Abstract
Superhydrophobic antibacterial cotton fabric can be widely applied in outdoor clothing, hospital bedding, and other fields. However, the existing manufacturing methods are difficult or complicated. Herein, a facile and straightforward fabrication strategy is proposed via a one-step hydrothermal method to construct micro-nanometer hierarchical structure with low surface energy on fabric. In an appropriate amount, 1H, 1H, 2H, 2H-perfluorooctyltriethoxysilane (PFOTES) and tetrabutyl titanate (TBT) were mixed in a hydrothermal reactor to generate titanium dioxide. Meanwhile, the PFOTES agent was hydrolyzed and condensed, bonded with titanium dioxide, and finally grafted onto the fiber together. Morphology and elements results demonstrated that the fabric surface was covered by the TiO2 nanoparticles with superhydrophobic coating. The chemical bonds of Si-O-Ti, Ti-O-C, and Ti-O-Ti revealed the structural relationship between TiO2 with PFOTES and cotton fibers. The water contact angle of the fabric obtained can reach to 168°. The fluorinated-TiO2 cotton fabric showed high antibacterial properties in visible light against E. coli and S. aureus. This simple method of preparing superhydrophobic and antibacterial fabric exhibited great potential in the field of functional textiles such as outdoor garments and hospital-related applications.
Introduction
Superhydrophobic property is always a welcomed subject for its practical applications in self-cleaning, anticorrosion, oil-water separation, and drag reduction.1–3 During the last two decades the project of superhydrophobic with other functions received much more attention. 4 Cotton fabrics are the most commonly used natural resources for their softness, moisture permeability, large scale of production, and low raw materials cost. However, the inherent hydrophilicity and fouling have restricted their application in some special fields. Superhydrophobic and antibacterial modification of cotton fabric can broaden usage.
Based on the superhydrophobic structure theory5–7 that the micro-nano hierarchical structure with low surface energy on a surface can effectively retain air at the bottom of the microstructure, and water droplets can exist on nanostructure in a spherical shape, thereby reducing the contact area and adhesion with the surface, obtaining superhydrophobic and self-cleaning effect. So far, numerous methods and techniques, such as sol-gel processing,8–14 polymer grafting,15,16 self-assembly,17,18 chemical etching, in-situ growth method,6,19 dip-coating,20,21 hydrothermal methods,13,22–25 plasma treatment,26–29 electrospinning,30,31 and so on,32–34 have been adopted in constructing hierarchical roughness structure with low surface energy on fabric. Unfortunately, the preparation process of these methods is always divided into two or three steps, namely, the fabric is first coated with nanoparticles to build a rough surface, and then low surface energy modification is performed, or other multiple steps are executed in sequence. The fabrication procedure of superhydrophobic fabric is complex or troublesome.35,36 While papers10,11 reported that they used a one-step sol-gel process to prepare superhydrophobic fabrics, but in fact, they needed to prepare the gel in advance and then modify the fabric. The process still requires two or more steps.
In addition, the micro-nano structures on the fabric are inherently vulnerable to mechanical forces, such as friction and abrasion. 37 The lack of chemical bonds between fibers, chemicals, and nanoparticles lead to susceptibility to mechanical damage. Therefore, establishing and ensuring strong chemical bonds between particles and fibers, particles and reagents is an important aspect of improving the durability and stability of superhydrophobic properties. All these problems need to be solved before the large-scale production of superhydrophobic cotton fabrics. Przybylak et al. 38 introduced a simple one-step chemical modification of bifunctional polysiloxane to prepare superhydrophobic cotton fabric. They demonstrated the bond between alkoxy silicon groups and fibers could enhance hydrophobicity and stability. The contact angle obtained was 152°. He et al. 39 conducted a two-step experiment to prepare covalently bonded superhydrophobic PET fabrics. First, pristine PET generates fluororadicals by fluorination. Then TFEMA, DVB, and MSiO2 were grafted to the fabric by ternary copolymerization. Superhydrophobicity was achieved via combination of hydrophobic chemical composition and physical structures. Han and Gong 40 adopted a simple one-pot method and in-situ growth process to prepare superhydrophobic cotton fabric. Firstly, the pH value was controlled to make DA generate benzoquinone, at the same time, APTES and FAE-17 were hydrolyzed and bonded. The quinone group in benzoquinone reacted with APTES to form nanocluster with low surface energy. This method is a good way to bond fibers, particles, and solvents to build durable super-hydrophobic cotton fabrics. Montazer 41 group introduced a one-pot method to prepare antibacterial and hydrophobic fabric by coating photocatalytic nano copper ferrite/myristic-lauric fatty acids on fabrics. Nano copper ferrite photocatalytic nanoparticles endowed fabric with antibacterial and photocatalytic properties at the same time. As a low surface energy material, myristic-lauric fatty acids combined with nanoparticles to help the fabric obtain higher hydrophobicity. WCA reached 143°. In short, antibacterial, photocatalytic, and superhydrophobic composite multifunctional materials have become a new application trend. 42
In this work, a simple one-step method was used to grow nanoparticles with low surface energy on cotton surface to achieve superhyddrophobic and antibacterial performance. In a hydrothermal reactor, Tetrabutyl titanate (TBT) experienced hydrolysis and condensation reaction to form nanoscale titanium dioxide particles. While the fluoro silane also performed hydrolysis and condensation to form branched oligomers and polymers or macromolecular networks. Silicone oxo macromolecular network reacted with TiOH2+ to create fluoro silane treated TiO2. The reaction of PFOTES with TiO2 can prevent the aggregation between titanium dioxide particles. At the same time, the abundant hydroxyl groups on the cotton surface bonded with hydroxyl groups of TiO2. These hybrid interactions of hydroxylsilanes with TiO2 can build a dense and robust water-repellent layer, which would increase the durability of hydrophobicity. The water contact angle of the prepared superhydrophobic cotton fabric is as high as 168°.
In addition, the cotton fabric prepared by one-step modification has proved to be a predictable antibacterial material, especially with the participation of TiO2 nanoparticles. 43 The superhydrophobic fabric showed excellent antibacterial properties against S. aureus and E. coli, making it a promising candidate material for outdoor clothing and hospital bedding.
Experimental procedures
Materials and chemicals
The Pure white cotton (Plain weave, 150 g m−2) and knitted cotton bleaching (32s, Single flat needled, 150 g m−2) fabrics were purchased from the local market in Shaanxi, China. Tetrabutyl titanate (Ti(OC4H9)4, ⩾99%) (TBT), sodium hydroxide (NaOH), 1H,1H,2H,2H-perfluo-rooctyltriethoxysilane (PFOTES), and ethanol-anhydrous alcohol (EtOH) were purchased from Aladdin Chemistry Co., Ltd., and deionized water was prepared in the laboratory. All chemicals and reagents used were without further purification. The bacteria studied were S. aureus and E. coli from the Shaanxi Institute of Microbiology, China.
The preparation process of PFOTES-TBT coated cotton fabric
The scouring process was to remove impurities from the fabrics. Cotton fabrics were immersed into 0.1 mol/L sodium hydroxide solution, treated 30 min at 80℃ by 1:80 weight ratio, then rinsed three times with deionized water and EtOH at 40℃, dried and set aside.
The one-step manufacturing process was illustrated in Scheme 1. First, TBT (0.4 mL) was added to ethanol (10 mL) to form solution A, and then mixed with PFOTES (0.8 mL) to form solution B. In order to better disperse the solution and ensure uniform distribution of particles after hydrothermal reaction, we firstly put the above clean cotton fabric (15 cm × 15 cm) immersed in solution B for 3 min to make sure the yarn and fiber inside the cotton fabric fully absorb the PFOTES/TBT mixture solution, then diluted the mixture to 100 mL with deionized water and disperse it by ultrasound (28 Hz and 100 W) for 3 min. The mixture solution was hydrolyzed on the fiber surface, and nanoparticles can be formed in situ on the fiber after hydrothermal reaction.
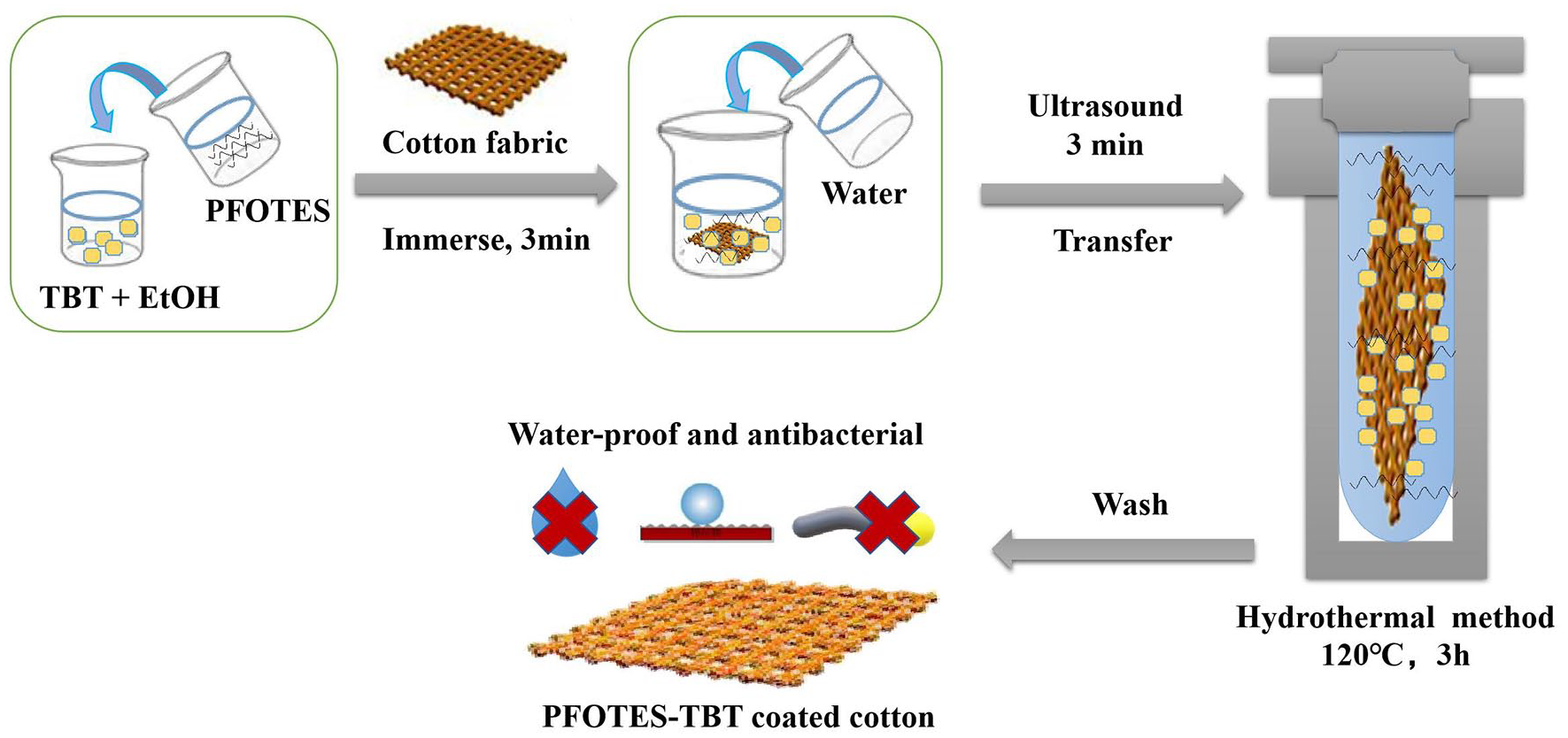
Schematic of preparation of PFOTES-TBT coated cotton fabric with waterproof and antibacterial properties.
Transferred the above materials to a stainless-steel autoclave lined with polytetrafluoroethylene. Heated to 120℃ for 3 h, rotated at the speed of 7 rpm, cooled to room temperature, and washed the fabric with absolute ethanol and deionized water at 40℃ and 80℃ respectively until the water clear. Here, the finished fabric was obtained. The following fabrics treated only by TBT or PFOTES were prepared in the same dose and procedure as above.
Characterization and instruments
Surface morphology and elemental information of pure and treated cotton fabrics were studied by using scanning electron microscopy (FEI Quanta-450-FEG SEM) and energy dispersive spectroscopy (Quanta-450-FEG EDS). The fabric samples were sputter-coated with gold and imaged at 20 kV accelerated voltage. Particles size on fibers were determined from the SEM images. Crystal phase of the fabric before and after treatment was analyzed by X-ray diffraction (XRD) with Cu-Kα radiation (Philips, MRD). The valence of elements were determined by X-ray photoelectron spectra (XPS, AXIS UltraDLD, Daojin, Kyoto, Japan).
Wettability and stability evaluation of the fabrics
Water contact angles (WCAs) were used to evaluate the hydrophobicity of the fabrics. The WCAs of untreated cotton fabric, pure TiO2 loaded fabric, PFOTES-only treated fabric, and TBT-PFOTES modified fabric were tested at room temperature by contact angle measurement instrument (Dataphysics Instrument GmbH, Germany). The volume of deionized water droplet was 2 μL. The average value was measured and calculated at three different positions on fabric. The self-cleaning property was also determined by placing dirty fabric on a flat surface with an inclination angle of 30° to observe the removal of the dust.
The mechanical abrasion and washing stability experiments were performed to test the possible situation during use. In short, the tests method of mechanical abrasion as follows: fabric was placed on sandpaper, a 200 g weight was placed on it. The fabric was dragged to a distance of 15 cm backward and forward 2, 4, 6, 8, 10 times. Then WCAs of cotton surface were measured. The durability of fabrics was detected on the basis of the standard (GB/T 8629 2017 China). The original and prepared cotton fabrics were washed 2, 4, 6, 8, 10 times by a laboratory washing machine, respectively. Each washing cycle was performed with 10 g soap. After drying in the air, the contact angle of the water droplets was measured.
Antibacterial activity
According to GB/T 20944.3-2008 (CHINA), antibacterial activity was tested with the shake flask method.43,44 The studied bacteria including S. aureus and E. coli. The bacteria were cultured in broth for 12 h in a 37℃ incubator. A single colony was selected and inoculated in fresh LB medium and incubated at 37℃ 150 rpm to logarithmic growth period. Cut 0.75 g of the pristine and treated fabric samples into fragments about 0.5 cm × 0.5 cm in size. Mix the sterilized fabric with 70 mL 0.3 mM PBS medium with the 1 × 105–4 × 105 CFU/mL bacteria suspension. Then shake the flask at 150 rpm at 24℃ for 18 h. After the treatment, the above solution was diluted by 1 mL and dispensed onto an agar plate, and incubated at 37℃ for 24 h. The knitted cotton bleaching fabric was the control. Counted the bacterial colonies and calculated the antibacterial rate as the following formula:
R is the antibacterial rate (%); λ0 is the average number of viable bacteria on the control sample; λt is the average number of viable bacteria on pristine and treated samples.
Results and discussion
Characterization of PFOTES-TiO2@cotton fabric and synthetic process
Surface morphology and element’s information
Typical SEM images of lotus leaf surface and its water-repellent principle were shown as Figure 1(a) to (c). The micron-level papillae and nanometer waxy villi texture constructed micro-nano hierarchical framework on lotus leaf surface. The SEM images of pristine and modified cotton fabric were exhibited in Figure 1(d) to (g). The picture showed that the raw cotton fiber has a natural curve and smooth surface, with a diameter of about 15 μm. However, because of the intensive hydrolysis and condensation of TBT with water, the cotton fiber modified with only TBT was covered with a large number of aggregated nanoparticles, as shown in Figure 1(e). The bulk aggregation of nanoparticles led to a failure of crystallization and a restriction of size growth. As for the fabric treated with PFOTES/TBT, the overall integrity of fibers had not changed significantly, but the surface of the fibers became obviously rough, and particles were evenly and densely distributed, of which size was ranging from 70 to 1000 nm, as indicated in Figure 1(f).

SEM images of (a and b) the lotus leaf surface at low and high magnification. 45 (c) Principle of water repellency on the lotus leaf surface. SEM images of (d1–d3) pristine cotton fibers, (e1–e3) only TBT prepared cotton fibers, (f1–f3) PFOTES/TBT treated cotton fabric, and (g) the inner surface of the cotton fabric. (h) Hydrophobic configuration of the treated yarns. (i) Element mapping of (f3) according to C/O/Ti/Si/F elements.
Moreover, the inner characters of fabric were also imaged in Figure 1(g). The fibers inside fabric structure were covered with a layer of uniform and compact particles, indicating that solvents passed through the fiber gaps and formed nanoparticles. The hierarchical structure, combining with nanoscale particles and numerous microlevel fibers, can fill more air and keep water droplets in a spherical shape.
Furthermore, we also tested the EDS mapping to verify elements and their distribution. The Figure 1(i) exhibited the EDS elemental mapping of Figure 1f3 according to C, O, Ti, Si, and F elements. The picture of F and Si signals originated from the PFOTES agent. And the majority of the C, O elements were aroused from the cotton and PFOTES. Ti and part of the O signals may indicate that TiO2 particles were distributed on the cotton fiber surface. Here, we proposed the nanoparticles on fibers were nano-TiO2, for the hydrothermal method is a standard way to synthesis TiO2.
Besides, compared with the only-TBT treated sample (Figure 1(e)), the nanoparticles produced from PFOTES/TBT reaction were scattered and dispersed very well. But the reaction bonds between TiO2, PFOTES, and fibers were not sure. X-ray diffraction (XRD) and X-ray photoelectron spectroscopy (XPS) were used to confirm the hydrothermal reaction of TiO2 and PFOTES modification further.
Crystal structure analysis
The phase of nanoparticles deposited on fibers was determined by X-ray diffraction (XRD). According to Figure 2, the diffraction peaks of pristine cotton displayed at 14.8°, 16.7°, 22.8°, and 33.2° were the characteristic peaks of cellulose. 32 While sample fabric treated with PFOTES/TBT showed the diffraction peaks located at 2θ = 25.4°, 37.8°, 48°, 55°, and 62.7°, which was coincided with the crystalline phase of anatase TiO2. 46

XRD tests of the pristine cotton fabric and PFOTES-TiO2@cotton fabric.
XRD analysis illustrated that TBT and PFOTES could synthesize TiO2 nanoparticles, and the fluoroalkyl silane was not involved in the crystallization reaction of TiO2 nanoparticles.
Chemical bonding forms
The X-ray photoelectron spectroscopy (XPS) was carried out to analyze bonding states. Figure 3(a) presented the crucial chemical compositions of pure and PFOTES-TiO2 coated cotton fabrics (PFOTES-TiO2@cotton). The spectrum of unfinished cotton showed only C 1s and O1s elements, while modified cotton can detect F 1s, O 1s, Ti 2p, C 1s, and Si 2p elements corresponding to binding energy at 688.97, 532.87, 459.37, 291.57, and 102.67 eV respectively.

XPS survey spectra of (a) the pristine fabric and fabric modified with PFTOES-TiO2 and (b–f) C 1s, O1s, Ti 2p, Si 2p, F 1s high-resolution spectra of the PFTOES-TiO2 treated fabric respectively.
The spectra of C 1s, Ti 2p, O 1s, and Si 2p were deconvoluted into multiple sub-peaks. The C 1s region showed four peaks in Figure 3(b) at 293.8, 291.5, 285.9, 284.8 eV, which were attributed to -CF3, -CF2-, C-O-C/C-O-H/C-O-Ti, and C-C respectively.11,32 The bonds of -CF3, -CF2-, and C-C were due to the long-chain of fluoroalkyl silane, while the possible bonds of C-O-Ti initially proved the combination between TiO2 and cellulose.
As shown in Figure 3(c), the O 1s binding energy peaks centered at 530.9, 532.4, and 533.2 eV, the first one was assigned to Ti-O-Ti, and probably to Si-O-Ti/Ti-O-C, for it was slightly higher than pure TiO2 (529–530 eV). The second one was attributed to Si-O-Si.47,48 The last one included cellulose O-C and C-O-C, which was also found in C 1s with a peak at 285.9 eV. The appearance of Si-O-Ti/Ti-O-C bonds demonstrated that TiO2 has closely bonded with cellulose and fluoroalkyl silane.
In Figure 3(d), the binds of Ti 2p displayed Ti 4+ and Ti3+ species, as four peaks at 459.4, 465.2, 458, 463 eV. The two high peaks illustrated the presence of Ti 2p1/2 and Ti 2p3/2 obtained from TiO2 particles. Furthermore, the space between the two peaks was 5.7 eV, which was consistent with reported Ti4+ values. 49
The energy bonding of Si 2p at 102.8 and 103.6 eV showed in Figure 3(e) was represented to Si-O-Si and Si-O-Ti, which was coincided with O 1s. Figure 3(f) shows that the energy of F 1s at 684.9 eV corresponded to organic fluorine, indicating that the F atom did not replace the TiO2 lattice.
The XPS analysis results concluded as follows: (1) the appearance of Ti-O-C and Ti-O-Ti bonds indicated that anatase TiO2 nanoparticles densely attached to fibers; (2) the long-chain groups of trifluoromethyl(-CF3) and difluoromethyl(-CF2-) in the PFOTES can endow cotton fabric with superhydrophobicity; (3) Si-O-Ti and Si-O-Si bonds proved the adhesion of TiO2 to PFOTES, and the formation of Si-O-Si 3D structure in PFOTES. In all, it can be said that the fluorinated TiO2 was grafted on the surface of the fibers and rendered the fabric with superhydrophobicity.
Mechanism of superhydrophobic PFOTES-TiO2@fabric
Based on the morphological structure and bonding analysis described above, we clarified the chemicals and mechanisms of the PFOTES-TiO2@fabric formation, as shown in Figure 4.
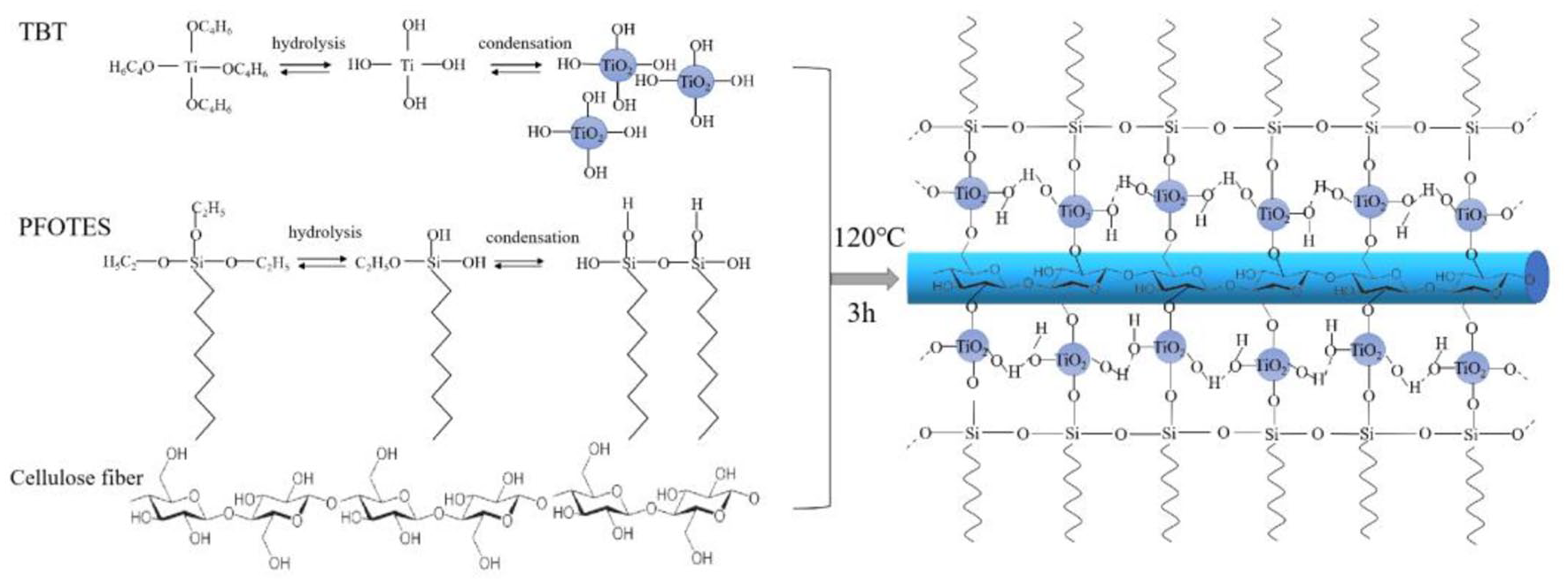
Reaction route of PFOTES-TiO2@fabric.
In the hydrothermal reaction, nanoscale titanium dioxide particles were formed by hydrolysis and condensation reaction. In comparison, fluoro silane also experienced hydrolysis and condensation to form branched oligomers and polymers or macromolecular networks. Silicone oxo macromolecular network reacted with TiOH2+ to create fluoro silane treated TiO2, and this reaction process had been demonstrated in Pazokifard et al. 46 and Razmjou et al. 50 In this stage, the hydroxyl groups in fluoro silane can form a hydrogen bond to the surface hydroxyl groups of TiO2 and eventually got the covalent bonds of Si-O-Ti. 23 During the reaction, the particles’ hydroxyl could condensate with the hydroxyl of cotton fabric to form a Ti-O-C chemical bond. These hybrid 3D interactions of hydroxylsilanes with TiO2 may lead to a dense and robust water-repellent layer, which would increase the durability of hydrophobicity. Besides, the existence of PFOTES can significantly reduce the aggregation of TiO2 nanoparticles and push the particles to adhere to fibers.
In this reaction system, the formation of titanium dioxide nanoparticles and the modification with PFOTES agent were carried out simultaneously. The treatment of TiO2 nanoparticles was mainly due to the hydrogen bonds between PFOTES and the favorable position on the TiO2 surface, as well as the condensation of the hydrolyzed PFOTES hydroxyl groups on the surface of TiO2. This hybrid process had shortened the experimental period and completed the superhydrophobic and antibacterial modification in one step.
Superhydrophobicity and self-cleaning property
The wetting properties of cotton fabrics were displayed in Figure 5. As shown in Figure 5(a), the original cotton fabric had an initial WCA of 86° and absorbed water entirely within 30 s. This result was consistent with our shared knowledge that cellulose is naturally hydrophilic. In Figure 5(b), however, the TiO2-coated fabric was utterly wet in a matter of seconds, the instantaneous WCA was only 33°, for the TiO2 particles introduced more hydrophilic -OH groups onto the fabric. Modified with PFOTES/TBT, the WCA of PFOTES-TiO2@fabric was 168° (Figure 5(c)), exhibited exceptionally waterproof. Staying for 5 min, the WCA remained 160°. Instead, the fabric treated with the PFOTES solvent (0.8 mL at the same dosage as the finished fabric) had an initial WCA of 150°. Five minutes later, the detected WCA reduced to 88°.
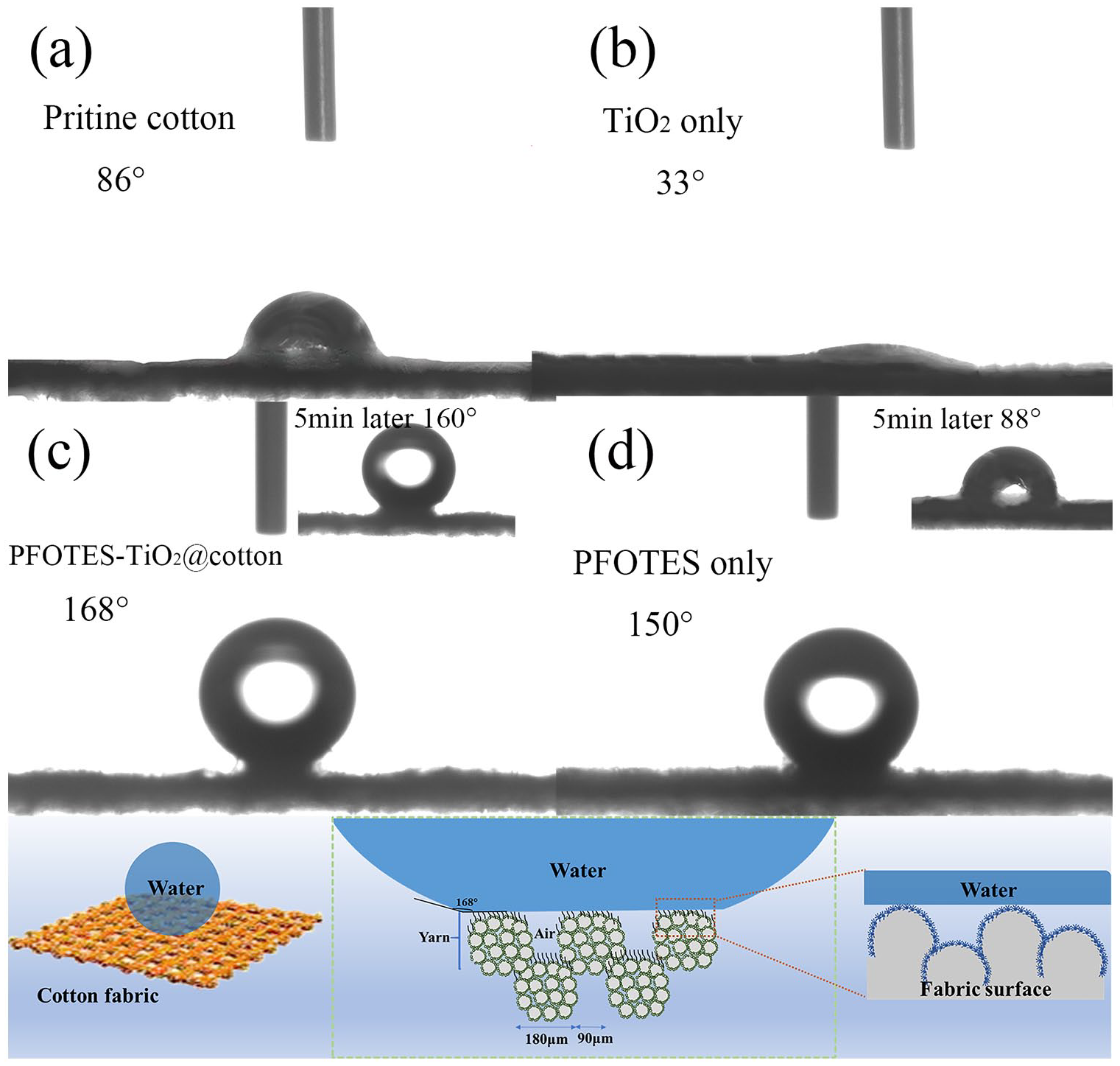
WACs measurement of (a) pristine cotton fabric, (b) only TiO2 coated cotton fabric, (c) PFOTES-TiO2 @cotton fabric, and (d) PFOTES only coated cotton fabric on 0 and 5 min. (e) Schematic diagram of the persistent superhydrophobic structure.
The persistent superhydrophobic structure schematic diagram was presented as Figure 5(e). Cloth weave, yarns, and fibers constituted the basic morphology of fabric. Although the macroscopic structure of weaves and yarns can reduce surface contact areas to some extent, it was evident that the dual system of nano-TiO2 and micro-fibers eventually led to superhydrophobicity. Compared with a smooth surface, such as the steel surface, the cotton fabric had plenty of gaps between particles, fibers, and yarns. This composite structure, covered with low energy solvent, can keep more air between water droplets and cotton fibers, form a water cushion, maintain water droplets’ spherical shape, and isolate the direct contact between fibers and water droplets.
Compared with PFOTES-TiO2 decorative fabric, the water on PFOTES finished fabric will reduce WCA after a period of time, as shown in Figure 5(d). Johnson and Dettre 51 proposed that when the hydrophobic surface roughness is low, there is no more nano gap to contain air, water droplets can not be lifted as a ball, and the solid-liquid contact has a certain contact angle. The large micron structure pores can be filled with water. Microns and larger pores allow water to penetrate into the internal structure of the fabric, resulting in low WCA. When the hydrophobic surface roughness is high, the water droplets contact the top of the nano particles and are lifted by the air layer maintained by the nano gap to form a sphere with a high contact angle.
Besides, the hydrophobicity of cotton fabric also brings a significant self-cleaning character. Water droplets slide under the action of gravity, and the dust will be taken away in the process of sliding.
As expected, in Figure 6(a), pure cotton absorbed moisture when water droplets passed through it, and dirt adhered to the fabric surface. In contrast, fluoridated-TiO2 coated fabric showed excellent water-repellent properties. Water rolled smoothly through the material, removed dust quickly, demonstrating extraordinary self-cleaning (Figure 6(b)). The principles of self-cleaning were shown in Figure 6(c). The superhydrophobicity endowed cloth with stain-less character. The water repellency was also detected by pressing the fabric into dye solution (Figure 6(d)) and spilling water droplets onto the fabric (Figure 6(e)). Cotton fabric floated above the liquid after releasing press and showed non-stick characteristics. Dye droplets can form ball shapes on the modified cloth.

Self-cleaning property and wettability tests. Photo images of (a) pristine and (b) PFOTES-TiO2@coated cotton fabric remove dirt, and (c) their principles. (d) Photo images of modified cloth on the water surface, in water under press, and floated onto water. (e) The wettability of pristine and PFOTES-TiO2 coated cotton fabric.
Abrasion resistance and durability tests
Hydrophobic durability is the most critical factor that determines the application scope of materials. The stability of superhydrophobic fabric was tested by abrasion resistance and washing tests. As shown in Figure 7(a), no noticeable decrease in water contact was observed after abrasion, and the water contact angles were all more than 150°. Besides, the sample exhibited super washing resistance after 10 washing cycles (Figure 7(b)). In all, these results indicated that the one-step fabricated superhydrophobic fabric possessed excellent stability.

Water contact values after (a) abrasion 10 times and (b) 10 washing cycles.
Antibacterial activities
Titanium dioxide nanoparticles are photo-active metallic nanoparticles; their photocatalysis has attracted much interest in the antibacterial application. When light energy more than the TiO2 bandgap, the electron (e−) and hole (h+) pairs generate and react with O2 and H2O to form O2- and ·OH, which is considered to be dominant factors of bacteria destruction.44,52
In order to study the antibacterial effect of our work, we carried out the anti-bacteria experiments. Pure cotton, TiO2-loaded cotton, and PFOTES- TiO2 coated cotton were tested against S. aureus and E. coli, respectively. Knitted bleaching cotton fabric was the control. As shown in Figure 8, the antibacterial ratio was 46.1% and 43.43% with TiO2 on cotton compared with pure cotton of 10.6% and 13.13% against S. aureus and E. coli. The PFOTES-TiO2@cotton showed the best killing efficiency reached 94% and 100% against S. aureus and E. coli, respectively. The antibacterial rate of S. aureus was lower than that of E. coli because of the structural differentiation of cell walls. Gram-negative bacteria like S. aureus have a rigid peptidoglycan layer in the cell wall to effectively resist the attack.

The antibacterial activities of pristine cotton, TiO2-loaded cotton, and PFOTES- TiO2 coated cotton against S. aureus and E. coli.
TiO2 nanoparticles were critical for antibacterial activities. However, PFOTES-TiO2@cotton prohibited an excellent antibacterial effect compared with the pure-TiO2 loaded cloth. The pure titanium dioxide particles had a low photocatalytic impact in visible light. In recent years, the doping of N, Ag, Fe, F, Si, and so on33,44,53 has vital significance to enhance the visible photocatalytic antibacterial effect of titanium dioxide. It was reported that Ti-O-Si hetero bonds in TiO2 were an effective method to improve photocatalytic efficiency. 23 Report 32 also proved that the composite cotton-anatase titanium dioxide had high photocatalytic activity in sunlight.
Based on the discussion above and analysis of SEM, XRD, and XPS, Ti-O-Si bonds in the constructed PFOTES-TiO2@cotton were the dominant factors enhancing antibacterial activity. The reasons for high antibacterial property illustrated as follows (diagrams were seen in Figure 9): (1) Ti-O-Si bonds in PFOTES-TiO2@cotton can effectively improve the photon efficiency of the TiO2 and stimulate the production of reactive oxygen species. 33 Also, the viscous dispersion of anatase TiO2 nanoparticles on cotton fibers can promote photocatalytic efficiency. (2) Bacteria may be easily trapped in the particles’ gaps and in close contact with titanium dioxide, thereby quickly sterilized. (3)When the bacteria were in contact with TiO2, visible light was absorbed by the bacteria, electron-hole pairs were generated. These excited electrons react with oxygen and water at the surface of TiO2, forming superoxide radical ROS that, in turn, decompose the bacteria. 54
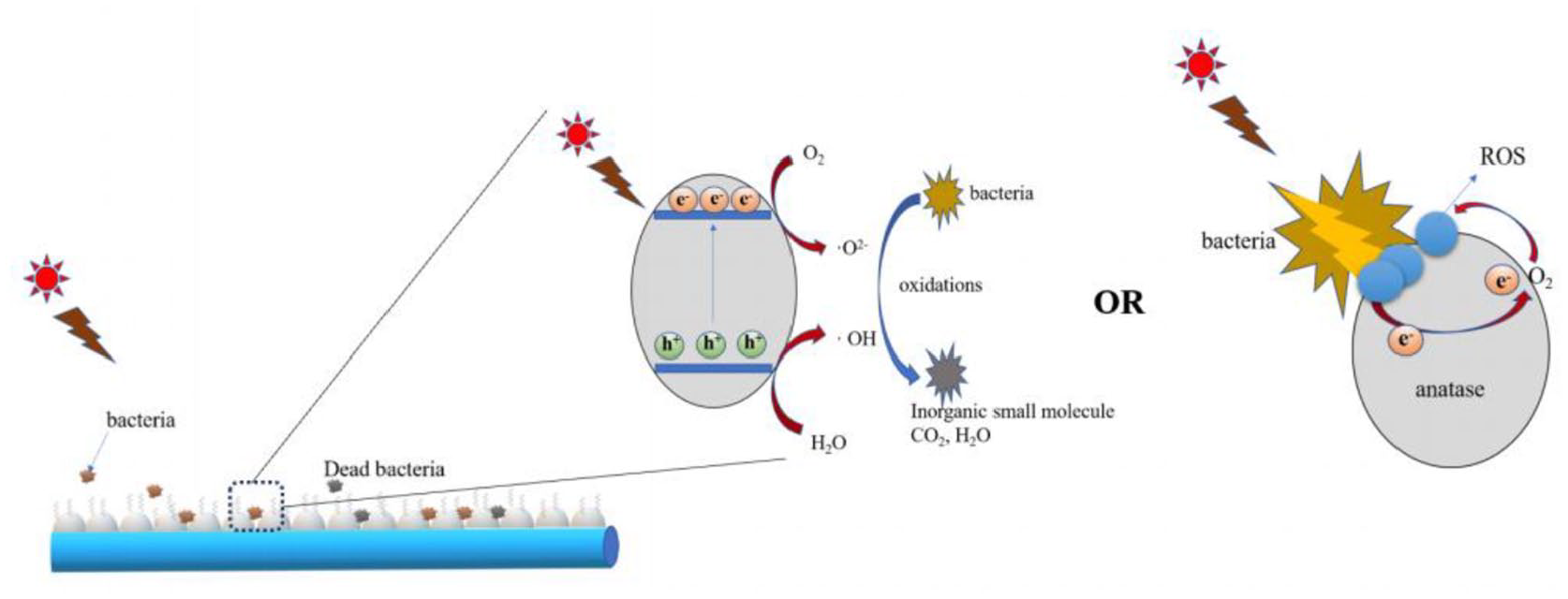
Mechanisms of the photocatalytic bacterial reaction of PFOTES-TiO2@cotton.
Conclusion
The simple one-step hydrothermal modification of cotton fabric with fluorinated titanium dioxide to form hierarchical rough structure and make fabric with excellent superhydrophobic and antibacterial properties.
In summary, we had prepared cotton fabric with superhydrophobic and antibacterial properties by a cost-efficient and time-saving approach with the combination of TBT and PFOTES. In this mild hydrothermal reaction system, TiO2 nanoparticles and PFOTES create a micro-nano roughness surface with low surface energy on cotton fibers in one step.
The structural study of chemical bonds demonstrated that the fluorinated-TiO2 nanoparticles were successfully fabricated on the fibers. Except for superhydrophobicity, the use of PFOTES also reduced the aggregation of nanoparticles and improved particle adhesion on cloth. This as-prepared fabric exhibited excellent superhydrophobicity and dust cleaning properties.
Furthermore, the obtained fabric sample inactivates both E. coli and S. aureus, attributing to the micro-structures and Si-O-Ti bonds. Finally, this robust superhydrophobic and antibacterial cotton fabric may have a fair application prospect in outdoor medical products with a cost-saving and straightforward process.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors were financially supported by the Scientific Research Program of Shaanxi Province Education Department (19JK0363), the Key R&D Plan in Shaanxi Province of China (2020ZDLGY13-08), and the Natural Science Basic Research Plan in Shaanxi Province of China (2021JQ-681).

