Abstract
Pitch-based carbon fiber, as one of the important engineering materials, has been widely used in aerospace, defense, sports, and other fields. In the long production process of pitch-based carbon fiber, the property of pitch precursor is significant for the mechanical performance of obtained carbon fiber. Thus, it is crucial to improve the property of pitch precursor by efficient means. Halogenation–dehalogenation is a newly developed method for the controllable synthesis of a pitch precursor from the molecular dimension, including fluorination–defluorination, chlorination–dechlorination, and bromination–debromination. This work reviewed the mechanism and application, as well as the advantages and disadvantages of each halogenation–dehalogenation method.
Graphical abstract

Introduction
Carbon fiber is an organic fiber with carbon as its main element and has been widely used in the field of aerospace industry, 1 defense industry, 2 sports goods, 3 medical instruments,4,5 etc. The industrial production process of carbon fiber is mature and stable currently in very few countries due to the technical barrier, reflecting mainly the inability to prepare pitch precursor with excellent spinnability. 6 Commercial carbon fibers can be divided into pitch-based carbon fibers and polyacrylonitrile-based carbon fibers according to the difference in precursors. Pitch-based carbon fiber uses pitch as a precursor, whereas polyacrylonitrile-based carbon fiber corresponds to polyacrylonitrile. However, the pitch-based carbon fiber precursor includes isotropic pitch and mesophase (anisotropic) pitch based on the difference in phase properties of pitch precursors, and are developed for general-purpose carbon fiber and high-performance carbon fiber, 7 respectively. General-purpose carbon fiber has low mechanical performance and low price, which is only applied in the fields of low-strength reinforcing, such as construction reinforcement. 8 On the contrary, high-performance carbon fiber can be applied in aerospace, aircraft, and other areas where high-strength performance is needed. 9 The development of pitch-based carbon fiber can be traced back to 1963 when polyvinyl chloride-derived isotropic pitch-based carbon fiber was prepared first by Otani, and it was realized in industrial production in 1970 in Japan. 10 The mesophase pitch-based carbon fiber was invented in 1970 and was commercially produced in 1975 in the USA. 11
Carbon fiber has the characteristics of being lightweight, having high strength, good thermal conductivity, high-temperature resistance, friction resistance, corrosion resistance, etc. 12 It can be used as reinforcing materials and lightweight materials. However, the price of carbon fiber is an important factor to affect its utilization. If the price of carbon fiber is reduced to an acceptable level, it can replace stainless steel, magnesium-aluminum alloy, glass fiber, polymer, and other materials in a wide range of applications. 13 Polyacrylonitrile-based carbon fibers are expensive, so it is scarcely possible to apply them in low value-added fields. In pitch-based carbon fiber, mesophase pitch-based carbon fiber has excellent mechanical performance (tensile strength: 1.5–7.0 GPa; Young’s modulus: 200–950 GPa) as well as excellent thermal conductivity and thermal expansion, while the high price and low output of mesophase pitch-based carbon fiber greatly limit its application in some fields with low value-added. 14 On the contrary, isotropic pitch-based carbon fiber has the advantage of low cost while its mechanical performance is very low (tensile strength: 0.5–1.1 GPa; Young’s modulus: 30–50 GPa). 15 With the rapid development and application of new energy vehicles and wind power generation, it is significant to light the weight of new energy vehicle bodies and wind turbine blades.16,17 The carbon fiber reinforced material based on isotropic pitch-based carbon fiber has become one of the preferred materials due to its low price. 16 However, the improvement of the mechanical performance of isotropic pitch-based carbon fiber needs to be largely improved to achieve the requirement of the car body and wind turbine blades.
Raw materials and synthesis methods are considered two key factors in determining the performance of pitch-based carbon fiber referring to the result of previous work. Low-cost raw materials are the main feedstock for the preparation of isotropic pitch-based carbon fiber to keep the price low, the byproducts of coal and petroleum processing for example.18,19 In addition, the byproducts of biomass processing20–22 and waste polymers 23 are also largely investigated to prepare isotropic pitch-based carbon fiber, although it is still in the exploratory stage in the laboratory. The byproducts of coal and petroleum processing have the disadvantages of heterogeneous properties, a wide molecular weight distribution, and complex components. It is difficult to improve the mechanical performance of as-prepared carbon fiber by adjusting the sorts and mixing ratios of the byproducts. Thus, the synthesis method is significant for improving the mechanical performance of isotropic pitch-based carbon fiber. Thermal condensation is the most common method for synthesizing isotropic pitch precursors since it is easy to operate. The mechanism of thermal condensation is that components in raw materials were involved in the spontaneous initiation of polycondensation under the thermal effect.24,25 Generally, the reaction is controlled by adjusting conditions including temperature, pressure, and atmosphere, therefore, thermal condensation reaction cannot be controlled effectively, especially from the molecular dimension. The mechanical performance of isotropic pitch-based carbon fiber cannot be greatly improved by using the thermal condensation method to synthesize pitch precursors. 26
Considering that carbon fiber is a one-dimensional material, if carbon fiber molecules can be designed to be linearly arranged along the one-dimensional direction, it will inevitably enhance strength performance. Currently, only the halogenation–dehalogenation method can accurately control the molecular structure for the preparation of pitch-based carbon fiber. 27 The molecular structure of the pitch precursor can be controlled in the synthesis process using the halogenation–dehalogenation method, making the molecular structure of the pitch precursor more linear and with higher orientation, which can also control the condensation degree of aromatic substances while increasing the average molecular weight.26,28 Furthermore, the pitch precursor synthesized by the halogenation–dehalogenation method has high softening point, excellent spinning property, good stabilized property, and high mechanical performance, which has been widely used in the development of pitch-based carbon fiber. 16 In this work, the mechanism and application of the halogenation–dehalogenation method were reviewed, the advantages and disadvantages of various halogen sources were revealed, and the future prospects of the halogenation–dehalogenation method were proposed.
Pitch-based carbon fiber production process
Currently, only Japan and the United States have mature technology for the industrial production of pitch-based carbon fiber, 6 while other countries still have many problems in production, particularly for pitch precursor synthesis. 29 The manufacturing process of pitch-based carbon fiber was fixed after decades of development, and isotropic pitch-based carbon fiber is prepared by almost the same process as mesophase pitch-based carbon fiber, including raw material pretreatment, pitch precursor synthesis, melt-spinning, stabilization, carbonization, and post-treatment, as shown in Figure 1. The precursors of isotropic pitch and mesophase pitch are derived from chemicals, polymers, and byproducts of coal, and petroleum, as well as biomass processing.30–33 These raw materials contain or can be converted to various polyaromatic hydrocarbons. Pure chemicals, naphthalene 34 and anthracene35,36 for example, have a relatively low reactivity to oligomerization and polycondensation. The various catalysts including HF/BF3 and bromine are necessary to induce a pitch formation reaction. On the contrary, the byproducts of the coal, petroleum, and biomass process are easier to oligomerize and condense under thermal effects, as they are composed of hundreds of organic compounds, which can induce various reactions of cyclization, aromatization, addition, etc. However, it is necessary to control the degree of oligomerization and polycondensation to prevent the formation of coke and other insoluble particles. Solid particles that cannot be completely melted during spinning have a large influence on melting spinnability, which tends to determine the feasibility of continuous spinning. In addition, the impurities in raw materials (inorganics, ash, etc.) 37 and special compounds (heavy components, quinoline insoluble, etc.)38,39 have a negative effect on the melt-spinning property of pitch precursors. Therefore, various pretreatments are needed to ensure the good spinnability of pitch precursors. Low-cost and practical pretreatment methods including pressurized treatment, 40 distillation, 41 extraction purification, 42 and centrifugal separation 32 are widely used in industrial production.
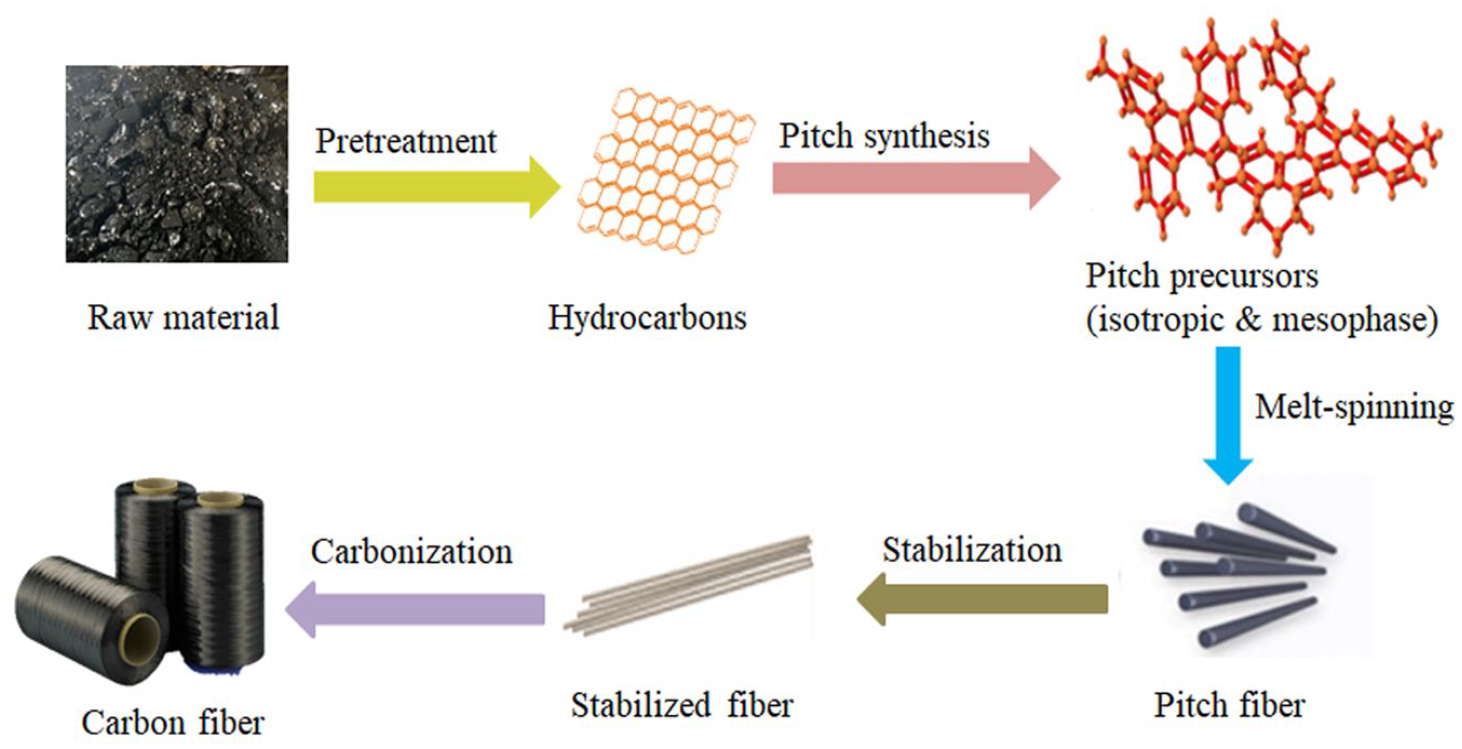
The process of pitch-based carbon fiber production.
The step of synthesis of pitch precursor involves the reactions of cyclization, aromatization, oligomerization, and polycondensation to enhance the degree of aromaticity and polymerization of raw materials. 19 The synthesis of a pitch precursor with excellent spinnability determines the stable and continuous preparation of pitch-based carbon fiber, and it is also the key step for optimizing the mechanical performance of the as-prepared carbon fiber by modifying the molecular structure and composition. The long production process of pitch-based carbon fiber, especially the synthesis of pitch precursors with excellent melt-spinning properties, has made it impossible for many countries to realize the industrial production of pitch-based carbon fiber or that the prepared carbon fiber products have poor mechanical performance. 19 The carbonized material has an extremely high melting point due to a certain degree of graphitization; it will be difficult for melting and forming a filament state after carbonization. Thus, the pitch precursor must be transformed into a monofilament state before carbonization, and melt-spinning is a common method to convert the pitch precursor into a monofilament.5,43 In melt-spinning, the pitch precursor was placed into a heating chamber, and it was melted completely by heating the chamber above the softening point of the pitch precursor. The molten pitch was extruded from a spinneret to transform a pitch fiber and the molecule was arranged along an axial direction. 39 Non-melting treatment is performed to prevent the pitch fiber from melting during the carbonization process since softening point is much lower than the carbonization temperature. 44 Therefore, stabilization is necessary before carbonization, which allows oxygen to enter the molecular structure of pitch fiber and form abundant oxygen-containing functional groups. 45 The oxygen will escape from the fiber slowly in the subsequent carbonization reaction, thus keeping the pitch fiber in the melt-free state. The purpose of stabilization is to change the pitch fiber from the thermoplastic state to the thermosetting state through the oxidation reaction.46,47 In the carbonization process, the side chains of non-carbon atoms are removed, and aromatic groups perform further polycondensation, thus shrinking the fiber diameter as well as enhancing the degree of aromaticity, carbonization, and graphitization. 48 In addition, the orientation of the carbon groups further aligned along the axial direction, forming a carbon content of more than 96% of the cross-layer graphite structure. Carbon fiber with certain mechanical, physical, and chemical properties is finally produced after carbonization. To improve the mechanical properties of carbon fibers, selective graphitization was carried out to promote the orderly transformation of thermodynamically unstable carbon atoms from disorderly layer structure to graphite crystal structure by thermal activation. 49 Therefore, the molecular structure of fiber has been graphitized, in addition, the carbon content is more than 99% and has a layered hexagonal lattice graphite structure. The final stage of treatment includes a surface treatment and sizing process that will change the surface of the carbon fiber and improve its interfacial properties. 5 The pitch-based carbon fiber production process is complex and time-consuming and each step has a significant impact on the quality of the final carbon fiber product. The most important one is the development of a pitch precursor with excellent spinning performance, linear molecular structure, and an appropriate degree of condensation.
Halogenation–dehalogenation
The development of the halogenation–dehalogenation method for pitch-based carbon fiber preparation can be traced to the application of fluorination–defluorination in mesophase pitch-based carbon fiber production by Mochida in the 1980s. 50 Since then, the halogenation–dehalogenation method has been used in the synthesis of pitch-based carbon fibers, including chlorination–dechlorination and bromination–debromination. Otani used the chlorination–dechlorination method when he first discovered the preparation of pitch-based carbon fiber, but he did not notice the effect and mechanism of chlorination–dechlorination. Polyvinyl chloride was used as a raw material to produce pitch-based carbon fiber by Otani, and the chlorine radicals released from polyvinyl chloride induced the chlorination–dechlorination reaction. 51
In the halogenation–dehalogenation reaction, a substitution reaction occurs in the presence of halogen, which can control the polymerization and polycondensation of polycyclic aromatic hydrocarbons, Halogenation–dehalogenation reaction process can be effectively controlled by adjusting the type, amount, and reaction conditions of halogens, to control the properties of pitch precursors and the final properties of carbon fibers. At present, halogenation–dehalogenation can improve the production efficiency and performance of carbon fibers, especially the production of mesophase pitch-based carbon fibers by the fluorination–defluorination method with methyl naphthalene as a raw material has been performed in industrial production, it plays an important role in the preparation of high-modulus carbon fibers. 36
Fluorination–defluorination
Fluorination–defluorination was the first to be developed and successfully applied to the industrial production of mesophase pitch-based carbon fiber among three halogenation–dehalogenation methods. In the fluorination reaction, the mixture of HF and BF3 is used as a composite catalyst, which is a widely used Lewis acid catalyst for the Friedel-Craft reaction. 52 AlCl3 was used as a catalyst to promote the formation of mesophase pitch before the application of HF/BF3 catalyst.53–56 However, it was difficult to completely remove AlCl3 from the as-synthesized pitch after the reaction was finished, thus the residual AlCl3 had a non-negligible effect on the melt-spinning property of the pitch precursor.57–59 Then, Mochida focused on the study of HF/BF3 catalyst that can provide a strongly acidic ion H+ with a bulky anion BF4. The nucleophilicity of BF4− is weak, while it acts as a proton donor to generate complexes with π-bonds on the aromatic ring at a relatively low temperature. Therefore, the electron cloud density of the aromatic ring is reduced and further forms a positively charged hydrocarbon. Then, the positively charged hydrocarbon attacks the other aromatic rings with the active site at the maximum active site to generate a dimer. The dimers continue to oligomerize to produce trimer, tetramer, and the other oligomers to a higher degree. The electron cloud density of each carbon atom changes gradually with the increase of the degree of oligomerization in the fluorination reaction, reducing the number of active sites. In addition, the complexing action between aromatic rings and proton donors is also decreased, so the reactivity of aromatic compounds with a high degree of oligomerization is largely reduced.50,60,61 The catalytic mechanism of fluorination–defluorination is shown in Figure 2. Finally, the product consists mainly of trimer-to-decamer oligomers, and the mass yield of oligomers is higher than 90% by weight. 62

The mechanism of fluorination–defluorination.
Compared with AlCl3, the HF/BF3 catalyst can be completely removed from the as-synthesized pitch by gas blowing due to the low boiling point. A large amount of inert gas blows through the fluorinated product to expel the HF/BF3 mixture and components with low molecular weight during defluorination. The method of vacuum distillation can greatly reduce the defluorination temperature and catalyst residence time. 63 The nucleophilicity of F− is strong so that the catalyst can be consumed in defluorination caused by ionization. In addition, the blown catalyst will mix with light components, and it is difficult to separate clearly, therefore, a constant supply of fresh catalyst is required. However, the acidity and corrosiveness of HF/BF3 catalyst are extremely demanding for the corrosion resistance of the equipment, which makes it an obstacle for application. 64 Currently, only the Mitsubishi Chemical Group of Japan had reported the successful industrial production of mesophase pitch-based carbon fiber using the fluorination–defluorination method derived from naphthalene series compounds. 65 The mesophase pitch synthesized by fluorination-defluorination has a large amount of naphthenic and alkyl groups, which can improve fluidity and viscosity. 54 Thus, it is beneficial to obtain a pitch precursor with excellent spinnability and stabilized reactivity. In fluorination–defluorination, cycloalkyl groups were introduced by non-dehydrogenation polycondensation of aromatic rings, and the previous study revealed that the structure and side chain of cycloalkyl was easily optimized by adjusting the reaction conditions and raw materials. Hence, the structure and composition of the pitch precursor can be controlled from the molecular dimensions.50,66
Previous research results show that HF and BF3 must be used in the preparation of mesophase pitch. In the process of catalytic condensation, if only HF is added, the aromatic degree of the product will be significantly improved, but the degree of aromatic ring substitution is low, so the number of naphthenic groups is reduced, which is not conducive to improving the fluidity of pitch. The function of HF is to trigger more of the condensation polymerization of the aromatic ring than to trigger the hydrogen transfer reaction. 67 In contrast, BF3 plays a more prominent role in the activation of the hydrogen transfer reaction. If only BF3 is used as a catalyst, a large number of substitution reactions will be induced, resulting in a low aromatic degree of the product and a large number of naphthenic groups. Therefore, the combination of HF/BF3 in a certain proportion can greatly improve the acidity of Lewis acid and provide large-volume proton donors with a weak nucleophile, so it has high catalytic activity. However, the corrosion of the two will be significantly enhanced after mixing, with greater risks to safety and environmental protection.68,69
Using naphthalene 70 and anthracene 71 as raw materials, Mochida successfully prepared the mesophase pitch for the precursor of high-modulus carbon fiber for the first time with the help of HF/BF3. The mesophase pitch has the phase property of long flow and large domain, and it also has good fluidity, excellent spinnability, as well as a high degree of polycondensation. The mesophase pitch precursor derived from naphthalene has a lower yield and softening point than that derived from anthracene, while the fluidity and spinnability of the naphthalene-derived pitch are better than the anthracene-derived pitch. Previous studies have shown that almost all methyl groups in methylnaphthalene can be retained in the final mesophase pitch, which is very effective in reducing the softening point and improving spinnability. Therefore, it is generally believed that the sidechains of aromatic groups can improve the spinnability and stability of pitch. The mesophase pitch prepared from methylnaphthalene contains a large amount of methyl group and naphthenic group induced by non-dehydrogenation polycondensation of naphthalene rings.72,73 Both the naphthalene group and methyl group have a positive influence on improving the spinnability of obtained pitch. 74
Bromination–debromination
The type of halogenated substitution reactions of side chains of aromatic compounds is typical of free radical chain reactions, and the necessary condition for the occurrence of free radical chain reactions is the need for a chain initiation agent. 75 In the bromine substitution reaction, monomeric bromine undergoes a homogeneous cleavage reaction to generate bromine radicals that can induce free radical chain reactions. 76 The molecular kinetic energy of bromine molecules increases by absorbing thermal energy at high temperatures, and the collision between bromine molecules converts the molecular kinetic energy into molecular internal energy; when the internal energy increases enough to overcome the bonding energy of covalent bonds of molecules, the bromine molecules are split into two bromine radicals. The generated bromine radicals selectively attack the hydrogen on the methyl group and then chain transfer occurs. Normally, the full homolytic cleavage of bromine molecules into bromine radicals requires a reaction temperature above 500℃, which is difficult to achieve in liquid-phase reactions. 77 Therefore, it is not possible to homolyze all bromine molecules into radicals at a lower reaction temperature, leading to a lower rate of methyl bromide in direct thermal bromination.78–81
The reaction mechanism of bromination–debromination is shown in Figure 3. 82 In the presence of bromine radicals, the hydrogen atom in the aliphatic sidechain of the aromatic ring is a readily radical substitution to form brominated compounds, which then bind to hydrogen on the other aromatic ring at high temperature to form hydrogen bromide. The produced hydrogen bromide is blown out of the reaction system by the inert gas flow. Aromatic ring oligomerization can be carried out by crosslinking of aliphatic side chains. When the amount of bromine is low, aromatic ring side chains are substituted by bromine monomers. 83 The aliphatic side chain of the aromatic ring forms a crosslinked structure by removing the hydrogen bromide, thus forming an oligomer with a low degree of oligomerization.

The mechanism of bromination–debromination and chlorination–dechlorination.
The bromination–debromination reaction promotes the formation of cycloalkyl and bridging groups, and these groups are well retained even after the oligomerization reaction. Cycloalkyl and bridging groups contribute to the formation of more linear and crosslinked microstructures of pitch precursors, and the degree of oligomerization can also be controlled by controlling the number of cycloalkyls and bridging groups.84–88 With increasing bromine addition, the molecular structure of isotropic pitch precursors gradually changed from a linear and crosslinked structure to a planar and macromolecular structure of condensed polycyclic aromatic hydrocarbons. Therefore, pitch precursors with linear and crosslinked molecular structures can be obtained by optimizing the bromine content, and such pitch precursors exhibit better melt-spinning properties.89,90 The molecular structure of the pitch precursors synthesized by the bromination–debromination method contains more cycloalkyl, bridging, linear, and crosslinked structures, leading to the better mechanical performance of as-prepared carbon fibers.27,91 Furthermore, the bromination–debromination reaction allows the relatively lighter components of feedstock to be converted into long side chains in aromatic compounds, and these long side chains undergo cyclization and aromatization reactions in debromination, which can improve the yield and aromaticity of as-synthesized pitch precursors. 92 With the increase of reaction temperature, the improvement of the kinetic energy of bromine molecules can be transformed into intramolecular energy by effective intermolecular collisions, which can lead to the increase of Br-Br bond homogeneity, therefore the increase of reaction temperature can promote the substitution reaction of bromine atoms. 93 The aromatic groups present in the reaction not only participate in the condensation reaction but also carry out the oligomerization reaction, which can yield more oligomers. It was proved experimentally that the components of the obtained brominated products do not change along with the increase in reaction time.94,95 The bromination–debromination method can achieve the controlled synthesis of pitch precursors from molecular dimension and effectively control excessive polycondensation to produce a linear and crosslinked molecular structure, thus improving the melt-spinning properties of pitch precursors and the mechanical performance of the final carbon fibers.
Ge 83 prepared a homogeneous isotropic pitch with a high softening point by bromination–debromination method using ethylene tar as raw material. Compared with the pitch precursors synthesized by the thermal condensation method, the bromination–debromination method increases the softening point of the pitch from 152℃ to 264℃, and the yield and coking value of pitch precursors are greatly improved. Moreover, the isotropic pitch produced by the bromination–debromination method has higher aromaticity and condensed degree. Ge 77 synthesized pitch precursor with abundant methylene bridged groups by bromination–debromination method using 1-methylnaphthalene as raw material, which has a high C/H ratio and average molecular weight, and it can be completely dissolved by toluene. Kim et al. 96 synthesized a unique isotropic pitch consisting of linear chains of condensed PAHs by bromination–debromination method using naphtha-cracked oil and coal tar as raw materials. The tensile strength of as-prepared pitch-based carbon fiber reached 2050 MPa. Yang et al. 97 successfully prepared general-purpose carbon fiber with a high softening point and good spinnability by bromination–debromination method using ethylene bottom oil and biotar as raw materials. The tensile strength of carbon fibers obtained by the bromination–debromination method reached 1200 MPa.
Chlorination–dechlorination
The mechanism of chlorination–dechlorination method is similar to that of the bromination–debromination, which requires chlorine radicals to initiate free radical chain reactions. Chlorine radicals can be generated from chlorine gas by a homolytic reaction or released from chlorine-containing polymers by thermal decomposition, polyvinyl chloride for example. 98 The chlorine radicals substitute the hydrogen on the side chain of the aromatic ring to form chlorinated aromatic hydrocarbons. As the reaction temperature increases, the chlorine atoms on the chlorinated aromatic hydrocarbons combine with the hydrogen atoms on other aromatic molecules during dechlorination to form hydrogen chloride, which is then purged from the reaction system by passing an inert gas. The chlorine gas in the reaction system can also be extracted by vacuum followed by air, or alternately vacuum and air can flow into the reaction system to ensure complete elimination. 99
Chlorination–dechlorination reactions are less likely to use high-purity chlorine, such as chlorine gas, as a chlorine source since the high-purity chlorine can produce highly acidic by-products that are corrosive for equipment. In addition, chlorine gas is highly toxic. Chlorine-containing polymers can be used as a chlorine source for the chlorination–dechlorination reaction. Chlorine-containing polymers can be decomposed into chlorine radicals and polyene radicals, of which chlorine radicals are easier to participate in various reactions and the existence time is very short.35,100 The chlorine radical substitutes the hydrogen on the methyl group of aromatic compounds when the reaction is conducted, after which the methylene group can be formed by dechlorinating the hydrogen. The pitch precursor contains more methylene and less condensed aromatic groups, so it can show better spinnability. The methyl side chains in the raw material are transformed into bridged groups connecting adjacent aromatic groups through chlorination–dechlorination, and the aromatic compounds are connected through methylene bridges, and the molecular structure develops into oligomers in a linear direction in a continuous and orderly manner. 35 The structure of pitch precursors is regulated by increasing the number of naphthene through aromatization, cyclization, and oligomerization. The evolution of the molecular structure of different intermediates in the carbon fiber production process is shown in Figure 4.

The molecular structure evolution of different intermediates in carbon fiber production process.
Liu et al. 45 used polyvinyl chloride as the chlorine source, and ethylene bottom oil was applied as the raw material to synthesize the pitch precursor by chlorination–dechlorination method. The obtained pitch precursor has a higher pitch yield and better spinnability in comparison to the pitch pretreated by simple atmospheric distillation. The coal liquefaction residue was used as raw material to controllably synthesize the pitch precursor for carbon fiber from the molecular dimension by chlorination–dechlorination method. 101 The C/H ratio, average molecular weight, and aromaticity were increased by chlorination–dechlorination. In addition, the pitch precursor derived by the chlorination–dechlorination method has a more linear and crosslinking molecular structure, and it exhibits better thermal stability and thermochemical properties due to the linear and crosslinking molecular structure.
The property and mechanical performance of carbon fiber derived from different halogenation–dehalogenation methods are summarized in Table 1. The carbon fiber synthesized by the fluorination–defluorination method is attributed to mesophase pitch-based carbon fiber, which has an excellent mechanical performance. The tensile strength and Young’s modulus are 4750 MPa and 975 GPa, respectively. The tensile strength and Young’s modulus of carbon fiber prepared by the bromination–debromination method can reach 2050 MPa and 54.5 GPa. The mechanical performance of carbon fiber obtained by chlorination–dechlorination is low compared to the other two methods, which is ascribed to the few studies on chlorination–dechlorination currently. The optimal reaction condition and halogen source still need further investigation.
Comparison of carbon fibers derived from three methods.

Conclusion and future perspective
Pitch is an important precursor for the preparation of diverse carbon materials, including carbon fiber, porous carbon, needle coke, high-oriented graphite, mesocarbon microbeads, carbon foam, etc. The composition, structure, and properties of pitch precursor play a decisive role in the properties and performance of as-prepared carbon materials. Currently, pitch precursor is mainly synthesized from polycyclic aromatic hydrocarbons by the thermal condensation method; however, the thermal condensation method has non-negligible limitations in optimizing the composition, structure, and properties of the obtained pitch precursor. The halogenation–dehalogenation method is a newly developed method for synthesizing pitch precursors. The halogenation–dehalogenation method can achieve controlled synthesis of pitch precursors from the molecular dimension by regulating the oligomerization to obtain linear and crosslinked structures, thus effectively controlling the degree of condensation of polycyclic aromatic hydrocarbons. In the future, the reaction mechanism of the halogenation–dehalogenation method needs to be further investigated, especially to reveal the effect of reaction conditions on the properties of as-synthesized pitch precursors.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support provided by the National Natural Science Foundation of China (Grant No. 22008254), and also supported by the Fundamental Research Funds for the Central Universities (Grant No. 2023ZKPYHH02), and the China University of Mining and Technology (Beijing) Yueqi Outstanding Scholar Project (Grant No. 2020JCB02).
