Abstract
In this work, willow fiber was extracted from willow inner bark and modified with alkali solution of various concentrations, temperature and time. The morphology, surface functional group, crystal and thermal stability were investigated by using a scanning electron microscopy, a Fourier transform infrared spectroscopy, an X-ray powder diffractometer, and a simultaneous thermal analyzer, respectively. The acid (H2SO4, 2 ml/l) extraction procedure removed the hemicellulose and part of lignin from willow bark, cleared the aggregation of WB, kept the crystal structure of willow fiber, dramatically increased the crystal length of fiber (from 18.46 to 30.15 nm), and enhanced the onset degradation temperature (from 262.23℃ to 297.62℃) and chemical reactivity (DTG: from 0.57%/s to 0.84%/s). The alkali treatment further removed lignin from willow fiber, smoothed the fiber surface, increased the intensity of cellulose
Introduction
Natural fiber materials were focused progressively due to the ever-growing global energy crisis and ecological risks. According to the study of life-cycle assessment, natural fibers reduced the usage or replaced the synthetic fibers due to their light weight, good renewability, large quantity, and low price. 1 Ramesh et al. 2 and Väisänen et al. 3 reviewed six kinds of plant fibers, including bark/stem, leaf, fruit/seed, stalk, reed/grass, and wood fiber, but did not mention willow fiber, which is inner bark fiber extracted from willow tree bark. Willow tree is Salicaceae plant, with 450 species in the world and 320 species in China, 4 and it often grows along pools, roads and streams. It is one of the most welcome tree and widely planted due to its unparalleled environmental adaptability and 20 times the growth rate of other wood species. 5
Willow bark (WB) is the bark of willow tree, which consists of inner bark and outer bark. Outer bark functions to protect willow tree from outside attack and to maintain moisture, which contains resins, latex, and other valuable substance. 6 Inner bark is the source of medicine extractives 6 and fibers. Salicin in inner bark can be absorbed by enzymes into salicylic acid and made into painkiller. 7 U. S. Pharmacopeial Convention reported that inner bark was able to cure pain, osteoarthritis, headaches, and inflammatory conditions. 8 In China, willow inner bark can also be used to treat detoxification, rheumatism, bronchitis, hypertension, water sores, and other diseases. 9 Additionally, inner bark fiber played a role as fillers to composite with polymers, such as poly (lactic acid) 10 and polyethylene.11,12
Therefore, it is necessary to separate WB into inner and outer bark, and it is inner bark that fiber can be extracted fiber from. Oktaee 13 has immersed the inner bark into hot water for 6 weeks to get willow fiber. However, this method might only be applicable to young willow tree, such as 3–4 years old willows. Wang and Wang 4 used two-steps chemical treatment with H2SO4 and NaOH respectively to obtain willow fiber. After the treatment, the cellulose ratio in inner bark was improved from 25 wt% to 43 wt%, while the hemicellulose weight ratio was reduced to 19% from 21%. Dou et al 6 investigated the chemical composition of willow fiber extracted through three methods (hot water, alkali, alkaline hydrogen peroxide) and found that lignin content was about 24%, 13%, and 8.5%, and sugar ratio was about 55%, 82%, and 91%, respectively. These results indicated that the application of acids and alkali could obtain willow fiber with high content of cellulose and sugar.
The alkali treatment is one of the most widely used chemical processing for natural fiber, which is able to remove a certain amount of lignin, hemicellulose, pectin, and other organic compounds from the surface of fibers. 14 Meanwhile, rougher fiber surface can be obtained by using alkali,15,16 which is very beneficial for fibers to compatible with polymer matrix. The mechanical properties of alkali treated Acacia Arabica fibers composite were enhanced due to its rough surface and well interfacial compatibility. 15 Bhoopathi and Ramesh 16 found that the thermal stability and crystallinity of eggshells were improved after being treated with alkali, and the compatibility between eggshell-matrix was excellent. NaOH treated Tapsi fiber possessed 43.02 and 160 MPa of tensile and flexural strengths, as well as high thermal stability. 17
However, the effect of treating concentration, temperature, and time of alkali on thermal and crystal behaviors was uncertain. Mishra et al. 18 treated sisal fibers with 5 and 10 wt% NaOH solution and found that 5 wt% NaOH showed greater improvement than 10 wt% NaOH in terms of tensile strength. Li et al 19 treated mulberry fiber by using 1 w/v% NaOH and 1 w/v% Na2S at 80℃ and 130℃, respectively, and concluded that the the cellulose content was higher, the hemicellulose and lignin content were lower at treating temperature of 130℃ than at 80℃. Wang et al. 20 applied NaOH to treat straw with conditions: 1−3 wt% of concentration, temperature at 45℃−85℃ and the treating time in range of 6–36 h, and obtained the optimal conditions in term of gas production rate: 3 wt%, 85℃ and 36 h. However, for willow fiber, the treating conditions were still not determined.
This paper investigated properties of willow fiber affected by alkali treatment. The main focus were: degumming the willow inner bark with acid, modifying the willow inner bark with alkali treatment with various concentration, temperature and time, investigating the morphology, surface functional group, crystal and thermal stability of the extracted willow fiber.
Material and methods
Material
Willow bark (WB, Figure 1(a)), obtained from a local planter in Guilin city, and was stripped from a 20 year-old willow tree. WB was brown and dimensioned about 20 mm in thickness, which includes outer bark and inner bark. The outer bark was very rough and loose in structure, cracked along the growth direction of willow tree, and was about half thickness of WB. The inner bark was smooth and dense, which would provide fibers. Therefore, the outer bark was reluctant in this study and should be separated from the inner bark. In order to debark easily, the willow bark was immersed in water for 3 days at room temperature (Figure 1(b) and (c)). The bark was manually separated into inner and outer bark with a putty knife (Figure 1(d)). An air hammer machine (20 kg, Tengzhou, China) was used to strike the inner bark for loosening its tissue (Figure 1(e)). An acid (H2SO4, 2 ml/l) degumming procedure was studied in hot water bath of 60℃ for 5 h with liquor ratio of 1:5 (Figure 1(f)). After degumming, inner bark fiber was obtained (Figure 1(g)), and then for further experiment (Figure 1(h)), it was ground into powder and labeled as WP.

Extraction process of willow bark fiber: (a) willow bark, (b) water-immerse bark fiber, (c) willow bark after being water-immersed, (d) willow inner bark, (e) striking inner bark, (f) water-bath, (g) inner bark fiber, and (h) inner bark fiber powder.
Modification of WP
The prepared WP showed strong aggregation, so that three kinds of NaOH treatments were carried out. A certain of WP was treated with: (1) 2 wt%, 5 wt%, 10 wt%, and 15 wt% concentration of NaOH under 30℃ in 5 h, and then obtained W2N30T, W5N30T, W10N30T, and W15N30T powders; (2) 5 wt% of NaOH under 30℃, 45℃, 60℃ and 75℃ in 5 h, and got W5N30T, W5N45T, W5N60T, and W5N75T powders; and (3) 5 wt% of NaOH under 30℃ in 1, 3, 5, and 7 h, acquired W5N1H, W5N3H, W5N5H, and W5N7H powders. Finally, the WP powder was treated by using a microwave under medium baking temperature for 1 min and got powder WPM.
Materials characterization
Effect of various treatment processes on the surface functional groups of WP were investigated by using a Fourier transform infrared spectrometer (FTIR-650, Gangdong, China) with a resolution of 1 cm−1. 2–3 mg dry WP was ground with 100–200 mg KBr powder in an agate crucible and then pressed into a tablet for testing in a range of 4000−400 cm−1.
A simultaneous thermal analyzer (WCT-121, China) was applied to investigate the thermal stability (TG). WP sample weighing 10–15 mg was heated to 500°C with a rate of 10°C/min, and then held at this temperature for 5 min, finally freely cooled. 20 mL/min flowing nitrogen was applied to the procedure.
Crystalline of WP were characterized with a X-ray powder diffractometer (RigaKu D/MAX 2500V), equipped with a Cu Kα radiation source (λ = 1.54060 nm), operating at a voltage of 40 kV and an intensity of 40 mA, over the incidence angle (2θ) in the range of 2°−50°, at the room temperature. The crystal index (Ic, %) was calculated by using Segal equation 21 (1), and the crystal grain size (Lc, nm) was calculated with Scherrer formula 22 (2):
Where I002 denoted the intensity of cellulose in the crystal region (counts), and Iamp denoted the intensity of amorphous peak (counts).
where K denoted Scherrer constant, which was in the range of 0.9–1 and is normally determined as 0.94 for fibers; β (rad) was the half-width of the calculated radial intensity distribution curve of the crystal plane.
The morphology of gold-coated WP was inspected by using a scanning electron microscope (SEM, HITACHI SU8020 system).
Results and discussions
Morphology of WP
Figure 2 demonstrates the morphology of the untreated and treated willow inner fiber, both of which showed as rods with diameters in range of 30–100 μm. Willow bark (WB) powder showed a very wide range of dimension and was aggregated strongly (Figure 2(a)), which could contribute to the hemicellulose, pectin, and impurities in WB. Aggregation would lead to uneven distribution of WB in polymer matrix and poor interfacial compatibility, consequently lowering down the thermal stability, and mechanical properties of WB-polymer composites. 16

Morphology of willow fibers: (a) WB, (b) WBM, (c) WP, (d) W5N1H, (e) W5N5H, and (f) W5N60T.
The aggregation (green circle) of WB was not been cleared but alleviated through microwave treatment, and loosen the cellulose structure of natural fibers by providing heater, shown in Figure 2(b). The WP fibers stayed separately without any aggregation (Figure 2(c)), indicating that the degumming procedure removed the hemicellulose, impurities and lignin from WB (further proof with FTIR test). However, the surface of WP fiber was rough, indicating matters residual (red circle), which would affect the compatibility between fibers and polymers.
An hour NaOH treatment smoothed the fiber surface (Figure 2(d)), indicating the residual matters were removed further. Extending the NaOH treatment to 5 h has loosened the fibers (Figure 2(e), purple arrow), which is beneficial for fibers to compatible with polymer matrix. Raising the temperature of aqueous alkali, some fibers were boiled into fragments (green circle), while others became thinner (Figure 2(f)), indicating that the higher the temperature is, the looser the fiber structure is.
Fourier transform infrared spectrum of WP
Fourier transform infrared spectrum

FTIR of willow fibers affected by various treatments: (a) degumming and microwave, (b) various treating time of alkali, (c) various treating temperature of alkali, and (d) various alkali concentration.
Figure 3(b) to (d) give the effect of various treating time (1, 3, 5 and 7 h), treating temperature (30℃, 45℃, 60℃ and 75℃) and concentration of alkali (2 wt%, 5 wt%, 10 wt%, and 15 wt%) on the spectra of WP, respectively. With the increase of treating time, the bands at 3418 cm−1 became broader and shallower than that of WP, which performed the most obviously at 5 h (sample W5N5H). This result indicated the number of -OH groups decreased. During the NaOH treatment, -OH groups of willow fiber reacted with NaOH, resulting in the bonding between Na+ and O-H of fiber, along with the water molecules (H-OH) product. 14 Meanwhile, Figure 2(e) proved that the structure of willow fibers was looser with 5 h alkali treatment than with 2 h due to the more removal of -OH groups in sample W5N5H. However, 7 h treatment might break the structure of cellulose, resulting in the wider of band 3418 cm−1. The peaks at 1063 cm−1 and 1247 cm−1 became weaker as the treating time extending, indicating that the lignin of willow fiber was further removed. Therefore, the optimal treating time was 5 h for willow fiber in this study. In other study, 2 h NaOH treated sugar palm fiber reinforced polyester composite had the optimum bending properties, when compared with 4 h, 6 h and 8 h. 24
As the treating temperature rose, the band around 3418 cm−1 and at 1063 cm−1 gradually became shallower, and got to the shallowest at 60℃. In higher temperature, the hydrolytic cleavage of glycosidic bonds due to the penetration of hydronium ions was more intense, resulting in the reduction of cellulose region. 19 Therefore, 60℃ was the optimum temperature for willow fiber.
Figure 3(d) showed that the band around 3418 cm−1 and at 1063 cm−1 was little shallower than that of WP at 2 wt%, 5 wt% and 15 wt% alkali. Only 10 wt% concentration of alkali functioned obviously on WP, which produced the shallowest band at 3418 cm−1 and almost level band at 1063 cm−1. Other researchers obtained various results: Radzi et al. 25 treated sugar palm fiber and found that the intensity of hydroxyl group (band around 3328 cm−1) was decreasing with the increase of alkali concentration from 0 wt% to 9 wt%; Orue et al. 26 found that 7.5 wt% alkali treated walnut shell possessed a weaker intensity at band 3350 cm−1; Rajeshkumar et al.28 treated Phoenix Sp. Fibers with 5%, 10%, and 15% NaOH, and obtained the highest tensile strength at 15% NaOH and the highest tensile modulus at 5% NaOH.
Crystalline of WP
The XRD curves of WP affected by various treatments are shown in Figure 4. The crystal index (Ic) and crystal grain size (Lc) of willow fiber calculated according to equation (1) and (2) were listed in Table 1. In Figure 4, the diffraction peaks at about 14.8°, 15.2°, 22.4°, 24.3°, 30.1°, 35.9°, and 38.2° presented the typical form of cellulose Ⅰ.4,19 Although XRD spectrum of the degumming product (WP) was similar to that of its original willow fiber (WB), the diffraction intensity at 22.4°of WP (646 counts) was much stronger than that of WB (301 counts), resulting in a dramatic increase of Lc (from 18.46 to 30.15 nm). Meanwhile, the similar spectrum indicated that the degumming did not change the crystal structure of willow fiber. WB consists of amorphous phase (hemicellulose and lignin) and crystal region (cellulose), the former one of which hampered the crystal process of WB, resulting in small crystal size. The degumming removed hemicellulose and part of the lignin in the amorphous region, resulting in the realignment cellulosic chains 19 and obtaining the higher WP diffraction intensity.
Crystal index and crystal grain size of willow fiber.

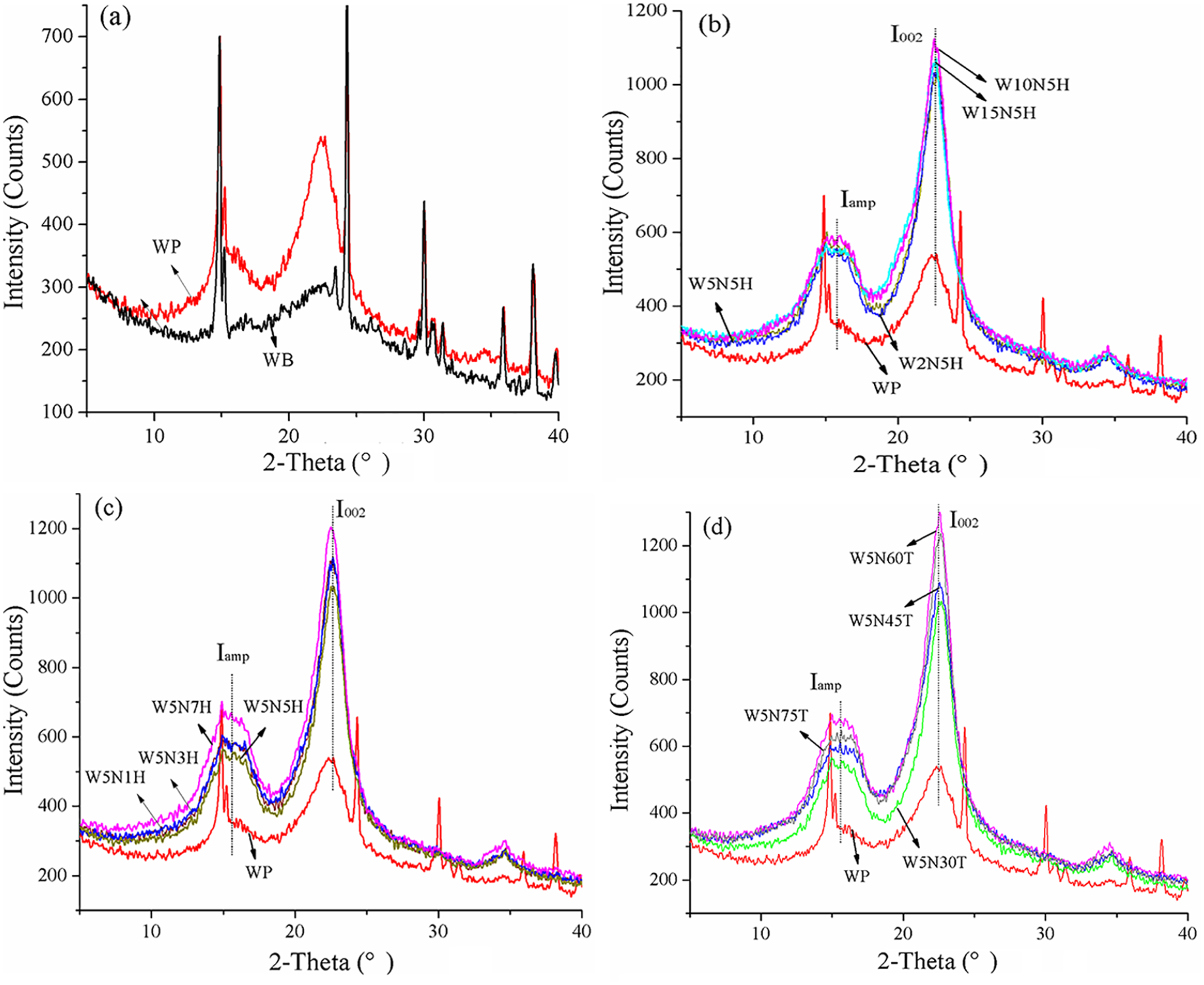
XRD of willow fibers affected by various treatments: (a) degumming, (b) alkali concentration, (c) treating time of alkali, and (d) treating temperature of alkali.
After being further treated by NaOH with various concentrations, temperature and time, all the crystal peaks (I002) of treated fiber became much sharper than that of WP, indicating an obvious increase of crystallinity. Meanwhile, only a main peak at 22.4° (I002) presenting the crystal region and a shoulder peak at about 15° (Iamp) representing amorphous region appeared on XRD curves of treated fiber, while other peaks at 24.3°, 30.1°, 35.9°, and 38.2° disappeared due to the enhancement of I002. This result contributed to the further removal of lignin, leading to the reduction of amorphous region and better packing of cellulose molecules. The crystal index enhancement due to the NaOH treatment has been reported in other literatures.19,21,26
Increasing the concentration of NaOH from 2 wt% to 15 wt%, Ic and Lc rose from 44% and 39.05 nm (W2N5H) to 47.21% and 39.5 nm (W5N5H), following by a steady drop to 45.94% and 37.65 nm (W15N5H), respectively (Figure 4(b)). Therefore, suitable concentration of alkali (5 wt%) was benefit to reduce the amorphous phase and get the lowest Iamp (549 counts). Further increase ratio of alkali broke the structure of cellulose, resulting in the enlargement of amorphous region and shortening of crystal grain size, the former got to the maximum with 591 counts of Iamp at 10 wt% alkali, and the latter reached to the valley at 15 wt% alkali, respectively. Similar results were observed for sisal fiber 27 and Phoenix Sp. Fiber. 28
Varying the treating temperature in the range of 30℃−75℃ (Figure 4(d)), Ic and Lc climbed to the peak of 48.68% and 41.84 nm at 60℃, respectively. Rising the treating temperature enhanced the penetration of hydronium ions and increased the amorphous phase, resulting in the increase of Iamp (663 counts at 60℃). Meanwhile, the lignin in WP was removed further, leading to the enhancement of crystal region and the rising of I002 (1292 counts at 60℃). However, the higher temperature (75℃) broken the crystal structure of WP, resulting in the reduction of crystal region (I002 was 1228 counts) and crystal length (Lc was 39.53 nm).
Extending the treating time from 1 h to 5 h reduced both crystal and amorphous region with I002 and Iamp of 1040 counts and 549 counts. On one side, further prolonging the time to 7 h, the reaction between NaOH and fiber was dramatic, resulting in the enlargement of amorphous region (Iamp was 666 counts). On the other side, the further removal of lignin and hemicellulose promoted the crystal of fiber, resulting in the rising of I002 (1205 counts). However, the former took the key role in the reducing of Ic and Lc.
Those results indicated that controlling the concentration and temperature of NaOH treatment beneficial to obtain good function of WB. A higher crystal index and larger crystal size signified good strength and stiffness. 22
Thermal stability of WP
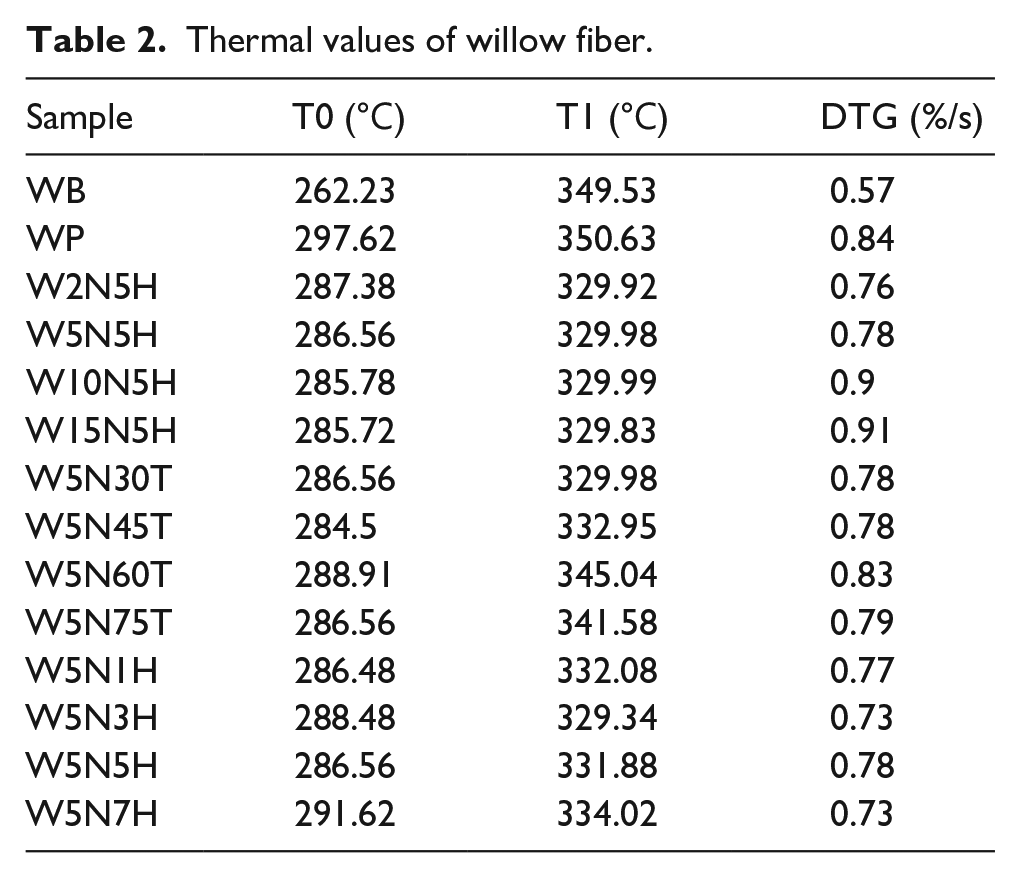
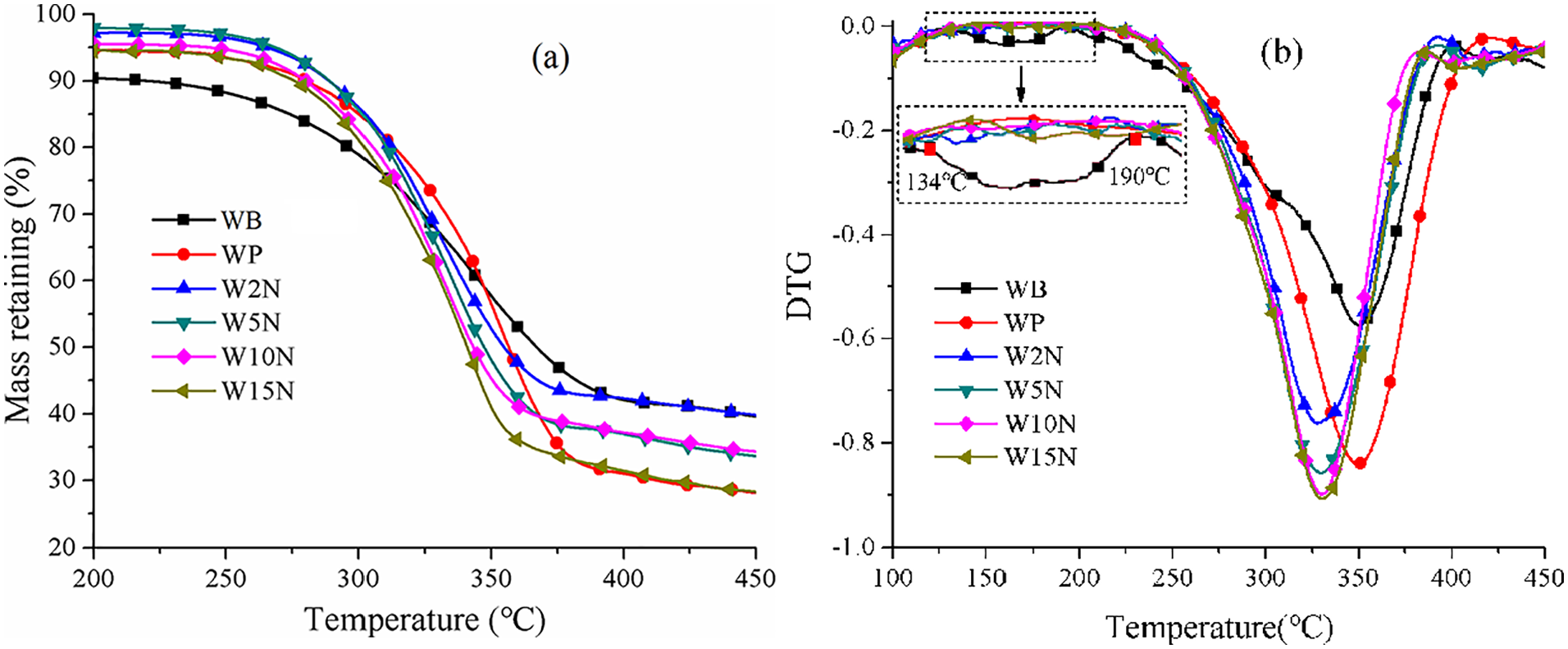
Figures 5 to 7, as well as Table 2 give the thermal stability of WP affected by various treatments with alkali. WB, its degumming fiber and modified fiber exhibited single-step degradation over the entire temperature range. WB experienced two endothermic transformations (Figure 5(b)), one for hemicellulose between 134℃−190℃ and another for cellulose in range of 190℃−400℃. The containing hemicellulose pushed it degrade from 262.23℃ (Figure 5(a)), embodying much inferior thermal stability. The hemicellulose peak did not show up on DTG curve of other treated fibers, indicating that the degumming removed the hemicellulose from WB. All the TG curve of treated fiber moved to the right of that of WB, while all DTG curves of treated fiber went downward, showing an improved thermal stability and reactivity.
Thermal values of willow fiber.
(a) TG and (b) DTG curves of willow fiber affected by alkali concentration.

(a) TG and (b) DTG of willow fiber affected by treating temperature of alkali.

(a) TG and (b) DTG of willow fiber affected by treating time of alkali.
After alkali treatment, the onset degradation temperature (T0) and the temperature at the highest degradation speed (T1) of fiber decreased from 297.62℃ and 350.63℃ to 284.5℃ and 329℃, respectively, indicating the thermal stability of WP was lowered by introducing alkali.
Increasing the concentration of alkali from 0 wt% to 15 wt%, T0 and T1 of treated fibers almost kept stable, but DTG of which increased from 0.84%/s to 0.91%/s. The higher concentration of alkali, the more lignin in WP was removed. Lignin degraded from 160℃ to 900℃, so that increasing concentration lowered the thermal stability of WP. Meanwhile, alkali penetrated into the cellulose structure and loosed it, resulting in enhanced reactivity, which was conductive to WP as fillers of polymers.
Varying the treating temperature between 30℃−75℃, T0, T1, and DTG of willow fiber reached to the climax values of 288.91℃, 345.04℃ and 0.83%/s at 60℃ (Figure 6(a) and (b)), indicating an improved thermal stability and reactivity. Appropriate increase of the treating temperature promoted the repacking and realignment of cellulosic chains, thus improved the thermal stability. However, continuous increasing the temperature would break the cellulose structure, leading to the reduction of thermal values. Figure 7 gives the effect of treating time on the thermal stability of willow fiber. It can be seen that a longer time (7 h) enhanced T0 and T1 to 291.62℃ and 334.02℃, respectively, although the values showed fluctuation.
In general, the choice of treating concentration, temperature and time functioned to the thermal stability of willow fiber. Here, the most suitable treating condition of alkali was: 5 wt% of concentration, 60℃ and 7 h.
Conclusion
In this study, willow fiber was extracted successfully from willow inner bark, which was a plentiful biomass planted widely in China. The acid (H2SO4, 2 ml/l) extraction procedure removed the hemicellulose and part of lignin from willow bark, and meanwhile cleared the aggregation of WB. The alkali treatment further removed lignin from willow fiber and smoothed the surface.
The result of FTIR showed that with the increase of the treating time, temperature or alkali concentration, the band around 3418 cm−1 and at 1063 cm−1 became shallow, and got to the shallowest at 5 h, 60℃ and 10 wt%, indicating that the number of -OH groups decreased and the lignin was removed furthest.
The XRD result showed the degumming did not change the crystal structure of willow fiber. The diffraction intensity at 22.4° of WP (646 counts) was much stronger than that of WB (301 counts), resulting in a dramatic increase of Lc (from 18.46 to 30.15 nm). After NaOH treatment, all the crystal peaks
Extraction procedure enhanced the thermal stability and reactivity of willow fiber, but introducing alkali lowered the thermal stability of WP. Varying the treating condition, the willow fiber got to the optimum thermal stability at 5 wt% of concentration, 60℃ of treating temperature and 7 h treating time.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was financially supported by the institution of National Natural Science Foundations of China (51705094 and 51965014), Natural Science Foundations of Guangxi province (2018JJA160268), Middle-aged and Young Teachers’ Basic Ability Promotion Project of Guangxi (2020KY21012) and Natural Science Foundations of Guilin University of Aerospace Technology.


