Abstract
Viscose dyeing is one of the major pollutants of water due to the large amount of salt in the dyeing effluent. This study paves the way for improving environmentally sustainable wool waste and highlights a promising invaluable application through salt-free viscose dyeing. The keratin hydrolysate (KH) was obtained using microwave (MW) alkaline hydrolysis then applied on the viscose fabric in the finishing bath formulation using the pad-cure technique. The rheology of the hydrolyzed wool fibers and the amino acids composition using high-performance liquid chromatograph (HPLC) was estimated; furthermore, the fourier transform infrared spectroscopy (FTIR) of freeze-drying keratin hydrolysate was evaluated. Microwave-assisted keratin hydrolysis leads to the breakdown of peptide bonds and the release of low molecular weight proteins and peptides. The color strength (K/S) of the dyed post-finished viscose fabric increased 75% compared with that dyed by conventional technique. FTIR, scanning electron microscopy (SEM) and energy disperse x-ray spectroscopy (EDX) demonstrated and confirmed the effective finishing of keratin hydrolysate. The tensile strength and elongation of viscose fabric did not change after finishing with KH, while the air permeability improved and the light fastness properties for the modified viscose fabrics.
Introduction
Reactive dyeing has maintained the highest annual cellulosic fabrics dye usage globally due to its shade brightness, variety of color, wet fastness, and high applicability. It is well known that the cellulosic fabric surface has a negative charge in the water, and the reactive dye is anionic, resulting in an electrostatic repulsion that causes difficulties in the dyeing process. Therefore electrolytes (such as sodium chloride and sodium sulfate) are applied to reduce the charge barrier. But the challenge in reactive dyeing is that about 70%–80% of the dye is exhausted (low dye use). Different rinses extract the rest of the electrolytes and non-fixed dye. This increase in the amounts of electrolytes in the wastewater harms aquatic life. Additionally, it has destructive effects on pipelines where alum-sulfate complexes penetrate and crack the concrete with a considerable amount of sodium sulfate as an electrolyte. This may lead to the emission of hydrogen sulfide gas under anaerobic conditions. The destruction of these sulfides and subsequent microbial oxidation may lead to sulfuric acid production. 1
Different techniques are being used to enhance color fixation and reduce environmental problems.2,3 As bio-polishing, cationization, ultrasonic, microwave (MW), and ultraviolet radiation (UV) have attained a great deal of interest. Modifying the cellulosic fabric surface is one of the favored strategies for reducing salt usage, like cationization of the fabric has achieved much consideration due to its simplicity. 4
The cellulosic-containing compounds are cationized through the introduction of amino groups for increasing the affinity to anionic dyes. In dye baths, such cationized fabric will be dyeable under neutral or mildly acidic conditions without electrolyte and alkali. For several years cationized material has been commercially available by many manufacturers for dyeing yarn, knits, woven, and apparel, reduce 50% of the chemicals required. 5
Such modification is performed using either polymer such as polyamide epichlorohydrin or compounds such as choline chloride or biopolymers such as chitosan, collagen hydrolysate (a byproduct of the leather industry), and keratin hydrolysate (KH) (a soluble natural fibrous protein byproduct that is commonly available in many industries, such as textile wool industry). 6
The published literature shows that keratin is a raw material to manufacture products with added value. High amounts of keratinous bio-waste generated by the food, wool, and livestock industries can be used as organic fertilizers to develop industrial-scale Keratin and its hydrolysates. Evaluating keratin waste will produce various consumer goods, improve environmental contamination, and raise the pharmaceutical, food, and cosmetic sectors. 7
While keratin hydrolysate (KH) is used in many applications, its consumption is relatively low, and substantial quantities are wasted and cause environmental problems. The circular economy aims to extend material life and encourage recycling to maximize material services while reducing environmental impact and resource utilization. 8 The cationization route using eco-friendly waste represents a step forward in this investigation, suggesting the possible application of KH to modify the surface of the viscose fabric and eliminate salt consumption throughout reactive dyeing. 9 To design different applications with increased green housing effect, the development of new and advanced ecological materials and research into keratin’s biochemistry, structure, physical, and chemistries is essential. 10
The textile industry consumed water and energy for wet processing, such as washing, dyeing, rinsing, desiccating, and bleaching. The energy costs are critical for developing the market competition like many applications such as food manufacturing, wood treatment, polymer, and biochemistry. MW, UV light radiation research has attempted to minimize processing time. It could be used in heating, drying, condensing, dyeing and pressing, finishing, changing surface textiles, and enhancing the product’s quality.11–13
Bhatti et al. used UV light to irradiate cotton fabric and dye solutions and dyeing the un-irradiated and irradiated cotton with un-irradiated and irradiated Reactive Blue 13. The results revealed that UV radiation for cotton and dye enhanced the grade of fastness, improved the irradiated fabrics’ color strength, and enhanced dyeing characteristics. Additionally, low irradiation time for dye solution and cotton fabric is more efficient for obtaining good color strength. In another study utilizes UV light to irradiate cotton and polyester textiles, and dyeing was done for irradiated material with a dye solution containing un-irradiated multifunctional reactive and dispersed dyes. UV light was proven to boost the color strength values of cotton and polyester textiles.14,15
Sustainable extraction of effective dyes using microwave radiations (green technology) from dye-yielding plants, such as Arjun bark’s natural colorant for mordant wool fabric dyeing, is gaining popularity due to its low-cost, short process time, and effectiveness. 16 It is found that microwaves enhanced the dynamic behavior of a natural coloring for wool dyeing, and acceptable fastness qualities are obtained under ideal conditions. In another recent study MW irradiation was administered to isolate natural dye from tea leaves under alkali conditions. It is found that MW energy has an excellent capacity to isolate colorant from tea leaves for dyeing cotton fabric at lowered ideal dyeing conditions. 17
Another study investigated the influence of microwave irradiation on the dyeing of cotton fabric with Reactive Blue 21. Irradiated and un-irradiated cotton textiles were colored under various circumstances using irradiated and un-irradiated dye solutions. The result revealed that Low irradiation time for cotton fabric and dye solution is more effective in producing good color strength and a bluer shade. 18
The present study is conducted to enlighten on the MW as a green technology for extracting keratin hydrolyzed (KH) of wool waste fiber under alkaline conditions and compared with hydrothermal heating through evaluating the color data of the dyeing process of the finished KH viscose fabric (free salt dyeing) and compared with the standard dyeing process containing necessary additives such as inorganic salt sodium chloride (NaCl). The use of KH has a dual function; the first one is bio-waste becomes degradable KH consequently reduces pollution, and the second one is a dyeing exhauster that enhances the affinity to reactive dye. Finally, to determine the dyeing quality, color yield, fastness to washing, and rubbing have been evaluated. In this study, the MW irradiation and hydrothermal influence on KH using amino acid analysis, apparent viscosity, and FTIR.
Experimental
Materials
Fabrics
One hundred percent wool waste fibers were used as source of keratin. Bleached viscose fabric100% (108 g/m2) plain weave 1/1.
Chemicals
Sodium hydroxide pellets, sodium carbonate (anhydrous powder) and sodium sulfate (anhydrous powder), were of laboratory grade chemicals from Sigma-Aldrich Chemie GmbH.
Dyes
Hetero bifunctional reactive dye; Reactobond Blue BRX.150% (C.I. Reactive Blue 221) (

Chemical structure of C.I. Reactive Blue 221.
Methods
Extraction of keratin hydrolysate
Microwave technique
Ten grams of wool waste fibers were mixed with 200 ml of sodium hydroxide (1 mole/L) and heated in the microwave oven (Samsung, model GE614ST May 2017, with 900 W out power operating at frequency of 2450 MHz) at different power intensities (300–900 W) for different duration times intervals (10–18 min.).
Hydrothermal technique
Ten grams of wool waste fibers were mixed with 200 ml of sodium hydroxide (1 mole/L) and heated at 80°C for 3 h. 1
Finishing of viscose fabrics with keratin hydrolysate
The extracted keratin was applied onto the viscose fabric using the pad-cure technique. The samples were padded through two dips and two nips to 100% wet pick up using a laboratory padder (ROACHES, UK) and then cured in an oven (ROACHES, UK). The samples were dry-cured (one step) at a temperature range (80–130°C) for time intervals (1–5 min). Different concentrations of the extracted solution were utilized (0.01–0.05 g/ml).
Dyeing of viscose fabrics
The finished viscose fabric was dyed with 1% (o.w.f) reactive dye (C. I. Reactive Blue 221) and 20 g/L (o.w.b) of sodium carbonate. The material was inserted into a bath with the dye at ambient temperature, steadily rising through 30 min to reach 60°C. The dyeing method provided by the manufacturer proceeded without salt to the end of the primary exhaustion, continued until the end of the fixation time (40 min.).
According to the manufacturer, the unfinished fabrics were dyed with salt 18 g/L (o.w.b.) in the same way to evaluate the effect of the finishing process (conventional dyeing method). At the end of the dyeing operation, the samples were washed with nonionic detergent (2 g/L) at 40°C for 20 min, then with water, and dried at ambient conditions.
Testing and analysis
Whiteness and yellowness
The whiteness (WI) and yellowness index (YI) of treated samples were measured according to ASTM E 313 standard test method, using Mini Scan TM XE Hunter-lab Universal.
Determination of color measurements
Color strength (K/S), CIE L*a*b* and ∆E of the dyed fabrics (blank and finished) were measured on Mini Scan XE spectrophotometer using Hunter lab universal software. Color strength was calculated based on Kubelka–Munk equation which stated below.
Where: K, S, and R are the absorption coefficient, scattering coefficient, and reflectance, respectively.
Nitrogen content
Nitrogen content of viscose fabrics was measured before and after finishing by the Kjeldhal method. 19
Scanning electron microscopy (SEM) and energy disperse X-ray spectroscopy (EDX)
Scanning electron microscopy (SEM) and Energy Disperse X-ray Spectroscopy (EDX) of viscose fabrics were evaluated by using A JEOL-Model JSM T20 scanning electron microscopy (SEM) operating at 19 kV were used to obtain photomicrographs of the fiber surface.
Fourier transform infrared spectroscopy (FTIR)
The chemical structure of the freeze – dried keratin hydrolysate for microwave and hydrothermal methods in addition to the untreated and finished fabrics were investigated under FTIR spectrophotometer (JASCO 4700, Japan) in the region of 4000–400 cm−1 with a spectral resolution of 4 cm−1. The freeze- drying of keratin hydrolysate was done under condition (−50°C, 0.1 mbar for 48 h with LABCONCO freeze dryer, Console, 12L, −50°C, Stoppering Tray Dryer, Free Zone, 240 V, Catalog No. 7754030, Serial No. 100931482, D, U.S.A.
Tensile strength
The tensile strength of the fabric was evaluated before and after finishing according to ASTM, breaking strength/elongation strip method 5035 standard test method.
Air permeability
The air permeability of viscose fabrics was evaluated before and after treatment with hydrolyzed wool according to ASTM procedure D737-04-2008.
Color fastness
The light, wash, perspiration and rubbing fastness properties of dyed samples was evaluated according to AATCC: Colorfastness to Light – 16-2004.2005 test method, AATCC: Colorfastness to Laundering, Home and Commercial Accelerated 61-2003.2005, AATCC: Colorfastness to Perspiration 15-2002.2005 test method and GB/T3920-2008, equal to ISO105-X12:2001 respectively.
Apparent viscosity
The Apparent viscosity of the hydrolyzed wool waste solution was assessed using Programmable Rheometer Model DV-III (Brookfield Engineering Labs. Stoughton, MA, USA). The solutions were determined with CP 41Cone Spindle. About 8 mL of each wool hydrolysate solution was used. The slurry temperature was maintained at 25°C with a jacketed water bath.
Amino acids analysis
Amino acids analysis of keratin hydrolysate was carried out by using high-performance liquid chromatograph (HPLC). HPLC analysis was carried out using an Agilent 1260 series. The separation was carried out using Eclipse Plus C18 column (4.6 mm × 250 mm i.d., 5 μm). The mobile phase consisted of buffer (sodium phosphate dibasic and sodium borate), pH 8.2 (A) and ACN:MeOH:H2O 45:45:10 (B) at a flow rate 1.5 ml/min.
Results and discussion
The extraction of keratin hydrolysate from wool waste fabric
The keratin hydrolysate was extracted by alkaline hydrolysis of wool waste fabric by microwave technique. The ionic nature of the acidic and basic wool side chains establishes salt interactions between the chains. Their formation depends on pH and at a limit of around pH 5.5 at the isoelectric point (pH value at which the wool fabric has the same cationic and anionic) and is thus neutral electrically. Wool binds to sodium hydroxide with swelling and concurrent degradation and undergoes degradation on heating. In alkaline conditions (pH > 9), the hydroxide ion transforms ammonium into amino groups, and the fibers are anionic, as shown in equation (1). 20
Cysteine and cystine decomposition is the most critical degradation reactions. Alkali attacks on wool peptide linkages contribute to the degradation of amino acids like arginine, serine, threonine, and cystine. Alkaline hydrolysis primarily attacks the asparagine and glutamine (amide bond) and breaks peptide bonds in the major chains result in amino acid racemization. The reactions schemes occur under alkaline hydrolysis, as shown in Scheme 1. 21
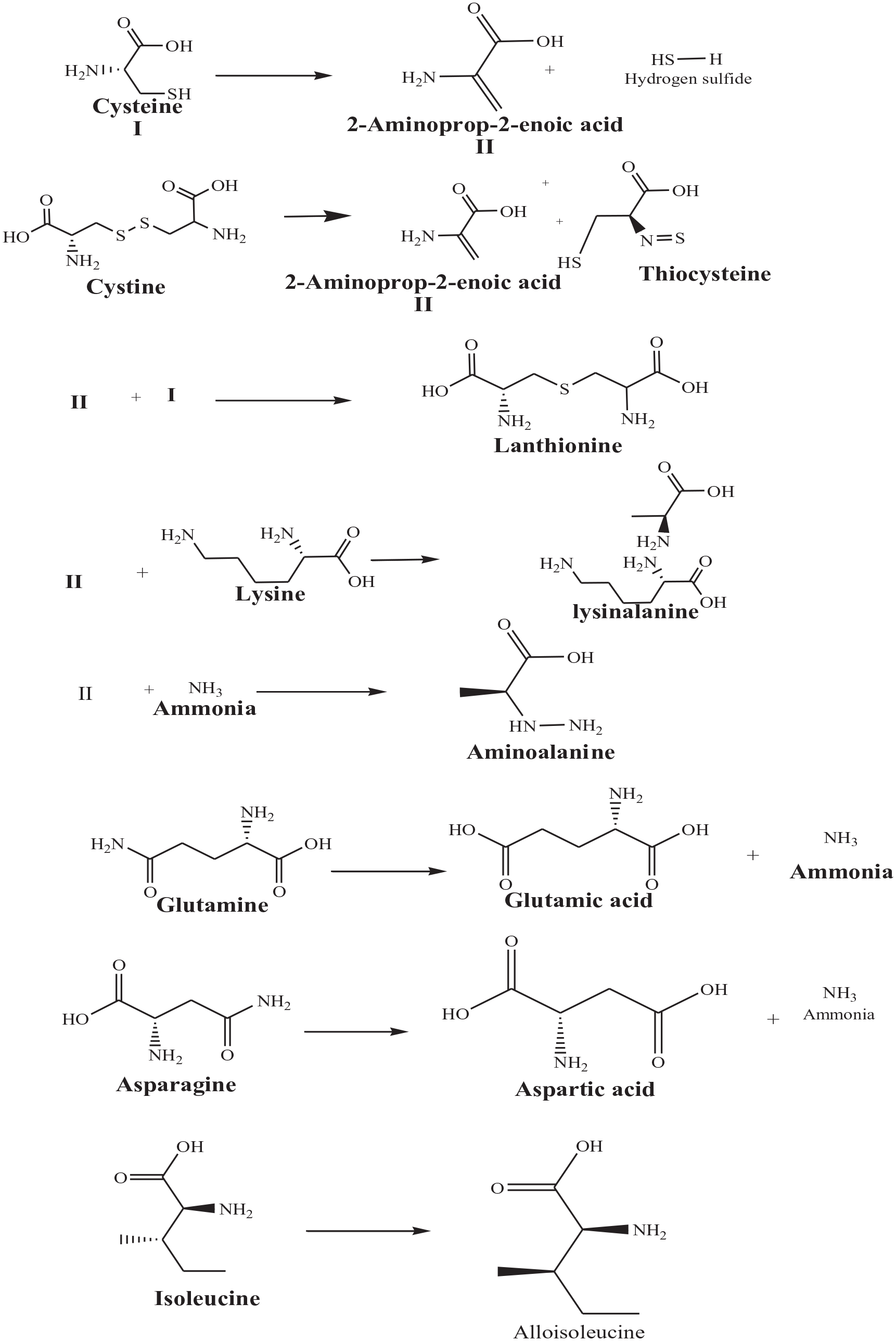
The reactions take place with alkaline wool hydrolysis.
Both bases and acids swell viscose fibers by splitting intermolecular hydrogen bonds and enhancing the mobility of the polymeric chain. Thus, it is believed that aqueous solutions will alter the intermolecular hydrogen bonding in cellulose under optimum conditions. 22 It was recognized that glycine is the simplest amino acid that could play a physical cross-linker. 23 Consequently, the amino acids constitute the keratin hydrolysate act as crosslinker in enhancing the dye -ability with reactive dye with salt-free dyeing. The structural changes in viscose fabric after finishing with keratin hydrolysate were confirmed by FTIR.
FTIR analysis
Figure 2 displays FTIR spectra of freeze-dried wool hydrolysate solutions (thermal heating and microwave) assisted hydrolysis before and after finishing in addition to the FTIR spectra of viscose fabric before and after finishing as well. FTIR analysis assessed the structural changes that could occur during the wool hydrolysis and the finishing of the viscose fabric also. The structure of the viscose fiber had to be studied to discuss the effect of finishing. There is a noticeable decrease in the peak intensities of O-H stretching vibration of viscose as shown in Figure 2, as a result of chemical interaction with sodium hydroxide, and more decrease in the peak intensities for the finished fabric.

FTIR of wool hydrolysate (different techniques) and viscose fabrics.
Also, the peak position shifted to the lower wavelength due to such chemical interaction leading to the structural change of viscose fabrics, and the peak broadening increased as well as a result of a variety of hydrogen bonds within the chemical structure. The same finding for the stretching vibration occurred in the range 1040 cm attributed to C–O of the primary alcohol.
The spectrum of finished viscose is characterized by a broad absorption band ranging from 3500 to 3200 cm−1; the wide absorption band can be assigned to the O-H and N-H bonds. The peptide vibration is predominantly Amide A, I, II. Amide A is due to the stretching vibration of N–H bonds at 3282 cm−1. 24 The absorption frequency of amide A and amide B in keratin hydrolysates at 3282 and 3065 cm−1 corresponds to N-H vibrations. A new absorption band appeared between 1737 cm−1 corresponding to C-O stretching vibration induces the amide I and the Amide II are from 1580 and 1480 cm−1, and it is due to the N-H bending and the C-N stretching vibration. The three classic areas are amide I, amide II, and amide III, among which amide I is the most significant protein absorption band. Amide 1 (1630–1650 cm−1) frequency of absorption is consistent with bonds of C = O stretching vibration. Amide II can be found in the range 1530–1550 cm−1 belongs to N-H bond bending vibrations. The amide III is related to the absorption frequency in the 1220–1240 cm−1 region (finished and freeze-dried hydrolysate solutions)
For the viscose fabric, the absorption values in the 3600–3100 cm−1 and 2800–3100 cm−1 correspond to –OH and –CH. 21 The larger absorption band that appeared at 3000–3600 cm−1 contributes to an OH stretching vibration that constitutes hydrogen bonds in the viscose configuration. The two small vibrations bands appeared at 3296 cm−1 (assigned to intermolecular hydrogen bonds), and 3341 cm−1 consist of the typical absorption band (attributed to intramolecular hydrogen bonds). The peaks corresponding to CH2 asymmetric – vibrations appeared at 2895–2908 cm−1. The absorption of water molecules, the level of absorption in the area 1633–1650 cm−1 equals H-OH-bending. The absorption band characterized by CH2- symmetrical bending occurs at different intensities in the spectra is 1424 cm−1. The FTIR demonstrates the effective finishing of keratin hydrolysate viscose. The noticeable results revealed are the difference between the FTIR spectra of the freeze-dried wool hydrolysate solutions assisted through thermal and microwave heating that results confirmed by the HPLC analysis and the rheological properties of the hydrolysate solutions and amino acid composition.
Viscosity of hydrolysate solutions
Viscosity is the flow resistance and the crucial rheological characteristic of coatings. Flow and deformation studies are essential for understanding coating utilization, treatment, and monitoring processes. Viscosity refers to the relationship between shear stress and shear rate when a liquid flows easily. Evaluation of the apparent viscosity to investigate the force-induced changes during processing in the application is a crucial component. 25
A fluid’s viscosity measures its resistance to flow (or flow resistance). It refers to the internal friction of a fluid in motion. Fluids with low viscosity have molecular structures that cause relatively negligible friction during moving. As a result of the molecular arrangement, the low-viscosity liquids flow easily. If the fluid is considered a set of parallel layers, the shear stress is the tangential force needed to move one horizontal plane relative to its adjacent one under a steady speed. Shear stress is created by fluid moving across the surface, and its amplitude is directly related to the fluid’s velocity.
The data of Table 1 reveals that the viscosity and shear stress of the wool hydrolysate solution using microwave is higher than that hydrolyzed by thermal heating. It appears that the internal friction in the fluid motion of wool hydrolysate by microwave techniques is greater than the internal friction of the fluid of wool hydrolysate.
Viscosity of wool hydrolysate solutions subjected to different shear rate.

Microwave extraction condition: 12 min, power 450 W and 1 M sodium hydroxide solution.
Amino acid composition
The solutions of the hydrolyzed wool (before and after finishing) are subjected to HPLC analysis to determine the effect of the heating methods on the amino acid compositions of the wool hydrolysates. Table 2 shows the result of hydrolysis, and the amino acid composition of hydrolysate is remarkably different from each other. After finishing, the amino acid concentration decreased due to thermal hydrolysis’s consumption, destruction, and racemization of some amino acids such as threonine, arginine, and cysteine. It breaks peptide bonds, which results in the production of low molecular weight proteins and peptides. Microwave radiation can penetrate deep into the folding layers of keratin to break the bonds accelerating the forward hydrolysis but to less extent than that occurred by thermal heating. Wide variations in the numbers of amino acids in the hydrolyzed solution can be associated with their thermal stability and secondarily with their solubility in water. The disulfide bonds are broken due to cysteine degradation, leading to many free amino acids.
Amino acid composition of keratin hydrolysate.

Microwave extraction condition: 12 min, power 450 W and 1 M sodium hydroxide solution.
Microwave-assisted hydrolysis of wool keratin produces a solution consisting of polypeptides and oligopeptides. The concentration of amino acids is lower than that of thermal-assisted hydrolysis of wool keratin. This conclusion agrees with the results of the rheological analysis of the claimed solution, where the solution has a greater viscosity than that subjected to thermal hydrolysis. 26 It is worth mentioning that keratin hydrolysis drives the disaggregation of protein structures, which entails breaking both amide (–CO–NH–) and disulfide bonds (–S–S–). The hydrolysis products have various molecular weights, depending on the hydrolytic circumstances; in extreme conditions, the peptide chains break down into amino acids. 27 This agrees with the results of HPLC analysis Table 2. Alkaline hydrolysis is used because of its efficiency at large scales and low cost. NaOH is the most common alkali used for keratin hydrolysis in practical applications. Hydrolysis in strong alkali cleavage breaks the disulfide bridges as shown in the following equations:

The effect of extraction method of keratin hydrolysate
The keratin of wool waste was dissolved converted into keratin hydrolysate (KH). The conversion occurred under two heating methods, hydrothermal (80°C for 3 h) and microwave (12 min, power 450 W) using 1 M sodium hydroxide solution. Table 3 showed that finishing viscose fabric with KH extracted using either MW or hydrothermal methods increases the fixation of reactive dye compared with other samples (sodium hydroxide treated and blank sample). This show furthers the potency of wool waste extract as a finishing agent for viscose fabric. Dyeing of virgin fabric with Reactobond blue BRX 150% (C.I. Reactive Blue 221) was performed as prescribed by the dye manufacturers (with salt). The K/S values of dyed fabric treated with alkali gave higher K/S than the blank sample but lower than the finished fabric. The finished fabric with KH extracted using MW gave higher K/S than that finished with KH extracted using the hydrothermal technique. This result encourages using MW as a heating technique for extraction KH to save energy and time. Microwave radiation is provided as an alternative to traditional hydrothermal techniques to shorten the processing time and energy, as well as uniform, efficient heating by allowing all particles to be simultaneously heated because it penetrates material atoms.
The effect of extraction method of keratin hydrolysate.

After washing.
Microwave extraction condition: 12 min, power 450 W and 1 M sodium hydroxide solution.
The results demonstrate the highly encouraging effects of sodium hydroxide to improve viscose dyeing. The application of caustic soda dissolving some molecular weight polymers and the fiber also can swell and cause disorder of the molecular alignment on the surface of fibers, thus making the dye molecules more accessible. The dye adsorption is attained after sodium hydroxide treatment, which could be attributed to the swelling of viscose by caustic, which agrees with the previous research. 28
The result showed that the highest color strength was achieved after KH finishing (without washing) before dyeing. The improved color strength of the finished fabric can be attributed to the numerous primary amino groups, thiol groups, carboxyl groups, and hydrophilic groups, which provide effective hydrogen bonding sites for the dye molecules in addition to the van der Waals force. These groups are essential contributors to the dye molecules’ substantivity and efficiency, increasing the attraction between the modified viscose and the reactive dye. Viscose fibers, like cotton fibers, comprise almost pure cellulose and so show very similar chemical properties as shown in Figure 3.29,30

Chemical structure of cotton cellulose.
Because cellulose’s hydroxyl groups are mildly acidic, absorption of hydroxide ions induces some dissociation, resulting in cellulosate ions. They react with the dye by nucleophilic substitution or addition. Reactive dyes interact with primary OH groups to form a stable covalent bond with the cellulose-containing fabric. The primary OH is more reactive than the two secondary groups; however, their relative reactivity depends on the molecule with which the cellulose-containing fabric interacts. The reactive dye reacts with the primary hydroxyl groups of cellulose-containing groups via a nucleophilic substitution or addition process. The reactions in each case need alkaline conditions; the dyeing process is usually carried out in an alkaline environment.20,29
The method of reactive dyeing comprises two stages: exhaustion and fixation. C.I. Reactive Blue 221 is a hetero-bifunctional dye (MCT/VS) that contains both vinyl sulfone group (VS) and mono-chlorotriazine group (MCT). It has higher fixation efficiency than the dyes with only a single reactive group. C.I. Reactive Blue 221 occupies a large share of reactive dyes, not only for cellulose fibers but also for wool fiber. The dye reacts rapidly at pH ~ 8–9, where amino groups (nucleophilic groups bind to VS groups. Due to different reactions of a variety of amino groups and hydroxyl groups with the MTC/VS reactive dyes, it should be straightforward to achieve dyeing with a high fixation for modified viscose fibers. 31
Effect of microwave power intensity and extraction time
Microwave power and time influence the extraction processes to a significant degree. Extraction yields were studied under various microwave power (350, 450, 600, and 900 W). However, the influence is analyzed individually, increasing from 350 to 450 W; the color strength of the finished keratin hydrolysate increased but decreased as the microwave power increased from 450 to 900 W. As seen in the Table 4, 450 W was the ideal power in this research. The use of high power would result in thermal damage and deterioration of the keratin hydrolysate as the power of the microwave increased from 450 to 900 W, the maximum color strength and lower lightness at 450W.32,33 The extraction time is one of the main factors in the extraction acquisition Table 4, which shows the effect of extraction time on the color strength of the dyed fabric. The result revealed that the ideal extraction time of keratin hydrolysate was 12 min. Longer extraction time allows the solution to penetrate the waste wool upon used power level. Also, more time means greater heat exposure to the mostly keratin hydrolysate, leading to degradation.
The effect of microwave power intensity and extraction time.
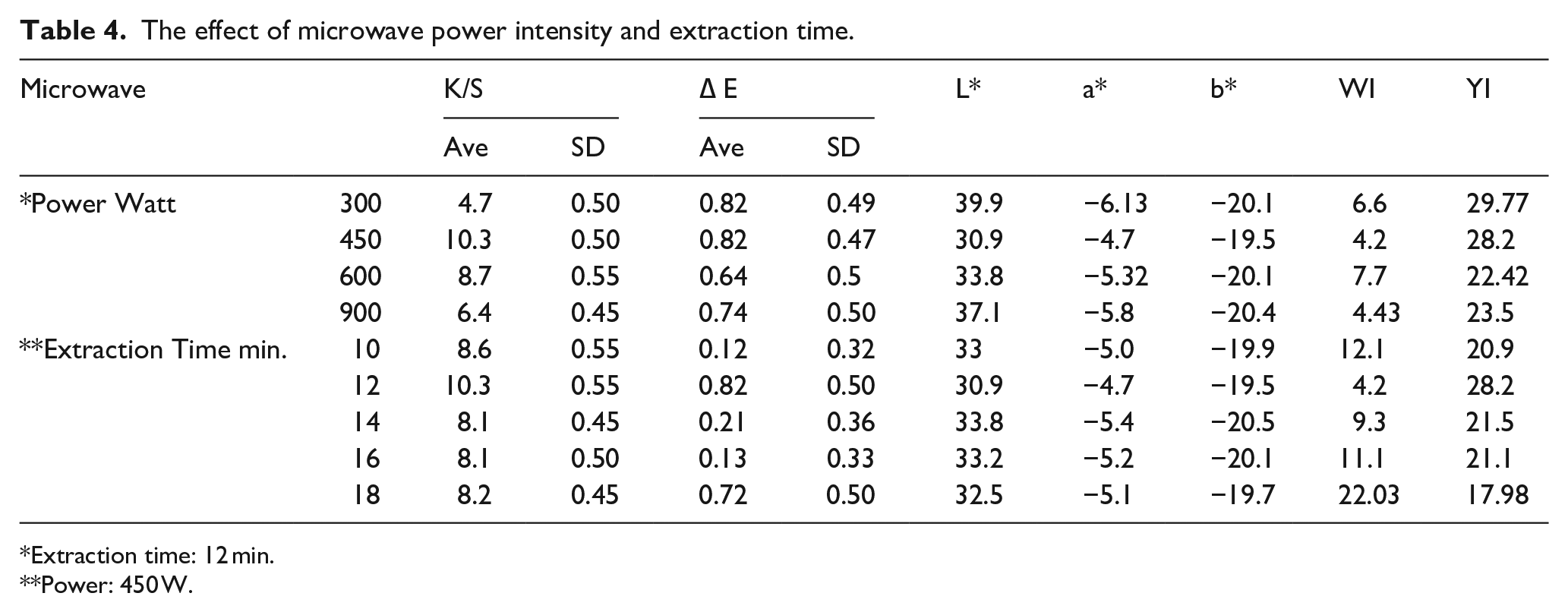
Extraction time: 12 min.
Power: 450 W.
Functional finishing of viscose fabric with extracted hydrolysate keratin
The effect of curing temp., and time
Viscose fabric was finished with KH by pad-cure technique under different curing conditions (temp. and time). From the data represented in Table 5, it is clear that increasing curing temp. Up to 130°C increased the color strength of dyed fabrics. This indicates that the curing temperature plays a role in the fixation of KH on the fabric surface and giving more reactive sites accessible to dye molecules leading to enhancement of the color strength of dyed fabric. 1 Besides, the results revealed that the best curing time for modifying viscose fabrics with KH was 3 min. The maximum color strength was 12.8, further increasing of the curing time led to a decrease in the K/S of dyed fabric. This may be correlated to the degradation of KH. These results are consistent with that found before. 2
The effect of curing temp. and time on K/S of viscose fabrics.

Curing time: 2 min.
Curing temp.: 130°C.
Concentration of extracted solution
Increasing keratin hydrolysate concentration enhances the color strength and decrease lightness. In comparison, the decrease of L* with an increase in KH add percentage means that fabrics are increasingly color strength as shown in Table 6. The color absorption is improved by increasing the KH weight to the viscose fabric. The rise in thiol and amine groups on KH-fixed viscose can be due to this condition. This finding reveals that the dye consumption compared with the control sample has risen to 75%. It is seen that KH can serve as an exhaustive source in reactive viscose textiles rather than salt. 6
The effect of concentration of extracted solution on nitrogen content and K/S of viscose fabrics.
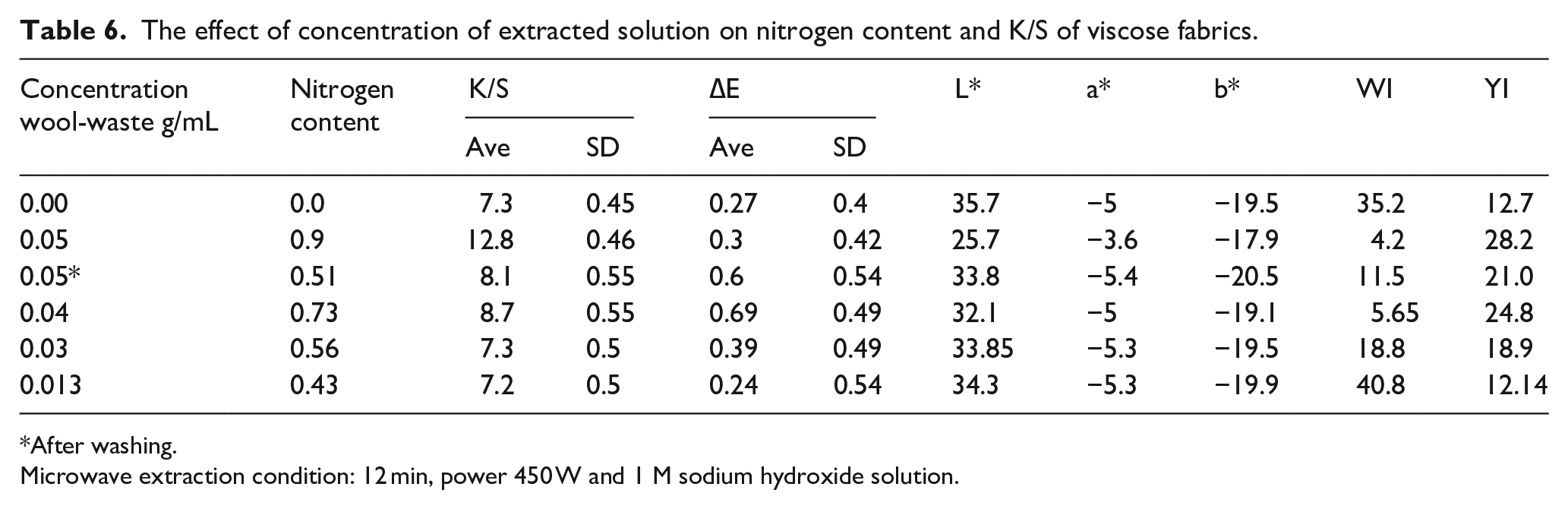
After washing.
Microwave extraction condition: 12 min, power 450 W and 1 M sodium hydroxide solution.
The decreased L* value, with a percentage increase in the KH applied, reveals that the fabrics’ color absorption improved without salt. Results revealed that the “L*” value (lightness) of unfinished and finished fabric was different, there was a lower whiteness index, which indicates the shade is higher in the keratin hydrolysate, this shows that the finished cloth retains high concentrations of dyes. 2 No noticeable variation in the (b*) between the KH finished viscose fabrics and unfinished fabric. Finished viscose fabrics had −ve (a*) means green value, and −ve (b*) means blueness, which was observed for low KH finished viscose fabric.
Fastness and physical properties of viscose fabrics
Colorfastness of dyed viscose fabric (light, washing, perspiration, rubbing), the tensile strength, elongation %, and air permeability were evaluated. The results of Table 7 indicate that colorfastness to light improved due to KH finishing, which may be interpreted by the increasing binding between the dye and KH finished viscose, enhancing light fastness properties compared to the untreated fabric. The reactive groups of KH finished fabric provide excellent hydrogen bonding sites for the dye molecules in addition to the van der Waals force. So, increasing the substantivity and efficiency of the dye molecules boosts the attraction between the modified viscose and the reactive dye, increasing the color strength of the finished viscose fabric and enhancing the fastness properties. 30 There were no changes in the washing, perspiration (acidic and alkaline), and the rubbing fastness properties of KH modified viscose fabrics compared with the conventional dyed fabric. In addition, the tensile strength and elongation % were not affected after finishing while improving the air permeability. The interpretation for this curious finding could be the extreme swelling of fibers, which could render the fabric structure tight, and KH finishing. 34
The effect of finishing on fastness properties and mechanical properties of viscose fabrics.

Microwave extraction condition: 12 min, power 450 W and 1 M sodium hydroxide solution.
Scanning electron microscopy (SEM) and Energy Disperse X-ray Spectroscopy (EDX) of viscose fabrics
Morphological characterization by SEM and EDX examination of pristine finished (before and after) washing and dyeing fabrics were presented in Figure 4(e) and (f) demonstrate that there is hydrolyzed keratin on the surface of viscose fabrics after dyeing. This verified that the keratin hydrolysate is linked with viscose (already has been confirmed by FT-IR). SEM photographs of finished fabric without washing are different from those after washing, and morphological changes are detected. The alkaline-finished viscose fabric tends to have distinct morphologies relative to the untreated viscose. The alkali resulted in the viscose fiber being cracks and pores formation, the significant differences in their morphologies caused by uniform and thin etching on the surface of viscose fiber, contributing to improved fiber dyeability.

SEM and EDX of viscose fabric: (a) blank sample, (b) Alkali finished viscose fabric, (c) KH finishing of viscose fabric, (d) KH after washing, (e) Dyed without washing after finishing, and (f) Dyed after washing of finished fabrics.
Figure 4(c) showed a sharp-needles shape on the surface of the alkali-finished viscose in addition to the cracks and pores. That sharp needles disappeared after dyeing. There is a noticeable difference between the surface of the finished fabric before and after dyeing. A comparatively even full coverage on the fabric surface has been found in this analysis. SEM and EDX analysis provide the most crucial surface parameters for evaluating keratin hydrolysate influence on the fiber surface. It demonstrated the ability of the keratin hydrolysate to connect with the fabric and affected both surface chemistry and roughness, detection of sulfur and nitrogen on the fabric surface good evidence for that connection.
Conclusion
This article presents the opportunity to establish environmentally sustainable keratin waste strategies and throws light on the possible application of wool- waste in added value textile finishing. The present research was extracted keratin hydrolysate from the wool waste fiber by microwave heating. The extracted KH was fixed to the viscose fabric and then dyed with reactive dye without salt. Keratin hydrolysis aided by microwave contains low molecular weight proteins and peptides due to the peptide bond breakage. The color depth of the post-finished fabric is higher than that of conventional dyed, by 75%. An increase in KH concentration within the range of study enhances dye exhaustion. The tensile strength of viscose fabric did not change after finishing with KH while the air permeability improved. Also, the light fastness properties for the modified viscose fabrics were upgrading.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.



