Abstract
In order to realize the immobilization of collagen, chitosan and sericin on the surface of polypropylene fabric, the fabric is treated by ammonia and nitrogen low temperature plasma to produce reactive groups, and then the epoxy compounds (ethylene glycol diglycidyl ether, trimethylolpropane triglycidyl ether) are used as crosslinkers for a bridging process. Single factor experiment is carried out to investigate the influence of crosslinking parameters (temperature, time and the dosage of crosslinker) on immobilization yield. Chemical composition changes on the surface of the treated samples is analyzed by infrared spectroscopy, which indicated that the ammonia plasma treatment can produce more active groups. The results showed that immobilization rate of samples crosslinked by ethylene glycol diglycidyl ether is superior to that of trimethylolpropane triglycidyl ether. It also can be found that the fabric had a better wettability and antimicrobial properties. The whiteness and comfort property of chitosan-polypropylene fabric decreased slightly.
Keywords
Introduction
Compared with other synthetic fibers, polypropylene (PP) fiber has unique competitiveness due to its convenient spinning technology, rich raw materials, low processing cost, and excellent properties including light texture, strong strength, good chemical corrosion resistance as well as high temperature resistance. 1 These characteristics make PP fiber used in many fields, such as carpets for interior decoration, workwear for clothing, blended fabrics, non-woven fabrics, and medical care dressing. 2 However, there is no polar group on the molecule chain of PP fiber, along with high crystallinity and compact structure, resulting in poor hydrophilicity and adhesion, which severely limits their application. In case some functional agents linked to PP fabric, the practicabllity of fiber will advance extremely. Owing to the inert nature of commercial PP fiber, it’s of great significance to modify it to obtain the desired reactive functional groups prior to attachment of a functional agent. 3
Several surface modification techniques such as wet chemical,4–6 ozone,7,8 plasma,9–12 and surface grafting13–16 have been developed to introduce polar groups on PP surface. However, when surface modification is a precursor to attaching a functional agent, these techniques must be tailored to introduce a specific functional group. Wet chemical and ozone treatment almost produce O-containing groups on polymer because of its oxidation process. Grafting modification needs extra initiation reaction, attached by the segment of bulk carbon chain, which sometimes influence the performance of polymer substrate. Plasma treatment is a rapid, clean and non-solvent process, is probably one of the most efficacious methods to introduce specific element or functional group onto the polymer surface by selecting a suitable gas.
Biological macromolecules are non-toxic and degradable with natural biocompatibility, which become new materials resources for the development and utilization as functional agents.17,18 Many biological macromolecules, including collagen, chitosan, and sericin, have been widely applied in the medical and cosmetics industry.19–25 Collagen is a kind of macromolecular protein found in skin, blood vessels and bones, which obtained from different animals has good health function.26,27 Chitosan have been identified as having universal antibacterial property, good coagulation ability and biocompatibility functional agent, it also has anticancer, anti-ulcer and anti-infection effects.28,29 Sericin and its hydrolysates have good pharmacological effects, such as antibacterial and anti-inflammatory, anticoagulant, anticancer, etc.30,31
Immobilization of biological molecules onto PP fabric has already made some progress. Degoutin et al. 32 reported immobilizing gentamicin and heparin on PP fabric surface by the means of plasma oxidation pre-treatment and acrylic acid grafting copolymerization. Lee et al. 33 introduced the use of plasma-coated PE/PP fibers for laccase immobilization as a dye decolorization filter. Wang et al. 34 underdone a coating of collagen/chitosan mixture on N-isopropyl acrylamide grafted PP fabric using glutaraldehyde as crosslinker. Peng et al. 35 described a four-step modification of PP nonwoven fabrics with “Piranha” solution, KH-550 and glutaraldehyde subsequently to bridge native type I collagen. Similarly, a antifouling membrane 36 is prepared by the coating of sericin to PP hollow fibre microfiltration membrane. The immobilization of chitosan or sericin on the crosslinker-impregnated PP fabric using supercritical fluid are also reported. 37 However, these processes are complex (three or more steps are required) or the results are mediocre, and even utilizing non-ecological chemicals which is not conducive to processing and health application, such as H2SO4 and glutaraldehyde. Even plasma technology is used in some of them, all the fibers are oxidized by plasma first and then grafted variable length carbon chain copolymers, resulting a unevenness surface prior to the biological molecules immobilization.
In order to immobilize biological macromolecules onto PP fabric in a simple, uniform, and green finishing process, an novel 2-step method is devised to prepare health fabric for medical packaging, workwear or hygienic decorative, as described in Figure 1. PP woven fabric is treated firstly by nitrogen and ammonia plasma to produce -NH2,38–41 which can be utilized as anchoring sites for binding functional molecules. Then, a series of immobilization of bio-functional agents (collagen, chitosan, sericin) onto the plasma-treated PP is performed using the eco-friendly crosslinker of ethylene glycol diglycidyl ether (EGDE) and trimethylolpropane triglycidyl ether (TTE). It is well known that the epoxy ring of these crosslinkers is reactive toward various nucleophilic groups of amines or hydroxyl without by-product elimination.42–44 Finally, the wettability, antimicrobial activity, whiteness and stiffness of the modified polypropylene woven fabric are analyzed, respectively.
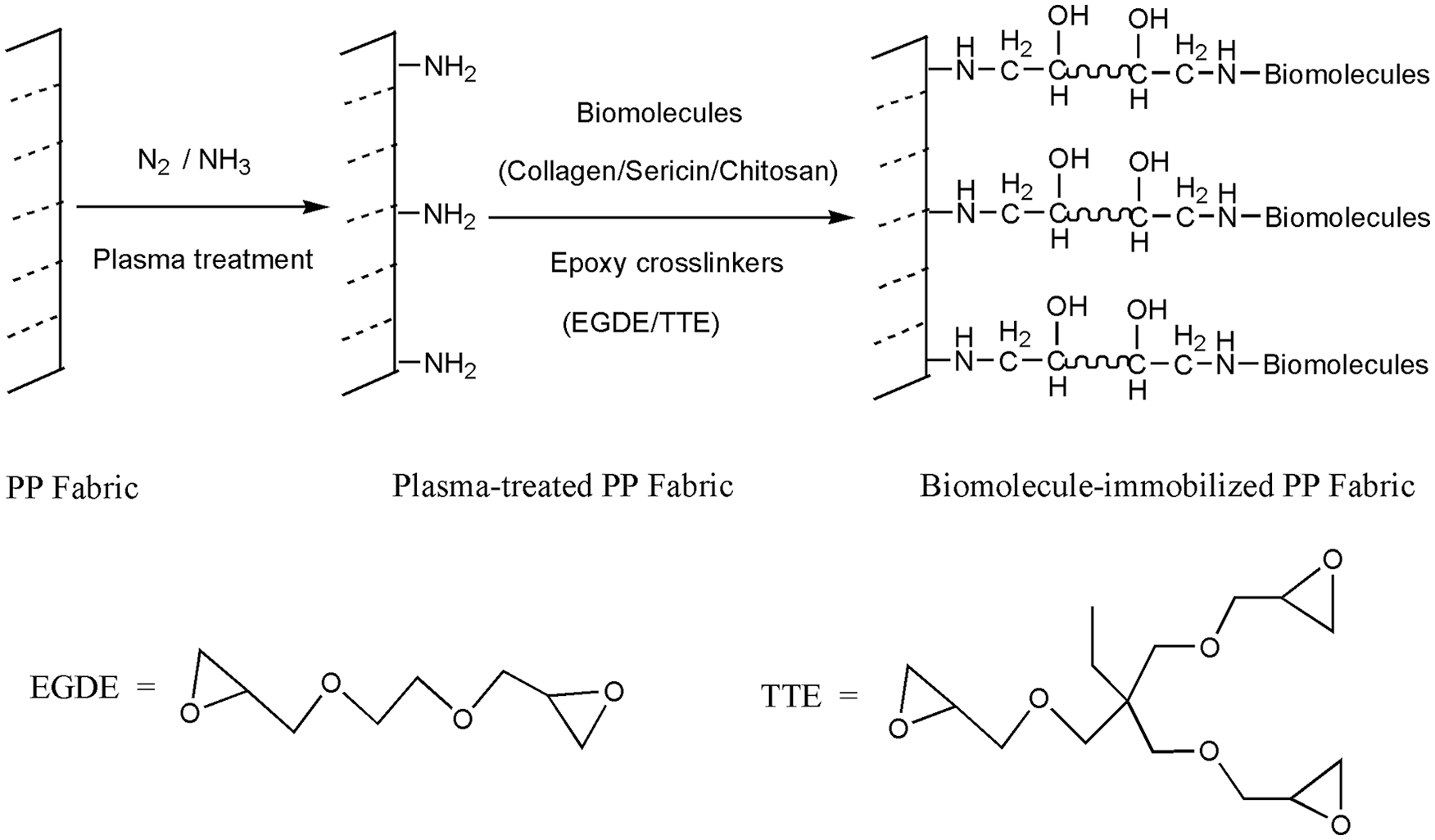
The schematic representation of biomolecules immobilization onto PP fabric surface by reacting with epoxy crosslinkers followed the pretreatment of PP substrate by N2/NH3 plasma.
Experiments
Experimental materials
PP woven fabric (warp and weft: 145/118; 200 g/m2; 118 μm thick) used as substrate polymer for modification is supplied by Anping Country Yongding filter cloth weaving Co., Ltd (China). The sample fabric was cut to the size of 8 × 8 cm (ca.4.0 g), ultrasonically cleaned in acetone and alcohol to remove organic material, dried in oven at 60°C for 1 h before being treated. Three biological macromolecules, collagen (Mw: 2000), chitosan (Mw: 50000), and sericin (Mw: 15,000) used as functional agents, were purchased by Fortuneibo-tech Co., Ltd (China) to immobilize onto the plasma-treated PP fabric. Crosslinkers of EGDE and TTE were purchased from Chengdu Aikeda Chemical Reagent Co., Ltd. Three kinds of acid dyes (C.I. Acid Red 14, C.I. Acid Yellow 44, and C.I. Acid Blue 25) supplied by JIHUA Group (China) are selected to dye PP fabric before and after modification. Other chemicals were guaranteed-grade reagents obtained from TCI Shanghai. Commercially available ultra-high purity gases (purity of NH3 ≧ 99.9%; N2 ≧ 99.9%) were used as plasma gas without further purification.
Plasma pretreatment
A laboratory-scale low-temperature plasma equipment (Omega DT-03, China) is used to treat the PP fabric. The discharge is glow discharge. Two types of atmosphere, ammonia (NH3) and nitrogen (N2) were used in the plasma pretreatment. The PP samples are treated in the plasma processing chamber in suitable conditions including the power, flow, and time. Table 1 showed the concrete process parameters.45,46
Process parameters of low temperature plasma on PP fabric.

Biological macromolecules immobilization onto pretreated PP fabric
The three types of solution with 0.5wt% chitosan, 2wt% collagen, and 2wt% sericin were prepared respectively based on previous research.37,44 Chitosan is dissolved with acetic acid (5vol %) at 30°C for a rapid dissolution, then NaOH was added until the solution approaching neutrality. For the solution of collagen and sericin, the pH values were adjusted to ca.10 with Na2B4O7·10H2O so that the amines became unprotonated. Plasma-treated PP sample and EGDE (or TTE) were put into the prepared solutions by stirring successively, the reaction were carried out at different temperature for a certain time. At the end of preset time, the fabrics were taken out and put it into the vacuum dryer at 60°C for 10 min. Finally, the samples were rinsed in distilled water, dried for testing.
Test method
Immobilization rate
The immobilization effect of biological macromolecules on PP fabric can be calculated by the weight gain formula (1):

where W1 is the weight of PP fabric treated by plasma, and W2 is the weight of PP fabric after immobilization process.
Infrared spectroscopy
Fourier transform infrared spectroscopy (IR Affinity-1, Shimadzu, Japan) equipped with an ATR accessory was used to analyze the surface chemical composition of the fabric samples. The total reflection prism is a ZnSe prism with a direction of 45° and a scanning range of 500–4000 cm−1. Each spectrum is scanned for 200 times at a resolution of 4 cm−1.
Scanning electron microscopy
The surface morphology of biological macromolecules-modified and untreated PP fabrics was observed by field emission scanning electron microscopy (FESEM) of SU 8220 (Hitachi, Japan). The acceleration voltage is 3 KV; and the resolution is 0.8 nm. Before the SEM observation, the samples were fixed onto a double-faced carbon tape adhered to a gold support and sputter-coated in an ion sputter instrument (E-1030 Hitachi).
Water contact angle and moisture regain
Static water contact angle was determined by the sessile drop method of placing a 5 μL distilled water droplet on the PP fabrics via a microsyringe. The drop images were taken after 20 s by a monochrome video camera, using PC-based control acquisition and data processing. The moisture weight of fabric was measured in a humidity chamber with a given environmental conditions (30°C, 95%RH) for 2 h. The weight gain is calculated by equation (2), in which Wm is the weight of moisturized sample, and Wd is the weight of dried sample in an oven (100°C, 2 h). All experiments were repeated three times to take the average value.

Antibacterial activity
A quantitative test method of AATCC 100-2004 is used to examine the antibacterial effect of functional PP fabric, Staphylococcus aureus (ATCC 6538) and Escherichia coli (ATCC 8099) were selected as test strains. PP fabrics were irradiated under ultraviolet lamp for 20 min to sterilize. A number of test tubes, each containing 5 mL nutrient broth, ca 0.1 g sample was placed in 5 mL liquid medium containing 10 µL concentrated bacterial culture medium. These solution were shaken for 24 h at 38°C and 160 rpm. After that, 100 µL solution was taken from each incubated sample to dilute until 105–106 CFU/mL bacterial concentration, the dilute solution were distributed over the nutritional agar plate, and were sealed. Finally, all plates were cultured at 38°C for 24 h and counted according to the number of colonies formed. The antimicrobial capacity of PP is shown in formula (3), where Y0 and Y1 are the number of colonies per unit in blank control and PP samples (CFU/mL).

Dyeing properties
Due to the introduction of amino groups on the surface of plasma modified PP fabric, acid dyes can be used for dyeing. The dyeing formula consist of 3% dye, 1:40 of bath ratio, 10% (NH4)2SO4, 10% Na2SO4 and moderate levelling agent. Before dyeing, PP sample is loaded with acetic acid to adjust pH value to 5.5. The dyeing process is shown in Figure 2. Dye uptake on fabric was obtained using the absorption value of dyeing bath measured with a visible spectrophotometer (UV1750, Shimadzu, Japan).
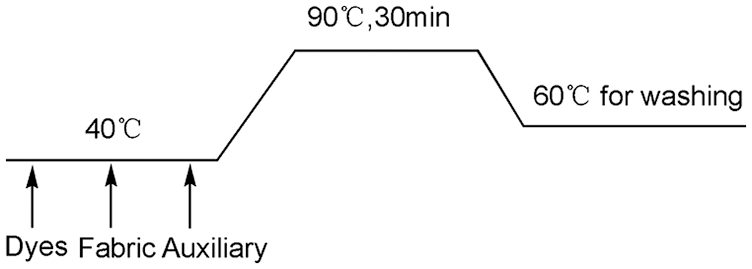
Dyeing process of plasma-treated PP fabric with acid dyes.
Stiffness and whiteness
The average bending length as the stiffness index of PP fabric is measured by inclined plane method according to the ASTM D 1388-96(2002) standard. A computer-controlled fabric stiffness tester of LLY-01 (Changzhou zhengda Co. Ltd, China) is used. Each sample is measured once at the left and right ends and on both sides of the specimen, the results are averaged four times.
Konica Minolta CM-3600 spectrophotometer (Japan) is used to measure the whiteness of PP fabrics in accordance with the AATCC 110-2000 test method. Each sample is tested from five different areas to get average numbers.
Results and discussion
Plasma modification of PP fabric
Infrared spectrum
PP fiber contains only carbon and hydrogen element, resulting in its simple IR spectra (Figure 3). Comparatively, regardless of the atmosphere of NH3 or N2, two new peaks of 3400 cm−1 and 1640 cm−1 can be seen from the spectra of plasma-treated PP, which can be attributed to the stretch vibration and bend vibration of -NH2. This indicated that -NH2 is introduced into the surface of PP after N2 and NH3 plasma modification. In addition, it is noted that the peak intensity of NH3 is higher than that of N2. This feature is a presage that NH3 plasma can produce more polar -NH2 relative to N2.

Infrared spectra of (a) untreated PP, (b) Plasma-NH3-treated PP, and (c) Plasma-N2-treated PP.
Dyeing performance
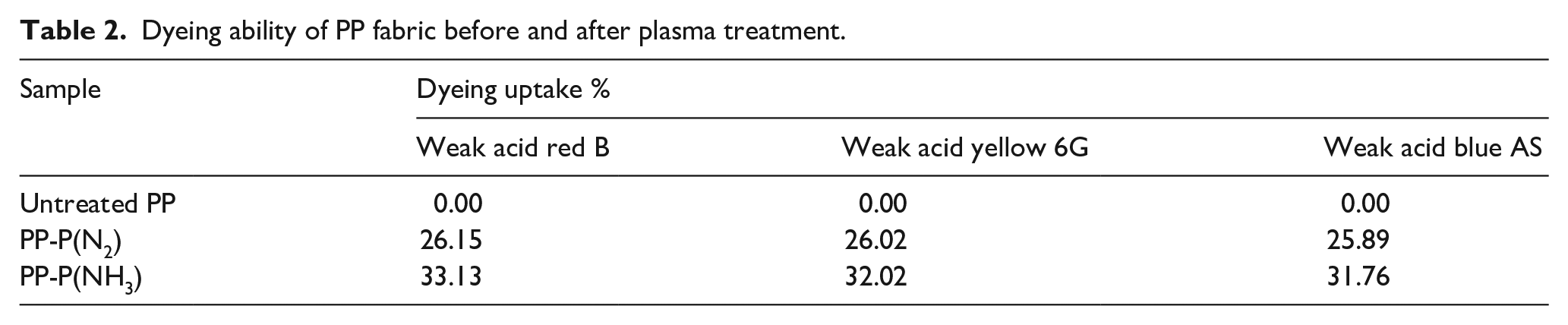
Dyeing ability of fabric is another important assessment means on whether or not the PP fabric is modified by plasma. In case of the -NH2 group existed on the fiber, weak acid dyes can be used to dye it. Table 2 shows the dyeing results of PP fabric. It can be seen that the dye uptake of plasma-modified PP is improved greatly compared with that of untreated PP. Among them, the color depth of PP treated with NH3 is better than that of N2-treated samples, as shown in Figure 4. This further proved amine group of plasma (NH3)-treated PP is better than that treated by N2 plasma.
Dyeing ability of PP fabric before and after plasma treatment.

Dyeing images of PP fabric by weak acid red B, weak acid yellow 6G and weak acid blue AS.
Immobilization of functional biomolecules
Because the effect of NH3 plasma treatment is better than that of N2, the immobilization of all of bio-functional agents was only implemented to the NH3-plasma treated PP fabric.
Effect of temperature on immobilization rate
In view of protein denaturation at high temperature, immobilization process is carried out under the temperature less than 70°C. From 25°C to 70°C, the reactions are performed with 1 g/L crosslinker for 4 h. Figure 5 showed the immobilization effect at different temperature. It can be seen that immobilization rate improves with the increase of reaction temperature. The immobilization rate of using EGDE as a crosslinker is better than TTE. For collagen immobilization, the weight gain rate decreases when the temperature exceeded 50°C. This reason may be caused by collagen degeneration, subsequently, 50°C is taken as its upper limit temperature in process. The suitable temperature for the immobilization of chitosan and sericin should exceed 60°C.

Influence of reaction temperature on immobilization rate with 1.0 g/L crosslinker for 4 h.
Effect of reaction time on immobilization rate
To investigate the relation of reaction time on immobilization rate, the experiments were carried out under the condition of 1.0 g/L crosslinker, 50°C for collagen and 60°C for the other biomolecules (sericin and chitosan), Figure 6 showed the immobilization curve after 2–12 h. The data revealed that chitosan reached the reaction equilibrium after 7 h, and the case of collagen and sericin need at least 10 h. This is considered that the reaction of epoxy group and primary is faster than secondary amine, as chitosan only has primary amine, collagen and sericin have both primary and secondary amines. In addition, it is obvious that EGDE is better than TTE. This may be due to the fact that higher molecular weight TTE is less soluble than EGDE.
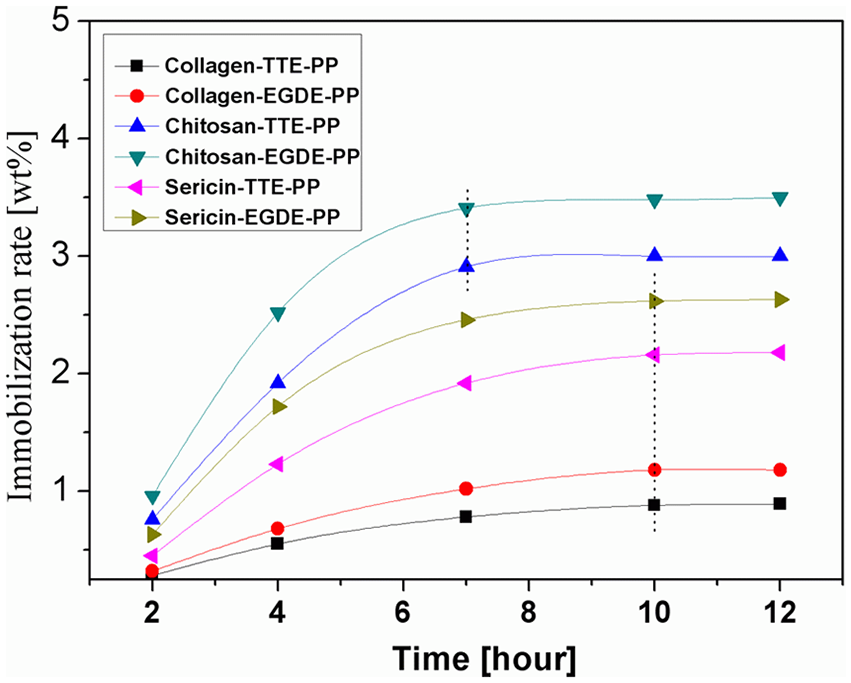
Effect of reaction time on immobilization rate with 1.0 g/L crosslinker at 50°C for collagen and 60°C for the other two (sericin and chitosan).
Effect of crosslinker dosage on immobilization rate
Crosslinker is vital auxiliary in permanent immobilization of biological molecules on plasma-treated PP for its multi-functional group can react with both biological molecules and activated PP by covalent bond. The proper crosslinker dosage can ensure effective grafting of functional agents onto PP fabric. Under the conditions of 7 h at 60°C for chitosan, and of 10 h at 50°C and 60°C for collagen and sericin, immobilization experiments were carried out with the amount of crosslinker as variable. Figure 7 showed the effect of the crosslinker dosage on the immobilization rate. It can be seen that the immobilization rate increased with the concentration of TTE until 1.2 g/L, but for EGDE, the proper concentration is 1.0 g/L. As the increase of the crosslinker dosage, the cross-linking reaction is more likely to occur in the functional agent itself, leading to the decrease of immobilization rate.

Effect of crosslinker dosage on immobilization rate.
Surface morphology of PP fabric
The surface morphology of PP fabric before and after modification was observed by field emission scanning electron microscopy. Figure 8 showed the photographs of untreated PP and PP grafted by three functional molecules using EGDE as crosslinker. Obviously, the surface of original PP fiber is smooth. After immobilization treatment, some micro-roughness can be seen on the fiber surface for three samples. These appearances exhibited that biological macromolecules were successfully grafted onto the surface of PP fabric after samples pre-treated with NH3 plasma. Among them, the impurity of sericin-PP and chitosan-PP were better than collagen-PP because of their higher immobilization rate. This case indicated that the immobilization rate is related to the molecular weight of biomolecules, as known, the lower the molecular weight, the lower the immobilization contact probability.

SEM images of (a) Untreated-PP, (b) Collagen-P(NH3)-PP, (c) Sericin-P(NH3)-PP, and (d) Chitosan-P(NH3)-PP fiber.
Hydrophilicity effect
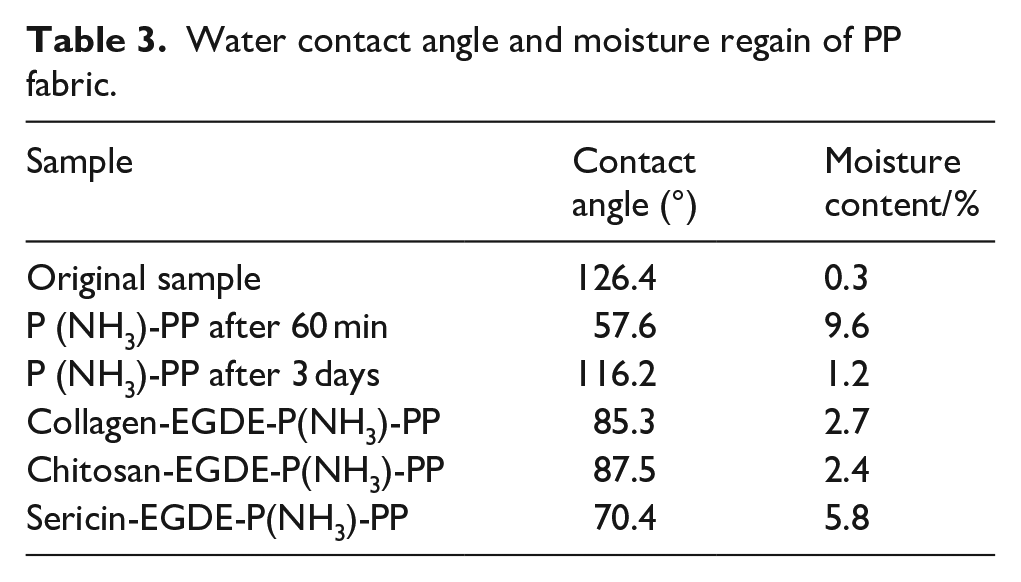
The immobilized PP fabrics with EGDE as crosslinker were selected to analyze the wettability and its equilibrium moisture content. Table 3 showed the hydrophilicity effects of untreated and treated PP fabric. It is found that PP fabric possessed good hydrophilicity after several treatment, and the hydrophilicity of PP grafted by bio-functional agents were better than the plasma-PP after long-term placement (3 days). This implied that the timeliness of plasma treatment can be improved by subsequent immobilization of functional agents. Although chitosan has the highest immobilization rate, the contact angle and moisture regain of chitosan-PP fabric is less prominent because its membrane exhibits poor hydrophilicity, which is consistent with the conclusion described by Wang et al. 34 Generally, the hydrophilicity of sericin-PP are the best with a 70.4° contact angle. These results are far superior to that of the biomolecules immobilized PP fabric using supercritical fluid. 37
Water contact angle and moisture regain of PP fabric.
Antibacterial performance of PP fabric
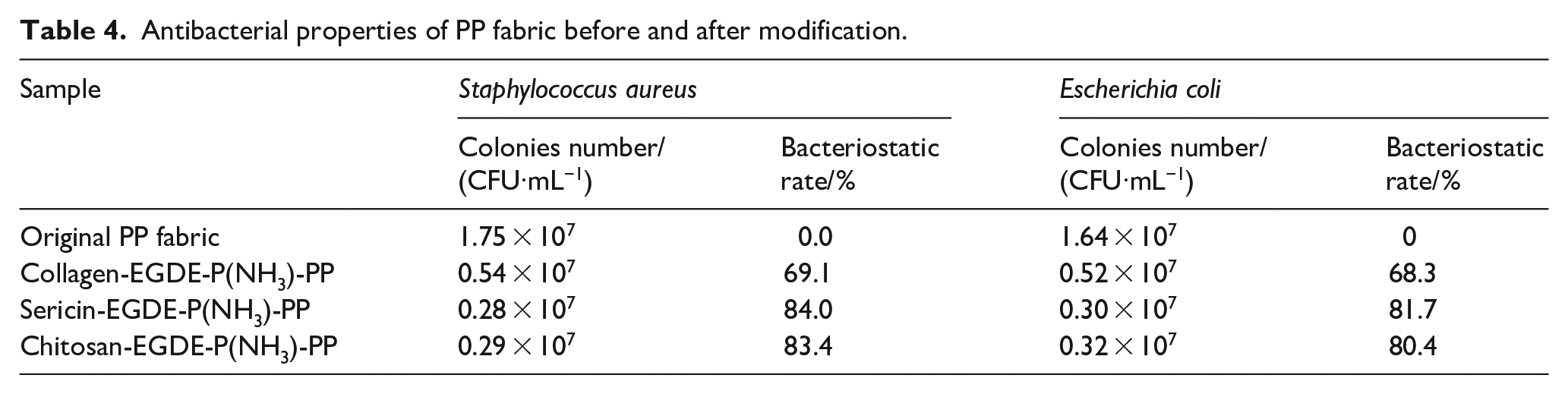
As is known, chitosan and sericin are good antimicrobial ability, that both are due to the ionic interactions between the protonated amino groups and the negatively charged surface of bacteria. 23 To investigate the antimicrobial change of these biological molecules after they are immobilized onto the PP fabric, the antimicrobial test of several PP fabrics was evaluated against representative Gram-positive bacteria and negative bacteria (Staphylococcus aureus and Escherichia coli), the results are presented on Table 4. As in other studies,32,34 untreated fabric has little antimicrobial activity. But the biomolecules-modified PP fabric reflected good antimicrobial activity, especially sericin-PP and chitosan-PP is superior to collagen-PP. It could be that the antimicrobial activity of collagen is inherently weaker than that of sericin and chitosan. 21 The mechanism of the antibacterial activity of the immobilized agent is speculated to be based on two factors. On the one hand, the active portion of biomolecules bounded to the PP fabric adsorbed the electronegative cell wall of bacteria, which could agglomerate them. On the other hand, it also formed a polymer membrane, which could prevent the transport of essential nutrients entering the bacteria cell.
Antibacterial properties of PP fabric before and after modification.
Whiteness and stiffness of PP fabric
The finished fabric often has influence on the subsequent process. It is desirable to keep the unchanged whiteness and stiffness after various modifications, the corresponding values of PP fabrics before and after modification with biomolecules (chitosan, collagen, and sericin) are listed in Table 5. It can be seen that the whiteness of PP fabrics grafted with collagen and chitosan decreased slightly, while that of PP fabrics grafted with sericin is not significantly different from that of untreated samples. For the stiffness, each functional PP fabric had an increase in bending length, but the handle feeling of fabric hardly had changes with the exception of chitosan-PP. This uncomfortable stiffness should be attributed to an higher cross-linking density of chitosan for its large molecular weight. Combined with previous hydrophilic and antibacterial results, the overall effect of finishing PP fabric with sericin is better than that of chitosan or collagen.
Whiteness and stiffness of PP fabric before and after biological macromolecules immobilization.

Conclusion
Biological macromolecules (collagen, chitosan, and sericin) were successfully immobilized onto the surface of PP fabric pre-treated by N2 and NH3 plasma respectively, with the aid of EDGE and TTE as crosslinkers. In plasma treatment, the effect of NH3 plasma is better than N2, deriving from the introduced amine group and the dyeing results with acid dyes. For the immobilization of bio-functional molecules on plasma-treated PP fabric, the suitable crosslinker dosage for EGDE and TTE is 1.0 g/L and 1.2 g/L, but immobilization rate using EGDE was higher than that of TTE. In addition, the optimal immobilization condition, 10 h at 50°C for collagen, 7 h at 60°C for chitosan and 10 h at 60°C for sericin were obtained respectively. After biological molecules immobilization, the PP fabric possessed good hydrophilicity and antimicrobial activity, but the finishing effect with collagen is not prominent. Moreover, the whiteness and handle feeling of chitosan-PP dropped somewhat severely. Overall comparatively, the application performance of sericin-PP is better than the other two.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work were supported by the Open Project Program of Fujian Key Laboratory of Novel Functional Textile Fibers and Materials, Minjiang University, China (No. FKLTFM1811); and the Research Program of Science and Technology at Universities, Inner Mongolia Autonomous Region, China (No. NJZY084).
