functional groups, specifically -OH and -COOH groups, contribute to the gelling, thickening, and stabilizing abilities of pectin, as well as its interactions.

Water absorption of pectin-based binder (PBB).
Rheological properties
The rheological properties of the commercial binder (CB) and pectin-based binder (PBB) were assessed and summarized in Table 4 and Figure 4. The rheological properties of the pectin-based binders and their viscosity are responsible for controlling dye penetration, depth of shade, sharpness of the print, and levelness, so it is of great interest to investigate the rheological properties of the prepared binders. The rheological properties and viscosity of the prepared binder are shown in Figure 4 and Table 4. Both samples examined exhibited non-Newtonian pseudoplastic behavior, with the up and down flow curves coincident.
The apparent viscosity of the pigment pastes containing the commercial binder, Pectin-based binder.


Rheological properties of pectin-based and commercial binders.
The apparent viscosities at different rates of shear were calculated using the shearing stress and rate of shear values. Table 4 displays the obtained data.
The chosen pastes were classified as a non-Newtonian pseudoplastic solution, as the relation between the shearing stress and rate of shear is not linear and the up and down flow curves are coincident. It can be seen that as the rate of shear increases, the apparent viscosity decreases (pectin-based binders show thinning down). The pectin-based binder’s rheogram curve was shifted away from the rate of the shear axis, indicating an increase in apparent viscosity. Non-Newtonian pseudo plastic behavior in a pectin-based binder as that of commercial binders indicates similar flow characteristics under different conditions. This behavior reduces viscosity as the shear rate increases, leading to shear-thinning. When up and down flow curves coincide, binders exhibit reversible behavior, returning to similar viscosity levels. 27 This rheological behavior affects processing and performance in binder applications. It facilitates processing, provides stable performance, and offers shear-thinning properties for uniform coverage. This allows for fine screen gauges to give fine designs where the pseudo shows shear thinning, but when the applied force is released, the pastes retain the consistency on the surface of the fabric and give prints with defined edges. Varied locations of rheograms show differences in viscosity as it is illustrated in Table 4 and Figure 4, and it is clear that printing pastes containing the prepared pectin-based binder are characterized by the same rheological properties as those containing the commercial binder.
Thermal analysis
The thermal properties of the prepared pectin-based binder were evaluated using Thermogravimetric analysis, as shown in Figure 5. Thermal characterization of the prepared binder using TGA is required to understand the nature of thermal dissociation of the pectin-based binder at drying and curing temperatures.

Relation between the decomposition temperature and weight loss by TGA.
The TGA curves in Figure 6 showed an initial small mass loss of around 100°C, which could be attributed to absorbed moisture evaporation from the pores of extracted pectin. Another significant weight loss occurred between 100°C and 172°C due to evaporation of volatile matter. Weight loss was observed between 232°C and 425°C due to macromolecule degradation of extracted components.

Effect of binder content in printed paste on color strength of printed cotton fabric.
Finally, above 536°C, there is degradation and conversion to ashes. It can be concluded that the extracted pectin binder can withstand temperatures of up to 232°C without losing weight. Figure 5 shows that the thermal stability of the pectin binder increased gradually with increasing temperature, which could be explained by the presence of various active groups in the PBB, such as carboxylic and hydroxyl groups, which generate new crosslinks that prevent degradation at higher temperatures. In this study Pectin’s carboxylic groups underwent esterification reactions with silver nitrate, resulting in the creation of nano pectinate particles, and because of the crosslinking between pectin molecules, the pectin-based binder has enhanced heat stability.
Effect of binder content in printing paste on color strength
The effect of binder content in printing paste was investigated. Figure 6 depicts the effect of binder (1%–30%) in printing paste on the color strength of printed cotton textiles containing pigment. Color strength decreases as the binder percentage in the printing paste increases until it reaches 15%. It was also discovered that as the binder content increased by 15%, the color strength value decreased. However, there is no statistically significant difference in color strength values between 12.5% and 15% binder content. As a result, the 12.5% binder content was chosen as the optimum binder concentration after considering binder cost, color strength, and effect on the fabric handle. As a result, it can be concluded that the optimal binder concentration was 12.5%.
Determination of the optimum fixation conditions
Figure 7 depicts the color strength values of printed fabrics at various fixation temperatures and times. The fabric printing technique was used, and the goods were dried in the air before being subjected to thermal treatment at various temperatures (120°C, 130°C, 140°C, 150°C) and time intervals (2, 3, 4, 5 min). According to the results, the pigment-printed cotton fabric had the highest color strength at a fixation temperature of 140°C for a fixation time of 3 min. It was also discovered that above the fixation temperature of 140°C, the color strength value of the printed fabric sample begins to decrease as it is shown in Figure 7. The relatively higher fixation temperatures of 140°C enhanced the binding capacity of the pectin binder with the cotton fibers improving the adhesion of the pigment to the fabric, which increased the percentage of fixation of the pigment on fabrics. Also, the relatively higher fixation time of 3 min provides a prolonged fixation time for the adhesion of the pigment ink to the cotton fibers, resulting in improved color strength. However excessive fixation temperature of more than 140°C and excessive fixation time of more than 3 min reduces the color strength of the lemon peel extracted pectin binder in pigment printed fabrics. Fixation temperature above 140°C can lead to the degradation of pectin, resulting in reduced binding efficiency and weaker adhesion of the pigment to the fabric. This can cause color strength. At a Prolonged fixation time above 3 min, there may be a possibility of color shift or change in the pigment’s original shade. The prolonged heat exposure during fixation may alter the pigment’s behavior, resulting in a different color outcome and reduced color strength than intended.

Effect of fixation temperature and time on color strength of printed cotton fabric.
Comparison of color strength of the different binder
The effect of pectin-based and commercial binders on the color strength values of pigment-printed cotton fabrics was evaluated and presented in Table 5. Three different sample fabrics were printed with different binders (pectin-based binder, mixture of 50% pectin-based binder, and 50% commercial binder, commercial binder) and the K/S values were determined to study the effect of binder type on color strength.
Comparison of K/S value of the different binders.

Effect of binder content on flexural rigidity and rubbing fastness
The influence of binder content in printing paste was studied. Table 6 shows the rubbing colorfastness and flexural rigidity (bending stiffness) of cotton fabrics printed with different binder concentrations. The binder concentration has a significant effect on the dry and wet rubbing fastness. The rubbing color fastness increases as the concentration of binder in the printing paste increases. This could be attributed to the binder’s increased cross-linking of the pigment on the fibers.
Effect of binder content on flexural rigidity and rubbing fastness.

According to the color strength assessment, the best performance of printed cotton fabrics was obtained with a 12.5% pectin-based binder and a curing temperature of 140°C for 3 min on cotton. When better color strength was obtained, pigment printing fabrics using the prepared binder performed well in dry and wet rubbing fastness tests. The printed cotton fabrics scored 4 and 3/4 in dry and wet rubbing fastness at this optimum binder concentration. Under the natural mild acidic pH and curing temperature, the binder with self-crosslinking groups aids in crosslinking and improves fastness properties. Crosslinking improves rubbing and washing fastness while degrading the fabric’s handle.
Table 6 shows that increasing the binder content in the printing paste increased the bending stiffness of the printed fabrics. Binder concentration increased fabric bending length and flexural rigidity in both the warp and weft directions. Bending length has a strong linear relationship with fabric flexural rigidity. In general, this study concludes that binder concentration has a significant impact on fabric handles.
The increase in flexural rigidity indicates that the fabric became slightly stiffer as a result of the pectin-based binder treatment of cotton. Furthermore, the bending stiffness of a fabric is highly dependent on its thickness; the thicker the fabric, the stiffer it is if all other factors remain constant.
Fastness properties
Commercial binder with a viscosity of 20 g/100 g poise has a higher viscosity than pectin-based binder with a viscosity of 12.5 g/100 g poise. To compare, equal amounts of paste from both binders were used to print cotton fabric samples. Equation (8) was used to calculate the amount of paste required to obtain 2 g of binder.
Table 7 shows the rating of color fastness to washing in terms of color change and staining on white. The fastness of samples printed with pectin-based binder and commercial binder was found to be superior to that of samples printed without binder (WB). Wash fastness was almost comparable when using commercial binder (CB) and pectin-based binder. This could be due to the strong H-bond reaction between pectin as a binder and the cotton fabric’s – OH group.
Comparison of color fastness to washing, light, and perspirations.

Commercial binder with a viscosity of 20 g/100 g poise has a higher viscosity than pectin-based binder with a viscosity of 12.5 g/100 g poise. To compare, equal amounts of paste from both binders were used to print cotton fabric samples. A viscosity measurement of “20 g/100 g poise” indicates a ratio of 20 g of a substance per 100 g of solution or paste and the 15 g/100 g poise indicates a ratio of 20 g of a substance per 100 g of solution or paste in terms of poise, which is a unit of dynamic viscosity. To calculate the amount of paste required to obtain 2 g of binder from the two types of binders with differing viscosities, equation (9) was employed.

As per equation (9) above 10 g of Commercial binder (20 g/100 g poise Paste) and 16 g of Pectin based binder (12.5 g/100 g poise Paste) were used to ensure that 2 g of binder in each case used for comparison.
According to the results in Table 7, cotton fabrics printed with a pectin-based binder and commercial binders have excellent light fastness ratings of 6–7 and 7, respectively, while cotton fabrics printed without binders only have a good light fastness rating of 4. According to the findings, light fastness is determined by the types of pigments rather than the types of binders.
Acidic and alkaline perspiration were measured in both pectin-based and commercial binder samples. As can be seen, the perspiration fastness of the PBB and CB binder printed sample fabric was nearly comparable.
Physical properties of the printed fabrics
Effect on flexural rigidity
The effect of different pectin binder concentrations on the flexural rigidity (bending stiffness) of the printed cotton fabric was presented and compared in Figure 8 as a value of the addition of warp (W) and weft (S) direction measurements. It demonstrates that these factors have a significant impact on the flexural rigidity of printed cotton fabrics. The stiffness of the printed samples increased as the binder concentration increased. Figure 8 shows and compares the effect of different types of binder on the flexural rigidity of printed cotton fabric.

Flexural rigidity of the printed cotton fabrics.
In both cases, commercial binder and pectin as a binder increase the flexural rigidity of printed samples when compared to control samples. This could be due to the increased stiffness. When the flexural rigidity of pectin-based binder and commercial binder printed cotton fabrics were compared, the commercial binder printed samples had a higher flexural rigidity. This means that the cotton sample printed with a commercial binder is stiffer than the sample printed with a pectin-based binder.
Effect on tear strength
The tear strength of printed cotton samples decreased with the addition of binder (commercial binder and pectin-based binder), as shown in Figure 9. This could be due to increased cross-linking of the binder with the substrate, which reduces yarn slippage and increases fabric flexural rigidity while decreasing tear strength. The tear strength of sample fabrics was greater in the warp direction than in the weft direction in all samples.

Comparison of tear strength of cotton fabrics concerning binder used.
The tear strength of the pectin-based binder and the commercial binder printed fabric is not significantly reduced.
Effect on tensile strength
As shown in Figure 10, the tensile strength of printed cotton samples decreased in the warp and weft directions with the addition of binder (commercial binder and pectin-based binder). This could be due to increased cross-linking of the binder with the substrate, which reduces yarn slippage and increases flexural rigidity while decreasing tensile strength. The tensile strength of the unprinted samples (control) was greater than that of the printed sample fabrics. Cotton fabric samples printed with pectin-based nanoparticle binder showed lower than samples printed with commercial binder. This could be because the commercial binder is stiffer than the pectin-based binder.

Comparison of tensile strength of cotton fabrics concerning binder used.
Air permeability
Figure 11 depicts the air permeability of printed cotton fabrics with commercial binders and pectin as a binder. The rate of air flow passing perpendicularly through a known area under a specified air pressure differential between two surfaces of a material is referred to as air permeability. The amount of air passing per second through a cm2 of fabric is expressed as air permeability. Fabrics printed with pectin-based binders and commercial binders are known to reduce the air permeability of cotton fabrics. The decrease in air permeability is expected because the printed portion will have binder and pigment filling the gaps between the fabric’s warp and weft. Furthermore, pectin-based binder-printed cotton fabrics have higher air permeability than commercial binder-printed fabrics.

Comparison of air permeability cotton fabrics concerning binder used.
Conclusion
In this study, pectin was extracted from lemon peel using acid hydrolysis methods. The extraction conditions were optimized using the OFAT experimental design in this extraction technique. The optimal extraction conditions were determined to be 95°C extraction temperature, 90 min extraction time, 1:40 MLR, and pH of 2. A higher percentage yield of pectin was obtained under these optimized extraction conditions. It was also discovered that ethanol precipitation produced a higher pectin yield than other solvents.
The chemical composition of pectin was determined, and a moisture content of 13.4%, volatile compounds of 82.3%, total ash content of 3.1%, fixed carbon of 14.6%, and pH of 3.5 at 30°C were obtained. The water absorption of the pectin-based binder film is greater than that of the commercial binder. The effect of binder content (1%–30%) in printing paste was investigated. 12.5% binder content was chosen as the optimum binder concentration after considering binder cost, color strength, and effect on the fabric handle.
This discovery demonstrated that as the concentration of binder in the printing paste increased, so did the rubbing color fastness. Binder concentration increased fabric bending length and flexural rigidity in both the warp and weft directions. The wash fastness of the commercial binder (CB) and the pectin-based binder were nearly comparable. Cotton fabrics printed with pectin-based and commercial binders had lightfastness ratings of 6–7 and 7, respectively. As can be seen, the perspiration fastness of the PBB and CB binder printed sample fabric was nearly comparable.
Commercial binder-printed samples had higher flexural rigidity than pectin-based binder-printed fabrics. The addition of binder reduced the tear and tensile strength of printed cotton (with commercial binder and pectin-based binder). It was discovered that printed cotton fabrics with no binder have the highest air permeability, whereas fabrics printed with pectin-based binder and commercial binder have the lowest air permeability in cotton fabrics.
In textile pigment printing, pectin-based binders can be used as an alternative to commercial binders. When the stability of pectin-based binders is compared to that of commercial binders; the stability of pectin-based binders decreases over time. Commercial binders are intended for increased stability and shelf life, as well as for long-term performance. Further research into the idea of substituting pectin-based binders for synthetic commercial binders, including stability testing and application requirements, is encouraged.
Footnotes
Acknowledgements
The authors wish to thank Kombolcha Institute of Technology (KIOT), Wollo University for financial support of the research, and Kombolcha Textile Share Company (KTSC) for providing the laboratory facilities.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
