Abstract
Environmental challenges, especially dye wastewater produced by printing and dyeing industry, pose a serious threat to global public health, and it is an urgent problem to realize harmless treatment of dye wastewater. Here, the combination of covalent organic framework materials (TpPa-1) and biological matrix materials (CF) was explored for the adsorption of dyes for the first time. The functional ACF-TpPa-1 composite adsorption materials were successfully prepared with collagen fiber (CF) made of leather waste as matrix, ethylenediamine (EDA) and covalent organic framework material (TpPa-1) as modified raw materials. It’s structure and properties were characterized by X-ray photoelectron spectroscopy (XPS), Fourier transform infrared spectroscopy (FTIR), Scanning electron microscopy (SEM). The experimental results showed that ACF-TpPa-1 had an adsorption capacity of 257.98 and 449.54 mg/g for acid fuchsia and reactive blue 19, respectively. It also showed excellent adsorption/desorption performance and repeatability after six cycles. As a collagen-based dye adsorbent with pH response, it has potential application prospects in the treatment of dye wastewater.
Introduction
With the rapid economic growth and the development of global industrialization, water pollution, especially the pollution caused by dye wastewater, has become one of the most serious threats facing mankind. 1 Most dyes are stable extremely, and difficult to degrade naturally. When they are discharged into the environmental water, they will affect the normal living activities of plants in the water, destroy the ecological balance of the environment, and even endanger the health of animals and human beings. Under such severe environmental conditions, the purification of dye wastewater is more challenging, finding an efficient and environmentally friendly dye wastewater treatment method is becoming increasingly urgent. There are some methods, such as biological method, physicochemical method, and physical-chemical-biological combined method are widely applied in industries according to the current research in the green chemistry field. 2 Organic dyes are compounds with complex and stubborn chemical structure, which have the characteristics of diverse types, poor biochemical properties, high biological toxicity, and difficult to condense and degrade. In this case, the adsorption method has become an attractive strategy because of its simple operation, strong adaptability, stable effect, and easy recovery.3,4
Correspondingly, the adsorption method has the limitation of large amount of adsorbent and high cost, the treatment of dye wastewater by adsorption method requires high performance of adsorbing materials. Therefore, an environmentally friendly and economically efficient method must be adopted to prepare functional materials to adsorb dyes in wastewater, numerous researches are carried out around the purpose of efficiently processing dyes.5–9 Here, we have carried on the beneficial exploration research to transform the collagen fiber waste into the functional adsorption material. Clearly, this is a potential win-win strategy that will greatly benefit the cleaner production of the leather industry and reasonably solve the environmental problems caused by dye wastewater.
A large amount of pre-tanned protein solid waste was produced in leather production, such as pruning and peeling processes. It will not only waste resources, but also pose a potential threat to the ecological environment.10,11 Therefore, it is necessary to convert these wastes into high value-added materials, collagen fibers (CF) derived from animal skin tissues are considered as a renewable biomass material with abundant active groups, such as −NH2, −C(O)NH2, −COOH, and −OH, making it a potential excellent adsorption material.12–15 However, it is still limited by the shortcomings of the material itself, such as limited active sites, easy swelling and low specific surface area. In view of this situation, many researchers have performed composite modification on CF to improve adsorption properties and make it more conducive to adsorption, such as cross-linking the CF matrix with polydimethylsiloxane (PDMS), loading Fe(III) on CF, preparing amino collagen fibers (ACF), and so on.16–22 It should be noted that using amino-rich ethylenediamine (EDA) to graft collagen fibers with amino groups can increase the number of amino groups and provide more active sites, which is of great benefit to the adsorption of dyes. 18
In order to improve the ability of CF to absorb dyes, we set our sights on covalent organic frameworks (COF), and explored the method of combining COF with biological matrix materials for dye adsorption for the first time. COF is a porous crystalline organic polymer formed by strong covalent bonds, with its good crystal structure, large specific surface area, permanent porosity, and high stability. COF has shown excellent potential in various applications such as gas storage and separation,23,24 extraction and adsorption,25,26 and photovoltaics, 27 making it an excellent material for removing organic dye pollutants. In addition, through the design and selection of monomers, active sites can also be introduced into the COF framework structure to adsorb dye molecules. Adsorption materials prepared based on COF with different structures have different adsorption characteristics for dyes. For example, COF materials of the Schiff base series form a network through the reaction of −CHO and −NH2, and the molecular structure contains nitrogen atoms, which is conducive to binding with dye molecules. These illustrates the broad prospects of COF adsorbed dye materials.25,26,28–33 However, in the application of COF adsorption materials, powdered crystalline materials can cause many problems, such as recycling and pipeline clogging, which results in the deterioration of separation ability, limiting the extent of their practical application. Therefore, the researchers often process the powder into a thin film or other molded body through the method of material compounding to facilitate the application of COF materials.34,35
Here, a functional ACF-TpPa-1 composite adsorption material that is expected to treat dye wastewater efficiently is introduced (Figure 1). CF is selected as the matrix of the material, and EDA is used to aminate CF to increase the number of –NH2 on the surface, TpPa-1 is used as a modifier to improve the adsorption performance. 36 Acid fuchsin and reactive blue 19 are selected for adsorption experiments. The performance of ACF-TpPa-1 as dye adsorbent is evaluated by fitting adsorption kinetics curve and isothermal adsorption curve. The results show that ACF-TpPa-1 has broad application prospects in the treatment of dye wastewater, which is beneficial to environmental protection.

Schematic diagram of material adsorption of acid fuchsin and reactive blue.
Experimental section
Materials and methods
The collagen fiber matrix (CF) is made from raw hides that come from a local tannery (Chengdu, China) (Detailed experimental procedures for preparing CF are described in Supporting Information Section S1). 37 Mesitylene (99%), 1,4-dioxane (99%), p-phenylenediamine (98%) (Pa-1) were purchased from Saen Chemical Technology Co., Ltd. (Shanghai, China); 1,3,5-Benzenetricarboxaldehyde (99%) (Tp) purchased from Jilin Chinese Academy of Sciences-Yanshen Technology Co., Ltd.(Jilin, China); N,N-dimethylformamide (DMF), Ethylenediamine (EDA), Cyanuric chloride (97.0%) (CNC) was purchased from Aladdin (Shanghai, China); CH3COOH (36%) and NaOH (96.0%) were purchased from Chengdu Kelon Chemical Co., Ltd. (Chengdu, China). Acetone (99.5%) was provided by Chengdu Changzheng Chemical Co., Ltd. (Chengdu, China). In particular, the experimental supplies are all analytically pure and can be used directly without further processing.
Fourier transform infrared (FTIR) spectra were recorded on KBr particles using DIGILAB FTS-3000 spectrometer; Powder X-ray diffraction (PXRD) patterns were obtained on X’Pert Pro MPD DY129 X-ray diffractometer; The surface composition analysis was performed by AXIS Supra X-ray photoelectron spectrometer (XPS); ULTRA Plus scanning electron microscope (SEM) was used for morphological analysis. The concentration of the dye solution was measured with a 722 UV-VIS spectrophotometer.
Synthesis of TpPa-1
The 1,3,5-trialdehyde phloroglucinol (0.3 mmol) and p-phenylenediamine (0.45 mmol) were added into the pressure tube (tube inner size = 10 × 8 mm2), followed by mesitylene and 1, 4-Dioxane (1:1, v/v, 4 mL), and ultrasonic treatment for 10 min at room temperature to make it uniformly dispersed. Subsequently, acetic acid (AcOH, 6 M, 0.6 mL) was added, and nitrogen was introduced into the pressure tube for 30 s to remove the air in the tube to create an oxygen-free environment for the reaction. The tube was quickly sealed and reacted at 120°C for 72 h. After the reaction, the orange-red precipitate was collected, and the product was thoroughly washed with N,N-dimethylformamide (DMF) and acetone. Finally, vacuum dried at 100°C for 12 h to obtain the target product TpPa-1(Yield 87.34%). Figure 2 shows schematic diagram of TpPa-1 preparation.

Schematic diagram of adsorbent preparation.
Synthesis of ACF and ACF-TpPa-1
About 2 g CF was mixed with distilled water in a 500 mL flask to fully swell it, filter to remove water, and then 0.008 mol cyanuric chloride dissolved in acetone was add slowly, react in ice-water bath for 3 h. During this period, 0.5 mol/L NaOH was continuously added to maintain the pH of the reaction solution at 6.0–6.5. After the reaction, the unreacted CNC was washed away with acetone, and the collagen fibers were washed thoroughly with water. The washed collagen fibers were put into a three-necked flask again, dissolve 0.016 mol of ethylenediamine in an appropriate amount of distilled water and 0.1740 g TpPa-1 was added at the same time, react at 45°C for 4 h and then heat up to 70°C for 3 h. After the reaction, the crude product was filtered and washed thoroughly with distilled water, and freeze-dried to obtain ACF-TpPa-1.
The preparation steps of ACF are similar to those of ACF-TpPa-1, except that TpPa-1 is not added. Figure 2 shows schematic diagram of adsorbent preparation. It should be noted that the use of amino-rich EDA to graft collagen fibers can increase the number of amino groups and provide more active sites in CF, which is of great benefit to the adsorption of dyes. 18
Results and discussion
Characterization of the TpPa-1
The infrared spectrum of the product is analyzed, 1604 cm−1 is the characteristic peak of C=O, and 1578 cm−1 is the characteristic peak of C=C. About 1450 cm−1 is caused by C=O tensile vibration, and the peak at 1275 cm−1 is caused by C–N tensile vibration (Figure 3(a)). These characteristic peaks are consistent with previous reports in the literature. 38 The spectrum obtained by PXRD analysis shows a peak at 4.7°, which corresponds to 100 reflection planes. In addition, there are several characteristic peaks at 8.1°, 11.1°, and 27°, corresponding to 200, 210, and 001 planes (Figure 3(b)), which are consistent with literature reports. 38 These two characterizations indicate that the TpPa-1 was successfully synthesized. In addition, the specific surface area of the material is 935.65 m2/g in the nitrogen adsorption test as shown in Supplemental Figure S1.

(a) FTIR spectrum of TpPa-1 and (b) PXRD spectrum of TpPa-1.
X-ray photoelectron spectroscopy (XPS) analysis
The element content on the surface of the material is characterized by XPS to obtain Figure 4. From the XPS spectra of CF, ACF, and ACF-TpPa-1, three characteristic peaks of 284, 532, and 400 ev can be seen, which are attributed to C, O, N element, respectively. The content of N element on the surface of ACF after amination modification increased significantly (Table 1), and the content of C element decreased, which is the reason for the increase of amino groups on the surface of the material. After continuing to introduce TpPa-1 into the material, the content of C and O elements increases, and the corresponding N element content decreases, which shows that the introduction of TpPa-1 does increase the oxygen-containing groups on the surface of the material. Further perform peak fitting of the carbon element spectrum, and divide the C element into four chemical environments: C–C/C=C, C–OH, C–N/C–O–C, and C=O/C(O)–N (Supplemental Figure S3). It can be seen from Table 2 that the content of C–N in ACF and C=O/C(O)–N in ACF-TpPa-1 increased. The increase in the content of these groups is beneficial to the adsorption of dyes.
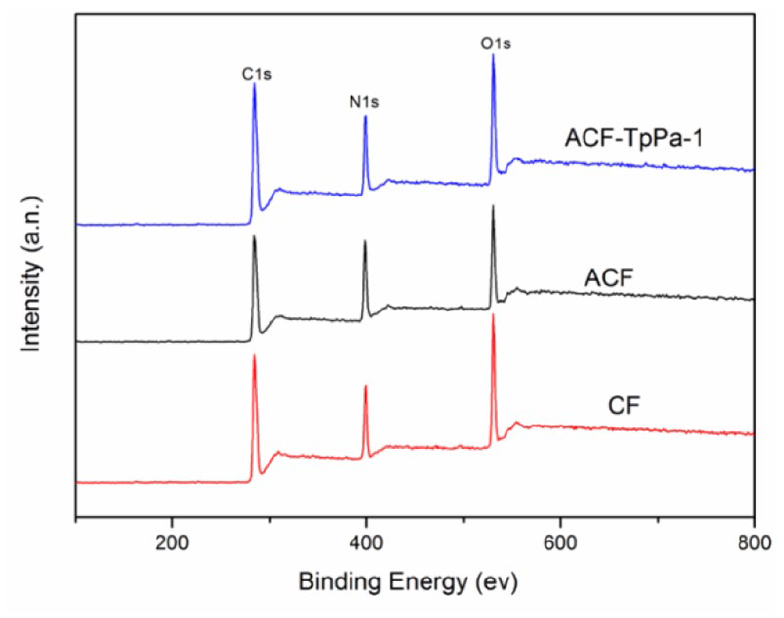
XPS spectrum of CF, ACF, and ACF-TpPa-1.
C, N, O element content of CF, ACF, and ACF-TpPa-1.
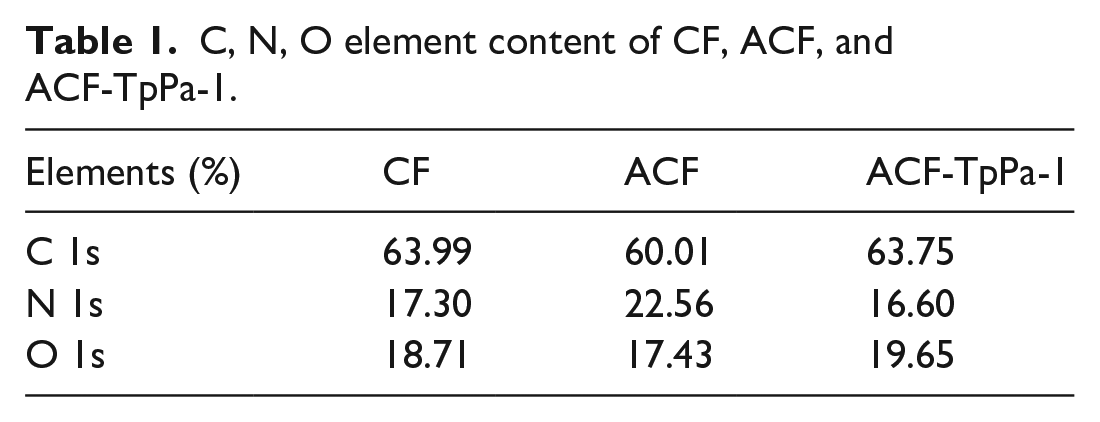
Peak fitting results of C1s of CF, ACF, and ACF-TpPa-1.
Scanning electron microscopy (SEM) morphology study
Figure 5(a) shows the bundled fiber structure of CF. Obviously, the looser gaps between the fibers can increase the specific surface area of the material and provide more active sites for dye adsorption, which fully indicates that CF as a matrix of composite materials is promising. Contrasting these figures, the fiber surface of the CF (Figure 5(a)) and ACF (Figure 5(b)) is relatively smooth and tight, after COF material modification, the fiber surface of the ACF-TpPa-1 (Figure 5(c)) is rougher and looser, which shows looser fiber bundles. The rougher surface and looser fiber bundles have more porosity and larger surface area, which may have better adsorption effect for dyes. From Figure 5(c), it can be clearly observed that the TpPa-1 powder crystals are dispersed on the surface of the fiber, which indicates that the surface of the the modified collagen fiber successfully grafted COF substance with large specific surface area and porosity, which is beneficial to improve the adsorption properties of composites. Supplemental Figure S2 shows the image of the TpPa-1 crystals on the surface of the ACF-TpPa-1.

SEM images of: (a) CF (×1000), (b) ACF (×1000), (c) ACF-TpPa-1 (×1000), (d) CF (×2000), (e) ACF (×2000), and (f) ACF-TpPa-1 (×2000).
Fourier transform infrared spectroscopy (FTIR) analysis
CF has several characteristic absorption bands of amide A (3306 cm−1), amide B (3079 cm−1), amide I (1631 cm−1), amide II (1552 cm−1), and amide III (1240 cm−1). 39 The absorption band of amide A due to the absorption peak of –NH2, and the amide I band is assigned to the C=O stretching vibration, the amide II represents the coupling of N–H stretching and bending vibration, and the amide III band is the absorption band produced by C–N. Figure 6 shows the FTIR spectrum of CF, ACF and ACF-TpPa-1. Through the analysis of amide A, amide I, amide II, and amide III, for ACF-TpPa-1, it can be seen that although the position of each characteristic peak did not change significantly, the intensity of these peaks is significantly enhanced compared to CF. The increase of the characteristic peak intensity can preliminarily indicate that the content of oxygen-containing and nitrogen-containing groups (C=O, N–H, C–N) in the modified composite material has increased, which will affect the adsorption properties of materials.
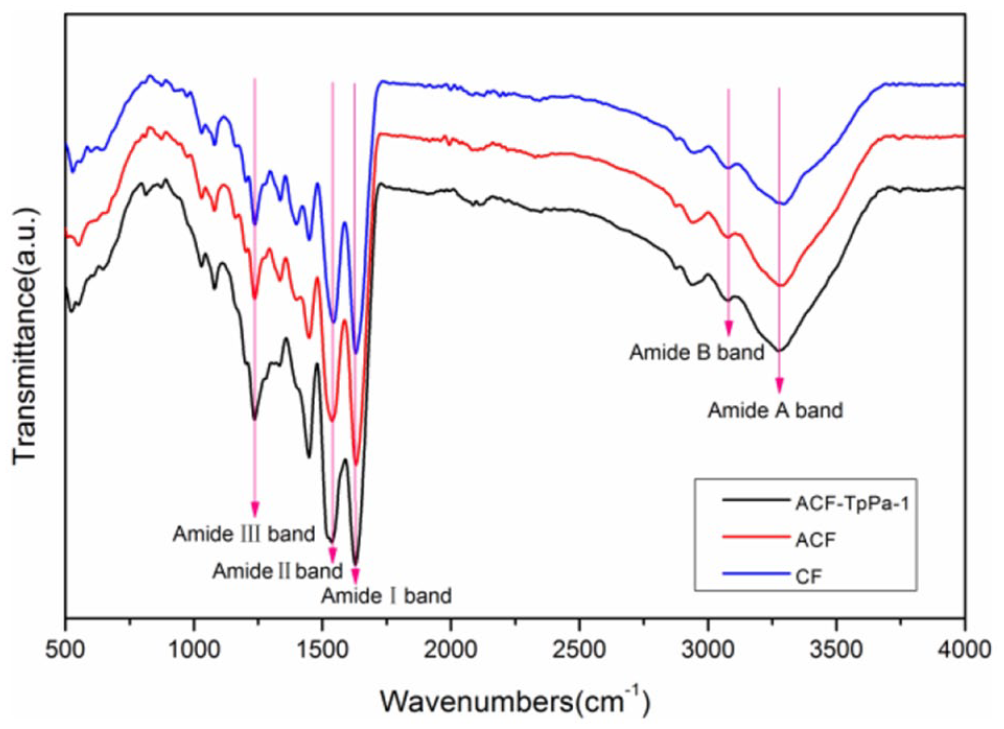
FTIR spectrum of CF, ACF, and ACF-TpPa-1.
Organic dye removal
Effect of pH on dye adsorption
The collagen-based adsorbent is a pH-sensitive material with an isoelectric point of 4.2. In the experiment, 0.0125 mg ACF-TpPa-1 is added to the dye solution of 100 mg/L, and the dye removal rate is measured under different pH values. Figure 7 shows that ACF-TpPa-1 has a good adsorption effect in the acidic environment of pH 1.5–3, and the best adsorption effect is around pH 2, the dye removal rate reach 97.41% and 92.85% respectively. The main reason is that reactive blue 19 molecules contain C=O, −OH, and −SO3−, negatively charged under neutral and alkaline conditions, while collagen fiber containing C=O, −NH2, and −COOH is negatively charged in alkaline environments. As a result, they are repulsive between the dye molecules and the adsorbent material, which is not conducive to the adsorption of reactive dyes. As the pH value decreases, the adsorbent is neutralized and protonated (the groups of –NH2, –COO–), causing positive charge enhancement, at the same time, accompanied by the reversible conversion between ketone form and enol form in TpPa-1, which is more conducive to the adsorption of reactive blue 19. And for acid fuchsin, it has similar results. Acid fuchsin can ionize in neutral and alkaline aqueous solutions. At lower pH values, the negative charge of acid fuchsin molecules weakens and is in a state of low charge density, while the degree of protonation of ACF-TpPa-1 increases, and the electrostatic effect is enhanced, thus increasing the adsorption capacity of acid fuchsin. The above results showed that ACF-TpPa-1 has good adsorption capacity for acid fuchsia and reactive blue 19 in acidic environments. As a dye adsorbent with pH response, its adsorption properties can be regulated by changing pH conditions.

Dye removal rate under different pH values.
Effects of modification of adsorbent materials on dye adsorption
This experiment is conducted under the conditions of pH 2 and initial concentrations of acid fuchsin and reactive blue of 150 and 100 mg/L, respectively. Figure 8 shows the comparison of the adsorption capacity of the three adsorption materials before and after modification under the same conditions. Obviously, the adsorption property of the adsorbents is indeed improved after amination modification and introduction of TpPa-1. In the adsorption of acid fuchsin (Figure 8(a)), the equilibrium adsorption capacity of ACF-TpPa-1 is increased by 18.80% compared with CF, and the adsorption curve gap of reactive blue 19 is more obvious (Figure 8(b)), and the equilibrium adsorption capacity is increased by 63.08%. This is because the amino modification and the addition of TpPa-1 increase the active sites of the adsorption material, and the interaction between the π–π conjugate structure of TpPa-1 and dye molecule is also beneficial to the adsorption of dye. In addition, the ACF-TpPa-1 material also has large specific surface area and porosity (see SEM Analysis). The combination of the above results significantly improves the adsorption properties of ACF-TpPa-1 materials.

Effects of modification of adsorbent materials on: (a) acid fuchsin and (b) reactive blue 19 absorption.
Effects of initial concentration and adsorption time on dye adsorption
About 15 mg ACF-TpPa-1 were added to acid fuchsin and reactive blue 19 solutions (pH = 2) with initial concentrations of 50, 100, 200, and 300 mg/L, respectively. It can be seen from Figure 9 that the adsorption amount increased sharply in the first 20 min of adsorption, which may be due to the van der Waals gravitational force between the adsorbent and the adsorbate, indicating that the initial stage of the adsorption process is dominated by rapid physical adsorption. Similarly, it can be observed that when the initial concentration of the dye is relatively low, the time to reach the adsorption equilibrium is earlier, and as the initial concentration increases, the time to reach the adsorption equilibrium is slightly longer. Within the initial concentration range of 50–300 mg/L, the adsorption capacity of ACF-TpPa-1 also increases with the increase of the initial concentration.

Effects of initial concentration and adsorption time on absorption capacity: (a) acid fuchsin and (b) reactive blue 19.
Recyclability test
Adsorption/desorption experiments were designed to evaluate the recyclability of ACF-TpPa-1. About 15 mg of adsorbents were added to 100 mg/L acid fuchsin and reactive blue 19 solution (pH = 2), the dye removal rate was measured after 3 h of adsorption. Dye desorption experiment is carried out in 0.01 mol/L NaOH (aq) and the next round of adsorption experiment was carried out hereafter. Repeat the above experimental steps to get the data shown in Figure 10. It can be observed that after repeating the above steps five times, the removal rate of acid fuchsin by the material is greater than 90% and still remains at a high level. For reactive blue 19, the removal rate decreased more obvious, and the removal rate decreased to 51% after five cycles, this is still a good performance, higher desorption rates may be achieved by using organic solvents such as methanol and acetone. Therefore, the experimental results show that ACF-TpPa-1 has excellent recyclability and sustainability for the two dyes. The tests also show that the new functional composite material can be used as a pH-responsive dye adsorbent, that is, the adsorption/desorption performance can be adjusted by the change of pH.
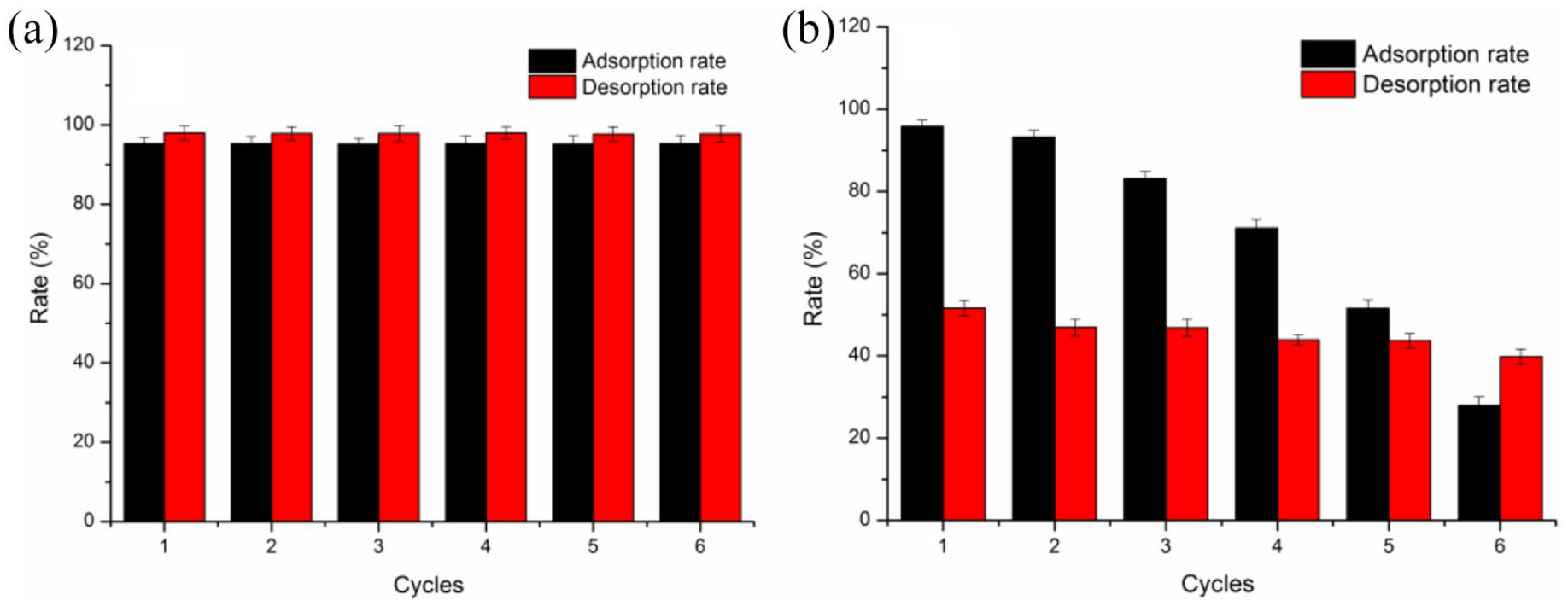
Adsorption rate and desorption rate under different cycles: (a) acid fuchsin and (b) reactive blue 19.
Adsorption kinetics
About 15 mg ACF-TpPa-1 samples are added to 30 mL of acid fuchsin solution and reactive blue 19 (solution pH = 2) at initial concentrations of 150 and 300 mg/L, respectively. The concentration of the dye remaining in the solution is measured at intervals until the residual concentration reach equilibrium. By using equation (1) to analyze the adsorption capacity qt (mg/g) of ACF-TpPa-1 on the dyes at time t, the adsorption kinetic curves of acid fuchsin and reactive blue 19 are obtained, respectively (Supplemental Figure S4).

In this equation, Co and Ct are the initial concentration and concentration at time t (mg/L), respectively. V is the volume (L) of the dye solution added, and M is the mass (g) of ACF-TpPa-1.
In this adsorption kinetics fitting, pseudo-first-order and pseudo-second-order kinetic models are used to explain the kinetic properties of the adsorption process.
For the pseudo first-order model, the equation is shown in equation (2).

For the pseudo-second-order kinetic model, the linear equation is defined as equation (3).

In equations (2) and (3), qe (mg/g) is the adsorption capacity at equilibrium, and qt (mg/g) is the adsorption capacity at time t. K1 (min-1) and K2 (g/mg/min) are the adsorption rate constants of the pseudo-first-order and pseudo-second-order respectively.
The kinetic fitting data is shown in Table 3. It can be seen that whether it is for acid fuchsin or reactive blue 19 dyes, the pseudo second-order kinetics have a higher correlation coefficient R2 than the pseudo first-order model. The pseudo-secondary adsorption capacity of the dye is 227.3 and 436.7 mg/g, which is very close to the actual equilibrium adsorption capacity, indicating that the pseudo-secondary kinetic model can better simulate the adsorption process of acid fuchsin and reactive blue 19. Figure 8 shows that in the first 20 min, the adsorption capacity increased significantly and the adsorption rate changed rapidly. This indicates that this stage mainly involves surface adsorption. After the surface layer is gradually saturated, the dye molecules begin to diffuse slowly through the surface of the material, causing the decrease of adsorption rate. In addition, the pseudo-second-order kinetic model assumes that the adsorption rate is controlled by chemical adsorption, including electron transfer and the sharing between the adsorbent, there are two types of binding sites on the adsorbent surface, which can also infer the chemical adsorption participates in the initial stage of the adsorption process.
The results of adsorption kinetics fitting of ACF-TpPa-1.

Adsorption isotherms
In the experiment, 15 mg of ACF-TpPa-1 samples were added to 30 mL of dye solutions with different initial concentrations (pH = 2) at room temperature until adsorption equilibrium was reached.
In the process of fitting adsorption data, two commonly used models, Langmuir and Freundlich, were used to calculate adsorption isotherms and adsorption equilibrium points (Supplemental Figure S5). The Langmuir isotherm model is usually used to obtain the single-layer adsorption equilibrium, while the Freundlich isotherm model, assuming the heterogeneous distribution and multi-layer adsorption of the adsorbent surface, is an empirical model of the heterogeneous system. Langmuir and Freundlich adsorption isotherm equations are expressed as equations (4) and (5), respectively.


In the equations, qe (mg/g) is the equilibrium adsorption capacity, qm is the maximum adsorption capacity, Ce (mg/L) is the equilibrium concentration of the dye, KL (L/mg) is the Langmuir constant of the reaction rate, KF (L/mg), and n are the rate constant and Freundlich’s adsorption strength, respectively.
The fitting data are shown in Table 4. The Langmuir model has better fitting effect than the Freundlich model for the two dyes. The Langmuir adsorption isotherm model is used for monolayer adsorption, and most of the adsorption sites have the same affinity for the dye molecules. In the fitting of Langmuir model, the extreme theoretical adsorption degrees of acid fuchsin and reactive blue 19 are 266.99 and 452.36 mg/g, respectively, which are close to the experimental values of the two dyes (257.98 and 449.54 mg/g). The KL value is 0–1, indicating that the adsorbent is beneficial to adsorb dyes; the KF value of the Freundlich model fitting results shows that ACF-TpPa-1 is suitable for the adsorption of dyes, and the adsorption strength 1/n reflects the advantages and capacity of adsorption, n > 1 also shows that the adsorption of the target dye is advantageous.
The results of adsorption isotherms fitting of ACF-TpPa-1.

Conclusions
In summary, this study innovatively designed a novel adsorption material, namely, ACF-TpPa-1, a functionalized composite from environmentally friendly biological matrix CF and covalent organic framework materials TpPa-1. It’s physicochemical properties were characterized by SEM, XPS, FTIR and so on, proven to be successful compounding. The adsorption results show that the ACF-TpPa-1 has an adsorption capacity of 257.98 and 449.54 mg/g for acid fuchsia and reactive blue 19, respectively, and also has excellent adsorption/desorption performance and reusability after six cycles. Compared with the previously published works and concluded that performance of the functional ACF-TpPa-1 is comparable and even superior to many other methods that have been reported for the dyes removal.15–19 This experiment successfully transformed collagen waste to high efficiency adsorbent, which embodies the green concept of turning waste into treasure, expands the application scope of covalent organic framework materials. As a collagen-based dye adsorbent with pH response, the adsorption-desorption properties can be adjusted by changing the pH conditions, so it has a realistic application in the treatment of dye wastewater.
Supplemental Material
sj-pdf-1-jef-10.1177_15589250211015898 – Supplemental material for Preparation and characterization of a novel ACF-TpPa-1 composite for dye adsorption
Supplemental material, sj-pdf-1-jef-10.1177_15589250211015898 for Preparation and characterization of a novel ACF-TpPa-1 composite for dye adsorption by Ying Han, Jiaxun Li, Bobing He and Lixin Li in Journal of Engineered Fibers and Fabrics
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to acknowledge the financial support to the project from National Natural Science Foundation of P.R. China. (Item No.21376152).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
