Abstract
Nanomaterials are favored choices for antibacterial applications. ZnTiO3 nanoparticles, characterized by advantageous properties like small size, large surface area, and unique physicochemical attributes, represent a novel class of antibacterial active materials. In this study, we synthesized and characterized ZnTiO3 nanoparticles using the molten-salt method (MSS). The effect of various parameters, such as reaction temperature, size, and purity of products, was investigated. Additionally, the impact of ZnTiO3 nanoparticles on antibacterial activity in natural fabrics was studied. The samples were characterized by X-ray diffraction (XRD), Field Emission Scanning Electron Microscopy (FESEM), and X-ray Energy Dispersive Spectroscopy (EDS). The XRD results revealed that the particles exhibited high phase purity at 100% and good crystallinity. A crystalline pure perovskite phase of ZnTiO3 was formed at 700°C. The particle size of ZnTiO3 powder at the optimum temperature is 119 nm with a spherical-like shape. The modified fabric with ZnTiO3 and B7 demonstrated an excellent reduction of 100% in bacterial colonies. The results of this study demonstrate that ZnTiO3 synthesis using MSS is a simple and environmentally friendly method for achieving antibacterial activity in natural fabrics.
Introduction
The fabric industry is experiencing consistent growth due to the development of new products that meet users’ expectations, expanding the value of fabric applications across various fields. Among the array of functional fabrics available, those with antibacterial qualities appear to be in high demand. Antimicrobial materials play a crucial role in preventing the reproduction and spread of germs. They find applications in disposable fabrics like aprons, hats, surgical drapes, and wound dressings, as well as in reusable materials such as bed sheets, towels, and work clothes. 1 Nanomaterials are considered the top priority of advanced materials providing miscellaneous novel applications such as innovative fabric compounds, food processing, and agricultural production to sophisticated medicinal techniques.2,3 Nanomaterials, leveraging their high surface-to-volume ratio (S/V) and the potential quantum effect, have found applications in inhibiting the growth of microorganisms in the fabric industry. 4 Zinc oxide (ZnO) and titanium dioxide (TiO2) are primarily characterized by their size, composition, crystallinity, and morphology. The reduction in the nanoscale size of ZnO and TiO2 can alter their chemical, mechanical, electrical, structural, morphological, and optical properties. 5 Interestingly, several studies report that ZnO and TiO2 nanoparticles are non-toxic to human cells and exhibit antibacterial properties.6,7 Furthermore, these nanoparticles are utilized for antibacterial activities in both the fabric and food industries.8,9 ZnO showed the strongest antibacterial activity against Escherichia coli and Staphylococcus aureus compared to microparticles, attributed to its high specific surface area. 10 Additionally, TiO2 nanoparticles, known for their high photocatalytic activity, are employed as photocatalysts, resulting in a self-cleaning effect on cotton fabric.10,11 Zinc titanate (ZnTiO3; ZT), derived from the combination of ZnO and TiO2, forms a perovskite material with versatile applications, including microwave dielectric, photocatalysis, ceramics, paint colors, CO & NO sensors, and photoluminescent devices. 12 Research by Rheima et al. 13 revealed that ZT possesses strong antimicrobial activities against high concentrations of Bacillus subtilis and Acinetobacter baumannii. In a study by Ruffolo and La Russa, 14 ZT nanopowders demonstrated high biocidal effectiveness as an antimicrobial coating for stone, particularly against Aspergillus niger. Various methods employed for synthesizing nanomaterials include sol-gel, combustion, solvothermal, hydrothermal, sonochemical, microemulsion, polyol, coprecipitation, and molten salt synthesis (MSS).2,15,16 MSS, introduced for producing metal oxide nanomaterials, offers advantages such as simplicity, environmental friendliness, reliability, scalability, and generalizability. 15 It can be seen as an enhancement of the solid-state reaction method (SSR) by using salt as a solvent once melted. This assists in dissolving solid reactants and solvating ions through intense polarization and the rapid movement of reactant species via convection and diffusion. The molten salts contribute to achieving phase-pure products with homogeneous powder characteristics and controlled size/shape, all at relatively low temperatures compared to the SSR. 17 To the best of our knowledge, no previous study has synthesized Zinc titanate using MSS and applied it to antibacterial performance on natural fabrics. Hence, the objective of this study is to synthesize and characterize ZT powder with a perovskite structure using the molten salt method, which proves to be a simple and environmentally friendly approach for imparting antibacterial activity to silk fabrics. The study investigates the impact of calcination temperature on the phase structure and microstructure of ZT powder. The optimal condition for ZT is determined for coating on natural fabric. Additionally, the study explores the effects of ZT powder on color measurement, color fastness, and antibacterial activities of fabrics.
Materials and methods
ZT powder preparation
ZT nanocrystalline powder was prepared by using the molten-salt method. Raw materials such as ZnO (99.9%), TiO2 (99.8%), KCl (99.8%), and NaCl (99.0%) were used to prepare. First, the oxide powders were weighed according to the stoichiometric proportion of the ZT composition. Subsequently, these powders were ball milled in alcohol for 24 h. After drying, the mixed powder was mixed with a KCl-NaCl salt in a 1:1 ratio 18 and calcinated between 600°C and 900°C for 2 h. Then, the calcined powder was washed with hot deionized water until there was no Cl- and then dried in an oven at 120°C.
The Streptomyces strain B7 cultivation and dye extraction
Streptomyces dyed is a pigment produced by bacteria of the genus Streptomyces. The Streptomyces strain B7 yields a purple-red powder, imparting a pink color to fabrics. To cultivate Streptomyces strain B7, sodium caseinate agar (SCA) was used as the medium, and the culture was incubated at 30°C for 5–7 days. The detailed procedures for dye production 19 and extraction have been previously presented and are summarized here. Initially, 30 g of broken-milled rice was soaked in tap water within a 250 ml Erlenmeyer flask at room temperature for 30 min. Subsequently, sterilization was carried out by autoclaving the mixture at 121°C for 15 min. The Streptomyces strain B7 was prepared as a spore suspension inoculum, matching the initial cell concentration to the standard solution of McFarland No. 0.5 (1.5 × 108 CFU/ml), measured at 625 nm with values ranging from 0.08 to 0.1. Then, 1 ml of the spore suspension was pipetted into the previously prepared sterile broken-milled rice, and 10 ml of distilled water was added. The mixture was then incubated at 30°C for 5–7 days. After the designated incubation period, the broken-milled rice was dried at 100°C for 60 min. Finally, the dried rice was crushed using a pestle and mortar (90 mm diameter Agate Top) and ground into a powder to obtain the pigment powder of actinobacteria (Streptomyces B7 dyed). The resulting Streptomyces B7 dyed was preserved in sterile plastic bags at room temperature. For the dyeing process, colorants were extracted from Streptomyces B7 dyed by adding 30 ml of 70% (v/v) ethanol to 5 g of the ground B7 in a 250 ml Erlenmeyer flask. The mixture was shaken at 120 rpm for 60 min. Following precipitation, the supernatant was separated and utilized as the natural dye.
For dyeing, 0.22 g of 100% silk fiber with a diameter of 0.98 mm was prepared. The silk was soaked in dye extract and shaken on a shaker at a speed of 120 rpm for 30 min. Subsequently, the silk fibers were twisted and left to dry at room temperature for 12 h. Afterward, the dyed silk fibers were washed with tap water and once again left to dry at room temperature for an additional 12 h.
Characterization
ZT powder
To identify the crystal structure and phase of calcined ZT powder, the X-ray diffraction (XRD) characterization was performed on an X-ray diffractometer (XRD, Philip PW 3040/60 X′Pert Pro, Eindhoven, The Netherlands) using Cu Kα radiation over a 2θ range from 10° to 80° with a step size of 0.04° at a scanning speed of 3°/min. The XRD data is fitted with the Rietveld refinement procedure employing the FullProf program.
The surface morphology of ZT calcined powders was observed using a field emission scanning electron microscope (FESEM) (FESEM Apreo S, Thermo Fisher Scientific Inc., Waltham, MA, USA). The calcined powder was mounted on aluminum stubs with conductive carbon tape adhesive and sputtered with gold before analysis. The microscope was operated at 10 kV in high-vacuum (HV) mode with an ETD detector beam current set to 0.05 nA. The FESEM morphological examination at 10,000–50,000 magnification was analyzed.
The Streptomyces strain B7 dye characterization
The reflectance values and corresponding CIELAB coordinates for both Streptomyces B7-dyed samples and those coated with ZT were measured using a colorimeter (CR-400 Chroma Meter, Konica Minolta Sensing Americas, Inc., USA). The color information is represented in CIELAB coordinates: L* corresponds to brightness (100 = white, 0 = black), a* (+/−) indicates the red/green ratio, and b* (+/−) indicates the yellow/blue ratio. 20
The color stability has been affected by washing. The quality of natural dyes derived from Streptomyces B7 extract has been investigated for stability. The Streptomyces B7 dyed samples and the Streptomyces B7 dyed samples coated with ZT were tested on the color fastness to washing according to ISO 105-C06 A2S: 2010 standard method. A grade of 5 is the best and a grade of 1 is poor. A color fastness rating of less than grade 3 indicates a considerable alteration in color after washing.21,22
Antibacterial studies
A 0.1 g sample of both B7-dyed and undyed fabrics was soaked in a solution containing 0.5% ZT in a 50 ml Erlenmeyer flask. The flask was placed in an ultrasonic cleaning bath (DH.WUC.D22H, DAIHAN Scientific, Korea) operating at a frequency of 40 kHz at 35°C for 60 min. Subsequently, the ZT-coated fabrics were squeezed to remove excess water and dried for approximately 10 min.23,24
Staphylococcus aureus, a microbial agent responsible for multiple infective diseases in humans and animals, was selected as the target organism for our test. Testing antibacterial agents against S. aureus is relevant because it is a common human pathogen. It is often responsible for skin and soft tissue infections, as well as more severe conditions such as pneumonia, endocarditis, and sepsis. 25
For bacterial strains and culture conditions, S. aureus was maintained on nutrient agar (NA) at 35°C for 24 h. Subsequently, S. aureus was prepared by selecting a single colony from the stock bacterial culture with a loop and inoculating 20 ml of sterile nutrient broth (NB) in a 100 ml Erlenmeyer flask. The flask was then incubated in a shaking incubator at 120 rpm at 35°C for 24 h. Liquid cultures were diluted to achieve bacterial cell concentrations of approximately 1.5 × 108 colony-forming units (CFU/ml) for the antibacterial test, corresponding to the McFarland standard No. 0.5. 25
The antibacterial properties of the fabrics were qualitatively assessed against S. aureus following the ATCC147 test standard using the disk diffusion method.9,26,27 This method is a relatively quick and easily executed qualitative test designed to determine the antibacterial activity of diffusible antimicrobial agents on treated textile materials.9,26,27 Four tested samples were employed for the evaluation: (1) control (undyed Streptomyces B7 and uncoated ZT); (2) coated with ZT; (3) Streptomyces B7 dyed; and (4) Streptomyces B7 dyed coating with ZT. These samples were placed on a nutrient agar layer in a Petri dish. The Petri dish was then incubated at 35°C for 24 h to observe inhibition zones. The growth of the tested microorganisms was constrained to a circular area or clear zone around the fabrics containing the tested material. Antimicrobial effects were determined and measured as the diameter of the clear zone in millimeters using a vernier caliper. All readings were recorded in triplicate.
Bacterial activity was quantitatively assessed against S. aureus using the colony counting method following the ATCC100 test standard. 26 The bacterial culture was grown using the same method as mentioned earlier. Sterile samples were placed in a 250 ml glass jar, dipped in 1 ml of bacteria with a concentration of 1000 CFU/ml, and then incubated at 35°C for 24 h. Following the incubation, 100 ml of sterilized distilled water was added to the jar and continuously shaken for 1 min. Subsequently, 0.1 ml of the solution was diluted and placed on 25 ml of nutrient agar, followed by incubation at 35°C for 24 h.
The results are expressed as bacterial reduction percentage (%R). The percentage reduction (%R) of bacterial colony-forming units (CFU) on the four tested samples was calculated at 0 and 24 h using equation (1) 28 :
Where
Results and discussion
ZT powder properties
The structure of ZT at different calcination temperatures (600°C and 900°C) is illustrated in Figure 1. At 600°C and 650°C, impurity phases (TiO2 and ZnO at 600°C, and TiO2, ZnO, and ZnTiO4 at 650°C) were observed. An increase of 50°C in the heat treatment temperature resulted in a more refined crystalline organization, evident from the significant increase and tightening of the peaks, particularly around 35°C. The ZT powders exhibited a pure perovskite phase with an increasing sintering temperature of ⩾700°C, which is lower than the ZT powder prepared by the solid-state method (calcined at 800°C for 24 h. Additionally, the ZT powder exhibits a pure perovskite phase with a rhombohedral structure, as confirmed by its alignment with the JCPDS file no. 00-026-1500. This is evident from the intensity peaks at (101), (012), (104), (110), (006), (113), (021), (202), (024), (107), (116), (211), (018), (214), (300), (208), (1010), (119), (217), consistent with findings from previous work. 29 Rietveld refinement, utilizing the Fullprof software, was employed to confirm the crystal structure of ZT powder at various calcination temperatures. The cell parameter, lattice constants, space group, and atomic functional positions can be cross-referenced with the Crystallography Open Database (COD) for simulation modeling. Additionally, the resulting refined parameters, such as chi-square (χ2) and the goodness of fit factor (GofF), should be less than 5, indicating higher crystallographic symmetry.30,31 χ2 and GofF can be determined from the expected and weighted profile R factors using χ2 = (Rwp/Rexp)2 and Rwp/Rexp, respectively. The obtained results suggest that the best refinement was achieved using the R3H (rhombohedral) space group. The low values of χ2 (~2.33 to 3.03) and GofF (~1.53 to 1.75) indicate good agreement between calculated and observed patterns. The percentage of the ZT powder phase at calcination temperatures of 700–900 is 100%, as shown in Figure 2 and listed in Table 1.

The XRD spectrum of ZT powder at different calcination temperatures.

Rietveld refined XRD patterns of ZT powder using R3H space group: (a) 700°C, (b) 750°C, (c) 800°C, (d) 850°C and (e) 900°C.
The profile parameters, lattice parameters a and c, chi-square (χ2), the goodness of fit factor (GofF), and the percentage of rhombohedral phase of ZT at differences of calcination temperature.

The surface morphology of ZT at temperatures ranging from 600°C to 900°C is illustrated in Figure 3(a)–(d). Between 600°C and 700°C, the particle size exhibited spherical-like morphology, appearing to be uniformly distributed. As the calcination temperature surpassed 700°C, the particle shape transformed from spherical-like to plate-like with a more fused. The average particle size increased with increasing calcination temperature, measuring 92, 114, 119, 250, 500, 1700, and 1850 nm for 600°C, 650°C, 700°C, 750°C, 800°C, 850°C, and 900°C, respectively. Changes in shape and size can be attributed to a nucleation speed faster than the growth speed of nanostructures at calcination temperatures ⩽700°C. Consequently, the particle products consisted of numerous separate, small, and uniform particles. Beyond 700°C, the growth speed surpasses the nucleation of nanostructures, resulting in more fused particles with larger sizes. 32 Another explanation is that the increase in particle sizes can be linked to enhanced atomic mobility at higher temperatures, leading to accelerated grain growth. 33 These results demonstrated that the optimum calcination temperature is 700°C, and corresponds with a previous work. 32 The improved crystallinity with calcination temperature observed in the X-ray diffraction analysis (seen in Figure 1) supports this phenomenon.

Surface morphology of ZT powders at different temperatures: (a) 600°C, (b) 700°C, (c) 800°C, and (d) 900°C.
To examine the surface morphology of ZT coated on silk fabrics, the optimal condition of 700°C was selected. The images of uncoated and ZT-coated silk fabrics were analyzed using FESEM, as illustrated in Figure 4. For the uncoated sample, both images displayed the surface morphology of the pristine fabric (Figure 4(a) and (c)). In contrast, small particles were observed on the surface of the ZT-coated silk fabric and the dyed fabric with ZT coating (Figure 4(b) and (d)). Confirmation of these small particles on the silk fabric was obtained through EDS, as depicted in Figure 5. The EDS results for the uncoated fabric revealed that most spectra comprised oxygen (O) with no detected zinc (Zn) and titanium (Ti) (Figure 5(a)). In contrast, the spectrum for ZT-coated samples exhibited three detected peaks corresponding to oxygen (O), zinc (Zn), and titanium (Ti) elements, with weight percentages of 45.8%, 34.4%, and 19.8%, respectively (Figure 5(b)). For the Streptomyces B7 dyed sample, the spectrum included carbon (C), oxygen (O), nitrogen (N), calcium (Ca), and aluminum (Al) elements resulting from the production of Streptomyces B7 forming rice grain (Figure 5(c)). With ZT nanopowders coated on Streptomyces B7 dyed silk fabric, the spectrum at spot 4 area showed oxygen (O), zinc (Zn), and titanium (Ti) elements, while the spectrum at spot 5 area included carbon (C), oxygen (O), nitrogen (N), zinc (Zn), and titanium (Ti) elements (Figure 5(d)). This indicates the persistence of ZT nanopowders on silk fabric, including Streptomyces B7 dyed samples.

Surface morphology of: (a) control (undyed Streptomyces B7 and uncoated ZT), (b) coated with ZT, (c) Streptomyces B7 dyed, and (d) Streptomyces B7 dyed coating with ZT.
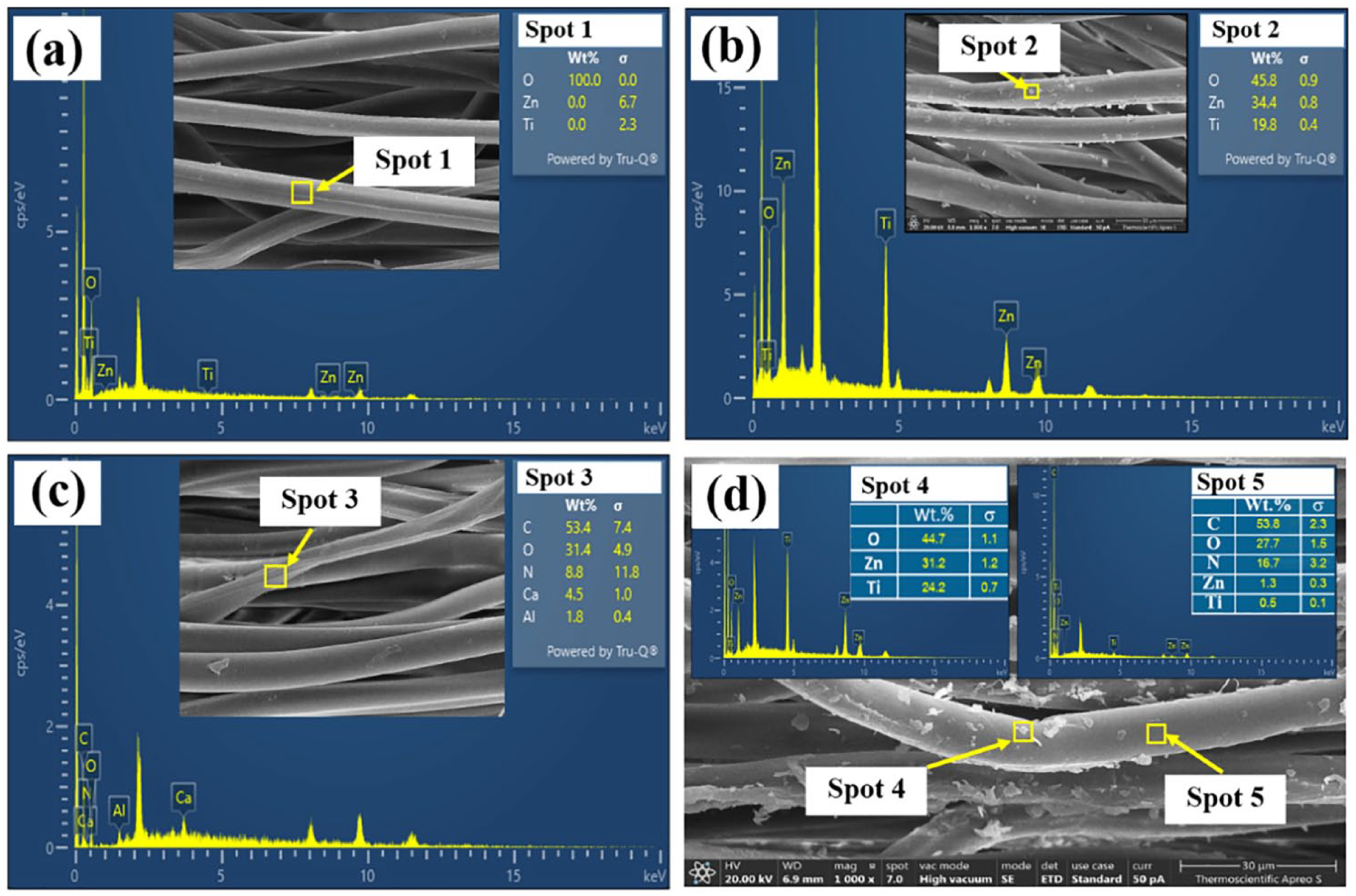
EDAX spectra of: (a) control (undyed Streptomyces B7 and uncoated ZT), (b) coated with ZT, (c) Streptomyces B7 dyed, and (d) Streptomyces B7 dyed coating with ZT.
The Streptomyces strain B7 dye properties
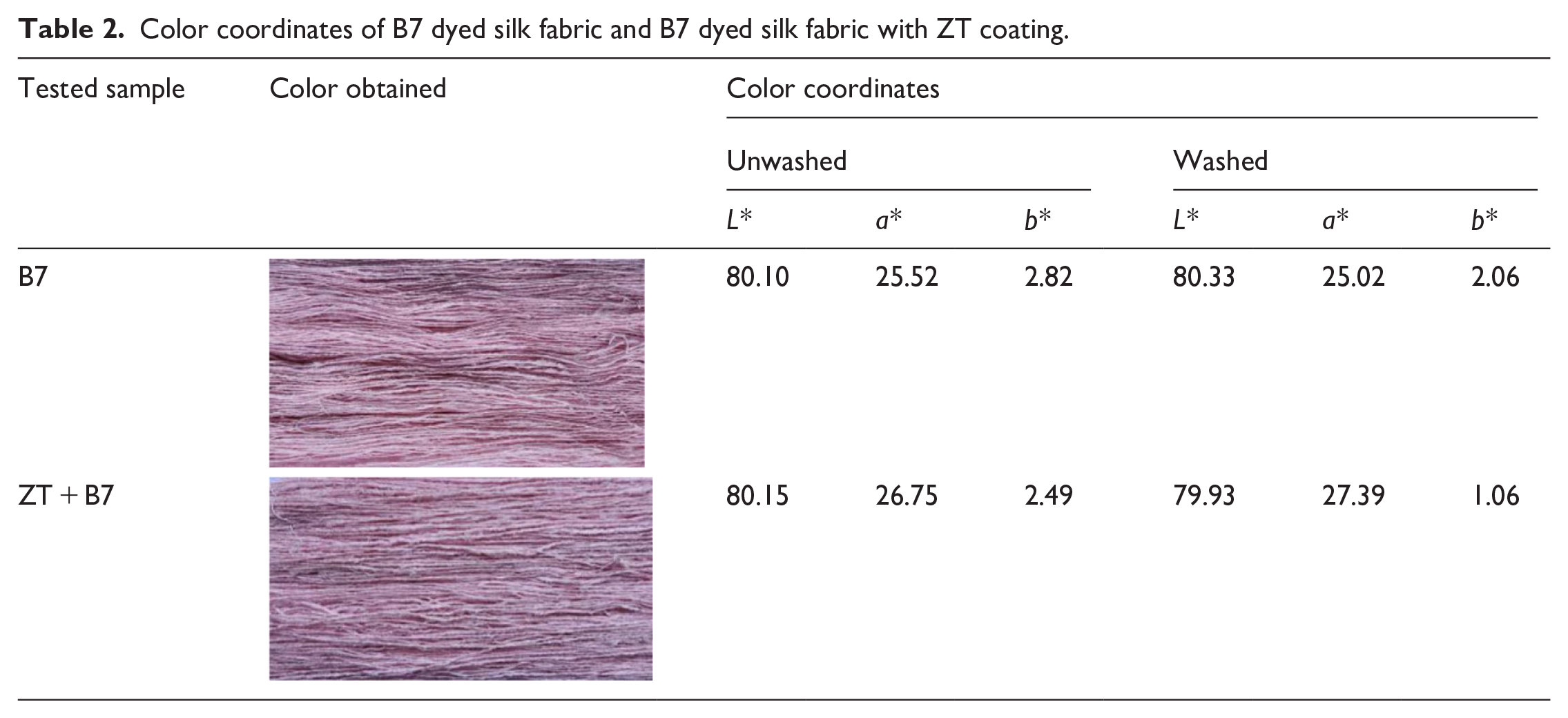
The color of the silk fabric dyed with Streptomyces B7 extraction and the Streptomyces B7-dyed fabric with ZT coating was examined, and the apparent color values are presented in Table 2. The shade was assessed by observing the L*, a*, and b* values of CIE-LAB color, as detailed in Table 2. For the fabric dyed with Streptomyces B7 extraction, the L*, a*, and b* values were 80.10, 25.52, and 2.83, respectively. After coating ZT powder on the silk fabric, the L*, a*, and b* values were 80.15, 26.75, and 2.49, respectively. There are minimal differences observed between the fabric dyed with Streptomyces B7 extraction and the Streptomyces B7-dyed fabric with ZT coating. Even after the washing process, the color coordinates of the B7-dyed fabric and the B7-dyed fabric with ZT coating remain the same. These results suggest that the ZT coating has not influenced the color change of B7-dyed fabrics.
Color coordinates of B7 dyed silk fabric and B7 dyed silk fabric with ZT coating.
Fabric colors may change due to exposure to sunlight or washing. According to the ISO105-C06 A2S:2010 standard testing, the washing color fastness properties of B7-dyed silk fabrics and B7-dyed silk fabrics coated with ZT are presented in Table 3. Fastness ratings on a gray scale ranging from 1 to 5 were assigned (1 for bad, 2 for fairly good, 3 for good, 4 for very good, and 5 for excellent). The color change after washing for both the B7-dyed sample and the B7-dyed sample coated with ZT was rated as “good.” Additionally, the color staining of cotton and silk after washing for both samples was rated as “very good” (4–5). Therefore, the color stability of B7-dyed silk fabrics and those coated with ZT remained unaffected by washing.
Color fastness properties of B7-dyed silk fabrics (B7) and B7-dyed silk fabrics coated with ZT (B7 + ZT).

Antibacterial activity
The antibacterial efficacy of the agent in inhibiting bacterial growth was assessed using a disk diffusion method. Four test samples were examined: (1) control (undyed Streptomyces B7 and uncoated ZT); (2) coated with ZT; (3) Streptomyces B7 dyed; and (4) Streptomyces B7 dyed fabric coated with ZT. The results are depicted in Figure 6 and summarized in Table 4. Figure 6 illustrates a clear zone of inhibition around both the B7-dyed fabric and the B7-dyed fabric coating with ZT, while the control fabric (undyed and uncoated with ZT) showed no signs of inhibition. The most significant inhibitory effect was observed against S. aureus in the B7-dyed fabric coating with ZT, with a zone of inhibition approximately 5.03 ± 0.06 mm in diameter. The antibacterial activity results after 24 h are presented in Table 5. The colony counting test results demonstrated enhanced activity against bacteria, leading to bacterial reduction in all treated fabrics, including ZT-coated fabric, B7-dyed fabric, and B7-dyed fabric coating with ZT. The percentage of reduction for ZT-coated fabric, B7-dyed fabric, and B7-dyed fabric coating with ZT was 99.9%, 100%, and 100%, respectively. Notably, B7-dyed fabric coating with ZT exhibited robust activity against bacteria in both qualitative and quantitative methods. A possible explanation could be the result of varied complex formation between metallic ions and functional groups of dye. 34 Furthermore, with varying calcination temperatures, the optimum calcination temperature is found to be 700°C. This temperature resulted in a crystalline pure perovskite phase of ZnTiO3 at 100%. A higher level of crystalline purity may contribute to enhanced antibacterial activity. A well-defined crystal structure with fewer defects and impurities can provide a more stable and reactive surface. Therefore, the synthesis of pure perovskite phase ZnTiO3 was successfully at 700°C achieved using the molten salt method, a simple and environmentally friendly approach for imparting antibacterial activity to silk fabrics.

Antibacterial activity of: (a) control, (b) ZT, (c) Streptomyces B7 dyed, and (d) Streptomyces B7 dyed fabric coating with ZT showing zone of inhibition against tested bacteria by a disk diffusion method.
Antibacterial assessment by disk diffusion method.

Bacterial reduction of treated and untreated fabric for S. aureus.

Conclusions
Nano-sized ZT powder was successfully prepared using the molten-salt method, a key novelty in our work. The pure perovskite phase was achieved at a low calcination temperature (700°C), surpassing the efficiency of the solid-state reaction method. The ZT structure aligns closely with the rhombohedral structure, and the particle size transitions from spherical to plate-like shapes with an increase in calcination temperature. Coating the modified B7-dyed fabric with ZT demonstrated an outstanding 100% reduction in bacterial colonies. Notably, this technique offers advantages such as low energy consumption, high purity, uniform particle size distribution, cost-effectiveness, and environmental friendliness. The outcomes of this study could find application in natural fabrics, contributing valuable insights into antibacterial activity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by The National Science, Research and Innovation Fund (NSRF) under the grant RDI-1-67-29 through Pibulsongkram Rajabhat University.

