Abstract
Bamboo pulping fabrics (BPFs), one of the most important textile materials for clothing industry, has many excellent characteristics such as soft, skin-friendly, and hygroscopic, etc., but some drawbacks especially mildewed, causing customers’ concerns. This experiment evaluated effects of two organic acids, acetic acid (AA) and citric acid (CA), as solvents for dissolving chitosan on the antibacterial properties of chitosan-treated BPFs with the intention of replacing AA with CA because previous research finding indicated the strong acid smelling of BPFs treated with chitosan dissolved in AA. Experimental results indicated that BPFs treated chitosan dissolved in CA had a significantly higher antibacterial rate than ones treated with chitosan dissolved in AA within each of five chitosan mass fraction levels investigated. Antibacterial rates of BPFs treated with chitosan dissolved in AA and CA increased as chitosan mass fraction increased, but increasing rates were different between BPFs treated with chitosan dissolved in AA and CA. No acid sour smell was detected for BPFs treated with chitosan dissolved in CA.
Introduction
In recent years, researching and developing functional fabrics such as heat conducting fabrics, conductive fabrics, antibacterial fabrics, UV resistant fabrics, etc. 1 have attracted much of attention. Antibacterial fabrics can be used for special purposes such as medical applications. Traditional antibacterial fabrics using silver ion, copper ion, and nano materials are incompatible with human body or environment. Chitosan is an environmentally friendly antibacterial agent 2 because its molecular structure (Figure 1) has many amino groups that are the main driving force of its antibacterial properties and make it soluble in acid medium, 3 and other reactive sites like hydroxyl for covalently bonding of polymer chains, chemical monomers or small molecules to make biomaterials with special properties.
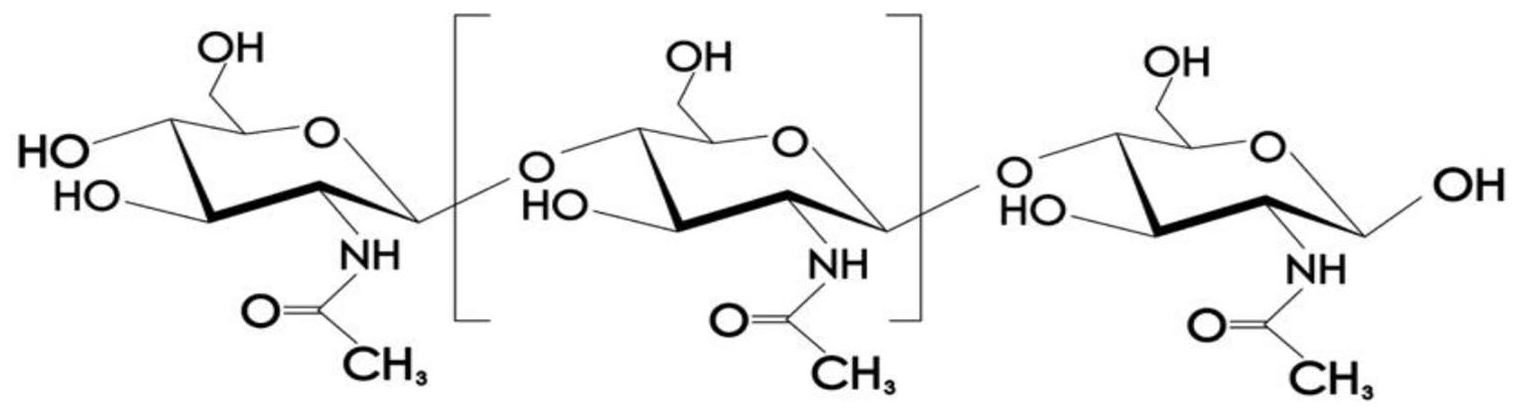
Molecular structure of chitosan.
Surface graft is an effective modification for the functionalization of targeted materials.4,5 Samanta and Bagchi 6 treated jute fabrics using chitosan dissolved in organic citric acid (CA). Samanta and Bagchi 6 modified polyester fabrics with chitosan for optimizing their antibacterial and hydrophobic properties.
Bamboo fiber is a kind of regenerated cellulose fiber made from bamboo by special processing. The chemical structure of cellulose fiber is not changed, but its physical structure is changed to optimize the properties of regenerated cellulose fiber. The first step to prepare BPFs is integrating fiber, when fiber extracted, bamboo fiber is converted into bamboo yarn. Then weaving in warp and weft direction. These unique structures lead BPFs having good moisture absorption and air permeability properties7–9 that could provide suitable conditions for the growth of microorganisms. Song 10 treated BPFs with chitosan through surface modification process and resulted in significant improvement of its antibacterial property.
The pH value of an acid is a good indicator of its antibacterial performance, that is, the ideal pH value range of an acid with the best antibacterial performance in terms of its antibacterial rate range 75%–100% 11 is 2.5–3.0. In genera organic acetic acid (AA) has a pH value of 4.8 while CA has its pH value of 3.5. Limited literature is found related to the antibacterial performance comparison between BPFs treated with chitosan dissolved in CA and AA, respectively. Meanwhile, the washability is an important index for evaluating fabrics’ performance as potential materials for apparel industries. Main factors on washable properties of BPFs treated with chitosan dissolved in CA and AA are crosslinking agent, immersion time, and fabrics’ treatment.12,13
In our previous research, the antibacterial BPFs were successfully fabricated using a lab prepared graphene/chitosan compounded solution with chitosan dissolved in AA as the solvent. However, the final treated BPFs had a strong acid smell mainly because there is no reaction occurring at carboxyl functional groups in AA. CA used as a solvent for dissolving lignin-carbohydrate complexes has less acid sour smelling when compared with AA in another research of us. 14 Therefore, the main objective of this study was to replace AA with CA as a solvent to dissolve chitosan to reduce acid smelling. Specifically, the antibacterial activity of BPFs treated with chitosan dissolved in CA was evaluated and compared with ones treated with chitosan dissolved in AA using the colony count method. In addition, the effects of pH values of organic CA and AA as solvents on the antibacterial performance and washability of BPFs treated with two acid dissolved chitosan were investigated. The mechanism of improved antibacterial performance of BPFs treated with acid dissolved chitosan was analyzed through washing resistance test, scanning electron microscopy (SEM), and Fourier Transform Infrared (FTIR) spectrometer.
Experimental procedure
Experimental design
A complete 2 × 6 factorial experiment with two replicates was conducted to evaluate factors on the antibacterial activity of BPFs treated with chitosan dissolved in organic acids as solvents, in terms of its antibacterial rate. The two factors were organic acid type (CA and AA) and chitosan mass fraction level (0%, 0.1%, 0.2%, 0.3%, 0.4%, and 0.5%). The antibacterial rate test was performed according to GB/T 20944.3–2008
15
based on the Escherichia coli method, a bacterial colony count method. The antibacterial rate,

where T₀ is the number of bacteria counted on a BPF before treated and T₁ is the number of bacteria counted on the corresponding BPF after treated with chitosan dissolved in organic acid and the completion of a preset number of washing cycle and one drying cycle.
In addition, the washability of BPFs treated with chitosan dissolved in organic AA and CA as solvents at a fixed chitosan mass fraction level of 0.3% was evaluated through washing a piece of BPF in a sequence of four different numbers of washing cycle (5, 10, 15, and 20), then followed by one drying cycle after the completion of each of four different numbers of washing cycle. The antibacterial rate of a piece of tested BPF before and after the completion of a preset number of washing cycle and one drying cycle was calculated using equation (1). Also, the acidity coefficient, pKa value 5 of chitosan, and pH values of CA and AA were measured, respectively.
Bamboo pulping fabrics’ preparation
The BPFs’ preparation started off with fully dissolving each of six loadings 0, 1, 2, 3, 4, and 5 g, of chitosan into 100 ml of each of two organic acids with their mass fraction of 2%, respectively, resulting in six chitosan and organic acid mixtures for each of AA and CA, respectively, that is, the chitosan mass fraction level was 0%, 0.1%, 0.2%, 0.3%, 0.4%, and 0.5% for each of AA and chitosan mixture (AACS) as well as CA and chitosan mixture (CACS), respectively. Each of six AACSs was coded with AACS0, AACS1, AACS2, AACS3, AACS4, and AACS5, for each of six chitosan mass fraction levels of 0%, 0.1%, 0.2%, 0.3%, 0.4%, and 0.5%, respectively, while each of six CACSs was coded with CACS0, CACS1, CACS2, CACS3, CACS4, and CACS5, for each of six chitosan mass fraction levels of 0%, 0.1%, 0.2%, 0.3%, 0.4%, and 0.5%, respectively. All tested BPFs with their surface areas measured 100 mm long × 100 mm wide were prepared with removing any burr from all their surfaces first and then flattening them at the room temperature controlled at 26°C, followed by oven-drying these BPFs at 60°C to constant weight. Oven-dried BPFs were treated through immersing them in a container with an acid dissolved chitosan solvent for 45 min, followed by removing, rinsing, and drying operations.
Antimicrobial test of factorial experiment
Each of BPFs tested in the factorial experiment went through one washing cycle and one drying cycle per GB/T 8629–2017 16 after being treated with chitosan dissolved in organic acids. The antimicrobial activities of treated BPFs were evaluated through counting and recording the number of bacteria observed on these BPFs before and after the completion of one washing cycle and one drying cycle. The antimicrobial rates of all tested BPFs were calculated using equation (1) based on three repeated counting.
Washing resistance test
Per GB/T 8629-2017 16 outlining washing and drying procedure for domestic textile products, a piece of treated BPFs went through a sequence of preset numbers of washing cycle, followed by one drying cycle after the completion of each preset number of washing cycle. The antimicrobial activities of treated BPFs were evaluated through counting and recording the number of bacteria observed on a fabric before and after a preset washing-and-drying cycle time completed. The antimicrobial rate for a tested BPFs was calculated using equation (1) based on three repeated counting. Each of BPFs tested in the washability evaluation went through a sequence of preset numbers of washing cycle followed by one drying cycle. The antimicrobial activities of treated BPFs through counting and recording the number of bacteria observed on a BPF before and after the completion of a preset washing-and-drying cycle.
Acid pH and chitosan pKa test
The pH values of CA and AA and pKa value of chitosan were measured using a pH tester (DL11-PHG-9506X, Beijing Zhong Xi Yuan Da Technology Co., Ltd., Beijing, China).
Scanning electron microscopy characterization
A evaluated BPF was first fixed on a metal bracket with conductive adhesive, then sprayed with platinum plating and gold plating, dried at room temperature, and finally observed using scanning electron microscopy (SEM) (FEI Company, NY, USA).
FTIR test
The surface functional groups of BPFs were detected using FTIR spectrometer (Avance 300, Bruker Company, Berlin, Germany). The test conditions is in general room temperature environment. The resolution of infrared spectrum is 1.5 cm−1. The FTIR test started off with completely drying BPFs first, followed by placing BPFs m on the top of a carrier sheet and then being flattened for infrared scanning. The transmission projective mode of the infrared microscope was selected with the frequency of spectrum scanning, resolution of infrared spectrum, and range of spectrum acquisition being set to 200 times, 1.5 cm−1, and 4000–500 cm−1, respectively.
Statistical analyses
A two-factor analysis of variance (ANOVA) general linear model (GLM) procedure was performed first to analyze two main effects and their interaction on the antibacterial rate of BPFs treated with chitosan dissolved in organic acids. In general, mean comparisons were performed using the protected least significant difference (LSD) multiple comparisons procedure if a significant two-way interaction was identified; otherwise, main effects were concluded. All of the statistical analyses were performed at the 5% significance level.
Results and discussion
Organic acid type and chitosan mass fraction effects
Figure 2 summarizes mean values of antibacterial rates of BPFs treated with chitosan dissolved in two organic acids. ANOVA results indicated that the two-way interaction (with a

Mean values of antibacterial rates of bamboo pulping fabrics treated with chitosan dissolved in acetic acid (AA) and citric acid (CA) as a function of chitosan mass fraction level and their corresponding mean comparisons for (1) organic acid type within each of five chitosan mass fraction levels, indicated by a lowercase letter; and (2) each chitosan mass fraction level within each of two organic acid types, indicated by an uppercase letter. Means with same letter or letters are not significantly different at the 5% significance level.
Mean comparisons for chitosan mass fraction level (Figure 2) indicated that within AA dissolved chitosan the antibacterial rate of BPFs treated with 0.5% dissolved chitosan was 97.8%, which was significantly higher than ones treated with 0.3% (96.8%), 0.2% (85.1%), and 0.1% (77.3%) dissolved chitosan, respectively, but not significantly higher than the ones treated with 0.4% (97.2%) dissolved chitosan. The antibacterial rate of BPFs treated with 0.4% dissolved chitosan was significantly higher than ones treated with 0.2% and 0.1% dissolved chitosan, respectively, but not significantly higher than ones treated with 0.3% dissolved chitosan. The antibacterial rate of BPFs treated with 0.3% dissolved chitosan was significantly higher than ones treated with 0.2% dissolved chitosan, followed by a significantly lower ones treated with 0.1% dissolved chitosan. Overall, the antibacterial rate of BPFs treated with AA dissolved chitosan shows a sharp increase trend as chitosan mass fraction level increased from 0.1% to 0.3% with an increment of 0.1%, but this increase was leveled off as chitosan mass fraction level continued its increase to 0.5% with an increment of 0.1%. The BPF treated with 0.3% dissolved chitosan, that is AACS3, if compared with one treated with AA only, that is, AACS0, had Escherichia coli almost disappeared (Figure 3), indicating that the sterilization effect can be increased with the increase of chitosan content.

Photographs show antibacterial activities of bamboo pulping fabrics before (CACS0, AACS0) and after (CACS3, CACS1, AACS3, and AACS1) treated with chitosan dissolved in acetic acid (AA) and citric acid (CA), respectively.
Mean comparisons for chitosan mass fraction level (Figure 2) indicated that within CA dissolved chitosan the antibacterial rate of BPFs treated with 0.5% dissolved chitosan was 99.4%, which was significantly higher than ones treated with 0.2% (98.3%) and 0.1% (98.1%) dissolved chitosan, respectively, but not significantly higher than ones treated with 0.4% (99.1%) and 0.3% (98.8%) dissolved chitosan. The antibacterial rate of BPFs treated with 0.4% dissolved chitosan was significantly higher than ones treated with 0.1% dissolved chitosan, but not significantly higher than ones treated with 0.3% and 0.2% dissolved chitosan. There was no significant difference in antibacterial rate among three BPFs treated with 0.3%, 0.2%, and 0.1% dissolved chitosan. Overall, the antibacterial rate of BPFs treated with CA dissolved chitosan shows an increase trend with a low constant rate if compared with AA dissolved chitosan as chitosan mass fraction level increased from 0.1% to 0.5% with an increment of 0.1%. The BPFs treated with 0.3% dissolved chitosan, that is, CACS3, if compared with one treated with CA only, CACS0, had Escherichia coli almost disappeared (Figure 3), indicating that the sterilization effect can be increased with the increase of chitosan content.
These differences in reducing rates could be caused by the difference in quantity of amide group and carboxyl group formed in AACS and CACS. The amide group and carboxyl group in AACS and CACS can be connected with BPFs through electrostatic force and van der Waals force. In addition, the carboxyl group in AACS and CACS can also be adsorbed on fibers of BPFs through -COO-, -NH, -OH, and other active groups. These connections can help to enhance the cross-linking between acid chitosan mixture (AACS or CACS) and BPFs. 17
Mean comparisons for organic acid type (Figure 2) indicated that BPFs treated with CA dissolved chitosan had a significantly higher antibacterial rate than ones treated with AA dissolved chitosan within each of five chitosan mass fraction levels, indicating that CA dissolved chitosan had better antibacterial effect than AA dissolved one. This was evidenced by the fact that (Figure 3) BPFs treated with 0.1% AA dissolved chitosan, that is, AACS1, had more coliform colonies than ones treated with 0.1% CA dissolved chitosan, that is, CACS1. The differences between antibacterial rates of BPFs treated with CA and AA dissolved chitosan were determined and the differences were expressed as ((CACS-AACS)/AACS) × 100% and plotted as a function of chitosan mass fraction level (Figure 4). Figure 4 indicated that the difference had a higher increase rate up to chitosan mass fraction level reaching 0.3%, then was leveled off.

Difference in antibacterial rate between bamboo pulping fabrics treated with chitosan dissolved in acetic acid (AA) and citric acid (CA) as a function of chitosan mass fraction.
Acid pH value effects
The measured pKa value of chitosan averaged 6.4 with its coefficient of variation (COV) of 2.5%, indicating that it can be dissolved only by acid with its pH value less than pKa value. 18 This is because when acid with its pH value is less than the chitosan pKa, its amino bands are positively charged. In this study, the average measured pH values of AA and CA were 4.75 and 3.13 with their COV values of 2.8% and 3.8%, respectively, implying that the ability of using CA to dissolve chitosan is higher than AA, that is, more chitosan citrates will be formed in CA if compared to AA. Therefore, no acid sour smell was detected when CA was used to dissolve chitosan.19,20
Electron microscopic characterization
SEM images (Figure 5) of treated BPFs indicated that the surface of BPFs treated with AA only (Figure 5(a)) was smooth, while the surface of BPFs treated with chitosan was showing many small etching stripes and grooves (Figure 5(b)–5(e)) and coarser fibers if compared to ones treated with only AA, indicating that BPFs’ surface were successfully grafted. These observations showed that a layer of chitosan film was coated on the fibers’ surface of chitosan treated BPFs, indicating that chitosan was chemically cross-linked on the fibers’ surface.21,22 This is mainly because the smaller molecular weight of the chitosan molecules not only deposited on the grooves and gaps on fibers’ surfaces, but also entered the loose area of BPFs and reacted with carboxyl group to cross-linking.

SEM images of bamboo pulping fabrics treated with chitosan dissolved in organic acetic acid (AA) and citric acid (CA): AACS0 (a); AACS1 (b); AACS5 (c); CACS1 (d); CACS5 (e); fabric photo (f).
FTIR analysis
The infrared spectrum of BPFs treated with chitosan dissolved in AA and CA (Figure 6) showed the wide absorption peak of 3335 cm−1–3338 cm−1, indicating the existence of stretching of -OH groups because of BPFs containing of celluloses. The characteristic acid absorption bands at 1156 cm−1 and 894 cm−1 were caused by C-O-C stretching.
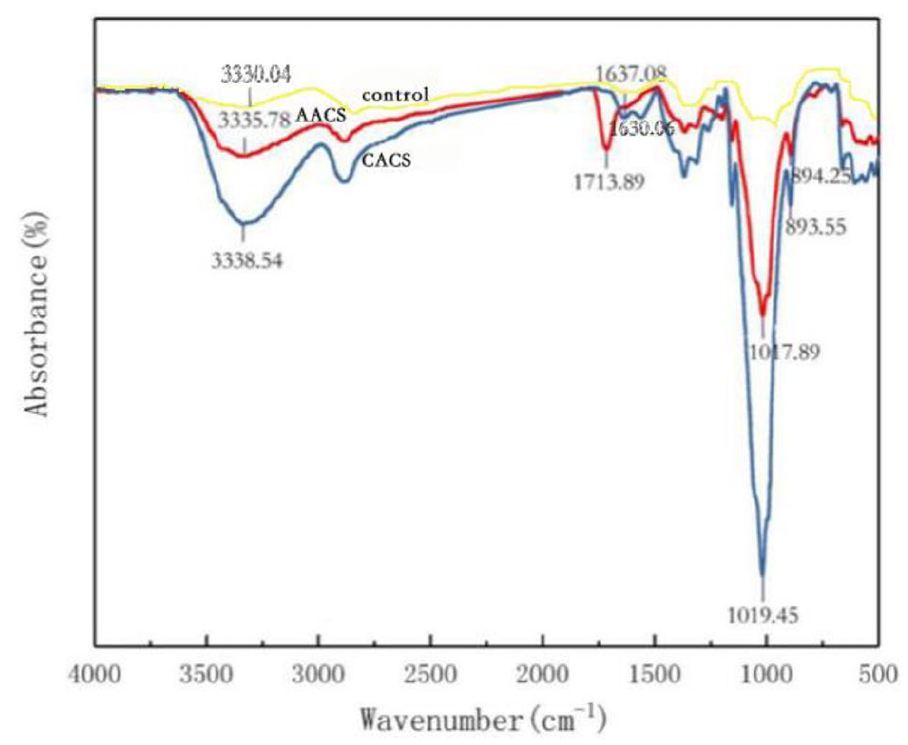
Infrared spectrum of bamboo pulping fabrics treated with control sample, chitosan dissolved in organic acetic acid (AA) and citric acid (CA), respectively.
The absorption band near 1636 cm−1 was the in-plane stretching vibration peak of amide -NH, indicating that the amino group in chitosan (Figure 1) was covalently bound to the free carboxyl group of organic acids (Figure 7) to form a salt bond through esterification, therefore realizing the fixation of chitosan on cotton fabrics, 23 that is, both AA and CA were successfully grafted on chitosan surfaces. However, a new absorption peak appeared at 1713 cm−1 for BPFs treated with chitosan dissolved in CA because of the absorption peak of C = O in the carboxyl group of CA, confirming the esterification reaction between the carboxylic acid in the cross-linked chitosan and the hydroxyl group on the cellulose. 19 The increase of these intensities is more obvious at the characteristic peaks of BPFs treated with chitosan dissolved in CA than AA, for instance, the stretching vibration absorption peaks of -OH at 1017 cm−1 and 3335 cm−1 of the cross-linked BPFs treated with chitosan dissolved in AA were higher than ones in CA (at 1019 cm−1 and 3338cm−1). The low wave number shift (red shift) and narrow peak width indicated that the cross-linking method of dissolving chitosan in CA enhances the interaction between functional groups on chitosan and BPFs. 24

Diagram showing the mechanism of organic acetic acid (AA) (a) and citric acid (CA) (b) dissolving chitosan.
Organic solution type and washing time effects on fabrics’ washability
Figure 8 shows there was a downward trend of the antibacterial rates of BPFs treated with chitosan dissolved in two organic acids as the number of washing cycle increased. The antibacterial rate of a BPF treated with chitosan dissolved in AA shows faster decreasing rates than one in CA as the number of washing cycle increased from 5 to 20 cycles with an increment of five cycles. Specifically, the antibacterial rate of BPFs treated with chitosan dissolved in CA was decreased by 6%, 6.4%, 2.6%, and 3% after being washed 5, 10, 15, and 20 times; while, the antibacterial rate of a BPF treated with chitosan dissolved in AA decreased 8.2%, 6.9%, 7.5%, and 12.3% after being washed 5, 10, 15, and 20 times.

The antibacterial rate of bamboo pulping fabrics treated with chitosan dissolved in organic acetic acid (AA) and citric acid (CA) as a function of washing time.
In this experiment, the numbers of amide group and carboxyl group in CACS increased by citrate acylated chitosan formed during the process of CA dissolving chitosan were higher than the ones in AACS done by acetate acylated chitosan formed during the process of AA dissolving chitosan. The washing resistance of BPFs treated with chitosan dissolved in CA is better than that of BPFs treated with chitosan dissolved in AA.
Conclusions
The major conclusions from experimental results of this study are followings:
(1) BPFs treated with chitosan dissolved in CA had a significantly higher antibacterial rate than ones treated with chitosan dissolved in AA within each of five chitosan mass fraction levels investigated.
(2) Antibacterial rates of BPFs treated with chitosan dissolved in AA and CA increased as chitosan mass fraction increased, but increasing rates were different between BPFs treated with chitosan dissolved in AA and CA.
(3) No acid sour smell was detected for BPFs treated with chitosan dissolved in CA.
(4) BPFs treated with chitosan dissolved in CA could yield higher washing resistance than ones treated with chitosan dissolved in AA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the project funded by the National Natural Science Foundation of China (32071687), the Special Scientific Research Fund of Construction of High-Level Teachers Project of Beijing Institute of Fashion Technology (BIFTQG201805), the Project of Science and Technology Plan of Beijing Municipal Education Commission (KM202010012001), “Nan Taihu Lake elite plan” project ([2018] No.2, Huzhou city, Zhejiang province) and the project from International Cooperation Joint Laboratory for Production, Education, Research and Application of Ecological Health Care on Home Furnishing.
