Abstract
The paper proposes a textile device for patients with large area burn wounds consisting of an assembly made up of a knitted backing layer, a hydrogel, with an antibiotic content, as a second layer and a membrane with controlled porosity located directly at the interface with the wound. The knit is the material support with a supporting clothing role, but also as a solution tank. The hydrogel has a keratin structure obtained from human hair with advanced swelling capacity and porosity. The study proposes the use of silver sulfadiazine (SSD), an antibiotic used in the topical therapy of burn wounds. The work optimizes the degree of swelling, evaluates the porosity of the hydrogel and designs the support garment for wound therapy. The optimal parameters to obtain the maximal swelling degree are: duration of hydrolytic reaction 180 min, volume of hydrogen peroxide 3.7 mL and duration of oxidation 90–125 min. Based on these optimal parameters one can obtain swelling degrees higher than 1000%. The device proposed is reusable and supports the SSD diffusion to wound.
Keywords
Introduction
Burn wounds represent a problem in many countries. 1 They are sensible to bacterial infections and represent a severe pathology modifying the general balance of the patient’s status. Burned skin opens a gate to bacterial infections and is the source of important loss of fluids, proteins, enzymes, electrolytes, altering the cardio-vascular, digestive, respiratory, and renal functions.2 –7 Due to their appropriate biochemical and mechanical properties, hydrogels have attractive advantages in the field of the wound dressings. Hydrogels performances imply advanced functions such as: antimicrobial, adhesion and hemostasis, anti-inflammatory and anti-oxidation, drug delivery, self-healing, and stimulus answer. 4 The common feature of its high standards is derived from water sorption capacity which is due to functional hydrophilic groups able to attach water molecules by means of hydrogen bonds.8 –11 They are used as wound dressings12 –15 with applications on derma levels, being biocompatible due to basic polymer structure and preservation of humidity at wound surface. Hydrogels have a viscous-plastic structure that adapts to various surface morphologies. Their performances are the result of the basic polymer structure, functional groups and cross-linking frequency.16,17 Hydrogel can be used as support for fabrics obtained through tissues engineering or as a mean for medicine diffusion through a systemic or dermal application.17,18
As healing action there are considered the following stages 12 : hemostasis, inflammation, proliferation and remodeling. Wound healing consists of a transient vasoconstriction of injured vessels. In the initial phase, damage to the skin exposes the subendothelial collagen and tissue factor, leading to platelet aggregation. 19 The final result is a strong blood clot. 20 During the inflammatory stage, the neutrophils express cytokines that recruit additional leukocytes (neutrophils, T cells, and macrophages) to the site of injury. 21 The proliferative stage consists of the epithelization, revascularization, and formation of granulation tissue. 22 The remodeling stage, considered as well as the maturation, is the longest stage of wound healing and is dependent on a delicate balance between synthesis and degradation. 23
Based on statements expressed in literature2 –7,24,25 the ideal dressing should provide a moisture environment, remove excess exudate, be able to provide antimicrobial properties, facilitate autolytic debridement, prevent contamination, be non-allergenic. Easily applied and removed and be cost effective.
Hydrogels can be characterized by evaluating the swelling degree, 26 cross-linking degree,27,28 porosity,29 –31 density, free fraction32,33 a.s.o., derived from the specific structure of the basic polymer.
As a biological potential, keratin pays attention of researchers as hydrogel34 –36 for biological purposes but also as a mixture with cellulose used for epithelization of burned wounds. 37 There is not up to now in the literature an optimization study for the conditions of obtaining the keratin hydrogel, although its applicability is certain. Another criterion 38 is to obtain the hydrogel with a swelling capacity as high as possible to include a quantity of drug to cover a therapeutically necessary period (the therapeutic dose for topical administration is 14.125 mg SSD/100 cm2/24 h).
It has been reported in Radu et al. 5 and it has also been known since time immemorial that for the healing of severe burns the Chinese harvested the hair by trimming the patient’s hair and then burned it to obtain a melted mass, which they ground. The powder obtained was deposited in the wound. Accelerated healing was achieved, without the effects of foreign body rejection (the hair comes from the patient) in aseptic conditions (the hair is charred, so sterilized).
The care of patients with burn wounds involves the daily change of the dressing and the aseptic preservation of the open wound interface - the environment, which favors the recovery of the tissue by re epithelialization. This involves severe skin discomfort or the need for a daily analgesic to reduce the pain. The aim of the work is to make a textile support in different forms, depending on the location of the wound, such as: cap, removable sleeves, blouse or pants, without buttons, zippers or seams, which contains the hydrogel with medicine and a membrane, with controlled porosity. This will help to alleviate the discomfort created by the daily detachment of the dressing from the recently closed wound.
The therapy of the burned patient involves the acute need to restore the level of fluid lost through the burned skin, a liquid that can be supplemented by the presence of reservoirs of saline formed by knitted structures that diffuse through hydrogel and entrain antibiotic or analgesic to the wound. Favorable conditions are obtained for the fast re epithelialization of the wound without septic risk, the recovery of the lost fluid as well as the maintenance of a moist environment in the wound which is essential for healing. The present work aimed to optimize the factors of the process of keratin transformation from human hair to a hydrogel. This process goes through the stages of hydrolytic transformation to keratose under the form of a low-molecular mass protein, and the oxidative transformation of the protein chains under the action of hydrogen peroxide. In this work, the hydrogel realization parameters that determine a maximum swelling degree have been optimized. The final stage implies insertion of a keratin hydrogel with medicine in a textile device able to improve the patient comfort and the therapy in the clinic. Other tests regarding chemical, morphological, in vivo changes and an histopathologic evaluation on keratin hydrogel have been published elsewhere. 5 The treated wounds with keratin hydrogel presented a favorable evolution. The process of scarring, with the necrotic tissue detersion and reconstruction of damaged structures by the action of the heat agent, was carried out much faster in comparison with control, which demonstrates the important role of keratin. The keratin played the role of an important barrier, with protection and support activities for the cells involved in detersion and regeneration.
Materials
Male undyed hair was received from a Hair styling center. The solvents CHCl3, CH3OH (96%), and H2O2 (32%) were of analytical grade and were purchased from Chimreactiv SRL and NaOH was from S.C Atochim SRL. For the textile experiments have been used cotton yarn with fineness of Count Number 70/1. Isotonic solution and SSD have been purchased from current pharmaceutical stores. Polyurethane membrane (PU) was provided by Novotex Company it has been used without any other pretreatments. Chitosan with molecular weight of 100,000–300,000 was purchased from Acros Oganics Company.
Methods
Hydrogel preparation
Hair samples were milled (Retsch PM 100), getting hair fragments shorter than 1 mm. Then, the samples were cleaned 24 h in a Soxhlet using as solvent the mixture of CHCl3 and CH3OH (2:1 v/v). Defatted hair samples were placed with distilled water, inside a sealed stainless steel capsule, at a solid/liquid ratio = 2:35 (w/v). The capsule was maintained for different durations at 150°C. By using this high level of temperature in water presence inside of capsule develops a pressure of 4.9 atm which increase denaturation level of proteins in hair. After heat treatment and cooling, the samples were centrifuged for 10 min at 10,000 r.p.m. Two phases were separated: a solid one, insoluble in water, considered as beta-keratose, and one soluble in water, gamma-keratose. To prepare the beta-keratose hydrogel, 2 g of solid phase was subjected to oxidation, by boiling it in a H2O2 solution with a liquor ratio = 1:11 (w/v) for 3 h. The solid compound obtained was cleaned in 96.6% ethanol and squeezed, by pressing with a filter paper. The resulting solid compound was treated with 0.25 g NaOH, 1.6 ml distilled water, and 18 ml of 96% ethanol at 70°C for 3 h in order to obtain the hydrogel, as we previously reported. 39 The liquid phase, gamma keratose has been used for other applications.
Hydrogel porosity
To determine the porosity and density of the hydrogel, solvent replacement is the main method for determining the porosity. The method involves immersing the dry hydrogel overnight in 96.6% ethanol and weighing after ethanol saturation of the surface.29,30 The porosity formula is:
where: M1 is the mass of the hydrogel before immersion in alcohol and M2 is the mass of the hydrogel after immersion in 96.6% ethanol; ρ is the density of ethanol and V is the volume of the hydrogel.
In order to determine the volumes and then the density of the dry hydrogel, dried hydrogel is weighed and then the hydrogel is immersed in a volume determined by hexane and the increase in volume of hexane is measured as volume of the hydrogel. 31
The density is calculated using the formula (2):
where: Mh is the mass of the hydrogel and V is the volume of the solvent displaced by the hydrogel.
Swelling degree of hydrogels
The hydrogel in dry state is weighed at 20°C relative humidity 65% (Mu) then the hydrogel is maintained 24 h in distilled water at 20°C and then is weighed (Mf). The swelling degree is computed with formula (3):
where: Mf = hydrogel mass after being maintained for 24 h in distilled water at 20°C; Mu = dry hydrogel mass at 20°C and 65% relative humidity. In the paper swelling degree was tested in water and ethanol.
Experimental plan for keratin hydrogel optimization
The independent parameters taken into account were: x1 – the duration of hair hydrolytic degradation (min) in water at 150°C, x2 – hydrogen peroxide concentration (number of milliliters of H2O2) of 32%, x3 – oxidation time at boiling temperature (min). The output parameter Y1 is the hydrogel swelling degree (%).
The swelling degree controls the capacity to maintain a humid medium within the acute wound and the maximum medicine quantity that can be enclosed in hydrogel.
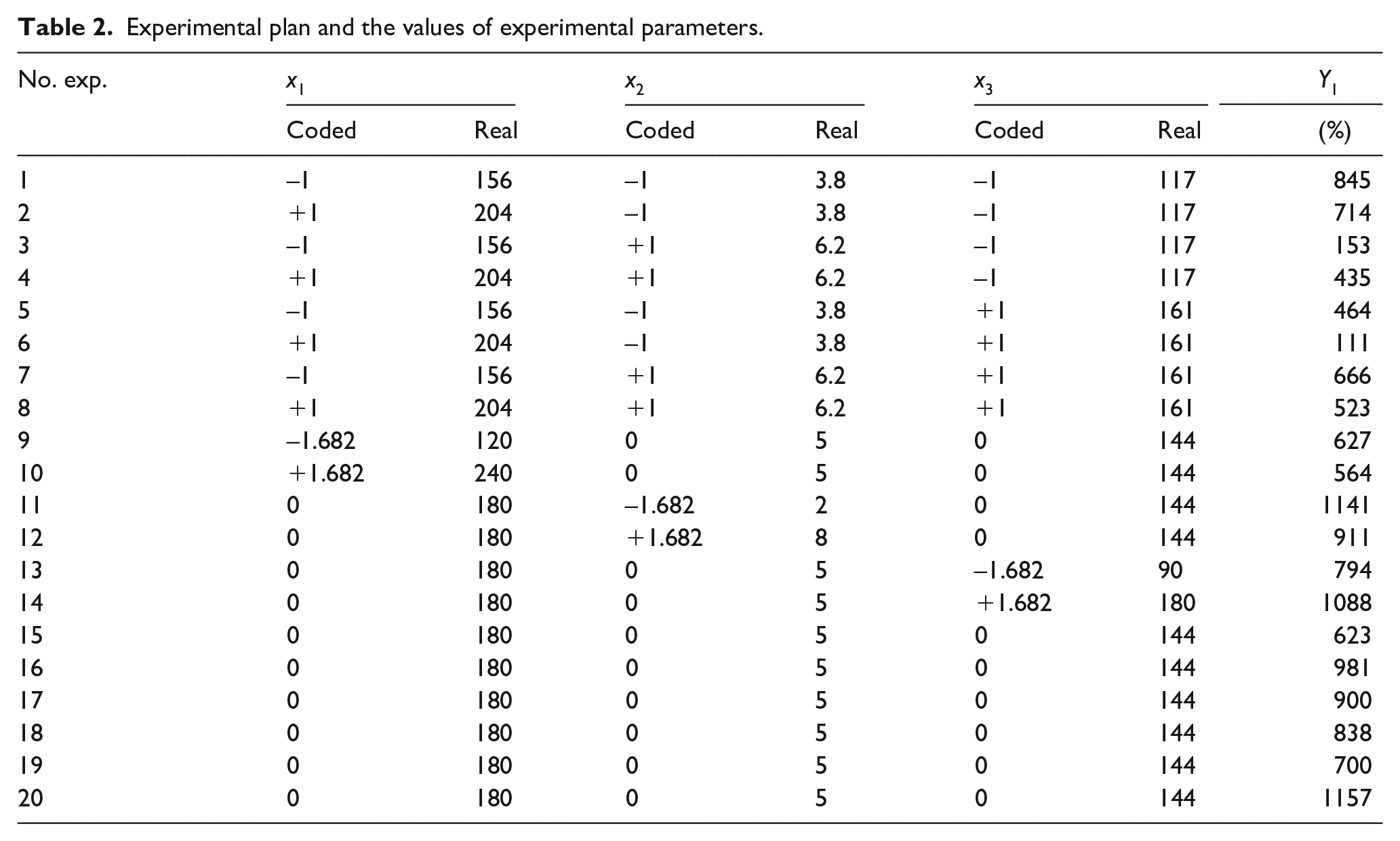
Tables 1 and 2 illustrate the range of variation and the coding of the variables x1, x2, and x3, as well as the values of swelling degree (Y1).
Limits of variation and coding independent variables.

Experimental plan and the values of experimental parameters.
Results and discussion
Experiments on swelling degree
In order to set up the experiments we have used a complex central rotable program of type 23. The independent factors were x1, x2, and x3. Y1 was considered as the output parameter. The Matlab software40 –43 was used for graphical representation. Tables 1 and 2 present the variation limits and the experimental design.
The equation that describes the interdependence between the proposed independent variables of the hydrogel production process and the considered dependent variable, Y1 respectively is given below:
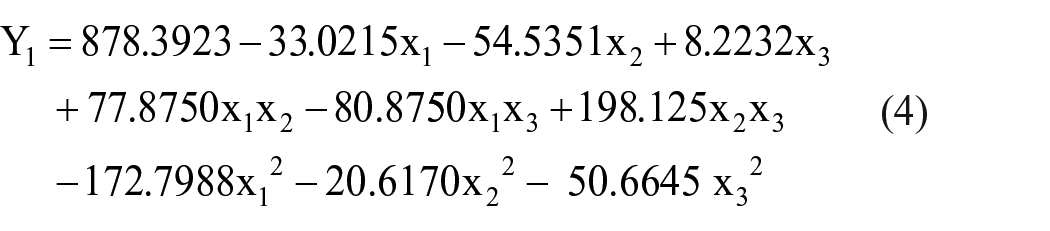
After have planned and carried out the 20 experiments, regression coefficients were determined as follows: bo = 878.3923; b1 = −33.0215; b2 = -54.5351; b3 = 8.2232; b12 = 77.8750; b13 = −80.8750; b23 = 198.125; b11 = −172.7988; b22 = 20.6170; b33 = −50.6645.
Figures 1–3 give a graphical representation of the influence of the factors: x2 and x3; x1 and x3, and x1 and x2 respectively, on the value of Y1.

Influence of independent variables x2 and x3 on hydrogel swelling degree (Y1): (a) response surface and (b) constant level curves.

Influence of independent variables x1 and x3 on hydrogel swelling degree (Y1): (a) response surface and (b) constant level curves.

Influence of independent variables x1 and x2 on hydrogel swelling degree (Y1): (a) responce surfaces and (b) constant level curves.
As seen in Figure 1, one can be observed that the maximal value of hydrogel swelling degree (1134%) is situated within the coded variation range of the parameter x2 from about −1.1 to −1.682, while the parameter x3 varies within the range from −1.682 to −0.7. This means, in real values, a variation of the values of hydrogen peroxide concentration from 2 to 3.7 ml for an oxidation time from 90 to 125 min, and a constant hydrolytic reaction time of 180 min of the hydrolytic reaction. In Figure 1, y23 stands for the influence of the factors x2 and x3 on Y1.
From the information illustrated in Figure 2(a) and (b), it follows that the maximal value of the swelling degree (781%) is the result of the coded path of the parameter x1 with values ranging between −1.1 and +0.8, and of the parameter x3 within the range from −1.5 to +1.682. In real terms, this means for x1 durations between 153 and 199 min for hydrolytic degradation, respectively for the parameter x3, oxidation times ranging between 100 and 180 min, for x2 = = constant = 5 ml hydrogen peroxyde.
From the data illustrated in Figure 3(a) and (b) it follows that the maximal value of the swelling degree (781%) is the result of the coded path of the parameter x1 with values ranging between −1.1 and +0.8, and of the parameter x3 within the range from −1.5 to +1.682. In real terms, this means for x1 a duration between 153 and 199 min hydrolytic degradation, respectively the parameter x3, the oxidation time ranging between 100 and 180 min, for x2 = constant = 5 ml hydrogen peroxide. In Figure 3, y13 represents the influence of the factors x1 and x2 on Y1.
By corroborating the data from Figures 1 to 3, one obtains the optimal variation values of the independent parameters from Table 3.
The range of independent variables optimal values and the swelling degree data obtained.

Statistical processing of data for hydrogel swelling degree optimization
Statistic data processing is meant to determine the confidence degree of presented experimental values44 –46 and it follows a procedure well known in literature. After running these stages, the confidence threshold of 99% and the obtained results are presented in Table 4.
Statistic values calculated for a 99% confidence level.

According with data illustrated in Table 4 one can notice that the parameters x1 and x3 have a Confidence Level (99%) of 15.9 and 13.2% but for x2 only 0.79%. According with these values for x1 and x3 the results are reasonable. In the case of x2 which it represents number of milliliters of H2O2 the effect is a chemical modification of keratin in products with more hydrophilic groups able to set water molecules on protein surface but in the whole procedure another step of oxidation was achieved by treating hair with water at 150°C. The processes implied meant protein denaturation by means of an oxidative stage. For this reason the parameter x2 it was not possible to be exactly evaluated in the work.
One considers that results presented are promising not only by numerical values but using hydrogen peroxide one can exploit its oxidative potential in a biocompatible way (the absence of other chemicals with an aversion to the biological environment).
Tests on porosity and degree of swelling in ethanol
For porosity tests, the main method is the solvent replacement, which implies immersion of hydrogel dried overnight, in 96.6% ethanol and then weighing the sample after surface saturation with ethanol.29 –31 Another portion of lyophilized hydrogel is immersed in hexane to determine the pores volume and then the porosity with the formula (1). Experimental data on porosity and swelling degree in ethanol were illustrated in Table 5.
Data on porosity and swelling degree of hydrogel in ethanol.
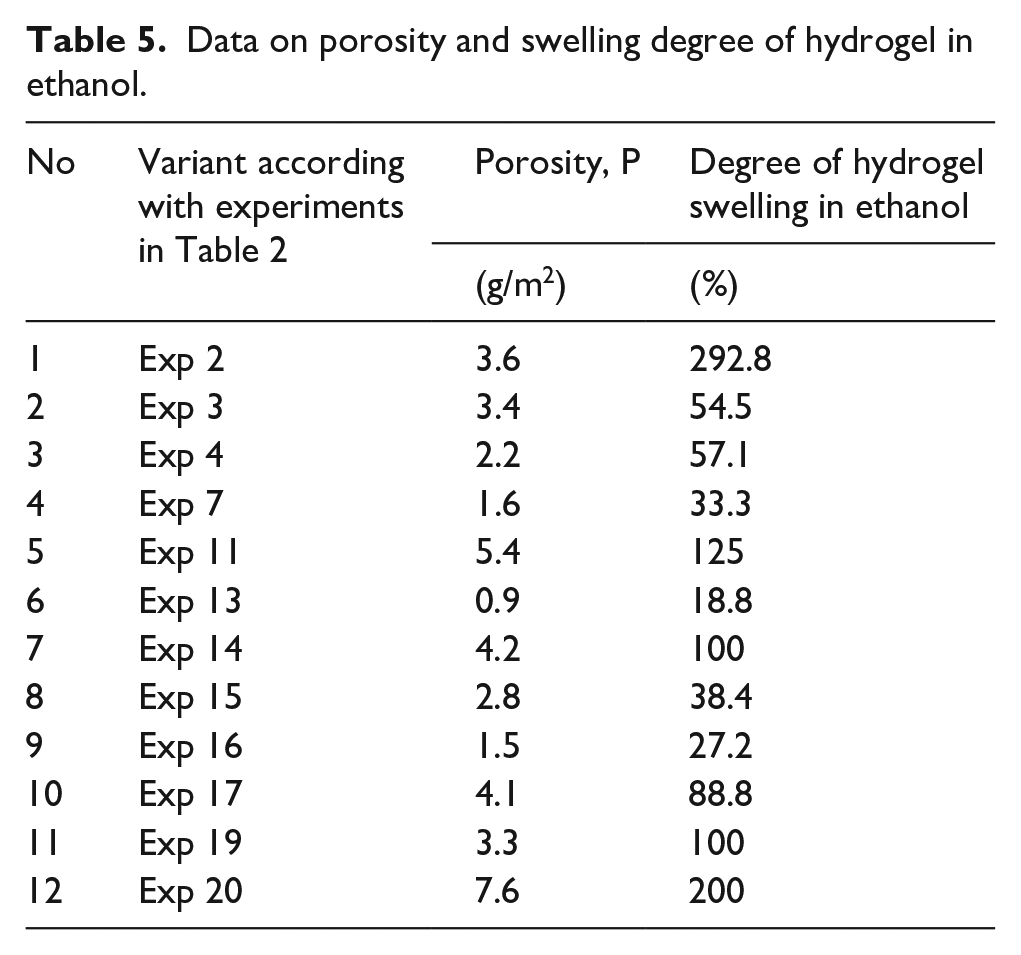
According to the values presented in Table 5, porosity covers a range of values between 0.9 and 7.6 g/m2. Because porosity is related to hydrogel swelling degree, the variants presented here follow theoretically the same variation pattern as that presented in figures illustrated in 1–3. The values (27% ÷ 292%) obtained for the swelling degree in 96.6% ethanol are much smaller than those determined in distilled water. This is the result to a much higher energetic value and frequency of the hydrogen bonds due to water, as compared to those between ethanol molecules. The significance of the latest results confirms a weaker behavior of the hydrogel in alcohol.
Other experimental considerations
The additional experiments which are presented for the first time have been performed with a keratin hydrogel, lyophilized and maintained dry as a powder at 20°C for 60 days; after immersing in distilled water and magnetic stirring at 500 r.p.m., the initial hydrogel has re-formed, with swelling degree ranging between 850% ÷ 950%. This behavior is similar to or derived from the phenomenon of “hydrogels self-healing” mentioned in literature.47 –50
Practical considerations
The determinations of the degree of swelling in ethanol have as practical support the fact some medicines are liposoluble and therefore the tests performed give an image on the efficacy of the hydrogel in the latter case. The degree of swelling in water of keratin hydrogel is significantly higher than in ethanol. Hydrogen bonds in the case of water are more effective than in the case of methanol and therefore the degree of swelling is higher in the first case. This is the reason the boiling point of ethanol is 70°C and water at 100°C.
In a different scientific context has been proved by in vivo tests and histopathological analysis 5 the biocompatibility of hydrogel in the current use.
Forming a solution tank in knit
In the first days of recovery, it is important to moisturize the wound, but also the correct hydration of the patient by infusion, to avoid unfavorable evolution of the wound in the sense of deepening, enlarging the area or infection and also in favor of improving the general condition of the patient. In order to hydrate the local lesions, in the structure of the material of which the patient’s protective pajamas is made, a free space can be created as a reservoir with a solution for wound hydration, a reservoir containing isotonic solution (0.9% NaCl, but also other components with standardized concentrations) prescribed by the clinician, which is distributed on the wound surface. The concept of isotonic refers to a solution of concentration equal to the normal physiological concentration. Infusion solutions are used to make up for fluid loss from burn / burns, to correct metabolic imbalances (diabetic acidosis) with or without burns, to supplement electrolyte, enzyme deficiency, diuresis disorders, edema reduction,6,51 –54 restoration of carbonized tissue.
For the textile support, a yarn with fineness of Count number = 70/1 of 100% cotton is used, which is knitted using interlock and jersey structures, as illustrated in Figure 4. Then the knit is boiled alkaline at a liquor ratio of 1: 100 using a recipe with: 3 g/L NaOH, 6 g/L Na2CO3, and 1 g/L anionic tensid for 2 h; then wash with hot distilled water (90°C) for 5 min twice; then a treatment with hydrogen peroxide using the recipe: 2 ml H2O2/L (32%) and 2 g/L Na2CO3 for 1 h; squeeze and dry at 120°C, 4 min. A bleached, hydrophilic and sterile knit is obtained.

Diagram of the knit and hydrogel assembly with the drug, respectively of the transfer of the solution to the dermis.
The diffusion of the liquid to the wound takes place through the pores or through the knitting stitches. Controlling the pores and the knitted structure is a difficult task which means a great deal of experimental effort; but it is possible to apply a membrane (e.g. polyurethane, Novotex type PU with a pore size of 0.2 μm) fixed on the knit by rolling at 120°C for 10 s, which achieves the required flow of liquid fluid by passing from the textile structure to the dermis. This stage takes place by following and supervising the procedure by the clinician. When using knitting systems, the diagram of the infusion tank and the diffusion to the wound is illustrated in Figure 4.
The use of several solution tanks is necessary to attenuate the effect of gravity, which makes the speed of the transfer of liquid through the pores at the bottom higher than at the top. This is due to the increased hydrostatic pressure when using a single tank. The entire system is customized according to the surface and location of the patient’s wounds. The isotonic solution diffuses through the mesh of the knit (see Figure 4) traverses the hydrogel with SSD extracting the drug and leads it through the pores of the membrane to the burned dermis.
The tanks can be included in the structure of a 100% cotton pajama made of cotton yarn with the fineness of Count Number = 70/1, consisting of blouse and pants, without buttons, zippers and seams to avoid any clothing discomfort for skin sensitized by the wound burned. As textile devices can be used: caps, detachable sleeves, blouse or pants depending on the surface and location of the affected area.
In the lines below, stages of obtaining the drug delivery device are presented.
The processing of textile structures by knitting;
Alkaline boiling and hydrogen peroxide treatment of knitted structure;
Washing and drying knitwear;
Mixing hydrogel with SSD;
Depositing the mixture into the pocket of the knitwear;
Isolation of the knitwear with a thermo-adhesive membrane;
Sterilization of the device and packing.
When applying the device onto the wound, the isotonic solution is injected into the tanks of the knitwear.
Textile devices for limiting sepsis and painful shock
Additional action can be taken to improve the condition of the burnt patient by using a mixed system consisting of a bacterial growth inhibitor and a second analgesic/antalgic agent by attaching a drug delivery system which is illustrated in Figure 4. Antalgic means a substance that relieves pain, and the analgesic stops the pain for a limited time.
The hydrogel with the drug is made of keratin, the parameters of which have been optimized in the work. The isotonic solution diffuses through the system and swells the hydrophilic areas it goes through, and drags the drug to the burned wound.
Thus, we can consider that the textile ensemble consists of a pajama consisting of a blouse and pants, without seams and customized to the particular dimensions of the patient. In the outer part there is a thermally insulating textile surface, and in the inner part the device from Figure 4 is assembled.
Conclusions
The optimal parameters for obtaining the maximal swelling degree (1134%) are the following: duration of hydrolytic reaction = 180 min, volume of hydrogen peroxide = 2.0 ÷ 3.7 ml, and duration of oxidation = 90 ÷ 125 min. The major influence one obtained on duration of hydrolytic and oxidation reaction.
The hydrogel performances are better in interaction with water compared with ethanol. This study proves that compared to other materials, hydrogel is a porous material with special properties in terms of swelling degree, humidity preservation within interstices or self-healing.
The designed textile assembly hydrates the wound and easily detaches the device for administering the drug; the textile assembly can be reused by loading with isotonic solution using a syringe. The use of hydrogel means a new amount of keratin hydrogel with an amount of SSD prescribed by the clinician depending on the evolution of the wound and the need for medication.
The proposed device facilitates the application and practical fixation of the hydrogel in the wound for the release of the antibiotic or for the use of an analgesic.
Footnotes
Acknowledgements
The authors are grateful to Prof. L Verestiuc and Dr. C Tamas from University of Medicine and Pharmacy “Gr. T. Popa” Iași from their important advices in research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was sponsored by Doctoral School of Technical University Tehnice “Gheorghe Asachi” Iași. The paper was a part of a Ph.D. thesis defended in 2021.



