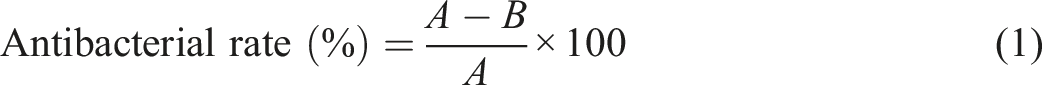Abstract
The fabrics of health care function have drawn increasing attention. Herein, AgNO3 was in-situ reduced into nano silver (AgNPs) by aloe extract (AVE) as reducing agent to gain AgNPs@hinokitiol-grafted chitosan (AgNPs@HTCS). The antibacterial and antioxidant cotton fabric were fabricated by LBL method using AgNPs@HTCS as positive layer and hyaluronic acid as negative layer. The results showed that the optimum preparation conditions of AgNPs@HTCS were as follows: AgNO3 10 mmol/L, HTCS 40 v/v %, AVE 15 mL, 80 °C, 6 h. MIC values of AgNPs@HTCS against S. aureus and E. coli attained 1.7 μg/mL and 5.3 μg/mL, respectively. The antibacterial ratio of cotton fabric reached the maximum 99.9 % at very low concentration of AgNPs@HTCS 0.25 g/L. After 3 LBL layers, the antibacterial properties of functional cotton fabric could withstand the acid or alkali washing. After 25 washing cycles, the antibacterial rate of LBL fabric still maintained above 96 %. Compared with the untreated fabric, antioxidant capacity of LBL functional cotton fabric raised approximately two times. LBL functional cotton fabric still retained the wear performances of the original fabric.
Introduction
With the pursuit of high-quality living, the demands for textiles were no longer confined to the warmth and comfort, but more inclined to pursue the health care protection. 1 Antibacterial and antioxidant function finishing has been regarded as an important way to improve the health care protection of textiles.
During antibacterial finishing, multi-factors should be considered in the selection of antibacterial agents, such as bactericidal efficiency and broad-spectrum, cost, toxicity, durability, and impacts on wearability of fabric.2–4 Up to now, antibacterial agents have been developing towards efficient, low toxic, durable, and environmentally friendly directions. 5 On the basis of composition, the commercial antibacterial agents for textiles covered the organic, 6 inorganic, 7 natural, 8 and composite. 9 However, there were only few antibacterial agents widely used in textiles. Generally speaking, quaternary ammonium salt with a chain of 12∼18 carbon was employed as bactericide and sanitizer for fibers. 10 Nevertheless, they have poor binding force with fibers, meanwhile exhibit the worse chemical stability, higher toxicity, and shorter antibacterial aging. Nano silver was also applied as antibacterial agent in textiles. 11 But it exists the defects of the large addition, high cost, uneven dispersion, and color change. With the increasing awareness of environmental protection, the natural antibacterial agents have been paid more attention. 12 The natural antibacterial agents of high molecules as chitosan (CS) have strong stability, but exhibit the weak antibacterial activity. While the natural antibacterial agents of small molecules as hinokitiol (HT) possess the stronger antibacterial activity, by show the problems of volatile and poor heat-resistance. Consequently, the compounding technology provides a good way to realize the antibacterial agent incorporated in high efficiency, stability, durability, and environmental protection.
There have been a number of studies regarding to the chemical modification of CS and the organic-inorganic composites.13,14 Focusing on the improvement of antibacterial activity and broad-spectrum of CS, several researches had tried to introduce kinds of small molecules with high antibacterial activity into CS structure, such as quaternary ammonium salt modification, 15 tea polyphenol modification, 16 protocatechuic modification, 13 geraniol modification, 17 curcumin modification, 18 lysine modification, 19 and so on. In our previous research, hinokitiol-grafted-chitosan (HTCS) was prepared and its MIC values against S. aureus and E. coli achieved the lower values 43 and 54 μg/mL, respectively. 20 Furthermore, metal ions or metal oxides were also incorporated into the organic structures to improve the antibacterial activity. Malini et al. combined the porous CS and nano ZnO to enhance antimicrobial activity. 14 Li et al. fabricated antibacterial fabric through immersing into CS@silver/sepiolite composites. 21
To the best of our knowledge, nano silver (AgNPs) possesses strong antibacterial activity with broad spectrum which can kill nearly 650 kinds of harmful microorganisms. 22 Many investigations showed that the design of AgNPs with strong antibacterial activity should consider the following: high dispersion stability, low chemical stability, positive surface charge, and relatively low particle size. 23 Generally, chemical synthesis of AgNPs leads to environmental pollution. Thus, green synthesis of AgNPs has been widely concerned. Golabiazar et al. reported the synthesis of silver nanoparticle with Pistacia atlantica leaf extract as a reductant, stabilizer, and capping agent. 24 Zhao et al. prepared uniform and stable AgNPs using hesperidin and pectin as reductants and stabilizers. 25 Bu et al. took advantage of AgNPs in-situ synthesis onto viscose fabric coated by tannic acid. 26 However, AgNPs has certain degree of toxicity which depends on the concentration of Ag+. As a result, the composite of AgNPs and organic antibacterial agents will be the best way to achieve the ideal antibacterial properties.
In this study, AgNO3 was in-situ reduced into nano silver (AgNPs) by aloe extract (AVE) as reducing agent to gain AgNPs@hinokitiol-grafted chitosan (AgNPs@HTCS). Effects of AgNO3 concentration, HTCS concentration, AVE dosage, reducing temperature and time on antibacterial activity of AgNPs@HTCS were studied. The antibacterial and antioxidation cotton fabric were fabricated by LBL method using AgNPs@HTCS as positive layer and hyaluronic acid as negative layer. The antibacterial and antioxidant properties, durability as well as wearability of LBL functional cotton fabric were investigated. It provided a simple and green method for the development and utilization of antibacterial and antioxidant health fabric.
Experimental
Materials
The bleached cotton fabrics (thread density 14.578 tex, warp 133 threads/inch, weft 72 threads/inch) were kind supported by Hualun Printing and Dyeing Mill, Shanghai, China.
Chitosan (CS, Viscosity 100∼200 mPa·s, Deacetylation degre≥95%), hinokitiol (HT), paraformaldehyde, dibasic sodium phosphate, polyethyleneimine (PEI), hyaluronic acid (HA), potassium persulfate, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), and potassium dihydrogen phosphate were obtained from Shanghai Macklin Biochemical Co., Ltd. Silver nitrate (AgNO3) was purchased from Guangdong Guanghua Sci-Tech Co., Ltd. Aloe vera leaf was purchased from Putian Hanjiang Jiangxia Aloe Development Co., LTD. Acetic acid and ethanol were provided by Hangzhou Gaojing Fine Chemical Industry Co., Ltd. Diethyl ether was supplied by Hangzhou Shuanglin Chemical Reagent Co., Ltd. Citric acid was supplied by Chengdu Dongjin Chemical Reagent Co., Ltd. Staphylococcus aureus (S. aureus, ATCC 6538) and Escherichia coli (E. coli, ATCC 25922) were supplied by Shanghai Luwei Technology Co., Ltd.
Preparation of AgNPs@HTCS antibacterial agent
AgNPs@HTCS antibacterial agent was prepared though two steps. First, HT was grafted onto CS via Mannich reaction to obtain HT-grafted CS (HTCS) according to our previous research.
20
Second, nano silver was complexed with HTCS and then in-situ reduced into AgNPs. Aloe leaves were mashed into pastes and mixed with water at the volume ratio 1: 4. The mixture was heated at 100 °C for 15 min, then cooled, and filtered to remove the solid. Aloe extract (AVE) as reducing agent was kept at 0 °C. 50 mL HTCS solution was poured into a three-flask. AgNO3 solution was added into the flask and stirred at certain temperature for 15 min. AVE was dropped into the flask by pipette and stirred for certain time. Finally, centrifuge thrice at 10000 r/min by supercentrifuge (CP70MX, Japan). The free AgNPs were removed to obtain AgNPs@HTCS. The preparation routes were presented in Figure 1. The preparation route of AgNPs@HTCS antibacterial agent.
Antibacterial finishing of cotton fabric
The cotton fabric was immersed into AgNPs@HTCS solution of different concentrations. The AgNPs@HTCS treated fabric was obtained after padding and drying at 80 °C for 2 min.
LBL coating cotton fabric
Cut the cotton fabric into a 10 × 10 cm2 square sample. Cotton fabric was impregnated into 2 mg/L PEI solution at a bath ratio of 1: 30 for 5 min and dried at 80°C to obtain the PEI-activated fabric. 0.25 g/L HA acted as an anionic reagent and 0.25 g/L AgNPs@HTCS solution acted as a cationic reagent. The pre-activated fabric was dipping into anionic and cationic solution in turn at a bath ratio of 1: 30 for 5 min and then dried at 80 °C. The process was repeated until the total of 1LBL、2LBL、3LBL、4LBL and 5LBL. The schematic diagram of functional fabric through LBL were shown in Figure 2. Schematic diagram of the construction of LBL functional cotton fabric.
UV–Vis spectrum
The state of AgNPs@HTCS was observed by UV–Vis spectrophotometer (TU-1901, China) using a quartz cell in the range of 350–600 nm.
Zeta potential of the fabric
The PEI-activated fabric and AgNPs@HTCS finished fabric were smashed and dispersed in water at different pH values. The charge of the PEI-treated fabric and AgNPs@HTCS finished fabric was determined by ZetaPALS nano particle size analyzer.
Antibacterial test
Minimum inhibitory concentration (MIC) of the antibacterial agents against S. aureus and E. coli were determined by a continuous dilution method according to the literature. 27 The minimum concentration at which the microplate reader could not observe the bacterial growth was taken as the MIC of antibacterial agent.
Antibacterial property of the treated cotton fabric was evaluated by the continuous dilution method according to AATCC test method 100–2012. The antibacterial rate of the treated cotton fabric was calculated as:
Oxidation resistance of LBL functional cotton fabric
ABTS radical cation decolorization was used to determine the oxidation resistance of fabrics.
28
Acid and alkali resistance stability of LBL functional cotton fabric
The LBL functional cotton fabrics were washed in the distilled water, 1 mol/L acetic acid, 1 mol/L ammonia water in a bath ratio of 1: 50 at 60 °C for 1 h, respectively. Correspondingly, the washed fabrics were marked as LBL-w, LBL-a and LBL-h, respectively. The antibacterial properties of LBL functional cotton fabric after washing were measured.
Wearability of LBL functional cotton fabric
The LBL functional cotton fabrics were washed according to AATCC test method 61–2013. Antibacterial durability was accessed by the antibacterial rate of the fabric after 5, 10, 15, 20, and 25 repeated washing cycles. According to GB/T 3923.1-1997, breaking strength of the fabric in both warp and weft directions were determined. According to GB/T 21655.1-2008, capillary effect of the fabric was tested. According to AATCC test method 202–2012, handle of the fabric was measured by a fabric sensory instrument (Phabromet, Nucybertek, USA). According to AATCC test method 110–2000, whiteness of the fabrics was tested.
Results and discussion
Preparation of AgNPs@HTCS antibacterial agent
Ag+ was complexed and in-situ reduced into HTCS by green reducing agent AVE so as to enhance the antibacterial activity. UV-Vis spectra can reflect the number of AgNPs via peak intensity and particle size distribution via peak width. Usually, the stronger the peak intensity is, the more the number of AgNPs. The narrower the peak width is, the more concentrated the particle size distribution of AgNPs. MIC value represents the antibacterial activity of the agents. The lower the MIC value is, the better the antibacterial activity. The primary factors which affected the properties of AgNPs@HTCS depended on the concentration of AgNO3, the concentration of HTCS, the dosage of AVE, reducing temperature and time.
Effect of AgNO3 concentration on antibacterial activity of AgNPs@HTCS.
Figure 3(a) shows the UV−Vis spectra of seven AgNPs@HTCS antibacterial agents at different concentrations of AgNO3. It was observed that HTCS had no characteristic absorption peak while AgNPs@HTCS owned the obvious peak at 429 nm. It suggested that AgNPs were successfully incorporated into HTCS. Moreover, the peak intensity of AgNPs@HTCS at 429 nm went through the remarkable increase with the concentration of AgNO3 increasing and arrived at the maximum at the concentration of AgNO3 10 mmol/L. It implied that there existed the most and the uniform and stable AgNPs at the concentration of AgNO3 10 mmol/L. (a) UV-Vis spectra and (b) MIC values of AgNPs@HTCS at different concentration of AgNO3; Ag-n means at the concentration of AgNO3 n mmol/L, n = 1, 2, 4, 6, 8, 10, 20; Preparation conditions: HTCS 40 v/v %, AVE 15 mL, 80 °C, 6 h.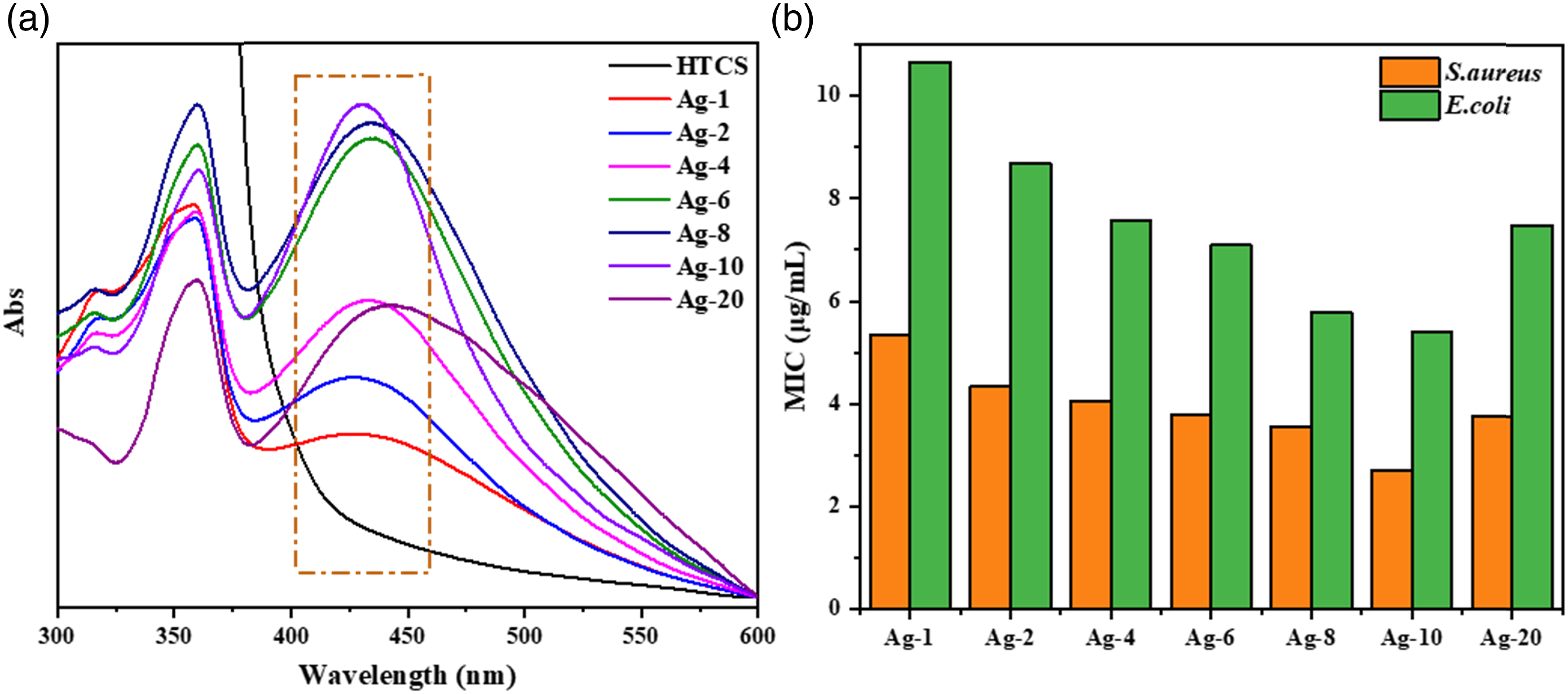
In order to explore the influence of AgNO3 concentration on antibacterial activity of AgNPs@HTCS, MIC values were presented in Figure 3(b). With the increase of AgNO3 concentration, MIC value of AgNPs@HTCS against S. aureus and E. coli decreased gradually, reached the lowest value at the concentration of AgNO3 10 mmol/L, and then increased. The smaller particle size and more concentrated distribution of AgNPs contributed to the enhancement of antibacterial activity. 23 With the concentration of AgNO3 increasing, the size distribution of AgNPs gradually decreased, and correspondingly the antibacterial activity of antibacterial agents gradually increased. It was consistent with the change trend of UV-Vis results. The excessive AgNO3 may lead to the increased size distribution of AgNPs and the weakened antibacterial activity of AgNPs@HTCS.
Effect of HTCS concentration on antibacterial activity of AgNPs@HTCS.
As seen from Figure 4, effects of HTCS concentration exhibited the same change trend as effects of AgNO3 concentration. With the increase of HTCS concentration, the peak intensity of AgNPs@HTCS at 429 nm had the obvious increase while MIC values of AgNPs@HTCS against S. aureus and E. coli decreased. It was attributed to the more complexation sites between HTCS and Ag+ as the concentration of HTSC increased. Therefore, more reduced AgNPs were obtained, leading to the increase of peak intensity. The peak intensity reached the maximum and MIC value got the minimum at the concentration of HTCS 40 v/v %. (a) UV-Vis spectra and (b) MIC values of AgNPs@HTCS at different concentration of HTCS; HTCS-n means at the concentration of HTCS n v/v %, n= 5, 10, 20, 40, 60, 80, 100; Preparation conditions: AgNO3 10 mmol/L, AVE 15 mL, 80 °C, 6 h.
Effect of AVE dosage on antibacterial activity of AgNPs@HTCS
It was observed from Figure 5 that when the dosage of AVE was 15 mL, the characteristic peak was the largest and narrowest while the MIC value of AgNPs@HTCS against S. aureus and E. coli got the lowest. It suggested that AVE reducing agent should be around the appropriated amount to obtain AgNPs@HTCS of the best antibacterial activity. (a) UV-Vis spectra and (b) MIC values of AgNPs@HTCS at different dosage of AVE; AVE-n means at the dosage of AVE n mL, n = 5, 10, 15, 20, 25, 30; Preparation conditions: AgNO3 10 mmol/L, HTCS 40 v/v %, 80 °C, 6 h.
Effect of reducing temperature and time on antibacterial activity of AgNPs@HTCS
As the same trends of the above factors in Figure 6, the optimum reducing temperature and time was at 80 °C for 6 h. The lower reducing temperature and the shorter time caused the insufficient reduction of Ag+ into AgNPs, resulting in the broader distribution of AgNPs and the lower antibacterial activity of AgNPs@HTCS. However, the higher reducing temperature and the longer time also displayed the adverse effects on the reduction of AgNPs and antibacterial activity of AgNPs@HTCS. (a) UV-Vis spectra and (b) MIC values of AgNPs@HTCS at different reducing temperatures and time; preparation conditions: AgNO3 10 mmol/L, HTCS 40 v/v %, AVE 15 mL.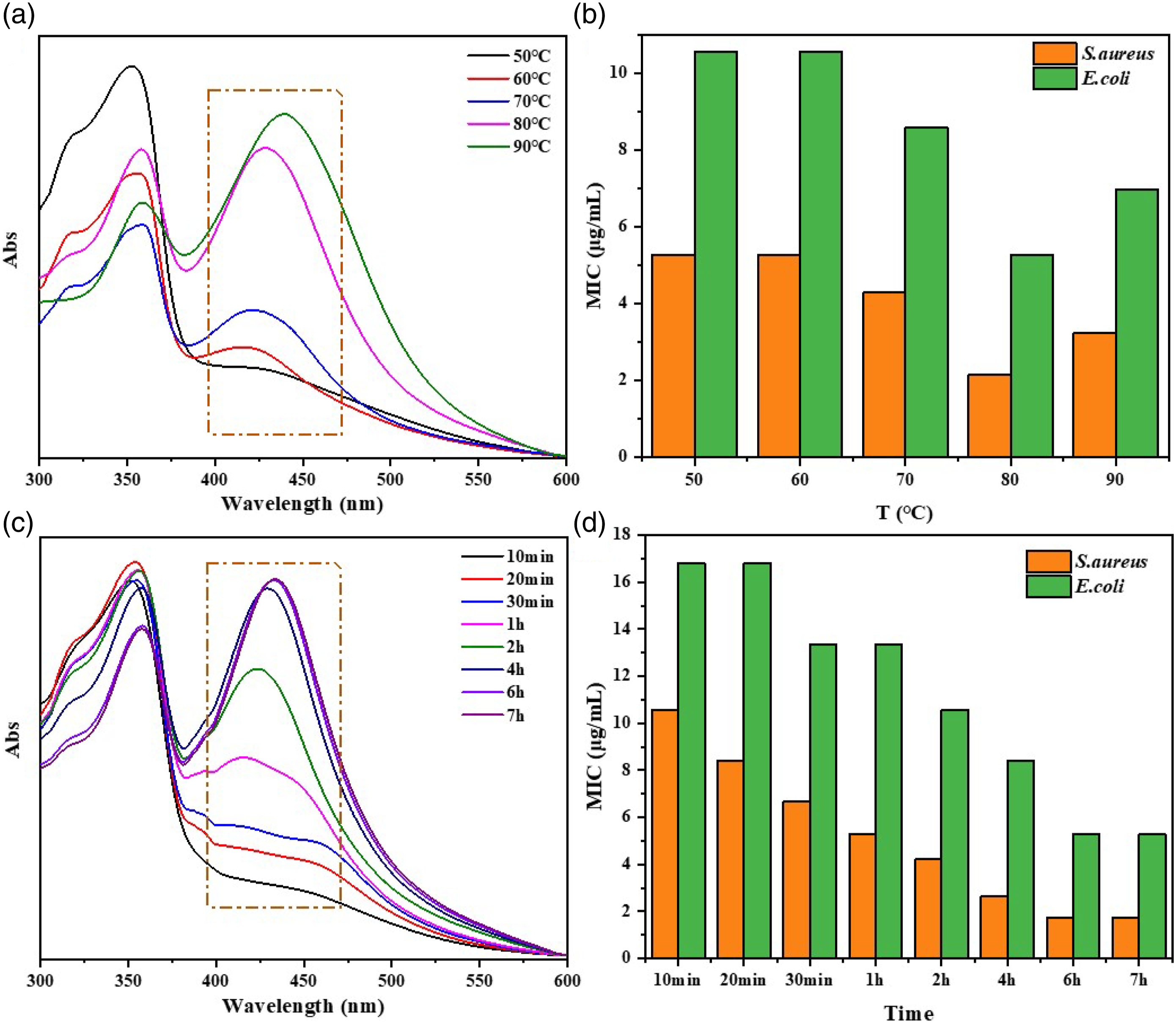
Above all, the optimum preparation conditions of AgNPs@HTCS were as follows: AgNO3 10 mmol/L, HTCS 40 v/v %, AVE 15 mL, 80 °C, 6 h. In our previous research, the average particle size of AgNPs was around 60 nm. 29
Comparison of antibacterial activity with other literature
MIC values of different antibacterial agents against S. aureus and E. coli.

AgNPs@HTCS finishing on cotton fabric
The concentration of AgNPs@HTCS dominates the antibacterial properties of cotton fabric. Seen in Figure 7, with the increase of AgNPs@HTCS concentration, the antibacterial ratio of cotton fabric had the remarkable increase and then remained stable. It arrived at the maximum value 99.9 % at the concentration of AgNPs@HTCS 0.25 g/L. Compared with CS treated cotton fabric, the antibacterial rates of AgNPs@HTCS treated cotton fabric against S. aureus and E. coli increased by around 58 % and 90 %. Compared with HT treated cotton fabric, those against S. aureus and E. coli increased by about 39% and 27 %. AgNPs@HTCS possessed the high antibacterial activity due to the synergistic antimicrobial mechanism of AgNPs, HT and CS incorporated into AgNPs@HTCS.29,34 Consequently, the low dosage of AgNPs@HTCS 0.25 g/L could impart the cotton fabric with high antibacterial property. Effect of AgNPs@HTCS concentration on antibacterial properties of cotton fabric: (a) The growth of S. aureus and E. coli on the solid culture medium; (b) Antibacterial ratios of cotton fabrics treated by different concentrations of AgNPs@HTCS.
Figures 8(a) and (b) display surface morphology of cotton fabrics before and after AgNPs@HTCS finishing. Compared with the pristine cotton fabric, finely white ground partiles evenly scattered onto the surface of antibacterial coton fabric. Besides, it was clearly observed from Figure 8(c) that Ag element was uniformly distributed onto cotton fabric. It not only demonstrated the even distribution of AgNPs@HTCS onto cotton fabric, but also confirmed the success complexation between nano silver and HTCS. SEM image of the untreated cotton fabric, (b) SEM image and (c) EDS image of the treated cotton fabrics by AgNPs@HTCS at the magnification ×10000.
Zeta potential of the treated cotton fabric
In order to pave the way of LBL treatment for cotton fabric, the zeta potential of cotton fabrics treated by PEI and AgNPs@HTCS were tested at different pH values. It was found from Figure 9 that the surface of pristine cotton fabric presented negative charges while the surface of PEI treated cotton fabric exhibited positive charges regardless of pH values. However, the surface of AgNPs@HTCS treated cotton fabric experienced the transformation from positive charge to negative charge with pH values increasing and performed the positive charge within pH 8.3. As a result, LBL method could be employed to impart the oxidation resistance by hyaluronic acid and the antibacterial properties by AgNPs@HTCS for cotton fabric. Zeta potential of (a) the untreated, (b) PEI treated, and (c) AgNPs@HTCS treated cotton fabrics at different pH values.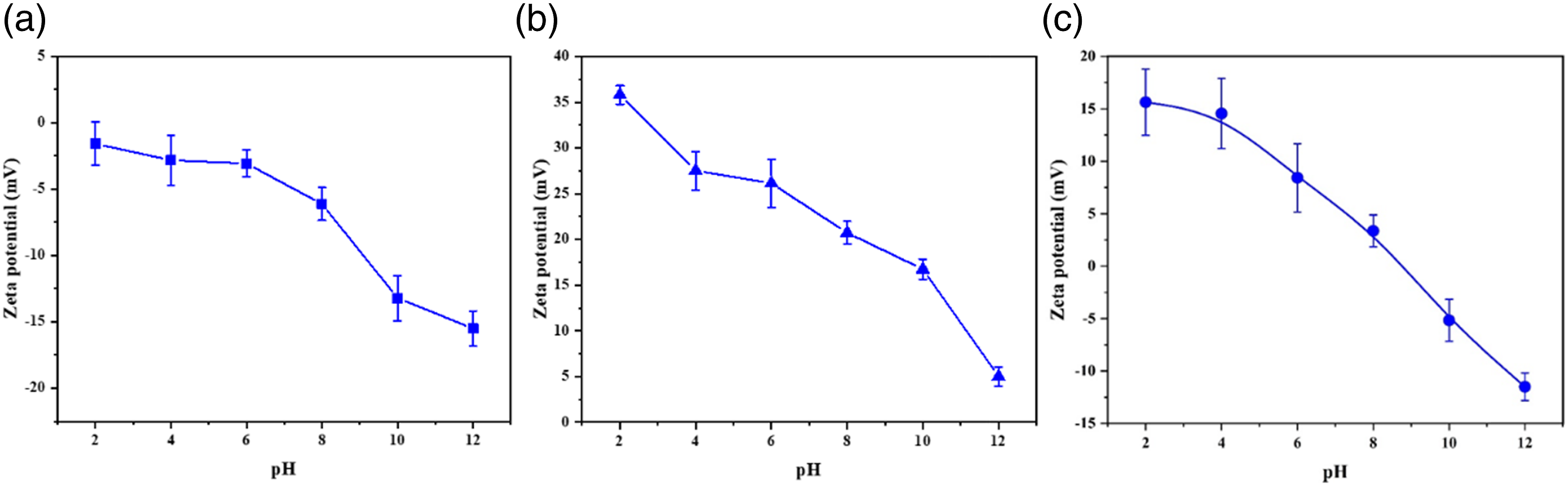
Antibacterial properties of LBL functional cotton fabric
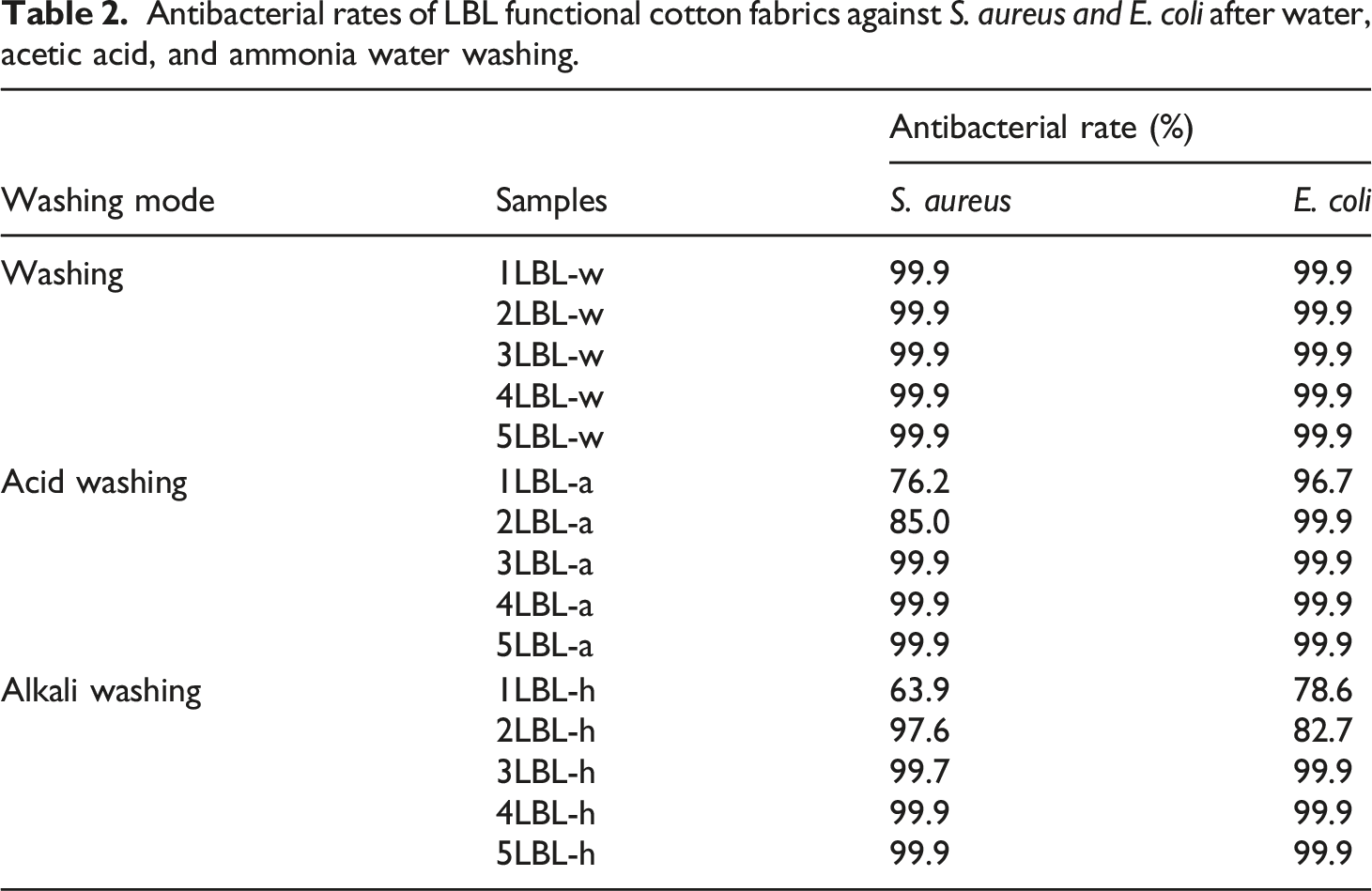
Functional cotton fabrics of antibacterial and anti-oxidation performances were fabricated by LBL method. LBL layers affected the antibacterial stability of functional cotton fabric. It was observed from Figures 10(a) and (b) that LBL functional cotton fabric possessed good antibacterial properties against S. aureus and E. coli from one layer to five layers before and after water washing. It demonstrated that LBL functional cotton fabrics exhibited the washing resistance due to the electrostatic force of LBL layers. Furthermore, those also achieved the acid- or alkali-resistance after 3 LBL layers, shown in Figure 10(c) and (d). Table 2 lists the antibacterial rates of functional cotton fabrics by different LBL layers after water, acid and alkali washing. It also illustrated that more than two LBL layers could impart cotton fabric good antibacterial stability against acid or alkali washing. The positive AgNPs@HTCS layer and negative hyaluronic acid layer interacted with electrostatic attraction. After 3 LBL layers, the relative stable interaction layers were formed onto the surface of cotton fabric, which could endure the external physical and chemical actions. Bacterial growth state of LBL functional cotton fabrics after different washing modes: (a) without washing; (b) water washing, (c) 1 mol/L acetic acid washing, and (d) 1 mol/L ammonia water washing. Antibacterial rates of LBL functional cotton fabrics against S. aureus and E. coli after water, acetic acid, and ammonia water washing.
Durability of LBL functional cotton fabric
The ideal functional cotton fabric should have certain durability and withstand washing many times. Figure 11 presents the antibacterial properties of 3LBL functional cotton fabric after different washing cycles. After 25 washing cycles, the antibacterial rate of functional cotton fabric against S. aureus and E. coli still maintained above 96 %. It manifested the excellent antibacterial durability of LBL functional cotton fabric. (a) The growth of S. aureus and E. coli on the solid culture medium and (b) Antibacterial rates of 3LBL functional cotton fabric after different washing cycles.
Oxidation resistance of LBL functional cotton fabric
LBL functional cotton fabric also possessed the antioxidant function. Compared with the untreated fabric, ABTS+ scavenging capacity of LBL functional cotton fabric raised approximately two times, shown in Figure 12. It confirmed the good oxidation resistance property of LBL functional cotton fabric. This was ascribed to the antioxidant function of hyaluronic acid (HA) layers (Figure 13). HA can clear oxygen free radical, including OH•, O2•, DPPH•, and so on.
35
Furthermore, the antibacterial properties of LBL functional cotton fabric were attributed to ternary synergistic antibacterial actions incorporated by AgNPs, HT, and CS. CS was liable to take on positive charge due to its amino groups, which could attract the bacterial cell membrane with negative charge and then rupture the cell membrane. In addition, HT, a tropolone compound, would also destroy the cell membrane and cause the loss of cytoplasm. HTCS exhibited the synergistic antibacterial actions of CS and HT. What’s more, the complexation of nano silver into HTCS further enhanced the antibacterial activity.
29
Ternary synergistic antibacterial actions impart LBL functional cotton fabric for high antibacterial properties. Oxidation resistance property of the untreated and LBL functional cotton fabric. Antibacterial and oxidation-resistance mechanism of LBL functional cotton fabric.

Wearability of LBL functional cotton fabric
The chemical treatment should have less impact on the wearability of fabric. The hydrophilicity, tensile, handle, and whiteness of LBL functional cotton fabric were investigated. Seen from Figure 14(a), the wicking height of LBL fabric improved in comparison with the untreated fabric. HA called as “natural moisturizing factor”, may impart better hydrophilicity of LBL fabric, which would increase the wear comfort of the fabric. Moreover, breaking elongation, handle, and whiteness of LBL fabric had little changes, shown in Figure 14(b)–(d). Since the concentration of AgNPs@HTCS and HA for LBL layers were very low, the wearability of LBL fabric could retain the original level. (a) Hydrophilicity, (b) Tensile properties, (c) Handle, and (d) Whiteness of the untreated cotton fabric and LBL functional cotton fabric.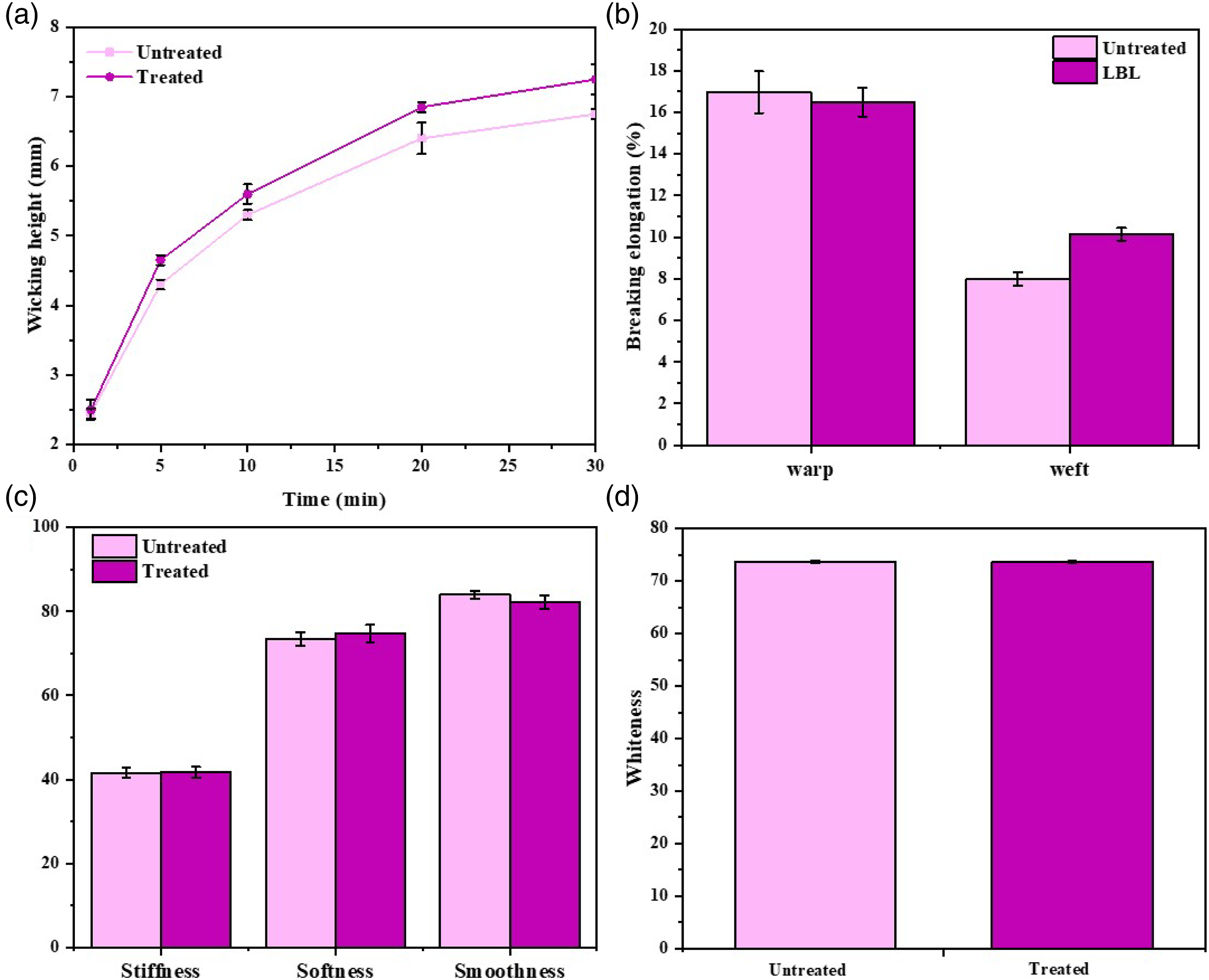
Conclusion
The optimum preparation conditions of AgNPs@HTCS were as follows: AgNO3 10 mmol/L, HTCS 40 v/v %, AVE 15 mL, 80 °C, 6 h. MIC values of AgNPs@HTCS against S. aureus and E. coli reached 1.7 μg/mL and 5.3 μg/mL, respectively. The antibacterial ratio of cotton fabric achieved the maximum value 99.9 % under very low concentration of AgNPs@HTCS 0.25 g/L. The antibacterial properties of functional cotton fabric after 3 LBL layers exhibited the resistance to acid/alkali washing and remained around 99.9 %. After 25 washing cycles, the antibacterial rate of LBL fabric still maintained above 96 %. Antioxidant capacity of LBL functional cotton fabric approximately tripled from the untreated fabric. In comparison with the untreated fabric, LBL functional cotton fabric had better hydrophilicity and retained the original tensile, handle, and whiteness. The antibacterial and antioxidant cotton fabric based on AgNPs@HTCS and HA possessed good potential application in industry. It provided a simple and green method for the development and utilization of multifunctional health fabric.
Footnotes
Acknowledgements
Much thanks are given for the financial support from Zhejiang Province Public Welfare Technology Application Research Project (No. LGG19E030001) and the Fundamental Research Funds of Zhejiang Sci-Tech University (No. 2021Q003).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Zhejiang Province Public Welfare Technology Application Research Project (No. LGG19E030001) and the Fundamental Research Funds of Zhejiang Sci-Tech University (No. 2021Q003).